Research on Minimum Ignition Energy Testing of Normal-Alkane Vapors
Abstract
1. Introduction
2. Apparatus and Testing Methodology
2.1. Experimental Materials
2.2. Apparatus
2.3. Testing Methodology
- (1)
- Initial Conditions: Temperature = 25 ± 1 °C, Absolute Pressure = 0.1 MPa, Relative Humidity = 45 ± 5%.
- (2)
- Electrode Specifications: Material: Tungsten; Tip Geometry: 30° conical tip; Gap Distance: 2.0 mm.
- (3)
- Surface Condition: Electrodes were polished with fine-grit sandpaper and cleaned with ethanol prior to each test series.
- (4)
- Discharge Circuit Parameters: Capacitance range: 5, 20, 80, and 320 pF; Corresponding discharge duration (as measured at half-peak current): 50–200 ns.
3. Results and Discussion
3.1. Determination of Critical Thresholds
3.1.1. Determination of Critical Capacitance Value
- (1)
- An initial energy value E0 = 0.30 mJ was set, with an energy step size of ΔE = 0.02 mJ (corresponding to 7% of E0). A total of 24 tests were conducted under these conditions. The results are summarized in Table 2.
- (2)
- The experimental data were organized in ascending order of energy level, with the lowest level set at 0.24 mJ. The sorted results are presented in Table 3.
3.1.2. Determination of Critical Optimal Electrode Gap
3.2. Integrated System Validation
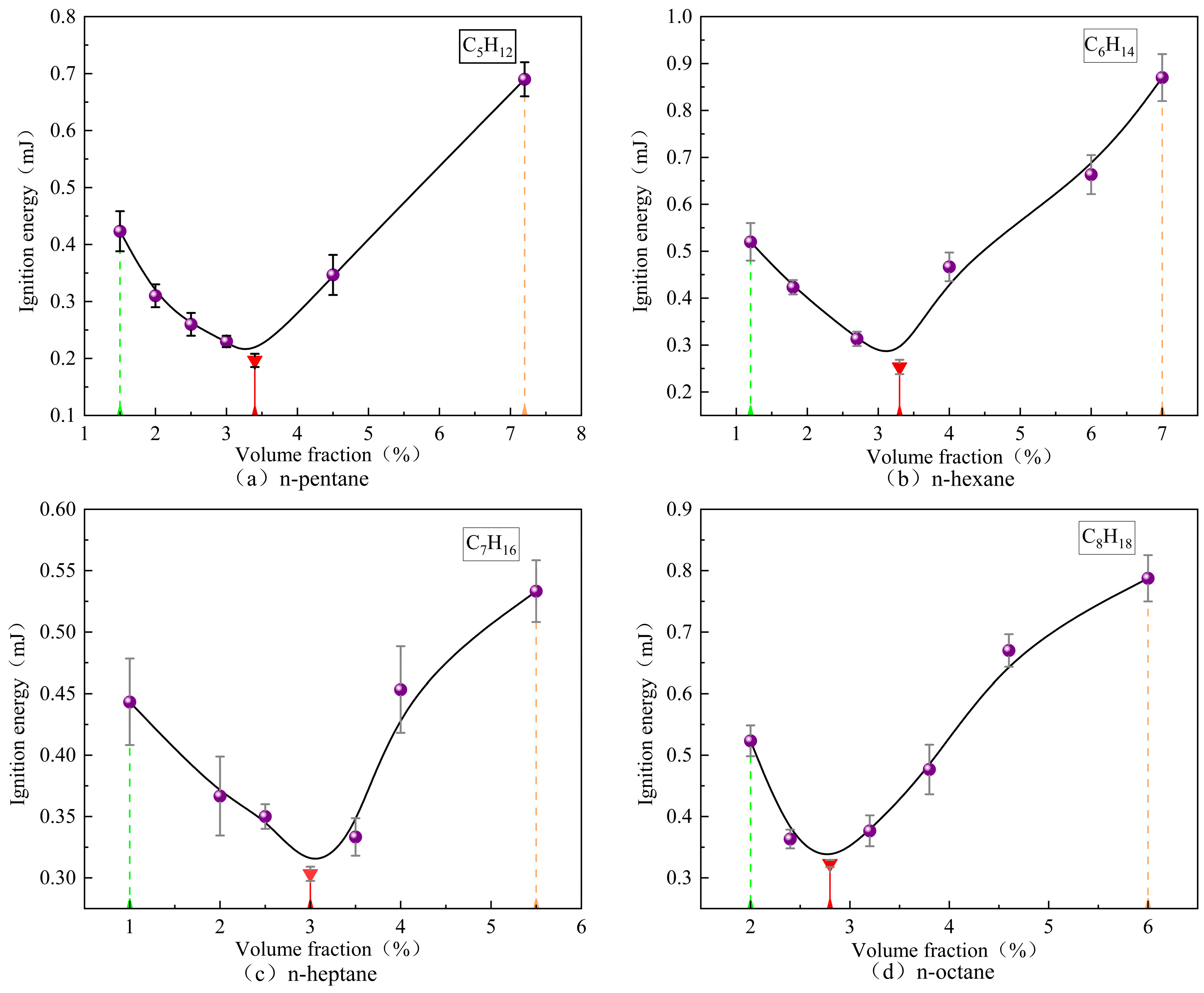
3.3. Influence of Volume Fraction on MIE of Alkane Vapors
- (1)
- Minor fluctuations in mixture homogeneity, particularly for high-boiling-point fuels where vapor condensation may occur;
- (2)
- The intrinsic stochastic nature of spark kernel development at energy levels approaching the minimum;
- (3)
- The measurement precision of the capacitive discharge circuit.
- (1)
- Theory of critical size for flame kernel:
- (2)
- Coupling of chemical kinetics and thermodynamics:
3.4. Correlation Between Alkane Carbon Chain Length and MIE
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Popa, C.; Nan, S.; Paraian, M.; Jurca, A.; Păun, F. Aspects of laboratory tests for the determination of the minimum ignition energy of the fuel/dust mixture. MATEC Web Conf. EDP Sci. 2021, 342, 04004. [Google Scholar] [CrossRef]
