The Effect of Ecophysiological Traits on Live Fuel Moisture Content
Abstract
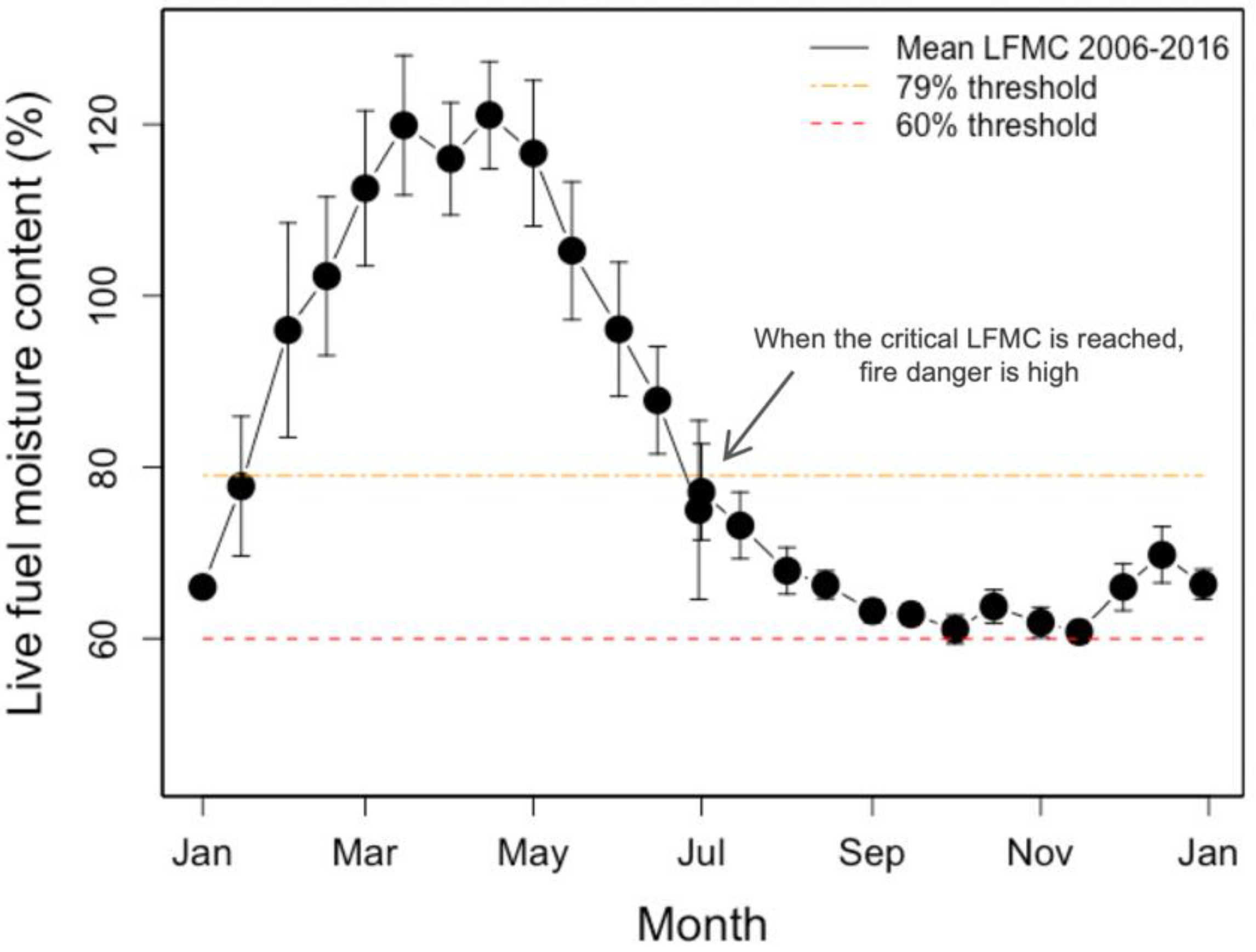
:1. Introduction
2. Methods and Materials
2.1. Study Site and Species
2.2. Live Fuel Moisture Content
