Organic Residues and Ammonium Effects on CO2 Emissions and Soil Quality Indicators in Limed Acid Tropical Soils
Abstract
1. Introduction
2. Materials and Methods
2.1. Collection and Preparation of Soils and Organic Residues
2.2. Analysis of Soils and Organic Residues
2.3. Experimental Treatments, Design, and CO2 Flux Measurement
2.4. Post-Incubation Sampling and Analyses
2.5. CO2 Calculations
2.6. Statistical Analysis
3. Results and Discussion
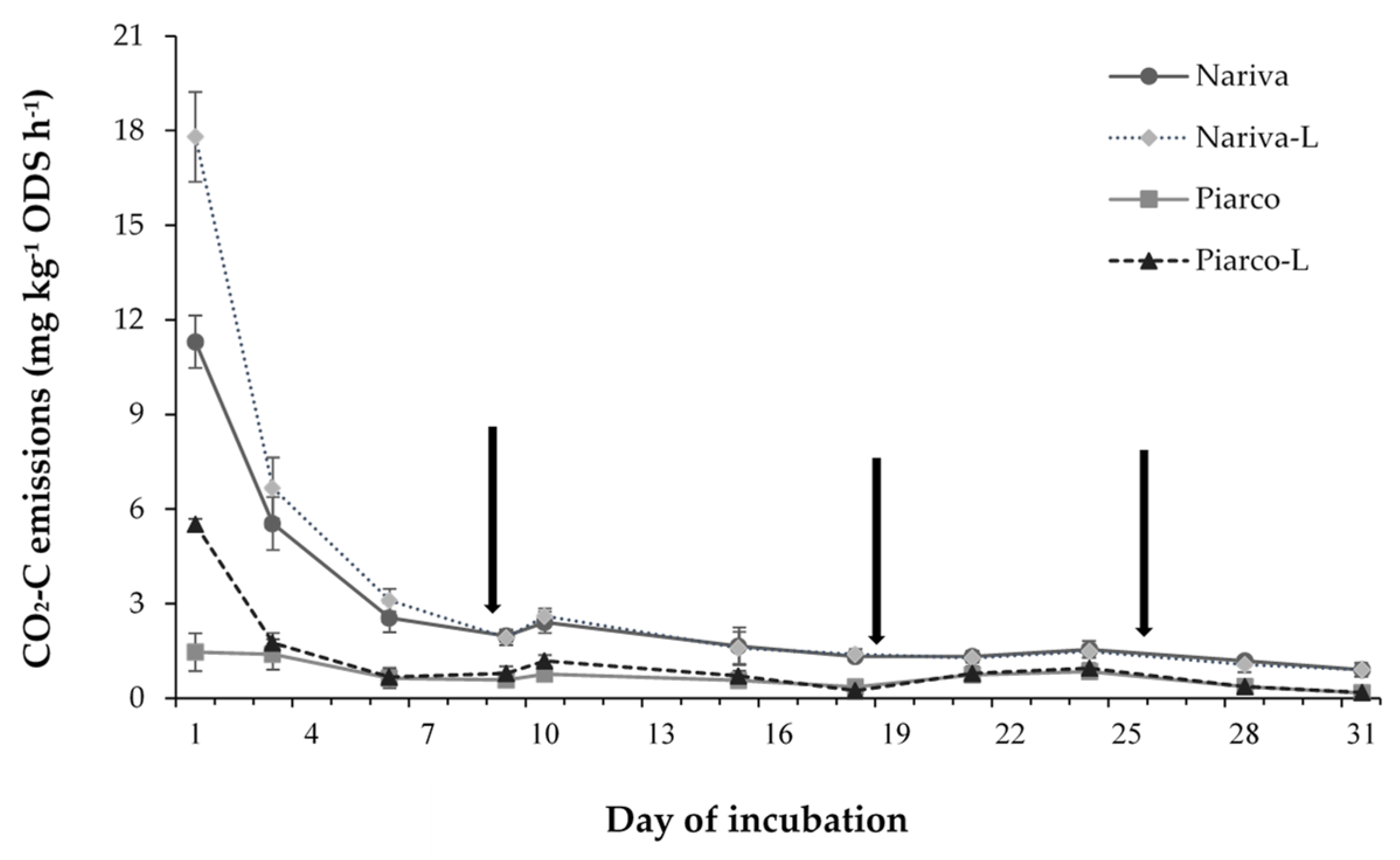
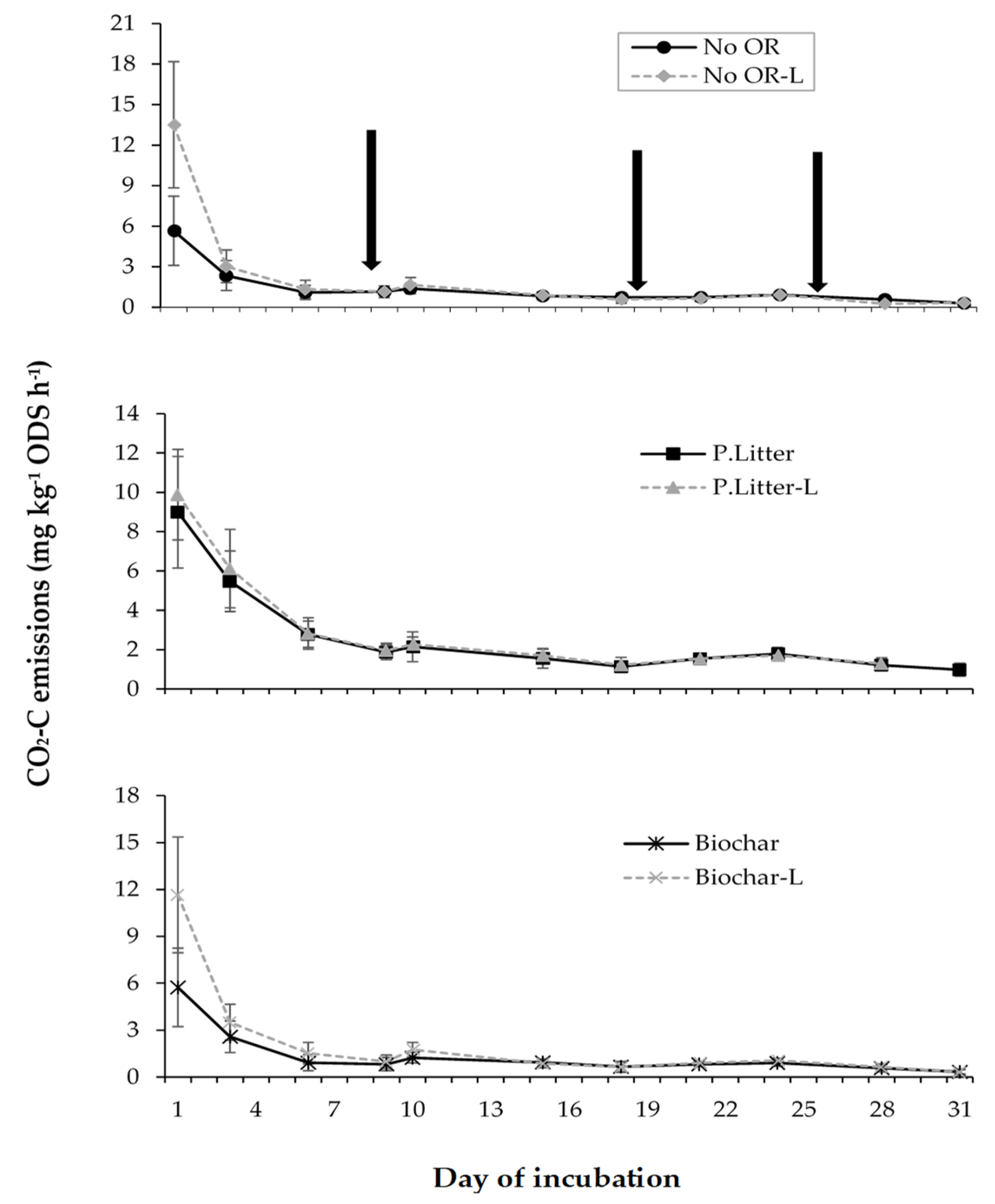
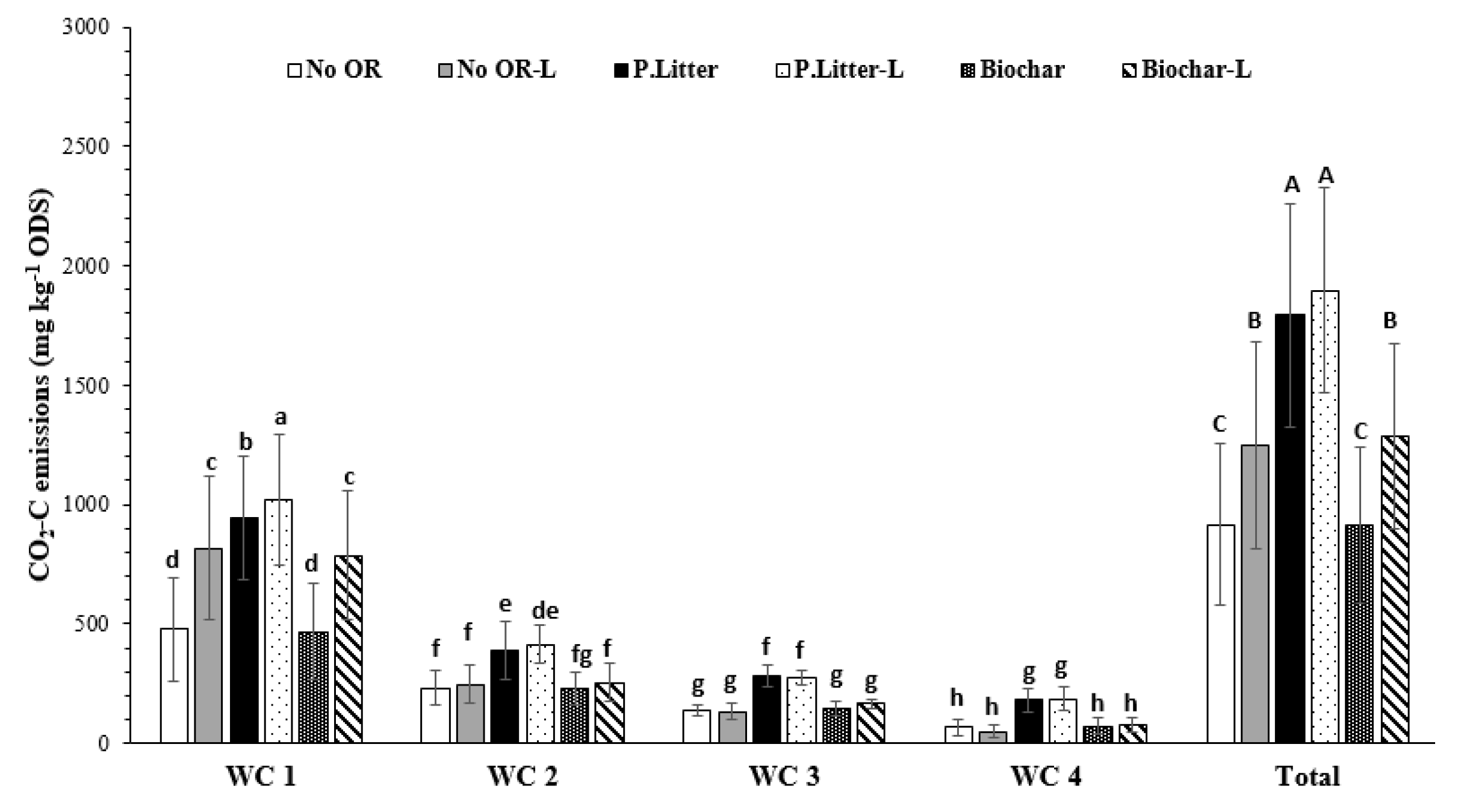
3.1. CO2 Emissions
3.1.1. Soil-Aglime Effects
3.1.2. Organic Residue-Aglime Effects
3.1.3. Ammonium-Aglime Effects
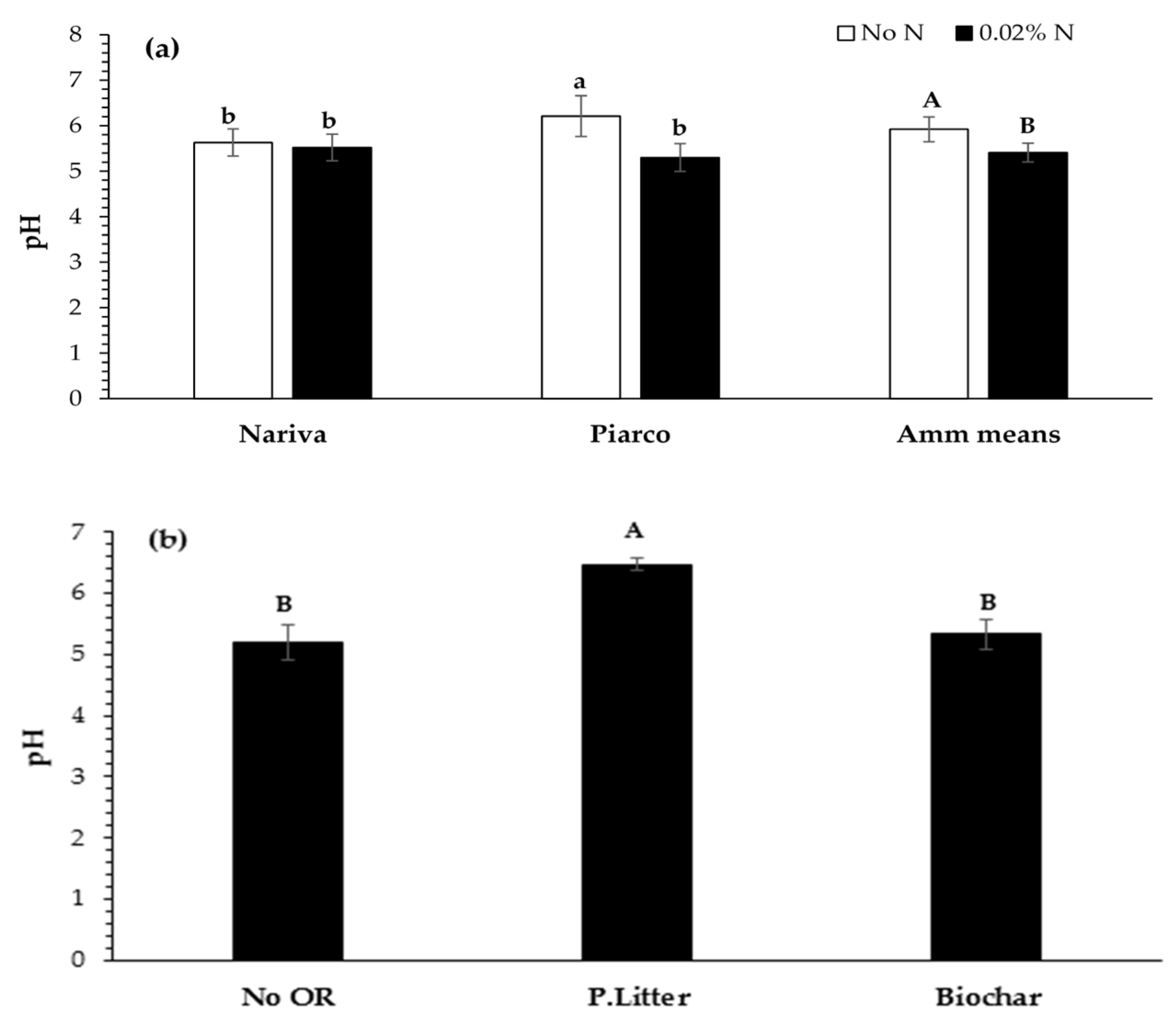
3.2. Soil Properties
3.2.1. Soil Electrical Conductivity (EC)
3.2.2. Soil pH
3.2.3. Available Nitrogen in the Soil
3.2.4. Soil Moisture Content
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bian, M.; Zhou, M.; Sun, D.; Li, C. Molecular approaches unravel the mechanism of acid soil tolerance in plants. Crop J. 2013, 1, 91–104. [Google Scholar] [CrossRef]
- von Uexküll, H.R.; Mutert, E. Global extent, development and economic impact of acid soils. Plant Soil 1995, 171, 1–15. [Google Scholar] [CrossRef]
- Brady, N.C.; Weil, R.R. The Nature and Properties of Soils, 14th ed.; Pearson Prentice Hall: Upper Saddle River, NJ, USA, 2008. [Google Scholar]
- da Costa, C.H.M.; Crusciol, C.A.C.; Neto, J.F.; Castro, G.S.A. Residual effects of superficial liming on tropical soil under no-tillage system. Pesq. Agropec. Bras. 2016, 51, 1633–1642. [Google Scholar] [CrossRef]
- Biasi, C.; Lind, S.E.; Pekkarinen, N.M.; Huttunen, J.T.; Shurpali, N.J.; Hyvönen, N.P.; Repo, M.E.; Martikainen, P.J. Direct experimental evidence for the contribution of lime to CO2 release from managed peat soil. Soil Biol. Biochem. 2008, 40, 2660–2669. [Google Scholar] [CrossRef]
- Dumale, W.A., Jr.; Miyazaki, T.; Hirai, K.; Nishimura, T. SOC Turnover and lime-CO2 evolution during liming of an acid Andisol and Ultisol. Open J. Soil Sci. 2011, 1, 49–53. [Google Scholar] [CrossRef]
