Effect of Fermented Rice Drink “Amazake” on Patients with Nonalcoholic Fatty Liver Disease and Periodontal Disease: A Pilot Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
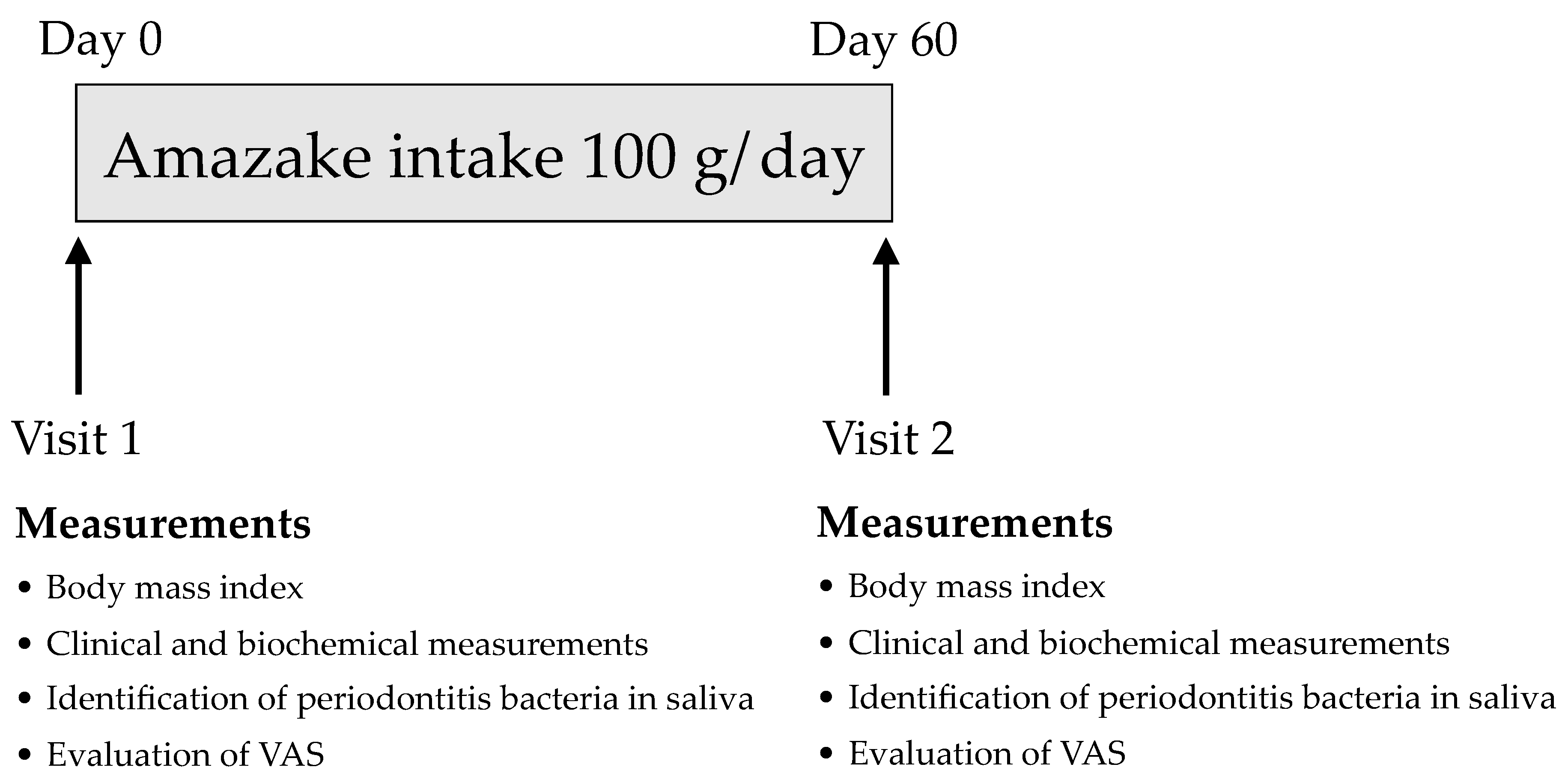
2.2. Study Assessment Protocol
2.3. Saliva Sample Collection
2.4. Identification of Periodontitis Bacteria and Detection of P. gingivalis fimA Genotypes
2.5. Safety Monitoring
2.6. Statistical Analysis
3. Results
3.1. Patient Characteristics
3.2. Effects of Amazake Intake on Biochemical Examinations, VAS, and Periodontitis Disease Bacteria
3.3. M2BPGi Levels and Effects of Amazake Intake
3.4. Adverse Events
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AFP | α-fetoprotein |
| ALP | alkaline phosphatase |
| ALT | alanine aminotransferase |
| Anti-HCV | antibodies to HCV |
| AST | aspartate aminotransferase |
| BCAA | branched-chain amino acids |
| BMI | body mass index |
| BUN | blood urea nitrogen |
| Crea | creatinine |
| CRP | C-reactive protein |
| D.Bil | direct bilirubin |
| DAA | direct-acting antiviral drug |
| Hb | hemoglobin |
| HbA1c | hemoglobin A1c |
| HBsAg | hepatitis B surface antigen |
| HBV | hepatitis B virus |
| HCC | hepatocellular carcinoma |
| HCV | hepatitis C virus |
| HDL | high-density lipoprotein |
| IL6 | interleukin-6 |
| IRI | immune reactive insulin |
| LDH | lactic dehydrogenase |
| LDL | low-density lipoprotein |
| LES | late evening snacks |
| M2BPGi | mac-2 binding protein glycosylated isomers |
| NAFLD | nonalcoholic fatty liver disease |
| NASH | nonalcoholic steatohepatitis |
| P-III-P | type III procollagen-N-peptide |
| PLT | platelets |
| QOL | quality of life |
| RBC | red blood cell |
| T. Bil | total bilirubin |
| T. cho | total cholesterol |
| TNFα | tumor necrosis factor α |
| Total P1NP | total procollagen type 1 amino-terminal propeptide |
| TRACP-5b | tartrate-resistant acid phosphatase-5b |
| VAS | visual analog scale |
| WBC | white blood cell |
| γ-GTP | gamma-glutamyl transpeptidase |
References
- Younossi, Z.M.; Otgonsuren, M.; Henry, L.; Venkatesan, C.; Mishra, A.; Erario, M.; Hunt, S. Association of nonalcoholic fatty liver disease (NAFLD) with hepatocellular carcinoma (HCC) in the United States from 2004 to 2009. Hepatology 2015, 62, 1723–1730. [Google Scholar] [CrossRef]
- Younossi, Z.M.; Koenig, A.B.; Abdelatif, D.; Fazel, Y.; Henry, L.; Wymer, M. Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology 2015, 64, 73–84. [Google Scholar] [CrossRef] [Green Version]
- Estes, C.; Razavi, H.; Loomba, R.; Younossi, Z.; Sanyal, A.J. Modeling the epidemic of nonalcoholic fatty liver disease demonstrates an exponential increase in burden of disease. Hepatology 2018, 67, 123–133. [Google Scholar] [CrossRef]
- Younossi, Z.M.; Golabi, P.; de Avila, L.; Paik, J.M.; Srishord, M.; Fukui, N.; Qiu, Y.; Burns, L.; Afendy, A.; Nader, F. The global epidemiology of NAFLD and NASH in patients with type 2 diabetes: A systematic review and meta-analysis. J. Hepatol. 2019, 71, 793–801. [Google Scholar] [CrossRef] [PubMed]
- Hamaguchi, M.; Kojima, T.; Takeda, N.; Nakagawa, T.; Taniguchi, H.; Fujii, K.; Omatsu, T.; Nakajima, T.; Sarui, H.; Shimazaki, M.; et al. The Metabolic Syndrome as a Predictor of Nonalcoholic Fatty Liver Disease. Ann. Intern. Med. 2005, 143, 722–728. [Google Scholar] [CrossRef]
- Eguchi, Y.; Nafld, J.-; Hyogo, H.; Ono, M.; Mizuta, T.; Ono, N.; Fujimoto, K.; Chayama, K.; Saibara, T. Prevalence and associated metabolic factors of nonalcoholic fatty liver disease in the general population from 2009 to 2010 in Japan: A multicenter large retrospective study. J. Gastroenterol. 2012, 47, 586–595. [Google Scholar] [CrossRef] [PubMed]