2.3. Plant Water Potential
2.4. Gas Exchange
2.5. Pressure-Volume Curves
2.6. Statistical Analyses
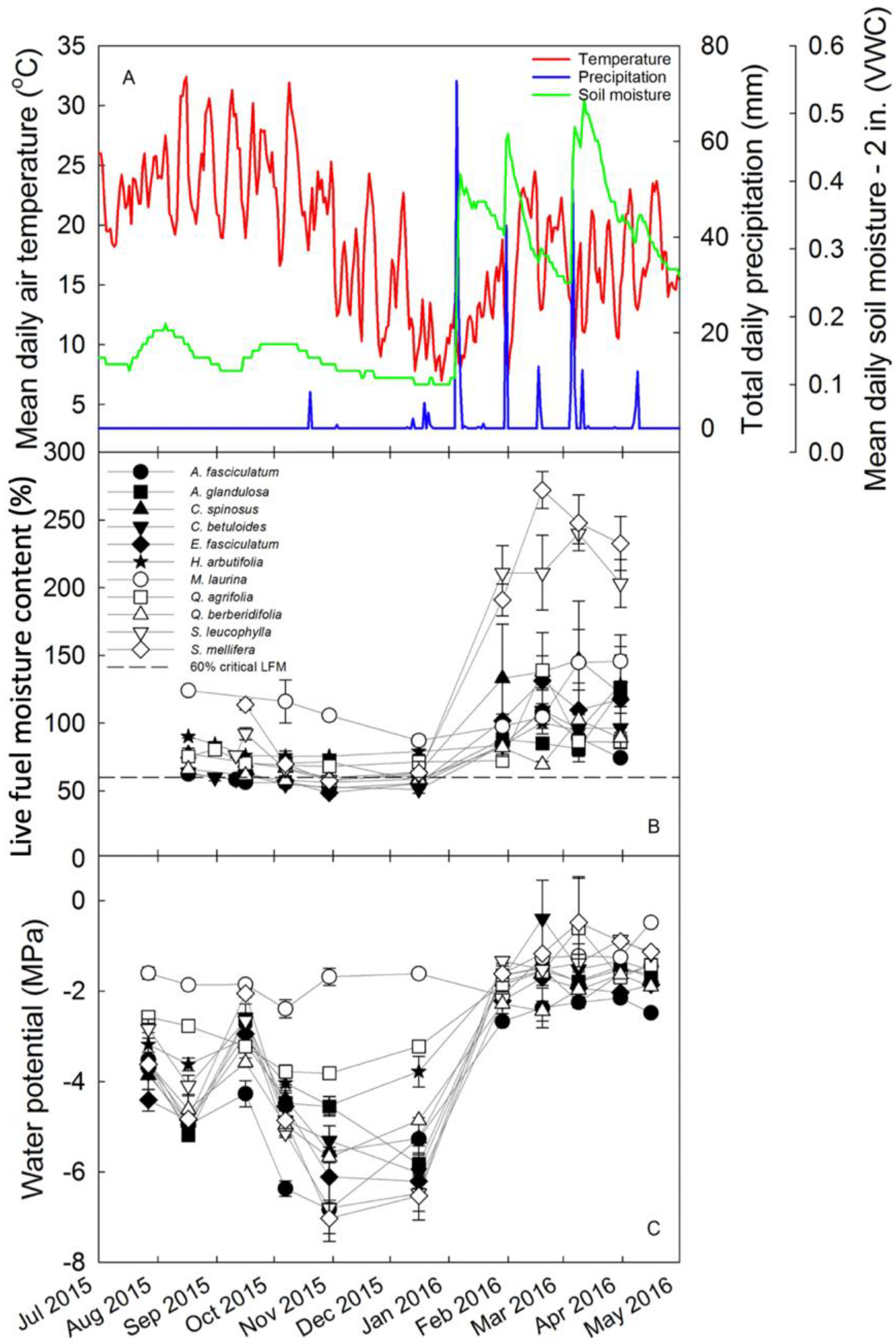
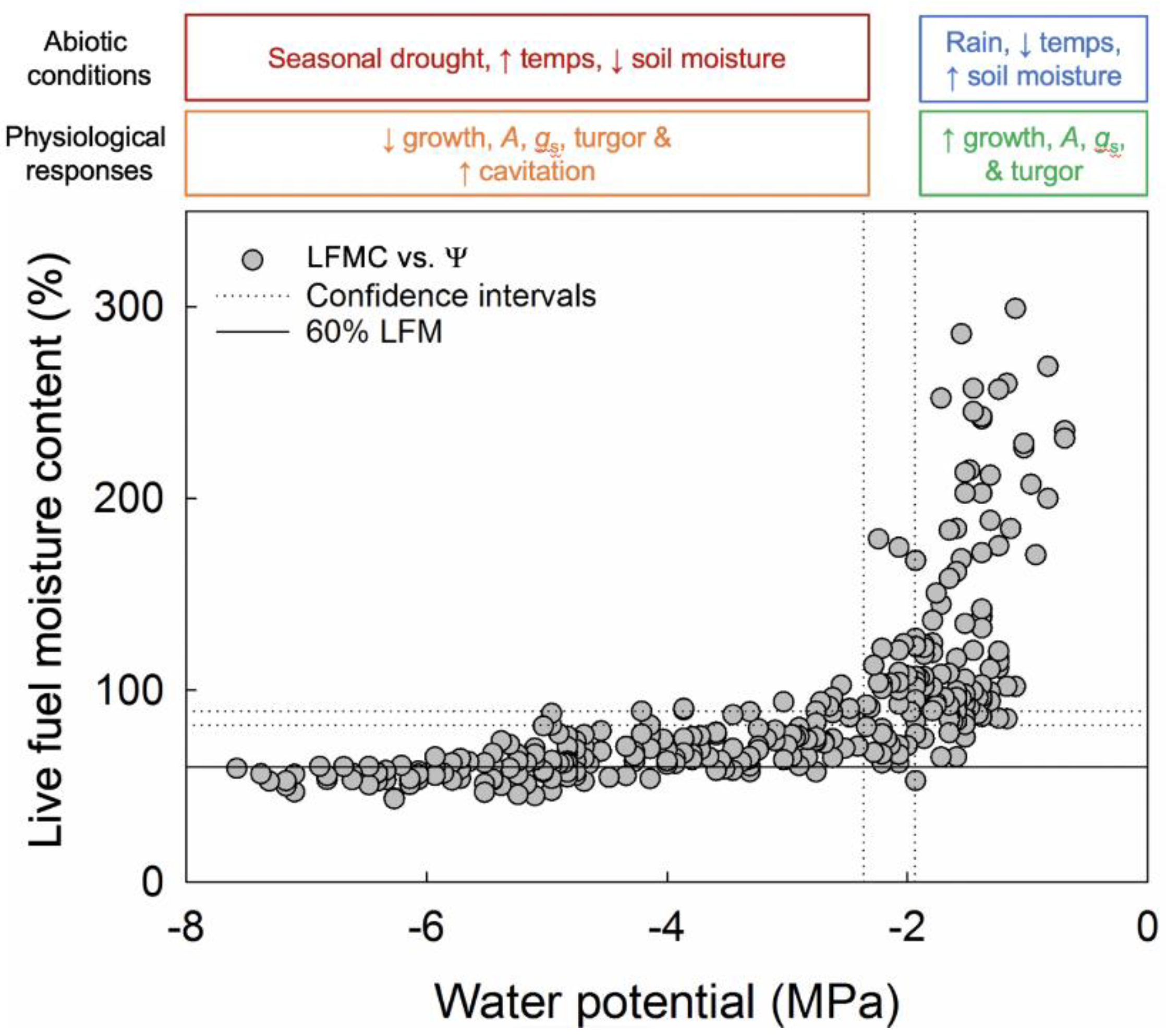