- Ahmad, W.; Singh, B.; Dijkstra, F.A.; Dalal, R.C. Inorganic and organic carbon dynamics in a limed acid soil are mediated by plants. Soil Biol. Biochem. 2013, 57, 549–555. [Google Scholar] [CrossRef]
- Ahmad, W.; Singh, B.; Dijkstra, F.A.; Dalal, R.C.; Geelan-Small, P. Temperature sensitivity and carbon release in an acidic soil amended with lime and mulch. Geoderma 2014, 214, 168–176. [Google Scholar] [CrossRef]
- IPCC. 2006 IPCC guidelines for national greenhouse gas inventories. In The National Greenhouse Gas Inventories Programme; Eggleston, S., Buendia, L., Miwa, K., Ngara, T., Tanabe, K., Eds.; Institute for Global Environmental Strategies: Hayama, Japan, 2006. [Google Scholar]
- Hamilton, S.K.; Kurzman, A.L.; Arango, C.; Jin, L.; Robertson, G.P. Evidence for carbon sequestration by agricultural liming. Glob. Biogeochem. Cycles 2007, 21, GB2021. [Google Scholar] [CrossRef]
- West, T.O.; McBride, A.C. The contribution of agricultural lime to carbon dioxide emissions in the United States: Dissolution, transport, and net emissions. Agric. Ecosyst. Environ. 2005, 108, 145–154. [Google Scholar] [CrossRef]
- Semhi, K.; Suchet, P.M.; Clauer, N.; Probst, J. Impact of nitrogen fertilizers on the natural weathering-erosion processes and fluvial transport in the Garonne basin. Appl. Geochem. 2000, 15, 865–878. [Google Scholar] [CrossRef]
- Khalil, M.I.; Hossain, M.B.; Schmidhalter, U. Carbon and nitrogen mineralization in different upland soils of the subtropics treated with organic materials. Soil Biol. Biochem. 2005, 37, 1507–1518. [Google Scholar] [CrossRef]
- Hossain, M.B.; Puteh, A.B. Emission of carbon dioxide influenced by different water levels from soil incubated organic residues. Sci. World J. 2013, 2013, 638582. [Google Scholar] [CrossRef] [PubMed]
- Carrington, E.M.; Hernes, P.J.; Dyda, R.Y.; Plante, A.F.; Six, J. Biochemical changes across a carbon saturation gradient: Lignin, cutin, and suberin decomposition and stabilization in fractionated carbon pools. Soil Biol. Biochem. 2012, 47, 179–190. [Google Scholar] [CrossRef]
- Schwendener, C.M.; Lehmann, J.; de Camargo, P.B.; Luizão, R.C.; Fernandes, E.C.M. Nitrogen transfer between high-and low-quality leaves on a nutrient-poor Oxisol determined by 15N enrichment. Soil Biol. Biochem. 2005, 37, 787–794. [Google Scholar] [CrossRef]