- Wang, Y.; Ding, J.; Yang, S.; Wang, Z.; Ye, W.; Ye, S. Study on the test method of minimum ignition energy of vapor under the gas-liquid coexistence condition with trace liquid. Fire Saf. Sci. 2023, 32, 77–84. [Google Scholar]
- Baalisampang, T.; Abbassi, R.; Garaniya, V.; Khan, F.; Dadashzadeh, M. Review and analysis of fire and explosion accidents in maritime transportation. Ocean. Eng. 2018, 158, 350–366. [Google Scholar] [CrossRef]
- Okamoto, K.; Ichikawa, T.; Fujimoto, J.; Kashiwagi, N.; Nakagawa, M.; Hagiwara, T.; Honma, M. Prediction of evaporative diffusion behavior and explosion damage in gasoline leakage accidents. Process Saf. Environ. Prot. 2021, 148, 893–902. [Google Scholar] [CrossRef]
- Fernández-Tarrazo, E.; Gómez-Miguel, R.; Sánchez-Sanz, M. Minimum ignition energy of hydrogen-ammonia blends in air. Fuel 2023, 337, 127128. [Google Scholar] [CrossRef]
- Movileanu, C.; Mitu, M.; Giurcan, V.; Razus, D.; Oancea, D. Quenching distances, minimum ignition energies and related properties of propane-air-diluent mixtures. Fuel 2020, 274, 117836. [Google Scholar] [CrossRef]
- Ghosh, A.; Munoz-Munoz, N.M.; Lacoste, D.A. Minimum ignition energy of hydrogen-air and methane-air mixtures at temperatures as low as 200 K. Int. J. Hydrogen Energy 2022, 47, 30653–30659. [Google Scholar] [CrossRef]
- Zhang, W.; Gou, X.; Chen, Z. Effects of water vapor dilution on the minimum ignition energy of methane, n-butane and n-decane at normal and reduced pressures. Fuel 2017, 187, 111–116. [Google Scholar] [CrossRef]
- Yu-ichiro, I.; Tomoya, S.; Hideshi, I.; Miyake, A.S. Determining the minimum ignitionenergy of toluene vapor containing hydrogen towards a risk assessment for liquid organic hydride storage in hydrogen refueling stations. Fuel 2022, 310, 122236. [Google Scholar]
- Ebina, W.; Liao, C.; Naito, H.; Yoshida, A. Effect of water mist on minimum ignition energy of propane/air mixture. Proc. Combust. Inst. 2017, 36, 3271–3278. [Google Scholar] [CrossRef]