- Estes, C.; Anstee, Q.M.; Arias-Loste, M.T.; Bantel, H.; Bellentani, S.; Caballeria, J.; Colombo, M.; Craxi, A.; Crespo, J.; Day, C.P.; et al. Modeling NAFLD disease burden in China, France, Germany, Italy, Japan, Spain, United Kingdom, and United States for the period 2016–2030. J. Hepatol. 2018, 69, 896–904. [Google Scholar] [CrossRef] [PubMed]
- Koda, M.; Kawakami, M.; Murawaki, Y.; Senda, M. The impact of visceral fat in nonalcoholic fatty liver disease: Cross-sectional and longitudinal studies. J. Gastroenterol. 2007, 42, 897–903. [Google Scholar] [CrossRef]
- Thoma, C.; Day, C.P.; Trenell, M. Lifestyle interventions for the treatment of non-alcoholic fatty liver disease in adults: A systematic review. J. Hepatol. 2012, 56, 255–266. [Google Scholar] [CrossRef]
- Glass, L.M.; Dickson, R.C.; Anderson, J.C.; Suriawinata, A.A.; Putra, J.; Berk, B.S.; Toor, A. Total Body Weight Loss of ≥10 % Is Associated with Improved Hepatic Fibrosis in Patients with Nonalcoholic Steatohepatitis. Dig. Dis. Sci. 2015, 60, 1024–1030. [Google Scholar] [CrossRef]
- El-Serag, H.B.; Tran, T.; Everhart, J.E. Diabetes increases the risk of chronic liver disease and hepatocellular carcinoma. Gastroenterology 2004, 126, 460–468. [Google Scholar] [CrossRef] [PubMed]
- David, K.; Kowdley, K.V.; Unalp, A.; Kanwal, F.; Brunt, E.M.; Schwimmer, J.B.; NASH CRN Research Group. Quality of life in adults with nonalcoholic fatty liver disease: Baseline data from the nonalcoholic steatohepatitis clinical research network. Hepatology 2009, 49, 1904–1912. [Google Scholar] [CrossRef] [Green Version]
- Beck, J.D.; Slade, G.; Offenbacher, S. Oral disease, cardiovascular disease and systemic inflammation. Periodontol. 2000 2000, 23, 110–120. [Google Scholar] [CrossRef] [PubMed]
- Mealey, B.L.; Oates, T.W.; American Academy of Periodontology. Diabetes Mellitus and Periodontal Diseases. J. Periodontol. 2006, 77, 1289–1303. [Google Scholar] [CrossRef] [PubMed]
- Falcao, A.; Bullón, P. A review of the influence of periodontal treatment in systemic diseases. Periodontol. 2000 2019, 79, 117–128. [Google Scholar] [CrossRef] [PubMed]
- Mealey, B.L.; Rethman, M.P. Periodontal disease and diabetes mellitus. Bidirectional relationship. Dent. Today 2003, 22, 107–113. [Google Scholar] [PubMed]
- Mei, F.; Xie, M.; Huang, X.; Long, Y.; Lu, X.; Wang, X.; Chen, L. Porphyromonas gingivalis and Its Systemic Impact: Current Status. Pathogens 2020, 9, 944. [Google Scholar] [CrossRef]
- Yoneda, M.; Naka, S.; Nakano, K.; Wada, K.; Endo, H.; Mawatari, H.; Imajo, K.; Nomura, R.; Hokamura, K.; Ono, M.; et al. Involvement of a periodontal pathogen, Porphyromonas gingivalis on the pathogenesis of non-alcoholic fatty liver disease. BMC Gastroenterol. 2012, 12, 16. [Google Scholar] [CrossRef] [Green Version]
- Alazawi, W.; Bernabe, E.; Tai, D.; Janicki, T.; Kemos, P.; Samsuddin, S.; Syn, W.-K.; Gillam, D.; Turner, W. Periodontitis is associated with significant hepatic fibrosis in patients with non-alcoholic fatty liver disease. PLoS ONE 2017, 12, e0185902. [Google Scholar] [CrossRef] [Green Version]
- Arimatsu, K.; Yamada, H.; Miyazawa, H.; Minagawa, T.; Nakajima, M.; Ryder, M.I.; Gotoh, K.; Motooka, D.; Nakamura, S.; Iida, T.; et al. Oral pathobiont induces systemic inflammation and metabolic changes associated with alteration of gut microbiota. Sci. Rep. 2015, 4, 4828. [Google Scholar] [CrossRef] [Green Version]
- Nagao, Y.; Kawahigashi, Y.; Sata, M. Association of Periodontal Diseases and Liver Fibrosis in Patients With HCV and/or HBV infection. Zahedan J. Res. Med Sci. 2014, 14, e23264. [Google Scholar] [CrossRef]
- Nagao, Y.; Tanigawa, T. Red complex periodontal pathogens are risk factors for liver cirrhosis. Biomed. Rep. 2019, 11, 199–206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sata, Y.N.A.M. Effect of a Late Evening Snack of Amazake in Patients with Liver Cirrhosis: A Pilot Study. J. Nutr. Food Sci. 2013, 3, 203. [Google Scholar] [CrossRef] [Green Version]