- Belfon, R. Sequestering Carbon and Improving Soil Fertility Using Organic Amendments. Ph.D. Thesis, The University of the West Indies, St. Augustine, Trinidad and Tobago, 2015. [Google Scholar]
- Tillman, D.; Fargione, J.; Wolff, B.; D’Antonio, C.; Dobson, A.; Howarth, R.; Schindler, D.; Schlesinger, W.H.; Simberloff, D.; Swackhamer, D. Forecasting agriculturally driven global environmental change. Science 2001, 292, 281–284. [Google Scholar] [CrossRef] [PubMed]
- Brown, C.B.; Bally, G.S. Land Capability of Trinidad and Tobago. Volume 3: Soils of Northern Trinidad; Government Printery: Port-of-Spain, Trinidad and Tobago, 1966.
- Brown, C.B.; Bally, G.S. Land Capability of Trinidad and Tobago. Volume 5: Soils of Southern Trinidad; Government Printery: Port-of-Spain, Trinidad and Tobago, 1968.
- Brown, C.B.; Bally, G.S. Land Capability of Trinidad and Tobago. Volume 4: Soils of Central Trinidad; Government Printery: Port-of-Spain, Trinidad and Tobago, 1970.
- McLean, E.O. Soil pH and lime requirement. In Methods of Soil Analysis. Part 2. Chemical and Microbiological Properties; Page, A.L., Miller, R.H., Keeney, D.R., Eds.; Soil Science Society of America, Inc.: Madison, WI, USA, 1982; pp. 199–223. [Google Scholar]
- Rhoades, J.D. Soluble salts. In Methods of Soil Analysis. Part 2. Chemical and Microbiological Properties; Page, A.L., Miller, R.H., Keeney, D.R., Eds.; Soil Science Society of America, Inc.: Madison, WI, USA, 1982; pp. 167–178. [Google Scholar]
- Hardy, F.; Lewis, A.H. A rapid electrometric method for measuring “lime requirements” of soils. J. Agri. Sci. 1929, 19, 17–25. [Google Scholar] [CrossRef]
- Gee, G.W.; Or, D. Particle-size analysis. In Methods of Soil Analysis. Part 4. Physical Methods; Dane, J.H., Topp, G.C., Eds.; Soil Science Society of America, Inc.: Madison, WI, USA, 2002; pp. 255–293. [Google Scholar]
- Rhoades, J.D. Cation exchange capacity. In Methods of Soil Analysis. Part 2. Chemical and Microbiological Properties; Page, A.L., Miller, R.H., Keeney, D.R., Eds.; Soil Science Society of America, Inc.: Madison, WI, USA, 1982; pp. 149–157. [Google Scholar]