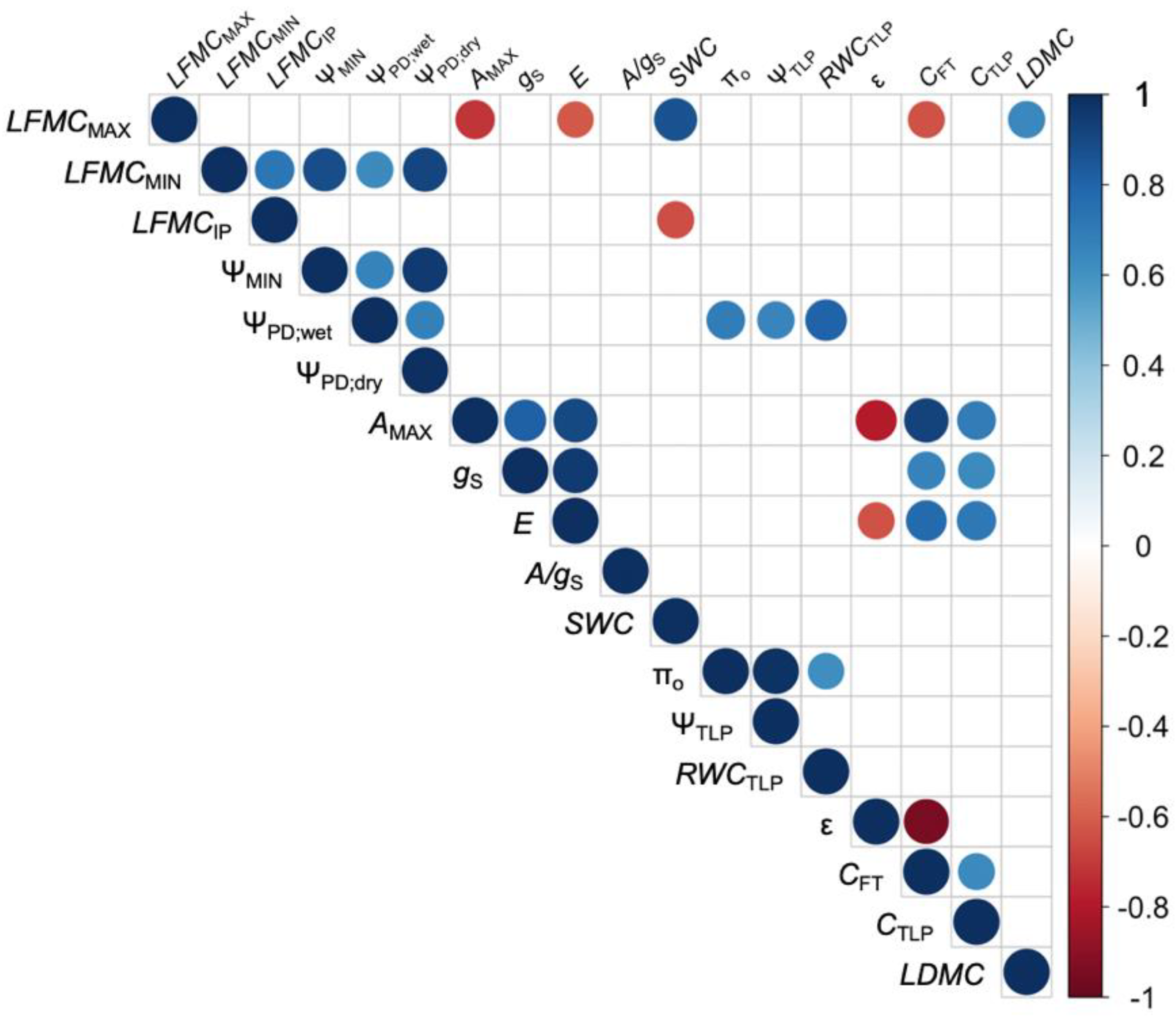
3. Results
4. Discussion
4.1. Overview
4.2. Physiological Controls of LFMC
4.3. Indicator Species Choice Can Impact Fire Danger Rating
5. Conclusions
The discipline of ecophysiology is rich and mostly unleveraged in live fuel research, yet it has the potential to link plant flammability traits at both the leaf and plant level to fundamental laws that govern how plants functions.
