Qualitative and Quantitative Coating Tests: A Comparison in Magnesia–Spinel Refractory Bricks
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
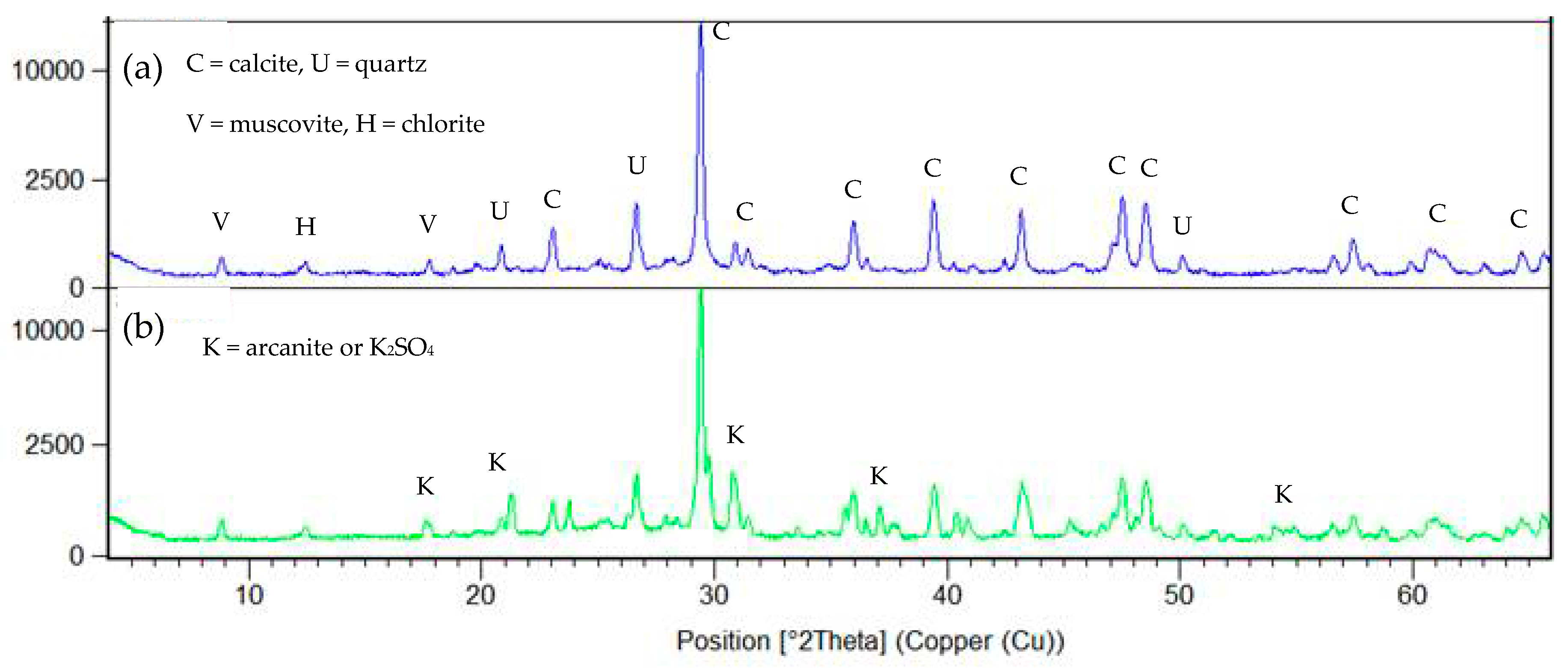
3.1. Characterization of Bricks and Cement Raw Meal
3.2. Qualitative Coating Test
3.3. Quantitative Coating Test
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Geith, M.; Jörg, S.; Krischanitz, R. Influence of flexibilisers on basic cement rotary kiln brick properties. In Proceedings of the UNITECR Congress, Santiago, Chile, 26–29 September 2017. [Google Scholar]
- Pacheco, G.R.C.; Fonseca, D.M. How to deal with alkali infiltration in mag-spinel bricks used in cement rotary kilns? In Proceedings of the Fifty-Fourth Annual Symposium on Refractories, St. Louis, MO, USA, 20–22 March 2018. [Google Scholar]
- Pacheco, G.R.C.; Fonseca, D.M.; Gonçalves, G.E. The role of zirconia in the alkali resistance of magnesia-spinel bricks. In Proceedings of the ALAFAR Congress, Medellín, Colombia, 30 September–3 October 2018. [Google Scholar]
- Pacheco, G.R.C.; Gonçalves, G.E. The cement industry. In Corrosion of Refractories: The Impacts of Corrosion, 1st ed.; Rigaud, M., Poirier, J., Eds.; Göller Verlag GmbH: Baden, Germany, 2018. [Google Scholar]
- Rigaud, M.; Guo, Z.; Palco, S. Coating formation on basic bricks in rotary cement kilns. In Proceedings of the ALAFAR Congress, Pucón, Chile, 2000. [Google Scholar]
- Guo, Z.; Palco, S.; Rigaud, M. Reaction Characteristics of Magnesia–Spinel Refractories with Cement Clinker. Int. J. Appl. Ceram. Technol. 2005, 2, 327–335. [Google Scholar] [CrossRef]
- Guo, Z.; Palco, S.; Rigaud, M. Bonding of Cement Clinker onto Doloma-Based Refractories. J. Am. Ceram. Soc. 2005, 88, 1481–1487. [Google Scholar] [CrossRef]
- Kosuka, H. New kind of chrome-free (MgO-CaO-ZrO2) bricks for burning zone of rotary cement kiln. In Proceedings of the UNITECR Congress, São Paulo, Brazil, 31 October–3 November 1993. [Google Scholar]
- Ghanbarnezhad, S. New development of spinel bonded chrome-free basic brick. J. Chem. Eng. Mater. Sci. 2013, 4, 7–12. [Google Scholar] [CrossRef][Green Version]
- Ohno, M. Magnesia-spinel brick with good coating adhesion and high resistance to corrosion and spalling for cement rotary kilns. In Proceedings of the UNITECR Congress, Santiago, Chile, 26–29 September 2017. [Google Scholar]
- Lin, X. Corrosion and adherence properties of cement clinker on porous periclase-spinel refractory aggregates with varying spinel content. Ceram. Int. 2017, 43, 4984–4991. [Google Scholar] [CrossRef]








