Bridging the Divide: Integrating Animal and Plant Paradigms to Secure the Future of Biodiversity in Fire-Prone Ecosystems
Abstract
1. Introduction
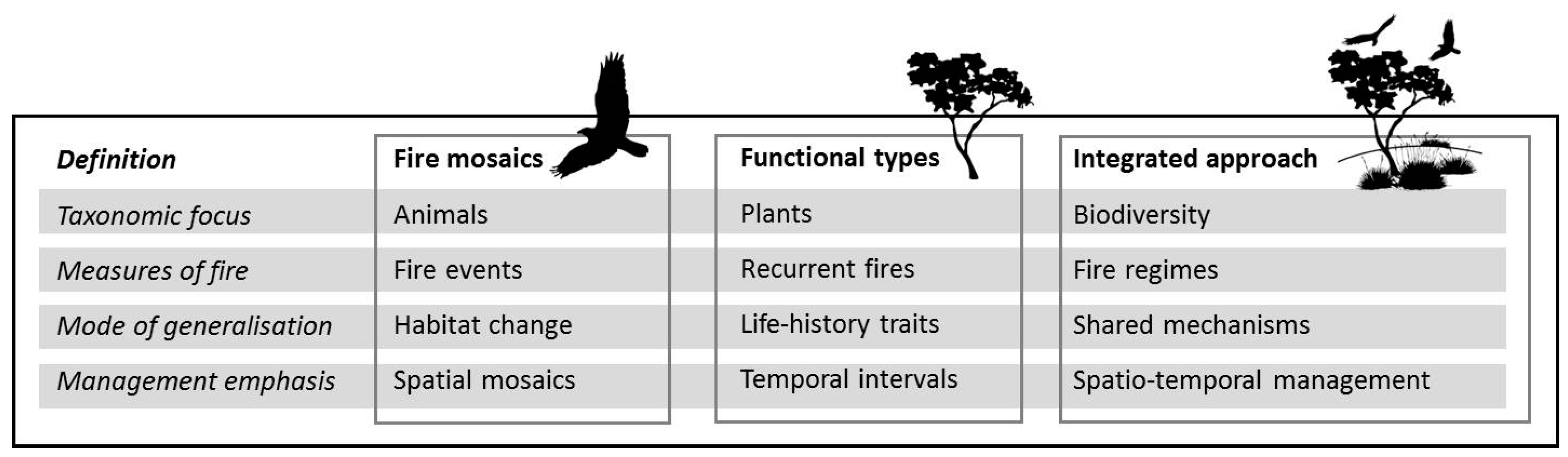
2. Animal and Plant Paradigms in Fire Ecology
2.1. Taxonomic Focus
2.2. Measures of Fire
2.3. Mode of Generalization
2.4. Management Emphasis
3. Towards a More Integrated Fire Ecology
3.1. Common Measures of Biodiversity
3.2. Multiple Components of Fire Regimes
3.3. Shared Mechanisms
3.4. Spatio-Temporal Fire Management
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Freeman, J.; Kobziar, L.; Rose, E.W.; Cropper, W. A critical evaluation of the historical fire regime concept in conservation. Conserv. Biol. 2017, 31, 976–985. [Google Scholar] [CrossRef] [PubMed]
- Bradstock, R.A.; Williams, R.J.; Gill, A.M. Future fire regimes of Australian ecosystems: new perspectives on enduring questions of management. In Flammable Australia: Fire Regimes, Biodiversity and Ecosystems in a Changing World; Bradstock, R.A., Gill, A.M., Williams, R.J., Eds.; CSIRO Publishing: Collingwood, Australia, 2012; pp. 307–324. ISBN 9780643104846. [Google Scholar]
- Kelly, L.T.; Brotons, L.; McCarthy, M.A. Putting pyrodiversity to work for animal conservation. Conserv. Biol. 2017, 31, 952–955. [Google Scholar] [CrossRef] [PubMed]
- Shedley, E.; Burrows, N.; Yates, C.J.; Coates, D.J. Using bioregional variation in fire history and fire response attributes as a basis for managing threatened flora in a fire-prone Mediterranean climate biodiversity hotspot. Aust. J. Bot. 2018, 66, 134–143. [Google Scholar] [CrossRef]
- Pausas, J.G.; Parr, C.L. Towards an understanding of the evolutionary role of fire in animals. Evol. Ecol. 2018, 1–13. [Google Scholar] [CrossRef]
- Keeley, J.E.; Bond, W.J.; Bradstock, R.A.; Pausas, J.G.; Rundel, P.W. Fire in Mediterranean Ecosystems: Ecology, Evolution and Management; Cambridge University Press: Cambridge, UK, 2012; ISBN 9780521824910. [Google Scholar]
- Puig-Gironès, R.; Clavero, M.; Pons, P. Importance of internal refuges and the external unburnt area in the recovery of rodent populations after wildfire. Int. J. Wildl. Fire 2018, 27, 425–436. [Google Scholar] [CrossRef]
- Gill, A.M.; McCarthy, M.A. Intervals between prescribed fires in Australia: What intrinsic variation should apply? Biol. Conserv. 1998, 85, 161–169. [Google Scholar] [CrossRef]
- Baker, W.L. Fire Ecology in Rocky Mountain Landscapes; Island Press: Washington, DC, USA, 2009; ISBN 9781597261821. [Google Scholar]
- Bond, W.J.; van Wilgen, B.W. Fire and Plants; Chapman and Hall: London, UK, 1996; ISBN 9789400914995. [Google Scholar]
- Foster, C.N.; Barton, P.S.; Robinson, N.M.; MacGregor, C.I.; Lindenmayer, D.B. Effects of a large wildfire on vegetation structure in a variable fire mosaic. Ecol. Appl. 2017, 27, 2369–2381. [Google Scholar] [CrossRef] [PubMed]
- Fox, B.J. Fire and mammalian secondary succession in an Australian coastal heath. Ecology 1982, 63, 1332–1341. [Google Scholar] [CrossRef]
- Smith, A.L. Successional changes in trophic interactions support a mechanistic model of post-fire population dynamics. Oecologia 2018, 186, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Noble, I.R.; Slatyer, R.O. The use of vital attributes to predict successional changes in plant communities subject to recurrent disturbances. Vegetatio 1980, 43, 5–21. [Google Scholar] [CrossRef]