- Coronel, S.; Mével, R.; Bane, S.; Shepherd, J. Experimental study of minimum ignition energy of lean H2-N2O mixtures. Proc. Combust. Inst. 2013, 34, 895–902. [Google Scholar] [CrossRef]
- Yu, D.; Chen, Z. Premixed flame ignition: Theoretical development. Prog. Energy Combust. Sci. 2024, 104, 101174. [Google Scholar] [CrossRef]
- GB/T 14288-1993; Determination of Minimum Ignition Energy of Combustible Gases and Flammable Liquid Vapors. National Technical Committee on Fire Safety of Standardization Administration of China: Beijing, China; China Standards Press: Beijing, China, 1993.
- IEC 61241-2-3-1994; Electrical Apparatus for Use in the Presence of Combustible Dust: Part 2 Test Methods, Section 3 Method of Determining Minimum Ignition Energy of Dust/Air Mixtures. International Electrotechnical Commission: Geneva, Switzerland, 1994; pp. 13–15.
- EN 13821; 2002 Potentially Explosive Atmosphere, Explosion Prevention and Protection, Determination of Minimum Ignition Energy of Dust/Air Mixtures. Comite Europeen de Normalisation: London, UK, 2002; pp. 5–14.
- Cirrone, D.; Makarov, D.; Proust, C.; Molkov, V. Minimum ignition energy of hydrogen-air mixtures at ambient and cryogenic temperatures. Int. J. Hydrogen Energy 2023, 48, 16530–16544. [Google Scholar] [CrossRef]
- Wang, B.; Zhou, L.; Xu, K.; Wang, Q. Fast prediction of minimum ignition energy from molecular structure using simple QSPR model. J. Loss Prev. Process Ind. 2017, 50, 290–294. [Google Scholar] [CrossRef]
- Su, B.; Dong, H.; Luo, Z.; Deng, J.; Wang, T.; Cheng, F. Research progress on explosion dynamics characteristics and mechanism of hybrid mixtures. CIESC J. 2024, 75, 2109–2122. [Google Scholar]
- Rimpf, L.M. Measurements of Spark Ignition Energy of n-Octane and i-Octane. Master’s Thesis, University of Toledo, Toledo, OH, USA, 2005. [Google Scholar]
- ASTM E582-21; Standard Test Method for Minimum Ignition Energy and Quenching Distance in Gaseous Mixtures. ASTM International: West Conshohocken, PA, USA, 2021.