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| Trait | Abbreviation | Units |
| Live fuel moisture content | LFMC | % |
| Minimum seasonal live fuel moisture content | LFMCMIN | % |
| Maximum seasonal live fuel moisture content | LFMCMAX | % |
| Live fuel moisture inflection point | LFMCIP | % |
| Water potential | Ψ | MPa |
| Wet season predawn water potential | ΨPD;wet | MPa |
| Dry season predawn water potential | ΨPD;dry | MPa |
| Minimum seasonal water potential | ΨMIN | MPa |
| Maximum seasonal water potential | ΨMAX | MPa |
| Maximum photosynthetic carbon gain | AMAX | µmol·m−2·s−1 |
| Stomatal conductance | gS | mol·m−2·s−2 |
| Transpiration | E | mol·m−2·s−2 |
| Water use efficiency | A/gS | µmol·mol−1 |
| Saturated water content | SWC | % |
| Water potential at turgor loss point | ΨTLP | MPa |
| Relative water content at turgor loss point | RWCTLP | % |
| Osmotic potential | πo | MPa |
| Modulus of elasticity | ε | MPa |
| Capacitance at full turgor | CFT | MPa−1 |
| Capacitance at turgor loss point | CTLP | MPa−1 |
| Leaf dry matter content | LDMC | g·g−1 |
References
- Andrews, P.L.; Bradshaw, L.S. Use of meteorological information for fire management in the United States. In Proceedings of Workshop on Meteorological Information for Forest Fire Management in the Western Mediterranean Region; WMO: Geneva, Switzerland, 1991; pp. 325–332. [Google Scholar]
- Rossa, C.G.; Fernandes, P.M. Short communication: On the effect of live fuel moisture content on fire-spread rate. For. Syst. 2017, 26, eSC08. [Google Scholar] [CrossRef]
- Rossa, C.; Fernandes, P. An Empirical Model for the Effect of Wind on Fire Spread Rate. Fire 2018, 1, 31. [Google Scholar] [CrossRef]
- Rossa, C.; Fernandes, P. Live Fuel Moisture Content: The ‘Pea Under the Mattress’ of Fire Spread Rate Modeling? Fire 2018, 1, 43. [Google Scholar] [CrossRef]
- Dimitrakopoulos, A.P.; Papaioannou, K.K. Flammability assessment of Mediterranean forest fuels. Fire Technol. 2001, 37, 143–152. [Google Scholar] [CrossRef]
- Bilgili, E.; Saglam, B. Fire behavior in maquis fuels in Turkey. For. Ecol. Manag. 2003, 184, 201–207. [Google Scholar] [CrossRef]
- Anderson, S.A.J.; Anderson, W.R. Ignition and fire spread thresholds in gorse (Ulex europaeus). Int. J. Wildl. Fire 2010, 19, 589–598. [Google Scholar] [CrossRef]
- Keeley, J.E.; Bond, W.J.; Bradstock, R.A.; Pausas, J.G.; Rundel, P.W. Fire in Mediterranean Ecosystems: Ecology, Evolution, and Management; Cambridge University Press: New York, NY, USA, 2012. [Google Scholar]
- Schoenberg, F.P.; Peng, R.; Huang, Z.; Rundel, P. Detection of non-linearities in the dependence of burn area on fuel age and climatic variables. Int. J. Wildl. Fire 2003, 12, 1–6. [Google Scholar] [CrossRef]
- Pimont, F.; Ruffault, J.; Martin-StPaul, N.; Dupuy, J.L. Why is the effect of live fuel moisture content on fire rate of spread underestimated in field experiments in shrublands? Int. J. Wildl. Fire 2019. [Google Scholar] [CrossRef]
- Keeley, J.E.; Fotheringham, C.J. Historic fire regime in southern California shrublands. Conserv. Biol. 2001, 15, 1536–1548. [Google Scholar] [CrossRef]
- Keeley, J.E. Future of California Floristics and Systematics: Wildfire Threats to the California Flora. Madrono 1995, 42, 175–179. [Google Scholar]
- Balch, J.K.; Bradley, B.A.; Abatzoglou, J.T.; Nagy, R.C.; Fusco, E.J.; Mahood, A.L. Human-started wildfires expand the fire niche across the United States. Proc. Natl. Acad. Sci. USA 2017, 114, 2946–2951. [Google Scholar] [CrossRef] [Green Version]
- Keeley, J.E.; Zedler, P.H. Large, high-intensity fire events in southern California shrublands: debunking the fine-grain age patch model. Ecol. Appl. 2009, 19, 69–94. [Google Scholar] [CrossRef]
- Santa Monica Mountains National Recreation Area. In Springs Fire Burned Area Emergency Response Plan; National Park Service, United States Department of the Interior: Washington, DC, USA, 2013.
