Delayed Onset of Dural Arteriovenous Fistula Following Trauma
Abstract
1. Introduction
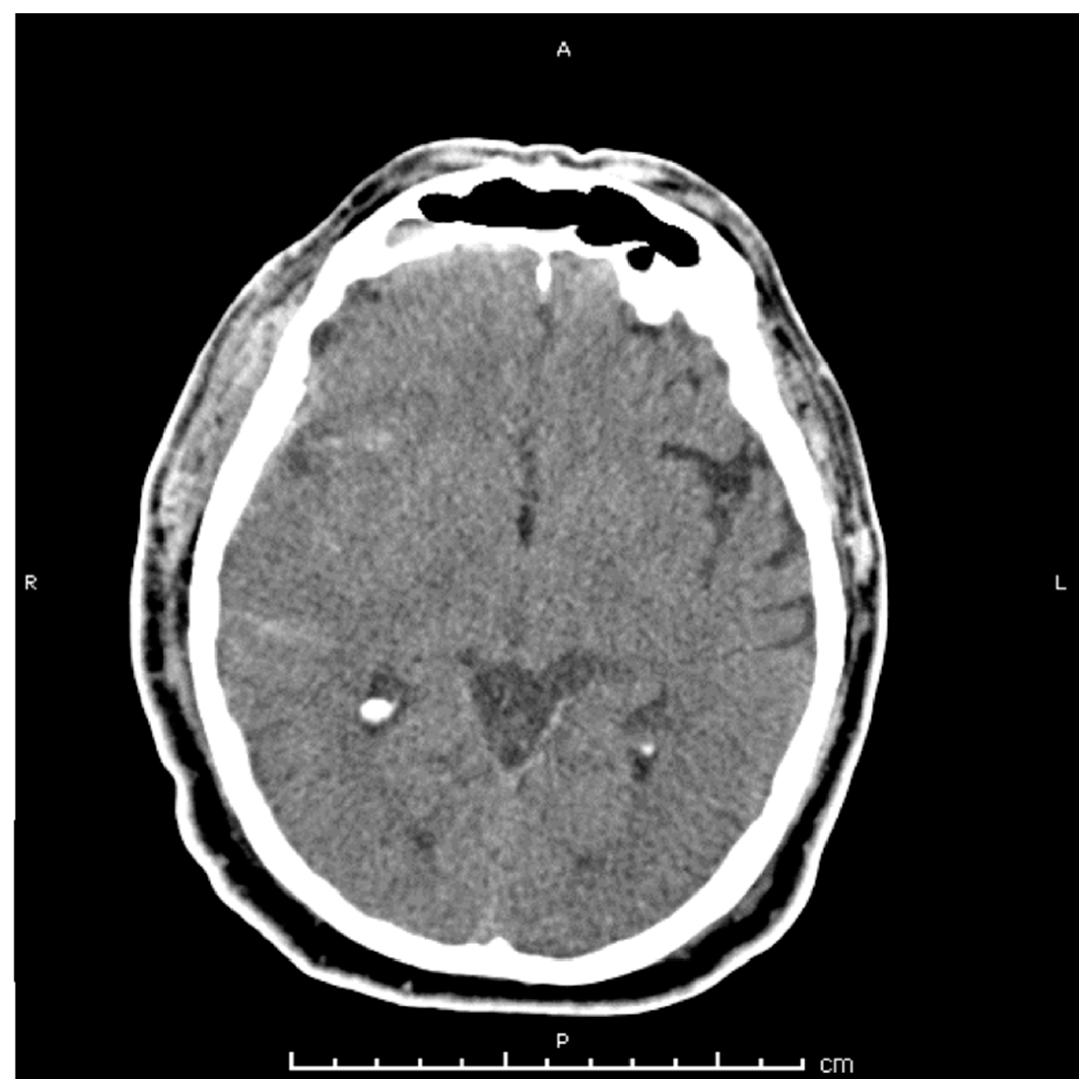
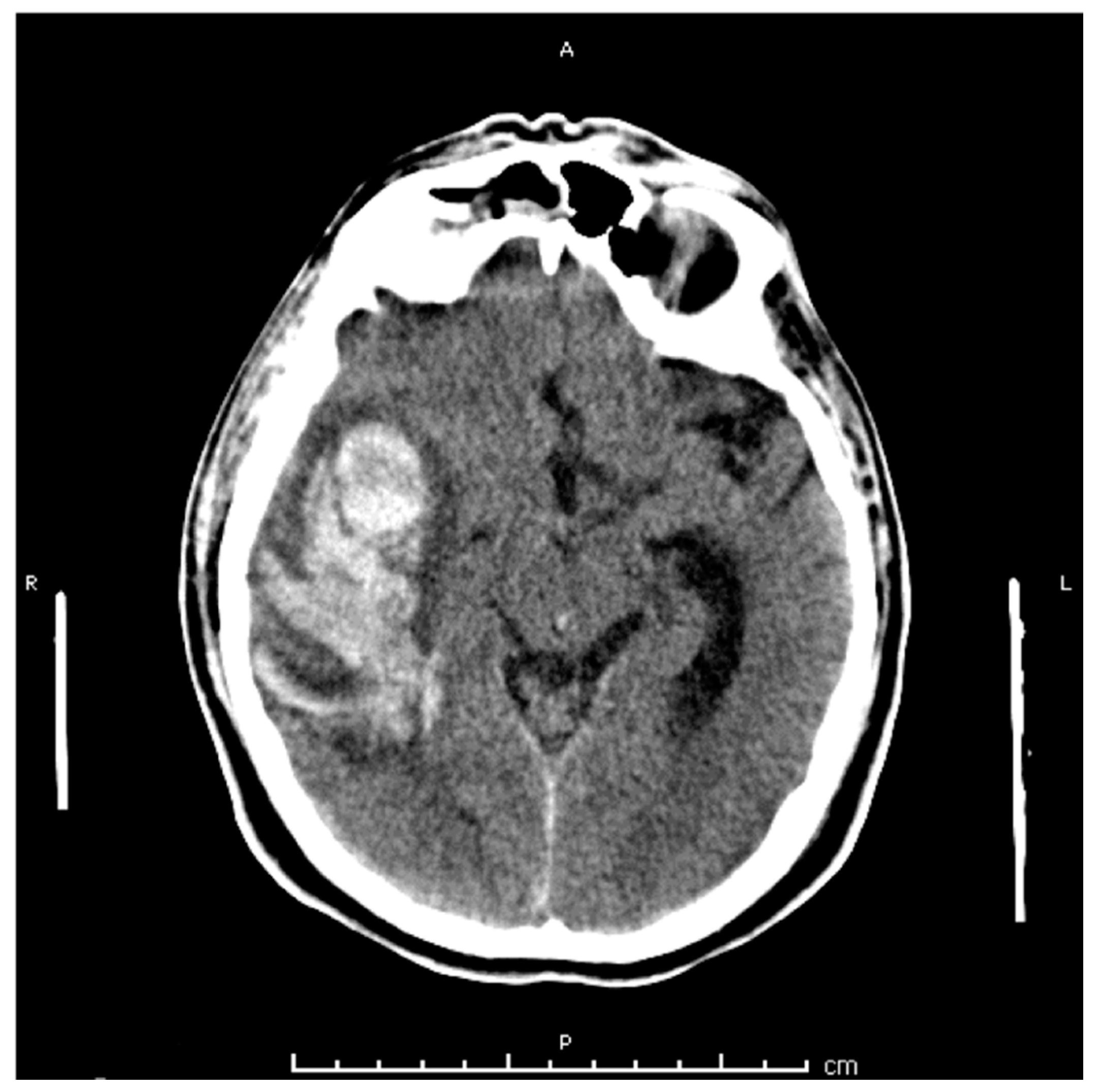
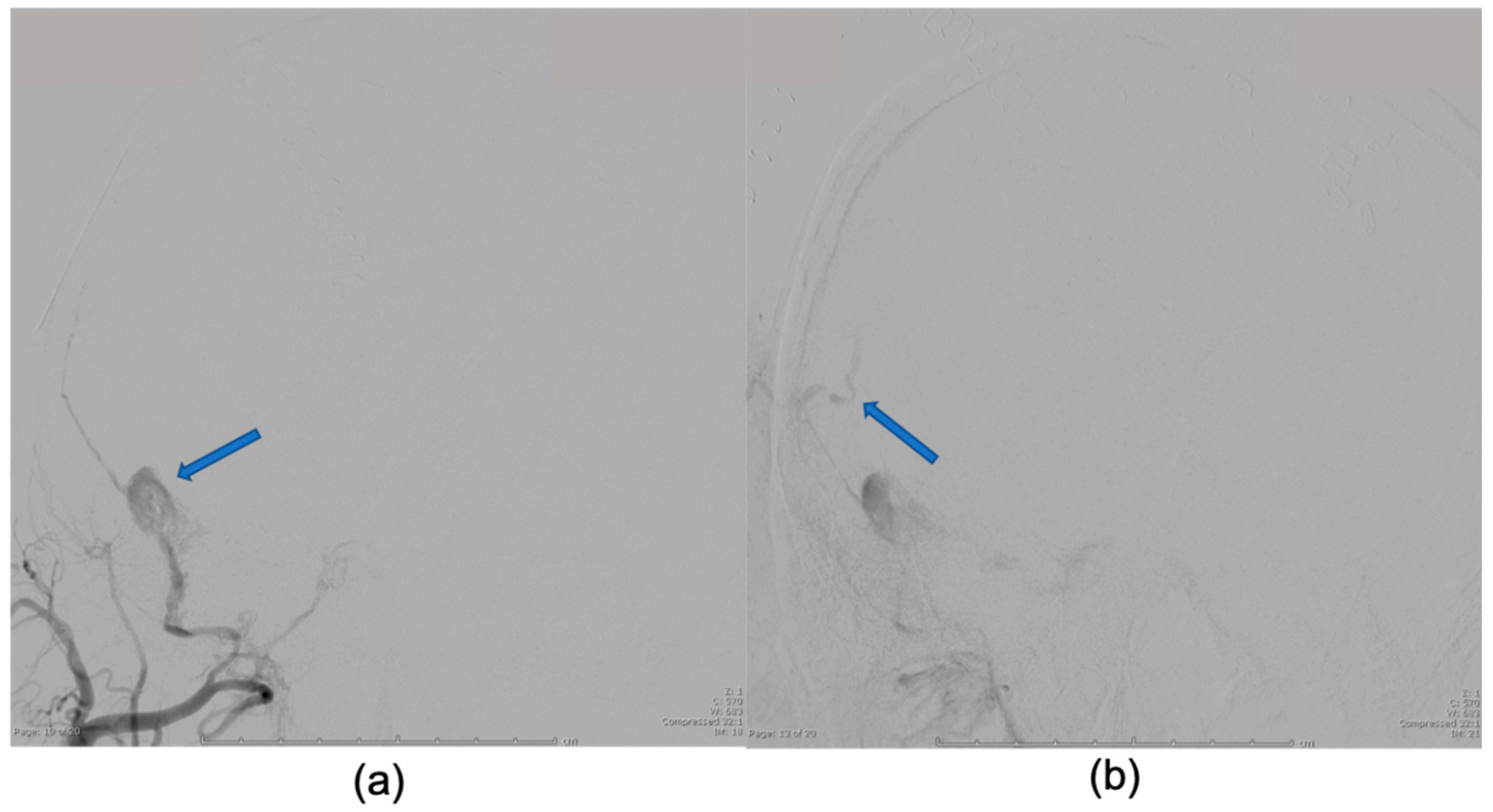
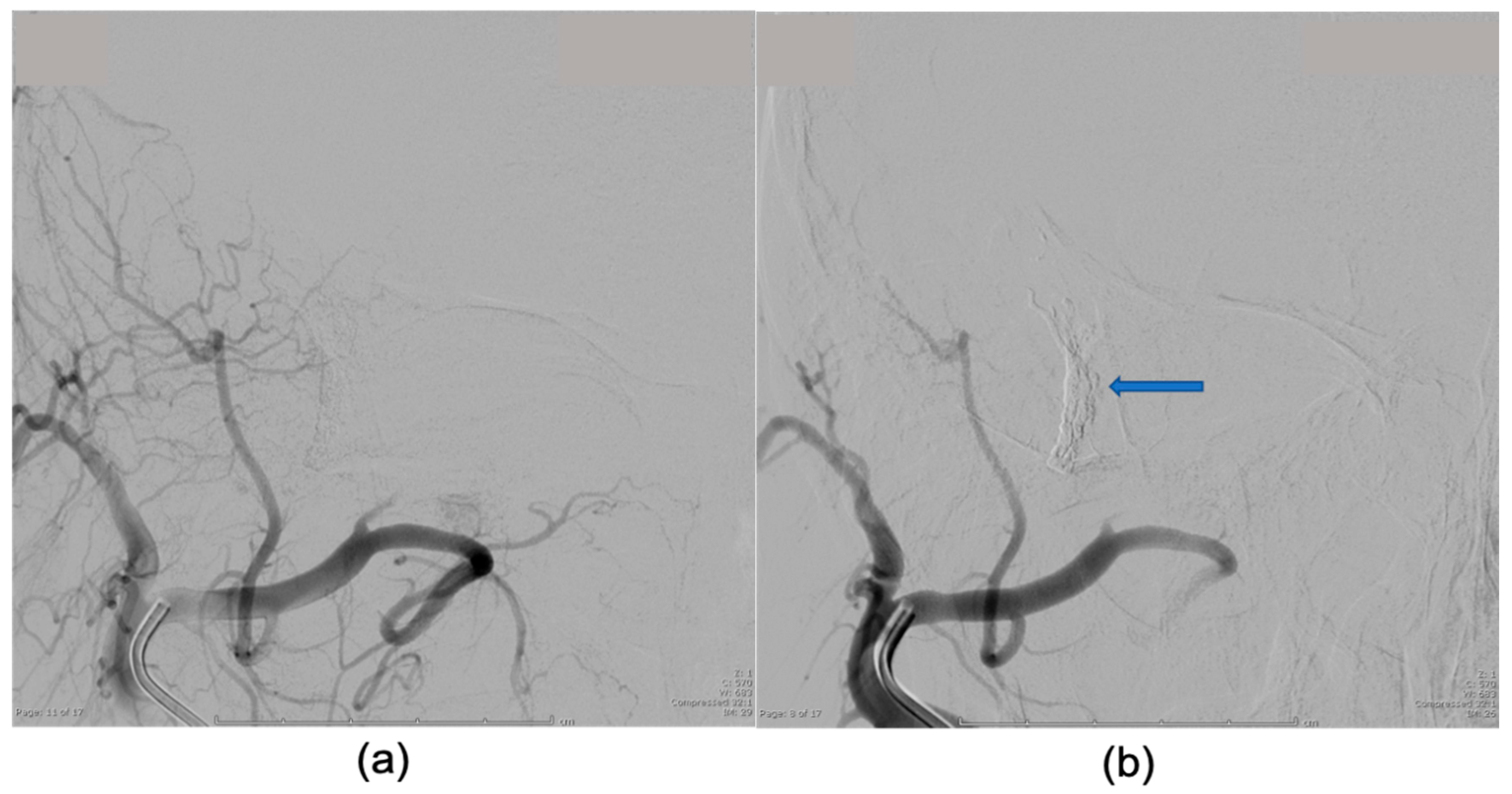
2. Case Presentation
3. Discussion
Author Contributions
Conflicts of Interest
References
- Miller, T.R.; Gandhi, D. Intracranial Dural Arteriovenous Fistulae. Stroke 2015, 46, 2017–2025. [Google Scholar] [CrossRef] [PubMed]
- Newton, T.H.; Cronqvist, S. Involvement of dural arteries in intracranial arteriovenous malformations. Radiology 1969, 93, 1071–1078. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, D.; Chen, J.; Pearl, M.; Huang, J.; Gemmete, J.J.; Kathuria, S. Intracranial Dural Arteriovenous Fistulas: Classification, Imaging Findings, and Treatment. Am. J. Neuroradiol. 2012, 33, 1007–1013. [Google Scholar] [CrossRef] [PubMed]