| Classification | Visual Aspect | Description |
|---|---|---|
| Bad |  | No coating adherence |
| Good |  | Coating agent easily removed |
| Very good |  | Coating agent well adhered |
| Brick | A | B |
|---|---|---|
| BD (g/cm3) | 3.03 | 3.01 |
| AP (%) | 14.0 | 15.5 |
| CCS (MPa) | 85 | 65 |
| HMOR at 1200 °C-3 h (MPa) | 9.0 | 8.5 |
| Permeability (µm2) | 0.13 | 1.20 |
| Chemical analysis (%) | ||
| MgO | 86.5 | 85.0 |
| Al2O3 | 11.5 | 11.0 |
| CaO | 0.8 | 0.9 |
| SiO2 | 0.3 | 0.4 |
| Fe2O3 | 0.5 | 0.5 |
| ZrO2 | 0.0 | 1.8 |
| XRD | MgO MgO.Al2O3 β 2CaO.SiO2 | MgO MgO. Al2O3 Cubic ZrO2 CaZrO3 |
| Cement Raw Meal | As Received | Modified |
|---|---|---|
| P.C.E. (°C) | 1520 | 1395 |
| Particle Size Distribution (µm) after grinding | ||
| d98 | 124.0 | 125.0 |
| d90 | 78.2 | 81.7 |
| d75 | 44.7 | 46.0 |
| d50 | 17.5 | 18.3 |
| d25 | 5.6 | 5.7 |
| d10 | 1.9 | 1.8 |
| Chemical analysis (%) | ||
| CaO | 66.7 | 36.1 |
| SiO2 | 22.7 | 14.1 |
| Al2O3 | 4.8 | 3.4 |
| Fe2O3 | 2.4 | 1.4 |
| MgO | 1.5 | 1.3 |
| Na2O | 0.1 | 0.1 |
| K2O | 0.8 | 24.6 |
| SO3 | 0.5 | 18.6 |
| SR | 3.2 | 2.9 |
| XRD | Calcite Quartz Muscovite Chlorite | Calcite Arcanite Quartz Muscovite Chlorite |
| Brick | A | B |
|---|---|---|
| Classification after 1st, 2nd and 3rd Cycle | ||
| Raw meal as received | Bad, Bad, Bad | Very good, Very good, Very good |
| Modified raw meal | Bad, Good, Good | Bad, Good, Good |
| Brick | A | B |
|---|---|---|
| CMOR (MPa) | ||
| Raw meal as received | 0.0/0.0/0.0 | 1.5/0.9/1.3 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pacheco, G.; Gonçalves, G.E.; Lins, V. Qualitative and Quantitative Coating Tests: A Comparison in Magnesia–Spinel Refractory Bricks. Ceramics 2020, 3, 144-154. https://doi.org/10.3390/ceramics3010014
Pacheco G, Gonçalves GE, Lins V. Qualitative and Quantitative Coating Tests: A Comparison in Magnesia–Spinel Refractory Bricks. Ceramics. 2020; 3(1):144-154. https://doi.org/10.3390/ceramics3010014
Chicago/Turabian StylePacheco, Graziella, Geraldo Eduardo Gonçalves, and Vanessa Lins. 2020. "Qualitative and Quantitative Coating Tests: A Comparison in Magnesia–Spinel Refractory Bricks" Ceramics 3, no. 1: 144-154. https://doi.org/10.3390/ceramics3010014
APA StylePacheco, G., Gonçalves, G. E., & Lins, V. (2020). Qualitative and Quantitative Coating Tests: A Comparison in Magnesia–Spinel Refractory Bricks. Ceramics, 3(1), 144-154. https://doi.org/10.3390/ceramics3010014