- Allen, C.D.; Breshears, D.D.; McDowell, N.G. ESA CENTENNIAL PAPER On underestimation of global vulnerability to tree mortality and forest die-off from hotter drought in the Anthropocene. Ecosphere 2015, 6, 1–55. [Google Scholar] [CrossRef]
- Pellizzaro, G.; Duce, P.; Ventura, A.; Zara, P. Seasonal variations of live moisture content and ignitability in shrubs of the Mediterranean Basin. Int. J. Wildl. Fire 2007, 16, 633–641. [Google Scholar] [CrossRef]
- Emery, N.C.; D’Antonio, C.M.; Still, C.J. Fog and live fuel moisture in coastal California shrublands. Ecosphere 2018, 9, e02167. [Google Scholar] [CrossRef]
- Nolan, R.H.; Hedo, J.; Arteaga, C.; Sugai, T.; Resco de Dios, V. Physiological drought responses improve predictions of live fuel moisture dynamics in a Mediterranean forest. Agric. For. Meteorol. 2018, 263, 417–427. [Google Scholar] [CrossRef] [Green Version]
- Ruffault, J.; Martin-StPaul, N.; Pimont, F.; Dupuy, J.L. How well do meteorological drought indices predict live fuel moisture content (LFMC)? An assessment for wildfire research and operations in Mediterranean ecosystems. Agric. For. Meteorol. 2018, 262, 391–401. [Google Scholar] [CrossRef]
- Jolly, W.M.; Johnson, D.M. Pyro-Ecophysiology: Shifting the Paradigm of Live Wildland Fuel Research. Fire 2018, 1, 8. [Google Scholar] [CrossRef]
- Weise, D.R.; Hartford, R.A.; Mahaffey, L. Assessing live fuel moisture for fire management applications. In Proceedings of the Fire in Ecosystem Management: Shifting the Paradigm from Suppression to Prescription; Pruden, T.L., Brennan, L.A., Eds.; Tall Timbers Research Station: Tallahassee, FL, USA, 1998; pp. 49–55. [Google Scholar]
- Burgan, R.E. Estimating Live Fuel Moisture for the 1978 National Fire Danger Rating System; Intermountain Forest and Range Experiment Station: Ogden, UT, USA, 1979. [Google Scholar]
- Koide, R.T.; Robichaux, R.H.; Morse, S.R.; Smith, C.M. Plant water status, hydraulic resistance and capacitance. In Plant Physiological Ecology: Field Methods and Instrumentation; Pearcy, R.W., Ehleringer, J.R., Mooney, H., Rundel, P.W., Eds.; Chapman and Hall: New York, NY, USA, 1991; pp. 161–183. [Google Scholar]
- Sack, L.; Cowan, P.D.; Jaikumar, N.; Holbrook, N.M. The “hydrology” of leaves: Co-ordination of structure and function in temperate woody species. Plant. Cell Environ. 2003, 26, 1343–1356. [Google Scholar] [CrossRef]
- Sack, L.; Pasquet-Kok, J.; Contributors. Leaf Pressure-Volume Curve Parameters. Available online: http://prometheuswiki.org/tiki-index.php?page=Leaf+pressure-volume+curve+parameters (accessed on 30 June 2013).
