Differentiated Thyroid Carcinoma and Late Onset of Lung Distant Metastasis. A Case Report
Abstract
1. Introduction
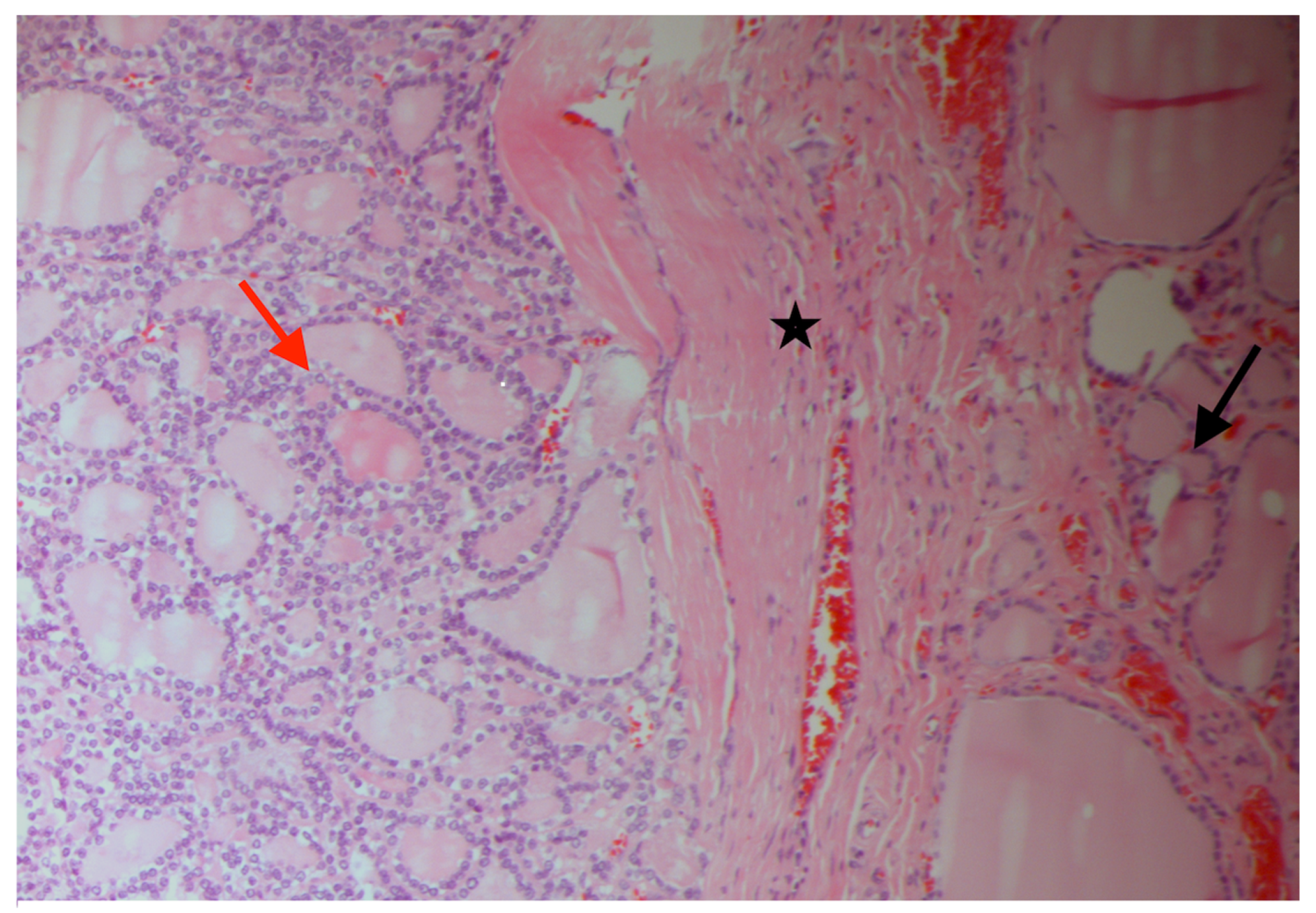
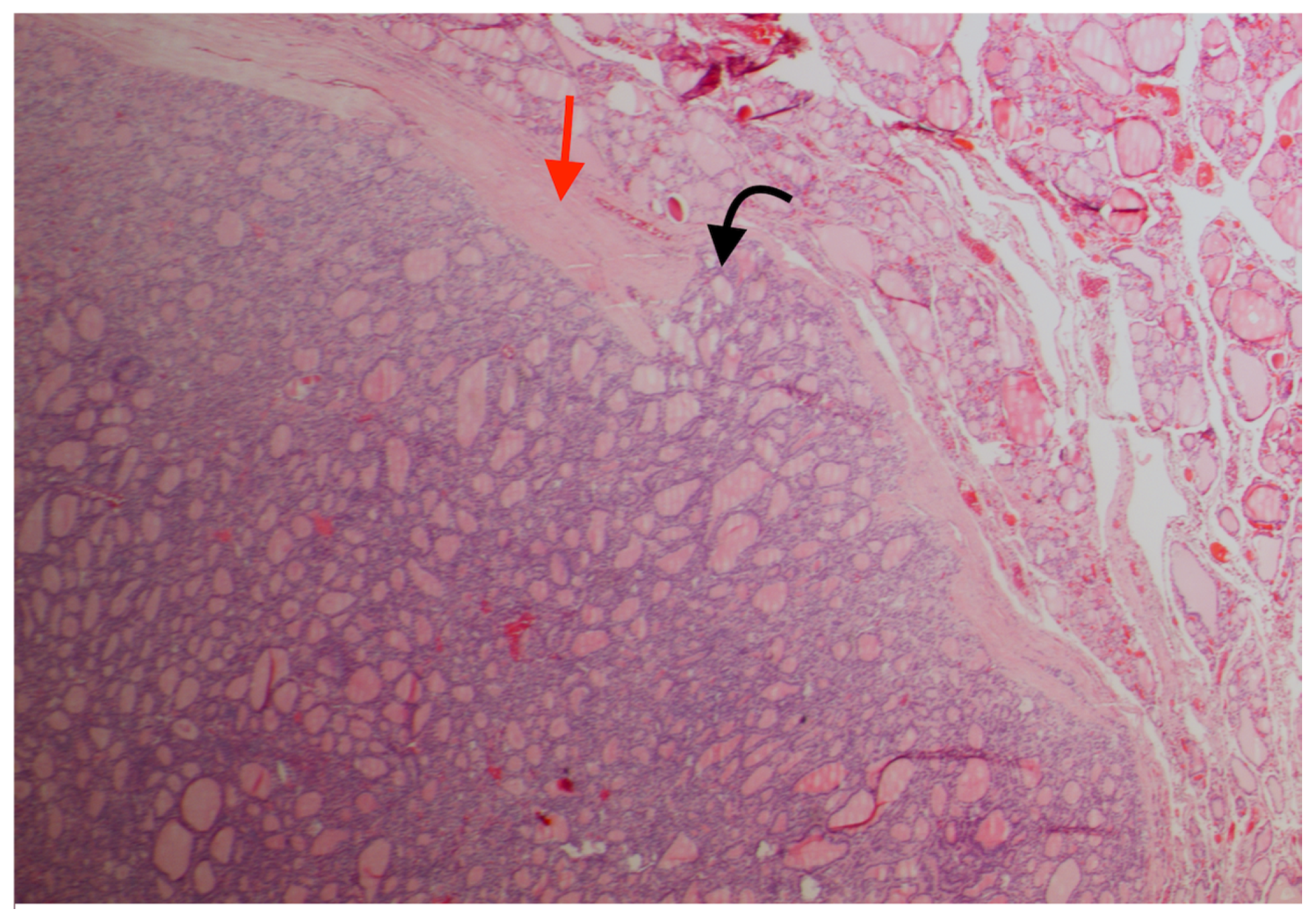
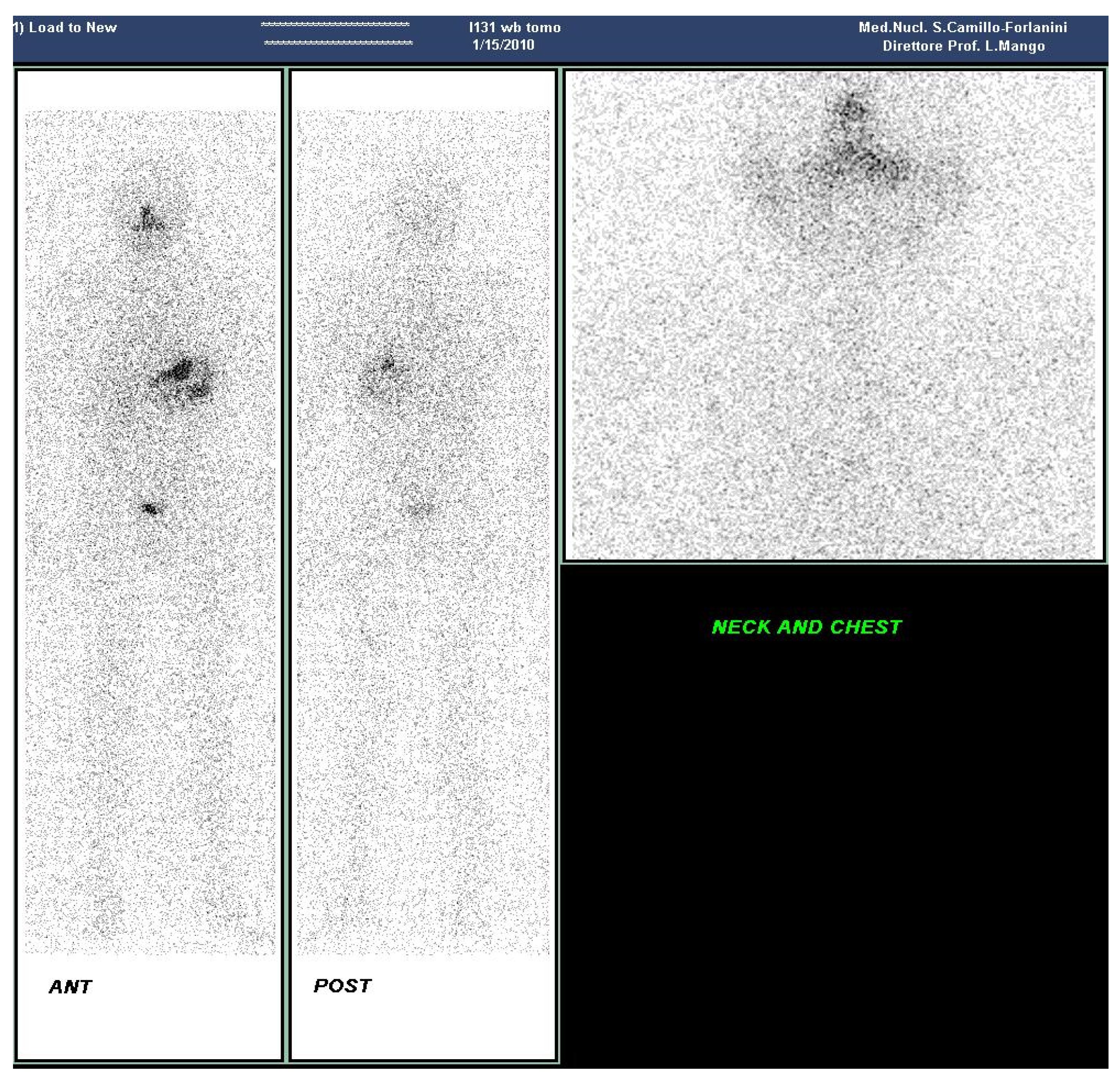
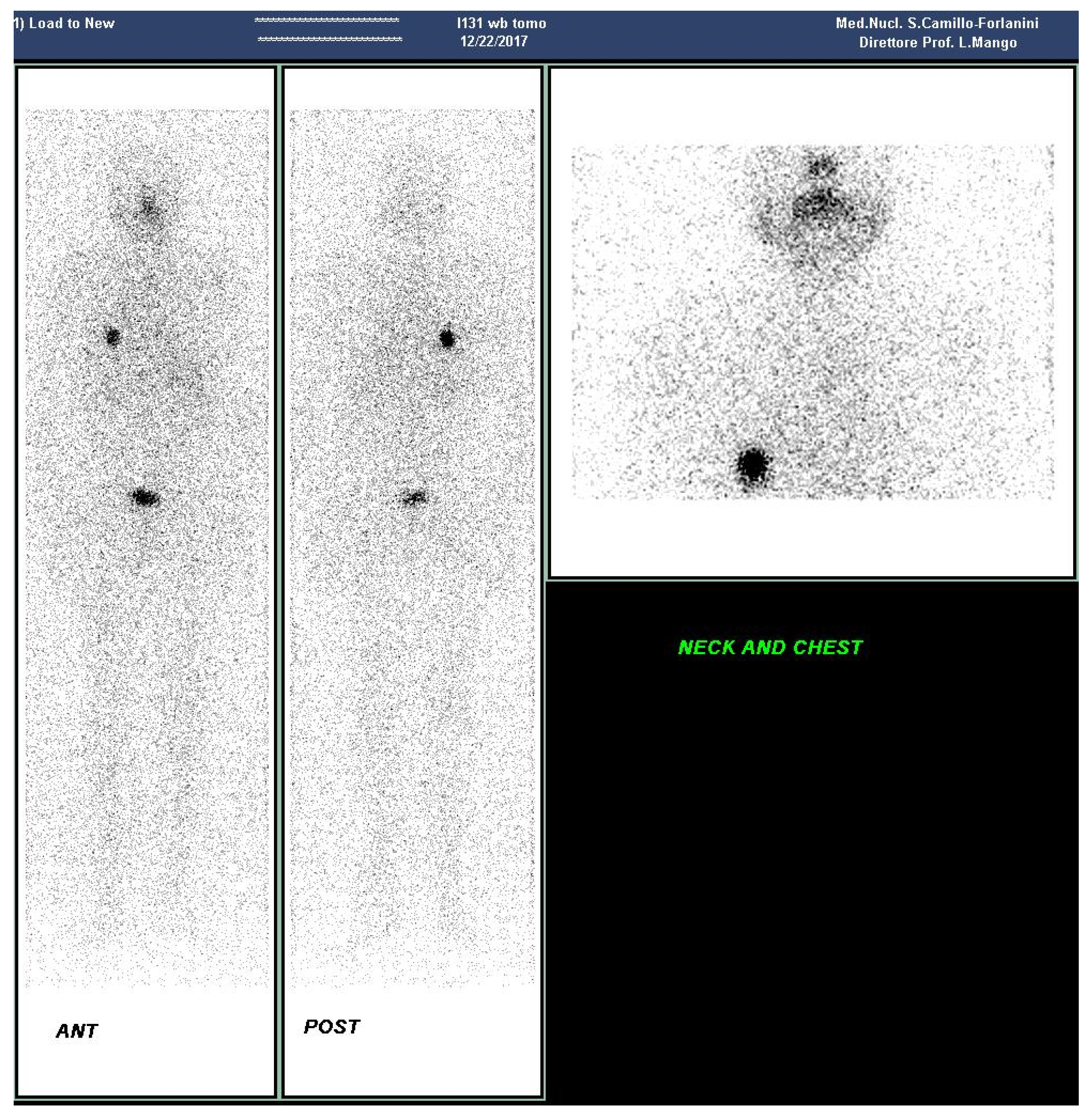
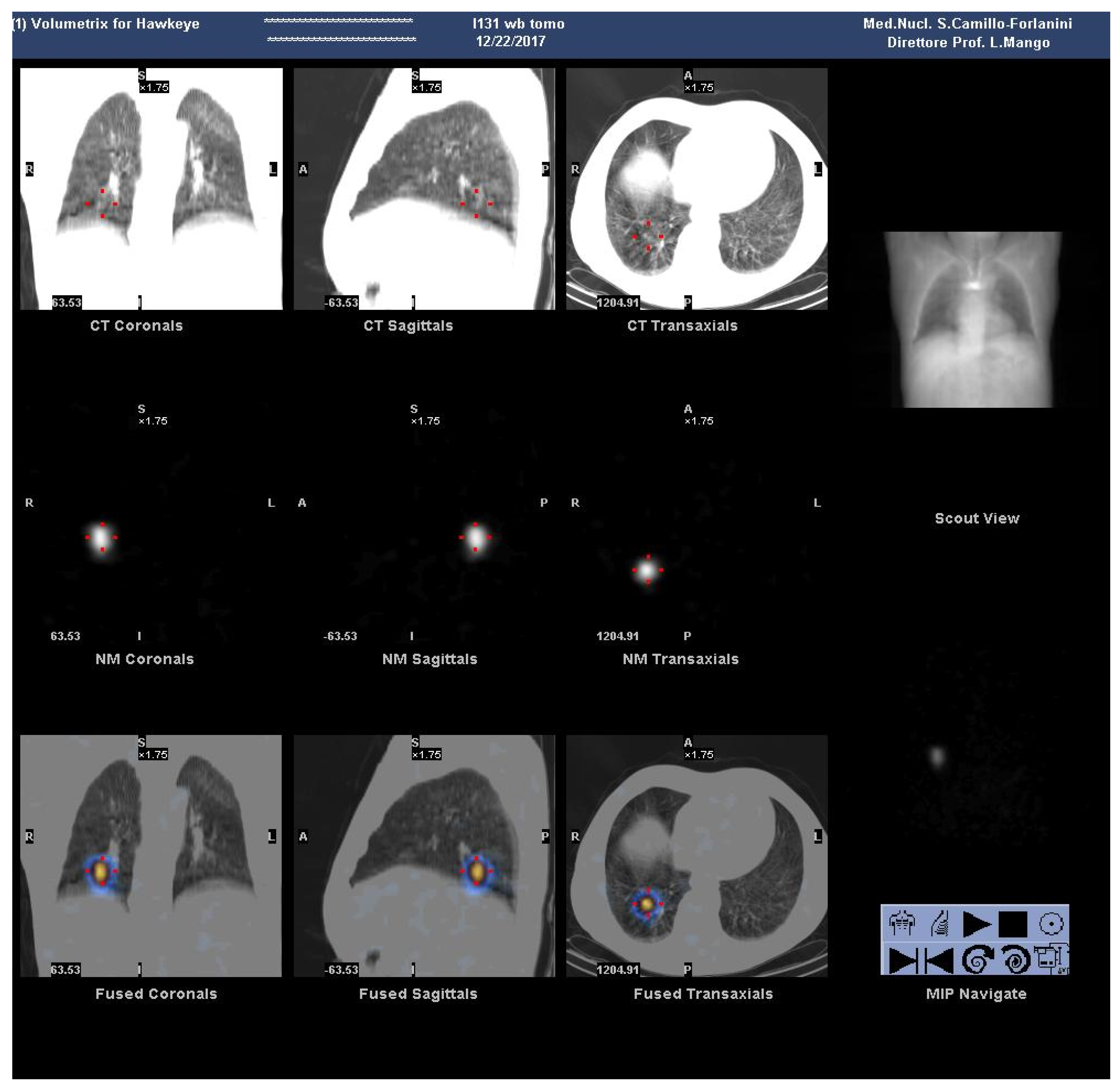
2. Case Presentation Section
3. Discussion
4. Conclusions
Acknowledgments
Funding
Conflicts of Interest
References
- Enache, O.S.; Martin, S.; Ion, O.; Grigore, A.; Bojoga, I.; Parfeni, O.; Sirbu, A.; Barbu, C.; Giulea, C.; Miron, A.; et al. Thyroid cancer prevalence and pathological features in thyroidectomy patients-five years experience. In Proceedings of the 20th European Congress of Endocrinology, Barcelona, Spain, 19–22 May 2018; p. 1161. [Google Scholar]
- Mazzaferri, E.L.; Jhiang, S.M. Long-term impact of initial surgical and medical therapy on papillary and follicular thyroid cancer. Am. J. Med. 1994, 97, 418–428. [Google Scholar] [CrossRef]
- Ronga, G.; Filesi, M.; Ferranti, E.; Mango, L.; Barone, R.; Procaccini, E.; Ventroni, G.; Signore, A.; Baschieri, I. Histology and survival in 1085 patients treated with 131I for thyroid cancer. Q. J. Nucl. Med. 1996, 40, 8. [Google Scholar]