- Zhang, Z.; Cai, P. Study on Affecting Factors of Minimum Ignition Energy (MIE) and Analysis on Its Calculation Error. China Saf. Sci. J. 2004, 14, 88–91. [Google Scholar]
- Christensen, D.; Novik, P.; Unneberg, E. Estimating sensitivity with the Bruceton method: Setting the record straight. Propellants, Explosives. Pyrotechnics 2024, 49, e202400022. [Google Scholar] [CrossRef]
- Kershaw, C. A comparison of estimators of the ED50 in up-and-down experiments. J. Stat. Comput. Simul. 1987, 27, 175–184. [Google Scholar] [CrossRef]
- Li, C.; Ma, Z.; Dong, Z.; Chen, Y.; Yuan, C.; Bai, R. Theoretical study on electric spark ignition sensitivity of methane/air mixture. J. Saf. Environ. 2022, 22, 1913–1918. [Google Scholar]
- Turquand d’Auzay, C.; Papapostolou, V.; Ahmed, S.F.; Chakraborty, N. On the minimum ignition energy and its transition in the localised forced ignition of turbulent homogeneous mixtures. Combust. Flame 2019, 201, 104–117. [Google Scholar] [CrossRef]
- Dorval, A.; Geraud, K.; Valensi, F.; Hamdan, A. Statistical analysis of pulsed spark discharges in water: Effects of gap distance, electrode material, and voltage polarity on discharge characteristics. J. Vac. Sci. Technol. A-Vac. Surf. Film. 2022, 40, 17. [Google Scholar] [CrossRef]
- Wu, F.; Liu, S.; Wang, D. Influence of needle electrode curvature radius on breakdown characteristics of micro-gap discharge in air. J. Xi’an Univ. Sci. Technol. 2023, 43, 1015–1024. [Google Scholar]
- Haasee, H. Electrostatic Hazards: Their Evaluation and Control; Verlag Chemie: Weinheim, Germany, 1977. [Google Scholar]
- Liu, X.L.; Zhang, Q. Experimental study on minimum ignition energy of n-Alkane mists. Trans. Beijing Inst. Technol. 2018, 38, 1252–1255+1320. [Google Scholar]
- Law, C.K.; Makino, A.; Lu, T.F. On the off-stoichiometric peaking of adiabatic flame temperature. Combust. Flame 2006, 145, 808–819. [Google Scholar] [CrossRef]
- Lewis, B.; von Elbe, G. Combustion, Flames and Explosions of Gases; Elsevier: Amsterdam, The Netherlands, 2012. [Google Scholar]
- Moorhouse, J.; Williams, A.; Maddison, T.E. An investigation of the minimum ignition energies of some C1 to C7 hydrocarbons. Combust. Flame 1974, 23, 203–213. [Google Scholar] [CrossRef]
- Kckhoff, E.R.; Ngo, M.; Olsen, W. On the minimum ignition energy (MIE) for propane/air. J. Hazard. Mater. 2010, 175, 293–297. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Van, K.; Lee, K.D.; Yoo, C.S.; Park, J.; Chung, S.H. Laminar flame speed, Markstein length, and cellular instability for spherically propagating methane/ethylene–air premixed flames. Combust. Flame 2020, 214, 464–474. [Google Scholar] [CrossRef]
- Chen, H.F.; Chen, C.C. A quantitative structure activity relationship model for predicting minimum ignition energy of organic substance. J. Loss Prev. Process Ind. 2020, 67, 104227. [Google Scholar] [CrossRef]