- Sarma, D.; Ter Brugge, K. Management of intracranial dural arteriovenous shunts in adults. Eur. J. Radiol. 2003, 46, 206–220. [Google Scholar] [CrossRef]
- Kwon, B.J.; Han, M.H.; Kang, H.-S.; Chang, K.-H. MR imaging findings of intracranial dural arteriovenous fistulas: Relations with venous drainage patterns. AJNR Am. J. Neuroradiol. 2005, 26, 2500–2507. [Google Scholar] [PubMed]
- Saito, A.; Takahashi, N.; Furuno, Y.; Kamiyama, H.; Nishimura, S.; Midorikawa, H.; Nishijima, M. Multiple isolated sinus dural arteriovenous fistulas associated with antithrombin III deficiency. Neurol Med. Chir. 2008, 48, 455–459. [Google Scholar] [CrossRef] [PubMed]
- Cooper, C.J.; Said, S.; Nunez, A.; Quansahm, R.; Khalillullah, S.; Hernandez, G.T. Dural arteriovenous fistula discovered in patient presenting with recent head trauma. Am. J. Case Rep. 2013, 14, 444–448. [Google Scholar] [CrossRef] [PubMed]
- Chung, S.J.; Kim, J.S.; Kim, J.C.; Lee, S.K.; Kwon, S.U.; Lee, M.C.; Suh, D.C. Intracranial dural arteriovenous fistulas: Analysis of 60 patients. Cerebrovasc. Dis. Basel Switz. 2002, 13, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Cognard, C.; Gobin, Y.P.; Pierot, L.; Bailly, A.L.; Houdart, E.; Casasco, A.; Chiras, J.; Merland, J.J. Cerebral dural arteriovenous fistulas: Clinical and angiographic correlation with a revised classification of venous drainage. Radiology 1995, 194, 671–680. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, M.R.; Lanzino, G.; Zipfel, G.J. Intracranial Dural Arteriovenous Fistulae. Stroke 2017, 48, 1424–1431. [Google Scholar] [CrossRef] [PubMed]
- Piippo, A.; Niemelä, M.; van Popta, J.; Kangasniemi, M.; Rinne, J.; Jääskeläinen, J.E.; Hernesniemi, J. Characteristics and long-term outcome of 251 patients with dural arteriovenous fistulas in a defined population: Clinical article. J. Neurosurg. 2013, 118, 923–934. [Google Scholar] [CrossRef] [PubMed]