- Maruki-Uchida, H.; Sai, M.; Yano, S.; Morita, M.; Maeda, K. Amazake made from sake cake and rice koji suppresses sebum content in differentiated hamster sebocytes and improves skin properties in humans. Biosci. Biotechnol. Biochem. 2020, 84, 1689–1695. [Google Scholar] [CrossRef] [PubMed]
- Hamajima, H.; Matsunaga, H.; Fujikawa, A.; Sato, T.; Mitsutake, S.; Yanagita, T.; Nagao, K.; Nakayama, J.; Kitagaki, H. Erratum to: Japanese traditional dietary fungus koji Aspergillus oryzae functions as a prebiotic for Blautia coccoides through glycosylceramide: Japanese dietary fungus koji is a new prebiotic. SpringerPlus 2016, 5, 1869. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hamajima, H.; Tanaka, M.; Miyagawa, M.; Sakamoto, M.; Nakamura, T.; Yanagita, T.; Nishimukai, M.; Mitsutake, S.; Nakayama, J.; Nagao, K.; et al. Koji glycosylceramide commonly contained in Japanese traditional fermented foods alters cholesterol metabolism in obese mice. Biosci. Biotechnol. Biochem. 2019, 83, 1514–1522. [Google Scholar] [CrossRef]
- American Diabetes Association 2. Classification and Diagnosis of Diabetes. Diabetes Care 2016, 39, S13–S22. [Google Scholar] [CrossRef] [Green Version]
- Kamada, Y.; Ono, M.; Hyogo, H.; Fujii, H.; Sumida, Y.; Mori, K.; Tanaka, S.; Yamada, M.; Akita, M.; Mizutani, K.; et al. A novel noninvasive diagnostic method for nonalcoholic steatohepatitis using two glycobiomarkers. Hepatology 2015, 62, 1433–1443. [Google Scholar] [CrossRef]
- Tada, A.; Takeuchi, H.; Shimizu, H.; Tadokoro, K.; Tanaka, K.; Kawamura, K.; Yamaguchi, T.; Egashira, T.; Nomura, Y.; Hanada, N. Quantification of Periodontopathic Bacteria in Saliva Using the Invader Assay. Jpn. J. Infect. Dis. 2012, 65, 415–423. [Google Scholar] [CrossRef] [Green Version]
- Tadokoro, K.; Yamaguchi, T.; Kawamura, K.; Shimizu, H.; Egashira, T.; Minabe, M.; Yoshino, T.; Oguchi, H. Rapid quantification of periodontitis-related bacteria using a novel modification of Invader PLUS technologies. Microbiol. Res. 2010, 165, 43–49. [Google Scholar] [CrossRef]
- Matsumoto, T. Sake in the ‘Engishiki’. J. Brew. Soc. Jpn. 1981, 76, 460–465. (In Japanese) [Google Scholar] [CrossRef]
- Koizumi, T. Food and Japanese wisdom. JJRM 2013, 61, 838–839. (In Japanese) [Google Scholar] [CrossRef]
- Yang, Y.; Sitanggang, N.V.; Kato, N.; Inoue, J.; Murakami, T.; Watanabe, T.; Iguchi, T.; Okazaki, Y. Beneficial effects of protease preparations derived from Aspergillus on the colonic luminal environment in rats consuming a high-fat diet. Biomed. Rep. 2015, 3, 715–720. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oura, S.; Suzuki, S.; Hata, Y.; Kawato, A.; Abe, Y. Evaluation of physiological functionalities of amazake in mice. J. Brew. Soc. Jpn. 2007, 102, 781–788. (In Japanese) [Google Scholar] [CrossRef] [Green Version]
- Yoshizaki, Y.; Kawasaki, C.; Cheng, K.-C.; Ushikai, M.; Amitani, H.; Asakawa, A.; Okutsu, K.; Sameshima, Y.; Takamine, K.; Inui, A. Ricekojireduced body weight gain, fat accumulation, and blood glucose level in high-fat diet-induced obese mice. PeerJ 2014, 2, e540. [Google Scholar] [CrossRef] [Green Version]
- Ashida, Y.; Saio, Y.; Kawato, A.; Suginami, K.; Imayasu, S. Effects of dietary sake cake on cholesterol metabolism in rats. Nippon Nogeikagaku Kaishi 1997, 71, 137–143. (In Japanese) [Google Scholar] [CrossRef] [Green Version]
- Kikushima, K.; Nakagawa, T.; Shimizu, K.; Ohnuki, K. Regular intake of Japanese traditional rice fermented beverage, koji amazake for 4 weeks decreases systolic blood pressure. Jpn. Pharmacol. Ther. 2020, 48, 305–312. Available online: http://www.pieronline.jp/content/article/0386-3603/48020/305 (accessed on 29 September 2021).