- Maynard, D.G.; Kalra, Y.P.; Crumbaugh, J.A. Nitrate and exchangeable ammonium nitrogen. In Soil Sampling and Methods of Analysis, 2nd ed.; Carter, M.R., Gregorich, E.G., Eds.; CRC Press: Boca Raton, FL, USA, 2008; pp. 181–196. [Google Scholar]
- Tamir, G.; Shenker, M.; Heller, H.; Bloom, P.R.; Fine, P.; Bar-Tal, A. Organic N mineralization and transformations in soils treated with animal waste in relation to carbonate dissolution and precipitation. Geoderma 2013, 209, 50–56. [Google Scholar] [CrossRef]
- Nelson, D.W.; Sommers, L.E. Total carbon, organic carbon, and organic matter. In Methods of Soil Analysis. Part 2. Chemical and Microbiological Properties; Page, A.L., Miller, R.H., Keeney, D.R., Eds.; Soil Science Society of America, Inc.: Madison, WI, USA, 1982; pp. 539–577. [Google Scholar]
- Bremner, J.M.; Mulvaney, C.S. Nitrogen—Total. In Methods of Soil Analysis. Part 2. Chemical and Microbiological Properties; Page, A.L., Miller, R.H., Keeney, D.R., Eds.; Soil Science Society of America, Inc.: Madison, WI, USA, 1982; pp. 595–624. [Google Scholar]
- Harris, D.; Horwáth, W.R.; van Kessel, C. Acid fumigation of soils to remove carbonates prior to total organic carbon or carbon-13 isotopic analysis. Soil Sci. Soc. Am. J. 2001, 65, 1853–1856. [Google Scholar] [CrossRef]
- Burford, J.R.; Bremner, J.M. Gas chromatographic determination of carbon dioxide evolved from soils in closed systems. Soil Biol. Biochem. 1972, 4, 191–197. [Google Scholar] [CrossRef]
- Nelson, R.E. Carbonate and gypsum. In Methods of Soil Analysis. Part 2. Chemical and Microbiological Properties; Page, A.L., Miller, R.H., Keeney, D.R., Eds.; Soil Science Society of America, Inc.: Madison, WI, USA, 1982; pp. 181–196. [Google Scholar]
- Gouveia, G.A.; Eudoxie, G.D. Distribution of fertiliser N among fixed ammonium fractions as affected by moisture and fertiliser source and rate. Biol. Fertil. Soils 2007, 44, 9–18. [Google Scholar] [CrossRef]
- Belfon, R.; Bekele, I.; Eudoxie, G.; Voroney, P.; Gouveia, G. Sequestering carbon and improving soil fertility; validation of an improved method for estimating CO2 flux. Geoderma 2014, 235, 323–328. [Google Scholar] [CrossRef]
