1. Introduction
Personal and industrial dosimeters provide a possibility to determine the radiation dose emitted by ionizing sources and radioactive materials. The technology is largely used by medical staff, atomic reactor maintainers, border control and scientists in areas where the absorbed dose must be controlled due to increased risk of developing harmful tumors and other injuries when working with ionizing radiation [
1,
2,
3].
The best known and widely used dosimeter material on alumina basis is carbon-doped aluminum oxide
(TLD-500) (band gap > 6 eV), which was proposed as a highly sensitive TL (thermoluminescent) and OSL (optically stimulated luminescence) material for personal dosimetry [
4]; it is now commercially available as a TL and OSL dosimeter (Landauer, Inc., Glenwood, IL, USA). Its main application is in OSL dosimetry for medicine, such as radiotherapy, radiodiagnostics, as well as heavy charged particle dosimetry [
5]. Although it has very attractive properties such as high sensitivity, stable signal at RT (room temperature) and a linear response over a wide range of doses (up to 50 Gy), this material shows disadvantages such as dependence of TL response on heating rate and overresponse to low-energy X-rays due to the effective atomic number—11.28. As a personal dosimeter, Al
2O
3:C is useful in the dose range 10 µGy–10 Gy but is not suitable at higher doses.
Efforts were undertaken to find other alumina-based dosimeter materials using different dopants. Some results were not as promising as others—several powdered doped
samples produced by solution combustion method were characterized for TL and OSL properties [
6]; however, they did not demonstrate attractive dosimeter properties. At the same time, Japanese researchers have found that doping with Cr enhances sensitivity and the fading characteristics of alumina, demonstrated in X-ray imaging, which makes this material suitable for two-dimensional TL slab dosimeters for robotic radiosurgery systems [
7]. As shown in [
8] doping of nanopowdered
with chromium makes it sensitive to higher γ-ray doses (100 Gy–20 kGy without saturation). Such properties might open novel application areas for
, such as radiation sterilization dosimetry and dealing with high doses during the irradiation of food products, seeds, medical instruments and agents.
Alumina has multiple phases, but in comparison to other alumina phases, the alpha phase is chemically inert and has a considerable hardness. It is also the most heat resistant phase, stable up to a temperature of 2051 °C; at higher temperatures it melts [
9,
10,
11,
12].
The most popular and widely used method for studying dosimetric properties is thermostimulated luminescence (TL). Ionizing radiation (in this study, X-rays were used) excites electrons from their ground level to the conduction band and this is followed by charge carriers geting trapped by the defects in the crystalline structure. Additional energy is necessary to release the charge carriers from the traps and to stimulate their recombination, thus causing the emission of a photon. The number of photons (registered luminescence intensity) correlates with the number of the trapped charge carriers, which in turn correlates with the absorbed radiation dose [
13].
Dosimeters must exhibit a linear correlation between absorbed radiation dose and TL response. In this article, we study Cr-doped alpha alumina as a potential material for a high dose dosimeter. The search for the optimal concentration during the sol-gel synthesis of Al2O3 and study of the dosimetric properties of the acquired Cr-doped alumina is presented. Comparison of luminescence intensity is done between microceramic powder and a ruby single crystal.
2. Materials and Methods
2.1. Materials
Aluminum nitrate nanohydrate , purity 99.6%; VWR Prolabo Chemicals, Singapore); and chromium (III) oxide were used as starting materials. Citric acid (, purity 99.5%) and ethylene glycol (, purity 99%) were used as chelating and polymerizable agents, and both were purchased from Sigma Aldrich. Nitric acid (, assay 65%) (Sigma Aldrich, Saint Louis, MO, USA) was used to dissolve . Analytical grade chemicals were used without any further purification.
2.2. Synthesis of Non-Doped and Cr-Doped Al2O3 Microceramic Powder
Pure and Cr-doped alumina samples were synthesized using the sol-gel polymerized complex method, which was also used in our previous work [
14]. Cr-doped alumina samples were prepared with varying concentrations (0.1, 0.2, 0.3, 0.35, 0.4, 0.7, 1 and 1.5 wt%) of the
. The molar ratio of metal ions, citric acid, and ethylene glycol was 1:1:4, respectively. Also, an appropriate amount of deionized water was added so that the molar concentration of all metals ions in the solution would be 0.2 M. When the necessary gel consistency was obtained, it was heated in an open oven at 400 °C for 2 h for nitric oxide elimination, and as a result a black powder was obtained. After synthesis, the obtained samples were calcined at 1400 °C for 4 h. In the end, white and pink powders were obtained.
2.3. Characterization Techniques
The crystalline phases of pure and doped samples after heat treatment were characterized by X-ray powder diffraction (XRD) using a PANalytical X’Pert Pro diffractometer. Cu Kα radiation (1.5418 Å) was used by setting the cathode voltage to 45 kV and current to 40 mA. Morphology and chemical composition of the pure and doped samples were characterized by scanning electron microscopy (SEM) using Tescan Lyra equipped with an energy dispersive X-ray (EDX) spectrometer operated at 15 kV. Before the examination, the samples were coated with a gold layer.
XRL (X-ray luminescence) and TL (thermostimulated luminescence) spectra were acquired using an ANDOR SR-303I-B monochromator with a 150 L/mm diffraction grating and a blaze wavelength of 800 nm. The monochromator was coupled with an ANDOR DU401A-BV CCD camera and an ANDOR SOLIS data logger. To avoid oxygen ionizing in the measurement chamber and the heater oxidizing during measurements, a vacuum pump ILMVAC GmbH CD 2015 was used, which provided a vacuum greater than Torr.