- Jeon, H.J.; Noda, M.; Maruyama, M.; Matoba, Y.; Kumagai, A.T.; Sugiyama, M. Identification and Kinetic Study of Tyrosinase Inhibitors Found in Sake Lees. J. Agric. Food Chem. 2006, 54, 9827–9833. [Google Scholar] [CrossRef]
- Duan, J.; Sugawara, T.; Hirose, M.; Aida, K.; Sakai, S.; Fujii, A.; Hirata, T. Dietary sphingolipids improve skin barrier functions via the upregulation of ceramide synthases in the epidermis. Exp. Dermatol. 2012, 21, 448–452. [Google Scholar] [CrossRef]
- Shimotsuma, S.; Yamamoto, T.; Mori, S.; Morita, M.; Yano, S.; Maeda, K. Effects of oral intake of rice koji amazake on skin condition -A randomized, double-blind, placebo-controlled, parallel-group, comparison study. Jpn. Pharmacol. Ther. 2019, 47, 759–765. Available online: http://www.pieronline.jp/content/article/0386-3603/47080/1269 (accessed on 29 September 2021). (In Japanese).
- Yang, Y.; Iwamoto, A.; Kumrungsee, T.; Okazaki, Y.; Kuroda, M.; Yamaguchi, S.; Kato, N. Consumption of an acid protease derived from Aspergillus oryzae causes bifidogenic effect in rats. Nutr. Res. 2017, 44, 60–66. [Google Scholar] [CrossRef] [PubMed]
- Mori, S.; Tanaka, Y.; Watabe, K.; Yamada, M.; Morita, M.; Matsuike, T. Amazake using the lees and rice koji promotes regular bowel movements -A randomized, placebo-controlled parallel-group comparison study. Jpn. Pharmacol. Ther. 2019, 47, 759–765. Available online: http://www.pieronline.jp/content/article/0386-3603/47050/759 (accessed on 29 September 2021). (In Japanese).
- Park, H.S.; Park, J.Y.; Yu, R. Relationship of obesity and visceral adiposity with serum concentrations of CRP, TNF-α and IL-6. Diabetes Res. Clin. Pract. 2005, 69, 29–35. [Google Scholar] [CrossRef]
- Hotamisligil, G.S.; Peraldi, P.; Budavari, A.; Ellis, R.; White, M.F.; Spiegelman, B.M. IRS-1-Mediated Inhibition of Insulin Receptor Tyrosine Kinase Activity in TNF-alpha- and Obesity-Induced Insulin Resistance. Science 1996, 271, 665–670. [Google Scholar] [CrossRef] [PubMed]
- Valenti, L.; Fracanzani, A.L.; Dongiovanni, P.; Santorelli, G.; Branchi, A.; Taioli, E.; Fiorelli, G.; Fargion, S. Tumor necrosis factor α promoter polymorphisms and insulin resistance in nonalcoholic fatty liver disease. Gastroenterology 2002, 122, 274–280. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.-K.; Feng, Z.-W.; Li, Y.-C.; Li, Q.-Y.; Tao, X.-Y. Association of tumor necrosis factor-α gene promoter polymorphism at sites -308 and -238 with non-alcoholic fatty liver disease: A meta-analysis. J. Gastroenterol. Hepatol. 2012, 27, 670–676. [Google Scholar] [CrossRef] [PubMed]
- Crespo, J.; Cayón, A.; Fernández-Gil, P.; Hernandez-Guerra, M.; Mayorga, M.; Domínguez-Díez, A.; Fernández-Escalante, J.C.; Pons-Romero, F. Gene expression of tumor necrosis factor [alpha ] and TNF-receptors, p55 and p75, in nonalcoholic steatohepatitis patients. Hepatology 2001, 34, 1158–1163. [Google Scholar] [CrossRef] [PubMed]
- Batchelor, P. Is periodontal disease a public health problem? Br. Dent. J. 2014, 217, 405–409. [Google Scholar] [CrossRef] [Green Version]
- Socransky, S.S.; Haffajee, A.D.; Cugini, M.A.; Smith, C.; Kent, R.L., Jr. Microbial complexes in subgingival plaque. J. Clin. Periodontol. 1998, 25, 134–144. [Google Scholar] [CrossRef]
- Nagasaki, A.; Sakamoto, S.; Chea, C.; Ishida, E.; Furusho, H.; Fujii, M.; Takata, T.; Miyauchi, M. Odontogenic infection by Porphyromonas gingivalis exacerbates fibrosis in NASH via hepatic stellate cell activation. Sci. Rep. 2020, 10, 4134. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hamada, S.; Fujiwara, T.; Morishima, S.; Takahashi, I.; Nakagawa, I.; Kimura, S.; Ogawa, T. Molecular and Immunological Characterization of the Fimbriae of Porphyromonas gingivalis. Microbiol. Immunol. 1994, 38, 921–930. [Google Scholar] [CrossRef] [PubMed]