- Pacini, F.; Schlumberger, M.; Harmer, C.; Berg, G.G.; Cohen, O.; Duntas, L.; Jamar, F.; Jarzab, B.; Limbert, E.; Lind, P.; et al. Post-surgical use of radioiodine (131I) in patients with papillary and follicular thyroid cancer and the issue of remnant ablation: A consensus report. Eur. J. Endocrinol. 2005, 153, 651–659. [Google Scholar] [CrossRef] [PubMed]
- Andresen, N.S.; Buatti, J.M.; Tewfik, H.H.; Pagedar, N.A.; Anderson, C.M.; Watkins, J.M. Radioiodine ablation following thyroidectomy for differentiated thyroid cancer: Literature review of utility, dose, and toxicity. Eur. Thyroid. J. 2017, 6, 187–196. [Google Scholar] [CrossRef]
- Thoresen, S.O.; Akslen, L.A.; Glattre, E.; Haldorsen, T.; Lund, E.V.; Schoultz, M. Survival and prognostic factors in differentiated thyroid carcinoma: A multivariate analysis of 1,055 cases. Br. J. Surg. 1989, 59, 231–235. [Google Scholar]
- Filesi, M.; Signore, A.; Mango, L.; Barbarossa, C.; Procaccini, E.; Righi, V.A.; Ronga, G.; Baschieri, I. 131I therapy and survival in young patients with differentiated thyroid carcinoma. Q. J. Nucl. Med. 1996, 40, 90. [Google Scholar]
- Filesi, M.; Mango, L.; Salerno, G.; Barbarossa, C.; Barone, R.; Ciancamerla, M.; Paserio, E.; Ventroni, G.; Ronga, G. Diagnostic value for metastases of first 131I WBS post thyroidectomy for differentiated cancer. Q. J. Nucl. Med. 1996, 40, 74. [Google Scholar]