- Ko, J.H.; Kim, Y.-J. Traumatic Pseudoaneurysm of the Middle Meningeal Artery with an Arteriovenous Fistula on a Non-Fractured Site. Interv. Neuroradiol. 2014, 20, 352–356. [Google Scholar] [CrossRef] [PubMed]
- Fujii, H.; Nagano, Y.; Hosomi, N.; Matsumoto, M. Dural arteriovenous fistula presenting with progressive dementia and parkinsonism. BMJ Case Rep. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Chang, W.S.; Jung, H.H.; Chang, J.W. Delayed Dural Arteriovenous Fistula after Microvascular Decompression for Hemifacial Spasm. J. Korean Neurosurg Soc. 2014, 56, 168–170. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Periakaruppan, A. Intracranial dural arteriovenous fistulas: A Review. Indian J. Radiol. Imaging 2009, 19, 43–48. [Google Scholar] [CrossRef] [PubMed]
| Borden Classification | Venous Drainage | CVD 1 | Risk |
|---|---|---|---|
| Type I | Meningeal veins or dural venous sinus | No | Benign |
| Type II | Meningeal veins or dural venous sinus | Yes | Aggressive |
| Type III | Subarachnoid veins or venous sinus | Yes | Aggressive |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Richardson, W.; Satarasinghe, P.; Koltz, M.T. Delayed Onset of Dural Arteriovenous Fistula Following Trauma. Reports 2019, 2, 14. https://doi.org/10.3390/reports2020014
Richardson W, Satarasinghe P, Koltz MT. Delayed Onset of Dural Arteriovenous Fistula Following Trauma. Reports. 2019; 2(2):14. https://doi.org/10.3390/reports2020014
Chicago/Turabian StyleRichardson, William, Praveen Satarasinghe, and Michael T. Koltz. 2019. "Delayed Onset of Dural Arteriovenous Fistula Following Trauma" Reports 2, no. 2: 14. https://doi.org/10.3390/reports2020014
APA StyleRichardson, W., Satarasinghe, P., & Koltz, M. T. (2019). Delayed Onset of Dural Arteriovenous Fistula Following Trauma. Reports, 2(2), 14. https://doi.org/10.3390/reports2020014