- Amano, A.; Nakagawa, I.; Kataoka, K.; Morisaki, I.; Hamada, S. Distribution of Porphyromonas gingivalis Strains with fimA Genotypes in Periodontitis Patients. J. Clin. Microbiol. 1999, 37, 1426–1430. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, D.; Molofsky, A.; Liang, H.-E.; Ricardo-Gonzalez, R.; Jouihan, H.A.; Bando, J.; Chawla, A.; Locksley, R.M. Eosinophils Sustain Adipose Alternatively Activated Macrophages Associated with Glucose Homeostasis. Science 2011, 332, 243–247. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calco, G.N.; Fryer, A.D.; Nie, Z. Unraveling the connection between eosinophils and obesity. J. Leukoc. Biol. 2020, 108, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Murata, A.; Hyogo, H.; Nonaka, M.; Sumioka, A.; Suehiro, Y.; Furudoi, A.; Fujimoto, Y.; Aisaka, Y.; Komatsu, H.; Tokumo, H. Overlooked muscle cramps in patients with chronic liver disease: In relation to the prevalence of muscle cramps. Eur. J. Gastroenterol. Hepatol. 2019, 31, 375–381. [Google Scholar] [CrossRef]
- Mehta, S.S.; Fallon, M.B. Muscle Cramps in Liver Disease. Clin. Gastroenterol. Hepatol. 2013, 11, 1385–1391. [Google Scholar] [CrossRef]
- Barbe, M.F.; Elliott, M.B.; Abdelmagid, S.M.; Amin, M.; Popoff, S.N.; Safadi, F.F.; Barr, A.E. Serum and tissue cytokines and chemokines increase with repetitive upper extremity tasks. J. Orthop. Res. 2008, 26, 1320–1326. [Google Scholar] [CrossRef]
- Mikova, O.; Yakimova, R.; Bosmans, E.; Kenis, G.; Maes, M. Increased serum tumor necrosis factor alpha concentrations in major depression and multiple sclerosis. Eur. Neuropsychopharmacol. 2001, 11, 203–208. [Google Scholar] [CrossRef]
- Strawbridge, R.; Arnone, D.; Danese, A.; Papadopoulos, A.S.; Vives, A.R.H.; Cleare, A.J. Inflammation and clinical response to treatment in depression: A meta-analysis. Eur. Neuropsychopharmacol. 2015, 25, 1532–1543. [Google Scholar] [CrossRef]
- Kawaguchi, T.; Taniguchi, E.; Itou, M.; Sumie, S.; Oriishi, T.; Matsuoka, H.; Nagao, Y.; Sata, M. Branched-chain amino acids improve insulin resistance in patients with hepatitis C virus-related liver disease: Report of two cases. Liver Int. 2007, 27, 1287–1292. [Google Scholar] [CrossRef]
- Kawaguchi, T.; Nagao, Y.; Matsuoka, H.; Ide, T.; Sata, M. Branched-chain amino acid-enriched supplementation improves insulin resistance in patients with chronic liver disease. Int. J. Mol. Med. 2008, 22, 105–112. [Google Scholar] [CrossRef] [Green Version]
- Nagao, Y.; Matsuoka, H.; Kawaguchi, T.; Sata, M. Aminofeel improves the sensitivity to taste in patients with HCV-infected liver disease. Med. Sci. Monit 2010, 16, 7–12. [Google Scholar]
- Nagao, Y.; Kawaguchi, T.; Kakuma, T.; Ide, T.; Sata, M. Post-marketing surveillance study for efficacy and safety of Aminofeel, a branched chain amino acids-enriched supplement including zinc. J. New Rem. Clin. 2011, 60, 1046–1063. Available online: https://mol.medicalonline.jp/archive/search?jo=aa9syrsc&vo=60&nu=5 (accessed on 29 September 2021). (In Japanese).
- Nagao, Y.; Kawaguchi, T.; Ide, T.; Sata, M. Effect of branched-chain amino acid-enriched nutritional supplementation on interferon therapy in Japanese patients with chronic hepatitis C virus infection: A retrospective study. Virol. J. 2012, 9, 282. [Google Scholar] [CrossRef] [Green Version]
- Kawaguchi, T.; Nagao, Y.; Abe, K.; Imazeki, F.; Honda, K.; Yamasaki, K.; Miyanishi, K.; Taniguchi, E.; Kakuma, T.; Kato, J.; et al. Effects of branched-chain amino acids and zinc-enriched nutrients on prognosticators in HCV-infected patients: A multicenter randomized controlled trial. Mol. Med. Rep. 2014, 11, 2159–2166. [Google Scholar] [CrossRef] [Green Version]
- Sako, K. Branched-chain amino acids supplements in the late evening decrease the frequency of muscle cramps with advanced hepatic cirrhosis. Hepatol. Res. 2003, 26, 327–329. [Google Scholar] [CrossRef]