- Tran, B.M.; Roshan, D.J.; Abraham, E.; Wang, L.Y.; Garibotto, N.; Wykes, J.; Campbell, P.; Ebrahimi, A. The Prognostic Impact of Tumor Size in Papillary Thyroid Carcinoma is Modified by Age. Thyroid 2018, 28. [Google Scholar] [CrossRef]
- Calabrese, G.; Dolcimascolo, A.; Torrisi, F.; Zappalà, A.; Gulino, R.; Parenti, R. MiR-19a Overexpression in FTC-133 Cell Line Induces a More De-Differentiated and Aggressive Phenotype. Int. J. Mol. Sci. 2018, 19, 3944. [Google Scholar] [CrossRef]
- Hu, J.; Li, C.; Liu, C.; Zhao, S.; Wang, Y.; Fu, Z. Expressions of miRNAs in papillary thyroid carcinoma and their associations with the clinical characteristics of PTC. Cancer Biomark 2017, Pre-press, 1–8. [Google Scholar] [CrossRef]
- Hundahl, S.A.; Fleming, I.D.; Fremgen, A.M.; Menck, H.R. A National Cancer Data Base report on 53,856 cases of thyroid carcinoma treated in the U.S., 1985–1995. Cancer. 1998, 83, 2638–2648. [Google Scholar] [CrossRef]
- Brennan, M.D.; Bergstralh, E.J.; van Heerden, J.A.; McConahey, W.M. Follicular thyroid cancer treated at the Mayo Clinic, 1946 through 1970: Initial manifestation, pathologic findings, therapy, and outcome. Mayo. Clin. Proc. 1991, 66, 11–22. [Google Scholar] [CrossRef]