- Anderson, J.P.E. Soil Respiration. In Methods of Soil Analysis. Part 2. Chemical and Microbiological Properties; Page, A.L., Miller, R.H., Keeney, D.R., Eds.; Soil Science Society of America, Inc.: Madison, WI, USA, 1982; pp. 831–871. [Google Scholar]
- Stotzky, G. Microbial Respiration. Methods of Soil Analysis. Part 2. Chemical and Microbiological Properties; Black, C.A., Evans, D.D., White, J.L., Eds.; American Society of Agronomy: Madison, WI, USA, 1965; pp. 1550–1572. [Google Scholar]
- Grover, S.P.; Butterly, C.R.; Wang, X.; Tang, C. The short-term effects of liming on organic carbon mineralisation in two acidic soils as affected by different rates and application depths of lime. Biol. Fert. Soils 2017, 53, 431–443. [Google Scholar] [CrossRef]
- Keller, J.K.; Bridgham, S.D.; Chapin, C.T.; Iversen, C.M. Limited effects of six years of fertilization on carbon mineralization dynamics in a Minnesota fen. Soil Biol. Biochem. 2005, 37, 1197–1204. [Google Scholar] [CrossRef]
- Miyazawa, M.; Takahashi, T.; Sato, T.; Kanno, H.; Nanzyo, M. Factors controlling accumulation and decomposition of organic carbon in humus horizons of Andosols. Biol. Fertil. Soils 2013, 49, 929–938. [Google Scholar] [CrossRef]
- Karberg, N.J.; Pregitzer, K.S.; King, J.S.; Friend, A.L.; Wood, J.R. Soil carbon dioxide partial pressure and dissolved inorganic carbonate chemistry under elevated carbon dioxide and ozone. Oecologia 2005, 142, 296–306. [Google Scholar] [CrossRef] [PubMed]
- Ramnarine, R.; Wagner-Riddle, C.; Dunfield, K.E.; Voroney, R.P. Contributions of carbonates to soil CO2 emissions. Can. J. Soil Sci. 2012, 92, 599–607. [Google Scholar] [CrossRef]
- Lardner, T.; George, S.; Tibbett, M. Interacting controls on innate sources of CO2 efflux from a calcareous arid zone soil under experimental acidification and wetting. J. Arid Environ. 2015, 122, 117–123. [Google Scholar] [CrossRef]
- Mukherjee, A.; Zimmerman, A.R.; Hamdan, R.; Cooper, W.T. Physicochemical changes in pyrogenic organic matter (biochar) after 15 months of field aging. Solid Earth 2014, 5, 693–704. [Google Scholar] [CrossRef]