- R Core Team. R: A Language and Environment for Statistical Computing; R Core Team: Vienna, Austria, 2016. [Google Scholar]
- Hollander, M.; Wolfe, D.A. Nonparametric Statistical Methods; Wiley: New York, NY, USA, 1973. [Google Scholar]
- Press, W.; BP, F.; Teukolsky, S.; Vetterling, W. Numerical Recipes in C; Cambridge University Press: Cambridge, UK, 1988. [Google Scholar]
- Royston, J.P. An Extension of Shapiro and Wilk’s W Test for Normality to Large Samples. Appl. Stat. 1982, 31, 115. [Google Scholar] [CrossRef]
- Pratt, R.B.; Jacobsen, A.L.; Mohla, R.; Ewers, F.W.; Davis, S.D. Linkage between water stress tolerance and life history type in seedlings of nine chaparral species (Rhamnaceae). J. Ecol. 2008, 96, 1252–1265. [Google Scholar] [CrossRef]
- Jacobsen, A.L.; Pratt, R.B.; Ewers, F.W.; Davis, S.D. Cavitation resistance among 26 chaparral species of southern California. Ecol. Monogr. 2007, 77, 99–115. [Google Scholar] [CrossRef]
- Malanson, G.P.; O’Leary, J.F. Post-fire regeneration strategies of Californian coastal sage shrubs. Oecologia 1982, 53, 355–358. [Google Scholar] [CrossRef]
- Bhaskar, R.; Ackerly, D.D. Ecological relevance of minimum seasonal water potentials. Physiol. Plant. 2006, 127, 353–359. [Google Scholar] [CrossRef]
- Martin-StPaul, N.; Delzon, S.; Cochard, H. Plant resistance to drought depends on timely stomatal closure. Ecol. Lett. 2017, 20, 1437–1447. [Google Scholar] [CrossRef]
- Green, L.R. Burning by Prescription in Chaparral; General Technical Report PSW-51; United States Department of Agriculture Forest Service: Berkeley, CA, USA, 1981.
- Pivovaroff, A.L.; Pasquini, S.C.; De Guzman, M.E.; Alstad, K.P.; Stemke, J.S.; Santiago, L.S. Multiple strategies for drought survival among woody plant species. Funct. Ecol. 2016, 30, 517–526. [Google Scholar] [CrossRef]
- Dennison, P.E.; Moritz, M.A. Critical live fuel moisture in chaparral ecosystems: a threshold for fire activity and its relationship to antecedent precipitation. Int. J. Wildl. Fire 2009, 18, 1021–1027. [Google Scholar] [CrossRef]
- Pimont, F.; Ruffault, J.; Martin-StPaul, N.; Dupuy, J.L. A cautionary note regarding the use of cumulative burnt areas for the determination of fire danger index breakpoints. Int. J. Wildl. Fire 2019, 254–258. [Google Scholar] [CrossRef]
- Emery, N.; Roth, K.; Pivovaroff, A.L. Flowering phenology associated with plant flammability. 2019; submitted. [Google Scholar]
- Jolly, W.M.; Hadlow, A.M.; Huguet, K. De-coupling seasonal changes in water content and dry matter to predict live conifer foliar moisture content. Int. J. Wildl. Fire 2014, 23, 480–489. [Google Scholar] [CrossRef] [Green Version]
- Qi, Y.; Dennison, P.E.; Jolly, W.M.; Kropp, R.C.; Brewer, S.C. Spectroscopic analysis of seasonal changes in live fuel moisture content and leaf dry mass. Remote Sens. Environ. 2014, 150, 198–206. [Google Scholar] [CrossRef] [Green Version]
- Tyree, M.T.; Yang, S. Water-storage capacity of Thuja, Tsuga and Acer stems measured by dehydration isotherms. Planta 1990, 182, 420–426. [Google Scholar] [CrossRef]
- Pivovaroff, A.L.; Burlett, R.; Lavigne, B.; Cochard, H.; Santiago, L.S.; Delzon, S. Testing the ‘microbubble effect’ using the Cavitron technique to measure xylem water extraction curves. AoB Plants 2016, 8, plw011. [Google Scholar] [CrossRef] [PubMed]
- Hanes, T.L. Succession after fire in the chaparral of southern California. Ecol. Monogr. 1971, 41, 27–52. [Google Scholar] [CrossRef]
- Philpot, C.W. Vegetative Features as Determinants of Fire Frequency and Intensity; United States Department of Agriculture Forest Service: Palo Alto, CA, USA, 1977.