- Jonathan, M.; Steward, D.L. Surgical Management of Low-Risk Papillary Thyroid Cancer. In Management of Thyroid Nodules and Differentiated Thyroid Cancer; Springer: Cham, Switzerland, 2017; pp. 231–240. [Google Scholar]
- Chan, T.W.; Hurley, J.R.; Fahey, T.J. Postoperative Radioactive Iodine for Differentiated Thyroid Cancer: A Historical Perspective. Clin. Oncol. 2017, 2, 1230. [Google Scholar]
- Baudin, E.D.C.C.; Cao, C.D.; Cailleux, A.F.; Leboulleux, S.; Travagli, J.P.; Schlumberger, M. Positive predictive value of serum thyroglobulin levels, measured during the first year of follow-up after thyroid hormone withdrawal, in thyroid cancer patients. J. Clin. Endocrinol. MeTab. 2003, 88, 1107–1111. [Google Scholar] [CrossRef] [PubMed]
- Alzahrani, A.S.; AlShaikh, O.; Tuli, M.; Al-Sugair, A.; Alamawi, R.; Al-Rasheed, M.M. Diagnostic value of recombinant human thyrotropin–stimulated 123I whole-body scintigraphy in the follow-up of patients with differentiated thyroid cancer. Clini. Nucl. Med. 2012, 37, 229–234. [Google Scholar] [CrossRef] [PubMed]