- Clough, T.J.; Condron, L.M.; Kammann, C.; Müller, C. A review of biochar and soil nitrogen dynamics. Agronomy 2013, 3, 275–293. [Google Scholar] [CrossRef]
- Yao, Y.; Gao, B.; Zhang, M.; Inyang, M.; Zimmerman, A.R. Effect of biochar amendment on sorption and leaching of nitrate, ammonium, and phosphate in a sandy soil. Chemosphere 2012, 89, 1467–1471. [Google Scholar] [CrossRef]
- Edmeades, D.C. Effects of lime on effective cation exchange capacity and exchangeable cations on a range of New Zealand soils. N. Z. J. Agric. Res. 1982, 25, 27–33. [Google Scholar] [CrossRef]
- Buni, A. Effects of liming acidic soils on improving soil properties and yield of haricot bean. J. Environ. Anal. Toxicol. 2014, 5, 248. [Google Scholar]
- Han, J.; Shi, J.; Zeng, L.; Xu, J.; Wu, L. Effects of nitrogen fertilization on the acidity and salinity of greenhouse soils. Environ. Sci. Pollut. Res. 2015, 22, 2976–2986. [Google Scholar] [CrossRef] [PubMed]
- Omari, R.A.; Bellingrath-Kimura, D.S.; Fujii, Y.; Sarkodee-Addo, E.; Sarpong, K.A.; Oikawa, Y. Nitrogen Mineralization and Microbial Biomass Dynamics in Different Tropical Soils Amended with Contrasting Organic Resources. Soil Syst. 2018, 2, 63. [Google Scholar] [CrossRef]
- Judge, A. The Effects of Surface-Applied Poultry Manure on Top-and Subsoil Acidity and Selected Soil Fertility Characteristics. Master’s Thesis, University of Natal, Pietermaritzburg, South Africa, 2001. [Google Scholar]
- Li, Z.; Wei, B.; Wang, X.; Zhang, Y.; Zhang, A. Response of soil organic carbon fractions and CO2 emissions to exogenous composted manure and calcium carbonate. J. Soils Sediments 2018, 18, 1832–1843. [Google Scholar] [CrossRef]
- Manangi, M.R.; Coon, C.N. The effect of calcium carbonate particle size and solubility on the utilization of phosphorus from phytase for broilers. Int. J. Pollut. Sci. 2007, 6, 85–90. [Google Scholar]
- Tamir, G.; Shenker, M.; Heller, H.; Bloom, P.R.; Fine, P.; Bar-Tal, A. Can soil carbonate dissolution lead to overestimation of soil respiration? Soil Sci. Soc. Am. J. 2011, 75, 1414–1422. [Google Scholar] [CrossRef]