- Countryman, C.M.; Philpot, C.W. Physical Characteristics of Chamise as a Wildland Fuel; Res. Pap. PSW-66; United States Department of Agriculture Forest Service: Berkeley, CA, USA, 1970.
- Dennison, P.E.; Brewer, S.D.; Arnold, J.D.; Moritz, M.A. Geophysical Research Letters. Geophys. Prospect. 2014, 41, 2928–2933. [Google Scholar]
| Species | Family | Leaf Phenology | Post-Fire Regeneration Type | LFMCIP | ΨTLP |
|---|---|---|---|---|---|
| Adenostoma fasciculatum | Rosaceae | Evergreen | Facultative sprouter 2 | 77% ± 5 | −3.40 ± 0.17 B |
| Arctostaphylos glandulosa | Ericaceae | Evergreen | Facultative sprouter 2 | 86% ± 4 | −2.51 ± 0.27 B,C |
| Ceanothus spinosus | Rhamnaceae | Evergreen | Facultative sprouter 1 | 121% ± 19 | −2.80 ± 0.29 B,C |
| Cercocarpus betuloides | Rosaceae | Evergreen | Obligate sprouter 2 | 71% ± 5 | −2.02 ± 0.09 A,C |
| Eriogonum fasciculatum | Polygonaceae | Semi-deciduous | Reseeder 3 | 67% ± 5 | −3.23 ± 0.20 B |
| Heteromeles arbutifolia | Rosaceae | Evergreen | Obligate sprouter 2 | 98% ± 14 | −2.70 ± 0.21 B,C |
| Malosma laurina | Anacardiaceae | Evergreen | Facultative sprouter 2 | 92% ± 10 | −2.07 ± 0.14 A,C |
| Quercus agrifolia | Fagaceae | Evergreen | Obligate sprouter 2 | 93% ± 12 | −2.61 ± 0.27 B,C |
| Quercus berberidifolia | Fagaceae | Evergreen | Obligate sprouter 2 | 81% ± 4 | −1.97 ± 0.22 A,C |
| Salvia leucophylla | Lamiaceae | Drought-deciduous | Reseeder 3 | 85% ± 4 | −1.47 ± 0.11 A |
| Salvia mellifera | Lamiaceae | Drought-deciduous | Reseeder 3 | 116% ± 8 | −2.08 ± 0.14 A,C |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pivovaroff, A.L.; Emery, N.; Sharifi, M.R.; Witter, M.; Keeley, J.E.; Rundel, P.W. The Effect of Ecophysiological Traits on Live Fuel Moisture Content. Fire 2019, 2, 28. https://doi.org/10.3390/fire2020028
Pivovaroff AL, Emery N, Sharifi MR, Witter M, Keeley JE, Rundel PW. The Effect of Ecophysiological Traits on Live Fuel Moisture Content. Fire. 2019; 2(2):28. https://doi.org/10.3390/fire2020028
Chicago/Turabian StylePivovaroff, Alexandria L., Nathan Emery, M. Rasoul Sharifi, Marti Witter, Jon E. Keeley, and Philip W. Rundel. 2019. "The Effect of Ecophysiological Traits on Live Fuel Moisture Content" Fire 2, no. 2: 28. https://doi.org/10.3390/fire2020028
APA StylePivovaroff, A. L., Emery, N., Sharifi, M. R., Witter, M., Keeley, J. E., & Rundel, P. W. (2019). The Effect of Ecophysiological Traits on Live Fuel Moisture Content. Fire, 2(2), 28. https://doi.org/10.3390/fire2020028