- Spencer, C.A.; Takeuchi, M.; Kazarosyan, M.; Wang, C.C.; Guttler, R.B.; Singer, P.A.; Fatemi, S.; LoPresti, J.S.; Nicoloff, J.T. Serum thyroglobulin autoantibodies: Prevalence, influence on serum thyroglobulin measurement, and prognostic significance in patients with differentiated thyroid carcinoma. J. Clin. Endocrinol. Metab. 1998, 83, 1121–1127. [Google Scholar] [CrossRef] [PubMed]
- Teo, K.W.; Yuan, N.K.; Tan, W.B.; Parameswaran, R. Comparison of prognostic scoring systems in follicular thyroid cancer. Ann. R Coll. Surg. Engl. 2017, 99, 479–484. [Google Scholar] [CrossRef] [PubMed]
- Ito, Y.; Miyauchi, A.; Kihara, M.; Fukushima, M.; Higashiyama, T.; Miya, A. Overall Survival of Papillary Thyroid Carcinoma Patients: A Single-Institution Long-Term Follow-Up of 5897 Patients. World J. Surg. 2018, 42, 615–622. [Google Scholar] [CrossRef] [PubMed]
- Hay, I.D.; Thompson, G.B.; Grant, C.S.; Bergstralh, E.J.; Dvorak, C.E.; Gorman, C.A.; Maurer, M.S.; McIver, B.; Mullan, B.P.; Oberg, A.L.; et al. Papillary thyroid carcinoma managed at the Mayo Clinic during six decades (1940–1999): Temporal trends in initial therapy and long-term outcome in 2444 consecutively treated patients. World J. Surg. 2002, 26, 879–885. [Google Scholar] [CrossRef] [PubMed]
- Soares, P.; Celestino, R.; Melo, M.; Fonseca, E.; Sobrinho-Simões, M. Prognostic biomarkers in thyroid cancer. Virchows. Arch. 2014, 464, 333. [Google Scholar]