- Keith, D.A. Functional traits: their roles in understanding and predicting biotic responses to fire regimes from individuals to landscapes. In Flammable Australia: Fire Regimes, Biodiversity and Ecosystems in a Changing World; Bradstock, R.A., Gill, A.M., Williams, R.J., Eds.; CSIRO Publishing: Collingwood, Australia, 2012; pp. 97–125. ISBN 9780643104846. [Google Scholar]
- Moritz, M.A.; Hurteau, M.D.; Suding, K.N.; D’Antonio, C.M. Bounded ranges of variation as a framework for future conservation and fire management. Ann. N. Y. Acad. Sci. 2013, 1286, 92–107. [Google Scholar] [CrossRef] [PubMed]
- Hessburg, P.F.; Spies, T.A.; Perry, D.A.; Skinner, C.N.; Taylor, A.H.; Brown, P.M.; Stephens, S.L.; Larson, A.J.; Churchill, D.J.; Povak, N.A.; et al. Tamm review: management of mixed-severity fire regime forests in Oregon, Washington, and Northern California. For. Ecol. Manag. 2016, 366, 221–250. [Google Scholar] [CrossRef]
- Kraaij, T.; Cowling, R.M.; van Wilgen, B.W.; Schutte-Vlok, A. Proteaceae juvenile periods and post-fire recruitment as indicators of minimum fire return interval in eastern coastal fynbos. Appl. Veg. Sci. 2013, 16, 84–94. [Google Scholar] [CrossRef]
- Menges, E.S. Integrating demography and fire management: An example from Florida scrub. Aust. J. Bot. 2007, 55, 261–272. [Google Scholar] [CrossRef]
- Pereira, H.M.; Ferrier, S.; Walters, M.; Geller, G.N.; Jongman, R.H.G.; Scholes, R.J.; Bruford, M.W.; Brummitt, N.; Butchart, S.H.M.; Cardoso, A.C.; et al. Essential biodiversity variables. Science 2013, 339, 277–278. [Google Scholar] [CrossRef] [PubMed]
- Einoder, L.D.; Southwell, D.M.; Gillespie, G.R.; Monfort, J.J.L.; Wintle, B.A. Optimising broad-scale monitoring for trend detection: review and re-design of a long-term program in northern Australia. In Monitoring Threatened Species and Ecological Communities; Legge, S., Lindenmayer, D., Robinson, N., Scheele, B., Southwell, D., Wintle, B., Eds.; CSIRO Publishing: Collingwood, Australia, 2018; pp. 271–280. ISBN 9781486307715. [Google Scholar]
- Thomas, F.M.; Vesk, P.A. Growth races in the Mallee: Height growth in woody plants examined with a trait-based model. Austral Ecol. 2017, 42, 790–800. [Google Scholar] [CrossRef]
- Talluto, M.V.; Benkman, C.W. Conflicting selection from fire and seed predation drives fine-scaled phenotypic variation in a widespread North American conifer. Proc. Natl. Acad. Sci. USA 2014, 111, 9543–9548. [Google Scholar] [CrossRef] [PubMed]
- Franklin, J.; Regan, H.M.; Syphard, A.D. Linking spatially explicit species distribution and population models to plan for the persistence of plant species under global change. Environ. Conserv. 2013, 41, 97–109. [Google Scholar] [CrossRef]
- Brotons, L.; De Cáceres, M.; Fall, A.; Fortin, M.J. Modeling bird species distribution change in fire prone Mediterranean landscapes: incorporating species dispersal and landscape dynamics. Ecography 2012, 35, 458–467. [Google Scholar] [CrossRef]
| Process | Benefit to Ecological Understanding and Fire Management | |
|---|---|---|
| Habitat development | Considering structural changes of plants provided new understanding of habitat provision over century-long time scales and showed that tolerable fire intervals based solely on plant occurrence were too short for animal conservation (Haslem et al. 2011) *. |  |