- Hidaka, H.; Nakazawa, T.; Kutsukake, S.; Yamazaki, Y.; Aoki, I.; Nakano, S.; Asaba, N.; Minamino, T.; Takada, J.; Tanaka, Y.; et al. The efficacy of nocturnal administration of branched-chain amino acid granules to improve quality of life in patients with cirrhosis. J. Gastroenterol. 2012, 48, 269–276. [Google Scholar] [CrossRef] [PubMed]
- Raman, M.; Ahmed, I.; Gillevet, P.M.; Probert, C.S.; Ratcliffe, N.M.; Smith, S.; Greenwood, R.; Sikaroodi, M.; Lam, V.; Crotty, P.; et al. Fecal Microbiome and Volatile Organic Compound Metabolome in Obese Humans with Nonalcoholic Fatty Liver Disease. Clin. Gastroenterol. Hepatol. 2013, 11, 868–875. [Google Scholar] [CrossRef] [PubMed]
- Goshima, S.; Kanematsu, M.; Kobayashi, T.; Furukawa, T.; Zhang, X.; Fujita, H.; Watanabe, H.; Kondo, H.; Moriyama, N.; Bae, K.T. Staging hepatic fibrosis: Computer-aided analysis of hepatic contours on gadolinium ethoxybenzyl diethylenetriaminepentaacetic acid-enhanced hepatocyte-phase magnetic resonance imaging. Hepatology 2011, 55, 328–329. [Google Scholar] [CrossRef] [Green Version]
- Miele, L.; Valenza, V.; La Torre, G.; Montalto, M.; Cammarota, G.; Ricci, R.; Mascianà, R.; Forgione, A.; Gabrieli, M.L.; Perotti, G.; et al. Increased intestinal permeability and tight junction alterations in nonalcoholic fatty liver disease. Hepatology 2009, 49, 1877–1887. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Variable | Total |
|---|---|---|
| Sex | Male/female | 2/8 |
| Age | Mean ± SD, years | 57.1 ± 19.2 |
| Diagnosis of liver diseases | NAFLD | 10 (100%) |
| Diabetes mellitus | Positive (%) | 2 (20%) |
| Dyslipidemia | Positive (%) | 7 (70%) |
| Hypertension | Positive (%) | 4 (40%) |
| Depression a | Positive (%) | 2 (20%) |
| History of extrahepatic malignant tumor b | Positive (%) | 2 (20%) |
| A habit of tooth brushing after meals | Positive (%) | 2 (20%) |
| Anti-HCV | Positive (%) | 0 (0%) |
| HBsAg | Positive (%) | 0 (0%) |
| Variable | Baseline | 60 Days after Amazake Intake | Difference in Means | LCL | UCL | p-Value |
|---|---|---|---|---|---|---|
| (Mean ± SD) | (Mean ± SD) | |||||
| BMI, kg/m2 | 27.39 ± 4.1 | 27.45 ± 4.1 | 0.06 | −0.37 | 0.49 | NS |
| Body fat percentage, % | 31.18 ± 5.2 | 31.99 ± 5.6 | 0.81 | −0.92 | 2.54 | NS |
| WBC, 103/µL | 5.55 ± 1.9 | 5.14 ± 1.8 | −0.41 | −1.07 | 0.25 | NS |
| Neutrophil count, % | 55.87 ± 11.7 | 56.85 ± 9.5 | 0.98 | −2.61 | 4.57 | NS |
| Lymphocyte counts, % | 33.90 ± 10.6 | 32.67 ± 8.5 | −1.23 | −4.30 | 1.84 | NS |
| Monocyte count, % | 6.85 ± 1.7 | 6.46 ± 1.5 | −0.39 | −1.44 | 0.66 | NS |
| Eosinophil count, % | 2.72 ± 1.4 | 3.27 ± 1.4 | 0.55 | 0.11 | 0.99 | 0.0203 |
| Basophil count, % | 0.66 ± 0.3 | 0.75 ± 0.4 | 0.09 | −0.06 | 0.24 | NS |
| RBC, 106/µL | 4.50 ± 0.5 | 4.49 ± 0.5 | −0.003 | −0.05 | 0.05 | NS |
| Hb, g/dL | 14.22 ± 1.7 | 14.12 ± 1.6 | −0.10 | −0.31 | 0.10 | NS |
| PLT, 104/µL | 22.39 ± 9.1 | 21.95 ± 9.1 | −0.44 | −1.21 | 0.33 | NS |
| AST, U/L | 47.6 ± 28.0 | 51.5 ± 40.1 | 3.9 | −7.91 | 15.71 | NS |
| ALT, U/L | 58.4 ± 38.1 | 56.2 ± 36.7 | −2.2 | −13.03 | 8.63 | NS |
| γ-GTP, U/L | 44.5 ± 29.9 | 47.8 ± 33.7 | 3.3 | −4.47 | 11.07 | NS |
| LDH, U/L | 196.2 ± 17.2 | 200.4 ± 26.4 | 4.2 | −8.16 | 16.56 | NS |
| ALP, U/L | 251.5 ± 83.6 | 252.8 ± 82.4 | 1.3 | −14.9 | 17.5 | NS |
| Total protein, g/dL | 7.47 ± 0.2 | 7.46 ± 0.3 | −0.01 | −0.25 | 0.23 | NS |
| Albumin, g/dL | 4.26 ± 0.3 | 4.26 ± 0.3 | 0 | −0.22 | 0.22 | NS |
| T. cho, mg/dL | 187.7 ± 25.2 | 190.3 ± 34.6 | 2.6 | −14.51 | 19.71 | NS |
| LDL, mg/dL | 125.3 ± 24.5 | 126.9 ± 31.6 | 1.6 | −13.6 | 16.8 | NS |
| HDL, mg/dL | 48.7 ± 9.9 | 49.6 ± 10.1 | 0.9 | −3.1 | 4.9 | NS |
| Triglyceride, mg/dL | 143.2 ± 86.6 | 130.2 ± 51.9 | −13 | −44.57 | 18.57 | NS |
| T.Bil, mg/dL | 0.80 ± 0.3 | 0.85 ± 0.3 | 0.05 | −0.12 | 0.22 | NS |