- Cailleux, A.F.; Baudin, E.; Travagli, J.P.; Ricard, M.; Schlumberger, M. Is diagnostic iodine-131 scanning useful after total thyroid ablation for differentiated thyroid cancer? J. Clin. Endocrinol. Metab. 2000, 85, 175–178. [Google Scholar] [CrossRef] [PubMed]
- Samaan, N.A.; Schultz, P.N.; Hickey, R.C.; Goepfert, H.; Haynie, T.P.; Johnston, D.A.; Ordonez, N.G. The results of various modalities of treatment of well differentiated thyroid carcinomas: A retrospective review of 1599 patients. J Clin Endocrinol Metab. 1992, 75, 714–720. [Google Scholar] [PubMed]
- Pacini, F.; Basolo, F.; Bellantone, R.; Boni, G.; Cannizzaro, M.A.; De Palma, M.; Durante, C.; Elisei, R.; Fadda, G.; Frasoldati, A.; et al. Italian consensus on diagnosis and treatment of differentiated thyroid cancer: Joint statements of six Italian societies. J. Endocrinol. Invest. 2018, 41, 849–876. [Google Scholar] [CrossRef] [PubMed]
- Mazzaferri, E.L. Thyroid remnant 131I ablation for papillary and follicular thyroid carcinoma. Thyroid 1997, 7, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Burch, H.B. Papillary Thyroid Cancer. In Thyroid Cancer; Humana Press: Totowa, NJ, USA, 2000; pp. 229–237. [Google Scholar]