| Pollination | Nest location and floral resource use mediate pollinator responses to fire. Plant traits such as growth form, phenology and bud location influence flowering responses to fire. Pollinators most vulnerable to changing fire regimes are predicted to be those that nest above-ground and have one brood per year (Brown et al. 2017). |  |
| Decomposition | Fossorial mammals affected fire behavior via an increase in the rate of organic matter breakdown. Reintroducing fossorial mammal species to landscapes where they have previously been extinct offers a new way to modify fire regimes for the benefit of plant and animal conservation (Hayward et al. 2016). |  |
| Seed dispersal | Colonization of holm oak in burnt forest is mediated by acorn dispersal by Eurasian jays. Post-fire salvage logging reduced the strength of this key plant-animal interaction. Management policies of non-intervention after forest fire are likely to increase the resilience of the ecosystem (Castro et al. 2012). |  |
| Seed predation | Mediterranean gorse seeds are predated upon by the weevil Exapion fasciolatum. Mediterranean gorse experienced lower seed damage in burnt compared to unburnt areas. Plants can benefit from fire through disruption of antagonistic interactions with seed predators and this might be one mechanism promoting success of fire-adapted plants (García et al. 2016). |  |
| Grazing | In periods of low rainfall, grazing by kangaroos and feral herbivores following prescribed fire had a large effect on the survival of seedlings and resprouting plants. This indicates that conservation of plants and animals will benefit from fire management that considers how herbivore management influences a foundation species of semi-arid Australia (Giljohann et al. 2017). |  |
| Community assembly | Spatial and temporal variation in fires influences community assembly globally (Kelly & Brotons 2017), and feedback among fires, biodiversity and ecological processes are central to understanding community-level changes (Bowman et al. 2016). For example, fire in combination with experimental exclusion of seed-eating rodents shifted a desert shrubland to a low-diversity, invasive grassland. In areas where they remained, rodents created biotic resistance to invasive plants, with cascading effects on plant diversity (St. Clair et al. 2016). |  |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kelly, L.T.; Brotons, L.; Giljohann, K.M.; McCarthy, M.A.; Pausas, J.G.; Smith, A.L. Bridging the Divide: Integrating Animal and Plant Paradigms to Secure the Future of Biodiversity in Fire-Prone Ecosystems. Fire 2018, 1, 29. https://doi.org/10.3390/fire1020029
Kelly LT, Brotons L, Giljohann KM, McCarthy MA, Pausas JG, Smith AL. Bridging the Divide: Integrating Animal and Plant Paradigms to Secure the Future of Biodiversity in Fire-Prone Ecosystems. Fire. 2018; 1(2):29. https://doi.org/10.3390/fire1020029
Chicago/Turabian StyleKelly, Luke T., Lluís Brotons, Katherine M. Giljohann, Michael A. McCarthy, Juli G. Pausas, and Annabel L. Smith. 2018. "Bridging the Divide: Integrating Animal and Plant Paradigms to Secure the Future of Biodiversity in Fire-Prone Ecosystems" Fire 1, no. 2: 29. https://doi.org/10.3390/fire1020029
APA StyleKelly, L. T., Brotons, L., Giljohann, K. M., McCarthy, M. A., Pausas, J. G., & Smith, A. L. (2018). Bridging the Divide: Integrating Animal and Plant Paradigms to Secure the Future of Biodiversity in Fire-Prone Ecosystems. Fire, 1(2), 29. https://doi.org/10.3390/fire1020029