| D.Bil, mg/dL | 0.10 ± 0 | 0.11 ± 0.1 | 0.01 | −0.03 | 0.05 | NS |
| Crea, mg/dL | 0.64 ± 0.2 | 0.63 ± 0.2 | −0.01 | −0.05 | 0.03 | NS |
| BUN, mg/dL | 12.80 ± 3.1 | 13.19 ± 2.8 | 0.39 | −1.57 | 2.35 | NS |
| Fe, µg/dL | 126.3 ± 37.8 | 117.6 ± 25.2 | −8.7 | −39.07 | 21.67 | NS |
| Ferritin, µg/dL | 231.9 ± 206.7 | 231.2 ± 196.2 | −0.7 | −22.38 | 20.98 | NS |
| Zinc, µg/dL | 80.6 ± 14.6 | 70.7 ± 7.6 | −9.9 | −20.74 | 0.94 | NS |
| Fasting plasma glucose, mg/dL | 108.2 ± 19.6 | 109.6 ± 28.4 | 1.4 | −14.48 | 17.28 | NS |
| HbA1c, % | 5.9 ± 0.5 | 6.1 ± 0.7 | 0.2 | −0.02 | 0.42 | NS |
| Fasting IRI, µU/mL | 16.18 ± 7.3 | 24.42 ± 10.0 | 8.24 | 2.87 | 13.61 | 0.0070 |
| HOMA-IR | 4.47 ± 2.5 | 6.72 ± 3.6 | 2.25 | 0.62 | 3.87 | 0.0122 |
| AFP, ng/mL | 4.54 ± 1.6 | 4.60 ± 1.9 | 0.06 | −0.88 | 1 | NS |
| Total P1NP, ng/mL | 58.88 ± 15.3 | 54.75 ± 19.0 | −4.13 | −9.17 | 0.91 | NS |
| P-III-P, ng/mL | 11.65 ± 5.3 | 11.98 ± 5.0 | 0.32 | −1.22 | 1.86 | NS |
| Type IV collagen 7s domain, ng/mL | 5.86 ± 2.3 | 6.00 ± 2.1 | 0.14 | −0.26 | 0.54 | NS |
| M2BPGi, COI | 1.61 ± 1.3 | 1.64 ± 1.3 | 0.03 | −0.23 | 0.3 | NS |
| TRACP−5b, mU/dL | 320.56 ± 147.5 | 354.40 ± 140.9 | 33.84 | −11.67 | 79.35 | NS |
| High molecular weight adiponectin, µg/dL | 2.65 ± 1.3 | 2.68 ± 1.5 | 0.03 | −0.17 | 0.22 | NS |
| Leptin, ng/mL | 25.61 ± 13.0 | 31.09 ± 19.9 | 5.48 | −3.06 | 14.02 | NS |
| IL-6, pg/mL | 2.16 ± 0.8 | 2.50 ± 1.2 | 0.34 | −0.35 | 1.03 | NS |
| High-sensitivity TNFα, pg/mL | 1.41 ± 0.4 | 1.21 ± 0.4 | −0.20 | −0.31 | −0.09 | 0.0025 |
| High-sensitivity CRP, ng/mL | 2344.5 ± 2266.7 | 2311.6 ± 3442.9 | −32.9 | −1191.25 | 1125.45 | NS |
| Total number of bacteria in saliva, copy/10 µL | 13,290,000 ± 7,130,131.7 | 11,560,000 ± 6,794,147.8 | −1,730,000 | −4,700,000 | 1,246,368 | NS |
| Number of A. actinomycetemcomitans in saliva, copy/10 µL | <10 | <10 | 0 | 0 | 0 | NS |
| Number of P. intermedia in saliva, copy/10 µL | 17.8 ± 22.6 | 34.7 ± 75.7 | 16.9 | −21.08 | 54.88 | NS |
| Number of P. gingivalis in saliva, copy/10 µL | 144.0 ± 294.4 | 54.0 ± 113.8 | −90 | −267.1 | 87.10 | NS |
| Number of T. forsythia in saliva, copy/10 µL | 330.0 ± 292.0 | 703.5 ± 1242 | 373.5 | −433.98 | 1180.98 | NS |
| Number of T. denticola in saliva, copy/10 µL | 369.0 ± 492.1 | 391.5 ± 609.3 | 22.5 | −188.40 | 233.40 | NS |
| Number of F. necleatum in saliva, copy/10 µL | 283,090 ± 274,895.9 | 243,044 ± 342,379.7 | −40,046 | −233,951 | 153,859 | NS |
| P. gingivalis fimA genotype II, positive (%) | 2 (20%) | 2 (20%) | n/a | n/a | n/a | NS |
| P. gingivalis fimA below detection sensitivity limits, positive (%) | 8 (80%) | 8 (80%) | n/a | n/a | n/a | NS |
| Sense of abdominal distension (VAS), mm | 25.8 ± 25.0 | 18.6 ± 28.9 | −7.2 | −20.1 | 5.7 | NS |
| Edema (VAS), mm | 27.2 ± 30.9 | 31.6 ± 37.7 | 4.4 | −6.48 | 15.28 | NS |
| Fatigue (VAS), mm | 37.2 ± 28.7 | 36.8 ± 29.6 | −0.4 | −7.02 | 6.22 | NS |
| Muscle cramps (VAS), mm | 24.1 ± 29.6 | 3.4 ± 7.2 | −20.7 | −39.62 | −1.78 | 0.0353 |
| Loss of appetite (VAS), mm | 24.4 ± 25.2 | 12.1 ± 9.8 | −12.3 | −31.47 | 6.87 | NS |
| Taste disorder (VAS), mm | 3.0 ± 6.9 | 0 | −3 | −7.93 | 1.93 | NS |
| Constipation (VAS), mm | 31.4 ± 30.0 | 18.2 ± 29.6 | −13.2 | −29.34 | 2.94 | NS |
| Diarrhea (VAS), mm | 16.6 ± 29.8 | 7.3 ± 12.5 | −9.3 | −28.42 | 9.82 | NS |
| Depression (VAS), mm | 30.8 ± 24.7 | 18.2 ± 21.6 | −12.6 | −23.52 | −1.68 | 0.0282 |
| Sleep disorder (VAS), mm | 25.3 ± 24.2 | 19.6 ± 24.1 | −5.7 | −25.06 | 13.66 | NS |
| M2BPGi | Group A | Group B | Group C |
|---|---|---|---|
| Negative (n = 5) | Positive 1 (n = 3) | Positive 2 (n = 2) | |
| Difference in Means | Difference in Means | Difference in Means | |
| BMI, kg/m2 | −0.08 | 0.23 | 0.15 |
| Body fat percentage, % | 1.76 | 0.03 | −0.40 |
| WBC, 103/μL | −0.76 | 0.20 | −0.45 |
| Neutrophil count, % | −1.9 | 5.2 | 1.8 |