- Webb, R.C.; Howard, R.S.; Stojadinovic, A.; Gaitonde, D.Y.; Wallace, M.K.; Ahmed, J.; Burch, H.B. The utility of serum thyroglobulin measurement at the time of remnant ablation for predicting disease-free status in patients with differentiated thyroid cancer: A meta-analysis involving 3947 patients. J. Clin Endocrinol Metab. 2012, 97, 2754–2763. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Liang, J.; Li, T.; Zhao, T.; Lin, Y. Preablative stimulated thyroglobulin correlates to new therapy response system in differentiated thyroid cancer. J. Clin Endocrinol Metab. 2016, 101, 1307–1313. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Cheng, L.; Jin, Y.; Ruan, M.; Sheng, S.; Chen, L. Predicting 131 I-avidity of metastases from differentiated thyroid cancer using 18 F-FDG PET/CT in postoperative patients with elevated thyroglobulin. Sci. Rep. 2018, 8, 4352. [Google Scholar] [CrossRef] [PubMed]
- Linee Guida SIE-AIMN-AIFM per il Trattamento e Follow-up del Carcinoma Differenziato della Tiroide. Available online: https://www.aimn.it/pubblicazioni/LG/LG_ca_tiroide.pdf (accessed on 1 February 2019).
- HaugenBryan, R.; AlexanderErik, K.; BibleKeith, C.; DohertyGerard, M.; MandelSusan, J.; NikiforovYuri, E.; Pacini, F.; Randolph, G.W.; Sawka, A.M.; Schlumberger, M.; et al. 2015 American Thyroid Association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: The American Thyroid Association guidelines task force on thyroid nodules and differentiated thyroid cancer. Thyroid. 2016, 26, 1–133. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Semprebene, A.; Mangano, A.M.; Ventroni, G.; Barone, R.; Piro, F.R.; Mango, L. Differentiated Thyroid Carcinoma and Late Onset of Lung Distant Metastasis. A Case Report. Reports 2019, 2, 6. https://doi.org/10.3390/reports2010006
Semprebene A, Mangano AM, Ventroni G, Barone R, Piro FR, Mango L. Differentiated Thyroid Carcinoma and Late Onset of Lung Distant Metastasis. A Case Report. Reports. 2019; 2(1):6. https://doi.org/10.3390/reports2010006
Chicago/Turabian StyleSemprebene, Alessandro, Anna Maria Mangano, Guido Ventroni, Raffaella Barone, Francesca Romana Piro, and Lucio Mango. 2019. "Differentiated Thyroid Carcinoma and Late Onset of Lung Distant Metastasis. A Case Report" Reports 2, no. 1: 6. https://doi.org/10.3390/reports2010006
APA StyleSemprebene, A., Mangano, A. M., Ventroni, G., Barone, R., Piro, F. R., & Mango, L. (2019). Differentiated Thyroid Carcinoma and Late Onset of Lung Distant Metastasis. A Case Report. Reports, 2(1), 6. https://doi.org/10.3390/reports2010006