- Rothrock, M.J.; Cook, K.L.; Warren, J.G.; Eiteman, M.A.; Sistani, K. Microbial mineralization of organic nitrogen forms in poultry litters. J. Environ. Qual. 2010, 39, 1848–1857. [Google Scholar] [CrossRef]
- Haynes, R.J.; Naidu, R. Influence of lime, fertilizer and manure applications on soil organic matter content and soil physical conditions: A review. Nutr. Cycl. Agroecosyst. 1998, 51, 123–137. [Google Scholar] [CrossRef]
- Sajjadi, S.A.; Mahmoodabadi, M. Aggregate breakdown and surface seal development influenced by rain intensity, slope gradient and soil particle size. Solid Earth 2015, 6, 311–321. [Google Scholar] [CrossRef]
- Tedeschi, A.; Zong, L.; Huang, C.; Vitale, L.; Volpe, M.; Xue, X. Effect of salinity on growth parameters, soil water potential and ion composition in Cucumis melo cv. Huanghemi in north-western China. J. Agron. Crop Sci. 2017, 203, 41–55. [Google Scholar] [CrossRef]
| Parameter | Nariva Soil | Piarco Soil | Poultry Litter | Biochar |
|---|---|---|---|---|
| pH (H2O) | 4.52 1 | 4.43 1 | 8.58 2 | 7.35 2 |
| pH (CaCl2) | 4.33 1 | 4.05 1 | 8.43 2 | 6.95 2 |
| ECe (dS m−1) | 2.08 | 0.793 | nd | nd |
| ECs (dS m−1) | 0.80 | 0.217 | 14.3 | 0.235 |
| LR (Mg ha−1) | 6.54 | 1.72 | nd | nd |
| Clay (%) | 47.1 | 12.6 | nd | nd |
| Sand (%) | 29.9 | 43.0 | nd | nd |
| Silt (%) | 23.0 | 44.4 | nd | nd |
| CEC (cmol kg−1) | 28.6 | 5.16 | nd | nd |
| WHC (g g−1) | 0.602 | 0.319 | nd | nd |
| TC (%) | 4.39 3 | 0.916 3 | 21.5 4 | 85.7 4 |
| IC (%) | --- | --- | 1.10 5 | --- |
| TN (%) | 0.470 6 | 0.092 6 | 3.58 4 | 0.16 4 |
| NH4+-N (mg kg−1) | 115 | 17.0 | 135 | nd |
| NO3−-N (mg kg−1) | 24.4 | 37.7 | 1387 | nd |
| C:N7 | 9.35 | 10.0 | 6.01 | 536 |
| CaCO3 (%) | --- | --- | 9.20 | --- |
| Soil Series | Soil × Organic Residue (OR) Means | Soil × Ammonium Means | Soil × Aglime Means | ||||
|---|---|---|---|---|---|---|---|
| No OR | Poultry Litter | Biochar | No N | 0.02 % N | No Aglime | 0.2 % Aglime | |
| Nariva | 1.38d | 2.37b | 1.54c | 1.45c | 2.07a | 1.84a | 1.67b |
| Piarco | 0.845f | 2.76a | 0.725f | 0.966d | 1.92b | 1.32c | 1.56b |
| MEM | 1.11B | 2.56A | 1.13B | 1.21B | 1.99A | ns | ns |