| Lymphocyte counts, % | 1.6 | −4.6 | −3.2 |
| Monocyte count, % | −0.2 | −1.1 | 0.2 |
| Eosinophil count, % | 0.38 | 0.57 | 0.95 |
| Basophil count, % | 0.1 | −0.1 | 0.3 |
| RBC, 106/μL | 0.01 | 0 | 0 |
| Hb, g/dL | 0.03 | −0.2 | −0.4 |
| PLT, 104/μL | 0.02 | −1.7 | 0.3 |
| AST, U/L | −3.8 | 15.3 | 6.0 |
| ALT, U/L | −6.2 | 2.0 | 1.5 |
| γ-GTP, U/L | 3.0 | 6.7 | −1.0 |
| LDH, U/L | −1.0 | 13.7 | 3.0 |
| ALP, U/L | −9.0 | 16.7 | 4.0 |
| Total protein, g/dL | 0.16 | −0.13 | −0.25 |
| Albumin, g/dL | 0.08 | −0.03 | −0.15 |
| T. cho, mg/dL | 5.8 | 10.0 | −16.5 |
| LDL, mg/dL | 3.8 | 6.7 | −11.5 |
| HDL, mg/dL | 1.8 | 1.3 | −2.0 |
| Triglyceride, mg/dL | −23.0 | −9.7 | 7.0 |
| T.Bil, mg/dL | 0.08 | −0.03 | 0.10 |
| D.Bil, mg/dL | 0 | 0.03 | 0 |
| Crea, mg/dL | 0 | −0.02 | −0.03 |
| BUN, mg/dL | 0.92 | 1.93 | −3.25 |
| Fe, μg/dL | −21.2 | 4.0 | 3.5 |
| Ferritin, μg/dL | −15.2 | 20.3 | 4.0 |
| Zinc, μg/dL | −6.6 | −18.3 | −5.5 |
| Fasting plasma glucose, mg/dL | −3.4 | 1.7 | 13.0 |
| HbA1c, % | 0.1 | 0.1 | 0.7 |
| Fasting IRI, μU/mL | 8.98 | 12.57 | −0.10 |
| HOMA-IR | 2.06 | 3.29 | 1.15 |
| AFP, ng/mL | −0.10 | 0.57 | −0.30 |
| Total P1NP, ng/mL | −8.48 | 0.47 | −0.15 |
| P-III-P, ng/mL | −0.17 | 2.56 | −1.80 |
| Type IV collagen 7s domain, ng/mL | 0.26 | 0 | 0.05 |
| TRACP-5b, mU/dL | 68.88 | 4.00 | −9.00 |
| High molecular weight adiponectin, µg/dL | −0.06 | 0.31 | −0.18 |
| Leptin, ng/mL | 5.40 | 11.27 | −3.00 |
| IL-6, pg/mL | −0.30 | 0.77 | 1.30 |
| High-sensitivity TNFα, pg/mL | −0.24 | −0.17 | −0.16 |
| High-sensitivity CRP, ng/mL | 305.40 | −208.00 | −616.00 |
| Total number of bacteria in saliva, copy/10 μL | −1,220,000 | −2,533,333 | −1,800,000 |
| Number of A. actinomycetemcomitans in saliva, copy/10 μL | 0 | 0 | 0 |
| Number of P. intermedia in saliva, copy/10 μL | 0.2 | 0 | 84 |
| Number of P. gingivalis in saliva, copy/10 μL | −18 | −263 | −10 |
| Number of T. forsythia, in saliva, copy/10 μL | 760 | −20 | −3 |
| Number of T. denticola in saliva, copy/10 μL | −14 | 96 | 4 |
| Number of F. necleatum in saliva, copy/10 μL | 64,200 | −216,567 | −35,880 |
| P. gingivalisfimA genotype II, positive (%) | n/a | n/a | n/a |
| P. gingivalis fimA below detection sensitivity limits, positive (%) | n/a | n/a | n/a |
| Sense of abdominal distension (VAS), mm | −15.2 | −8.3 | 14.5 |
| Edema (VAS), mm | 9.4 | 7.7 | −13.0 |
| Fatigue (VAS), mm | 2.6 | −1.7 | −6.0 |
| Muscle cramps (VAS), mm | −11.4 | −22.3 | −41.5 |
| Loss of appetite (VAS), mm | −13.6 | 3.0 | −32.0 |
| Taste disorder (VAS), mm | −5.4 | −1.0 | 0 |
| Constipation (VAS), mm | −28.8 | 1.3 | 4.0 |
| Diarrhea (VAS), mm | −17.8 | −1.3 | 0 |
| Depression (VAS), mm | −16.0 | −13.0 | −3.5 |
| Sleep disorder (VAS), mm | 1.2 | −14.3 | −10.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nagao, Y.; Takahashi, H.; Kawaguchi, A.; Kitagaki, H. Effect of Fermented Rice Drink “Amazake” on Patients with Nonalcoholic Fatty Liver Disease and Periodontal Disease: A Pilot Study. Reports 2021, 4, 36. https://doi.org/10.3390/reports4040036
Nagao Y, Takahashi H, Kawaguchi A, Kitagaki H. Effect of Fermented Rice Drink “Amazake” on Patients with Nonalcoholic Fatty Liver Disease and Periodontal Disease: A Pilot Study. Reports. 2021; 4(4):36. https://doi.org/10.3390/reports4040036
Chicago/Turabian StyleNagao, Yumiko, Hirokazu Takahashi, Atsushi Kawaguchi, and Hiroshi Kitagaki. 2021. "Effect of Fermented Rice Drink “Amazake” on Patients with Nonalcoholic Fatty Liver Disease and Periodontal Disease: A Pilot Study" Reports 4, no. 4: 36. https://doi.org/10.3390/reports4040036
APA StyleNagao, Y., Takahashi, H., Kawaguchi, A., & Kitagaki, H. (2021). Effect of Fermented Rice Drink “Amazake” on Patients with Nonalcoholic Fatty Liver Disease and Periodontal Disease: A Pilot Study. Reports, 4(4), 36. https://doi.org/10.3390/reports4040036