- Hori, T.; Kamino, T.; Yoshimoto, Y.; Takagi, S.; Kinefuchi, I. Mutual influence of molecular diffusion in gas and surface phases. Phys. Rev. E 2018, 97, 013101. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Gong, S.; Wang, L.; Zhang, X.; Liu, G. High pressure pyrolysis mechanism and kinetics of a strained-caged hydrocarbon fuel quadricyclane. Fuel 2019, 239, 935–945. [Google Scholar] [CrossRef]






| Test Sample | Purity | Manufacturer |
|---|---|---|
| Propane | 99.9% | Guanghan Mingyuan Gas Co., Ltd., Guanghan, China. |
| Dry Air | 99.9% | Chengdu Jinkexing Gas Co., Ltd., Chengdu, China. |
| n-Pentane, n-Hexane, n-Heptane, n-Octane | 99.5% | Chengdu Jinshudu Scientific Supply Co., Ltd., Chengdu, China. |
| n-Nonane, n-Decane, n-Undecane | 99.2% | Chengdu Kelong Chemical Co., Ltd., Chengdu, China. |
| Test Number | Ignition Energy (mJ) | Results | Adjustment |
|---|---|---|---|
| 1 | 0.30 | S | reduce ΔE |
| 2 | 0.28 | F | increase ΔE |
| 3 | 0.30 | S | reduce ΔE |
| 4 | 0.28 | S | reduce ΔE |
| 5 | 0.26 | F | increase ΔE |
| 6 | 0.28 | S | reduce ΔE |
| 7 | 0.26 | S | reduce ΔE |
| 8 | 0.24 | F | increase ΔE |
| 9 | 0.26 | S | reduce ΔE |
| 10 | 0.24 | F | increase ΔE |
| … | … | … | … |
| 23 | 0.28 | S | reduce ΔE |
| 24 | 0.26 | F | over |
| Energy Level (mJ) | Level Number i | Total Number of Times ni | Number of Successful Attempts | Number of Failed Attempts | i·ni |
|---|---|---|---|---|---|
| 0.24 | 0 | 6 | 0 | 6 | 0 |
| 0.26 | 1 | 10 | 6 | 4 | 10 |
| 0.28 | 2 | 5 | 4 | 1 | 10 |
| 0.30 | 3 | 3 | 3 | 0 | 9 |
| Total | / | 24 | 13 | 11 | 29 |
| Alkane Vapors | Experimentally Determined MIE (mJ) | Literature-Reported MIE (mJ) | Absolute Deviation (mJ) | Relative Error (%) |
|---|---|---|---|---|
| C5H12 | 0.197 | 0.220 [28] | 0.023 | 10.45 |
| C6H14 | 0.253 | 0.248 [29] | 0.005 | 2.02 |
| C7H16 | 0.303 | 0.240 [28] | 0.063 | 26.25 |
| C8H18 | 0.323 | / | / | / |
| Alkane Vapors | Flammability Limit Range (%) | Stoichiometric Volume Fractions (%) | Critical Volume Fraction (%) |
|---|---|---|---|
| C5H12 | 1.4~7.8 | 2.6 | 3.4 |
| C6H14 | 1.1~7.5 | 2.2 | 3.3 |
| C7H16 | 1.1~6.7 | 1.9 | 3.0 |
| C8H18 | 1.8~6.5 | 1.7 | 2.8 |
| Alkane Vapors | Flammability Limit Range (%) | MIE (mJ) | Critical Volume Fraction (%) |
|---|---|---|---|
| C9H20 | 0.7~5.6 | 0.523 | 2.8 |
| C10H22 | 0.8~5.4 | 0.857 | 2.5 |
| C11H24 | 0.6~6.5 | 1.127 | 2.0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xiong, C.; Jia, X.; Chow, W.K.; Li, W. Research on Minimum Ignition Energy Testing of Normal-Alkane Vapors. Fire 2025, 8, 451. https://doi.org/10.3390/fire8120451
Xiong C, Jia X, Chow WK, Li W. Research on Minimum Ignition Energy Testing of Normal-Alkane Vapors. Fire. 2025; 8(12):451. https://doi.org/10.3390/fire8120451
Chicago/Turabian StyleXiong, Caizhi, Xuhong Jia, Wan Ki Chow, and Wenbing Li. 2025. "Research on Minimum Ignition Energy Testing of Normal-Alkane Vapors" Fire 8, no. 12: 451. https://doi.org/10.3390/fire8120451
APA StyleXiong, C., Jia, X., Chow, W. K., & Li, W. (2025). Research on Minimum Ignition Energy Testing of Normal-Alkane Vapors. Fire, 8(12), 451. https://doi.org/10.3390/fire8120451