| NH4+-N (mg kg−1 ODS) | NO3−-N (mg kg−1 ODS) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Soil × OR Means | Soil × Ammonium Means | Soil × OR Means | Soil × Ammonium Means | Soil × Aglime Means | ||||||||
| Soil Series | No OR | P.Litter | Biochar | No N | 0.02% N | No OR | P.Litter | Biochar | No N | 0.02% N | No Aglime | 0.2% Aglime |
| Nariva | 342b | 505a | 320b | 313b | 466a | 0.143c | 34.1bc | 0.982c | 10.4c | 13.1c | 14.3c | 9.14c |
| Piarco | 77.3c | 36.6c | 51.1c | 20.3d | 89.7c | 62.5b | 252a | 71.6b | 91.7b | 166a | 100b | 157a |
| MEM | 210B | 271A | 186B | 167B | 278A | 31.3B | 143A | 36.3B | 51B | 89.4A | ns | ns |
| Wetting Cycle (WC) | Nariva | Piarco | ||
|---|---|---|---|---|
| No N | 0.02% N | No N | 0.02% N | |
| 1 | 63.5c | 69.0b | 24.3g | 44.7f |
| 2 | 56.3d | 61.8c | 11.2i | 20.1gh |
| 3 | 69.6b | 70.4b | 19.1h | 40.0f |
| 4 | 82.0a | 80.4a | 50.9e | 62.9c |
| Soil × Amm means | 67.8A | 70.4A | 26.4C | 41.9B |
| Ammonium | Aglime | |
|---|---|---|
| No Aglime | 0.02% Aglime | |
| No N | 43.3c | 50.9b |
| 0.02% N | 56.3a | 56.0a |
| Aglime means | 49.8B | 53.4A |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bramble, D.S.E.; Gouveia, G.A.; Ramnarine, R. Organic Residues and Ammonium Effects on CO2 Emissions and Soil Quality Indicators in Limed Acid Tropical Soils. Soil Syst. 2019, 3, 16. https://doi.org/10.3390/soilsystems3010016
Bramble DSE, Gouveia GA, Ramnarine R. Organic Residues and Ammonium Effects on CO2 Emissions and Soil Quality Indicators in Limed Acid Tropical Soils. Soil Systems. 2019; 3(1):16. https://doi.org/10.3390/soilsystems3010016
Chicago/Turabian StyleBramble, De Shorn E., Gregory A. Gouveia, and Ravindra Ramnarine. 2019. "Organic Residues and Ammonium Effects on CO2 Emissions and Soil Quality Indicators in Limed Acid Tropical Soils" Soil Systems 3, no. 1: 16. https://doi.org/10.3390/soilsystems3010016
APA StyleBramble, D. S. E., Gouveia, G. A., & Ramnarine, R. (2019). Organic Residues and Ammonium Effects on CO2 Emissions and Soil Quality Indicators in Limed Acid Tropical Soils. Soil Systems, 3(1), 16. https://doi.org/10.3390/soilsystems3010016