XRF (X-ray fluorescence) measurements were performed using an EAGLE Probe III X-ray fluorescence spectrometer. The X-ray lamp was set to a current of 10 A and a voltage of 40 kV. Rays were focused on a circular area with a diameter of 200 µm. Since dopant concentrations were small, the XRF spectrometer was operated at the detection limit. This meant that the measurement errors were rather large, as was determined by repeatedly measuring the same sample in different surface locations and calculating the average value and standard deviation. Measurement error was determined for one sample and the authors assume that it will stay consistent with all other samples in the series.
3. Results
3.1. XRD Measurements
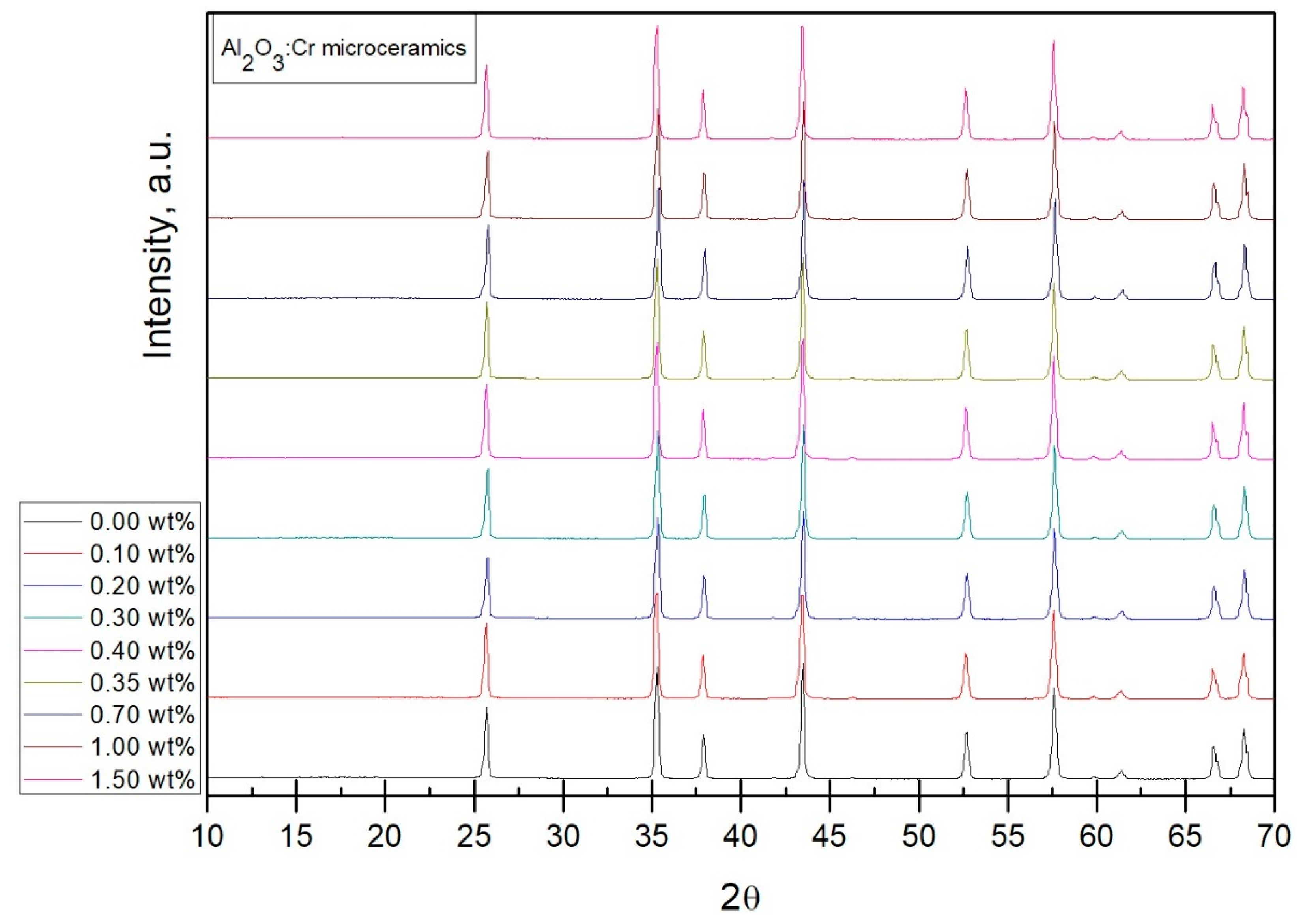
Results of the X-ray diffraction measurements displayed in
Figure 1 show that all samples contain the same alpha alumina phase [
15,
16]. Since no changes in the X-ray diffraction pattern are noticeable, one can assume that
at the studied concentrations does not cause any major disruptions in the crystalline lattice of the host matrix.
3.2. Composition Analysis
Comparisons between the initial and post-synthesis
concentrations were made using an EDX and XRF and results can be observed in
Figure 2. The main conclusions that can be drawn are that although the detected concentration grows by increasing the initial concentration, the efficiency at which
incorporates into the crystal lattice is not straightforward and has to be further studied. The obtained average EDX readouts are closer to the initially added Cr concentrations than the XRF results. This might be explained by the technological differences in measurement devices. Since the area studied in XRF measurements is larger, it might lead to erroneous results due to the sample inhomogeneity.
3.3. SEM Measurements
The results of the SEM measurements are shown in
Figure 3. From these results, we propose that a 4 h long heat treatment at 1400 °C is enough for nanoparticles to start joining together, forming porous microceramics. These results suggest that a lower temperature and shorter heat treatment duration could create nanoparticles if a way was found to eliminate all organic matter traces after synthesis with such conditions.
3.4. Luminescence Measurements
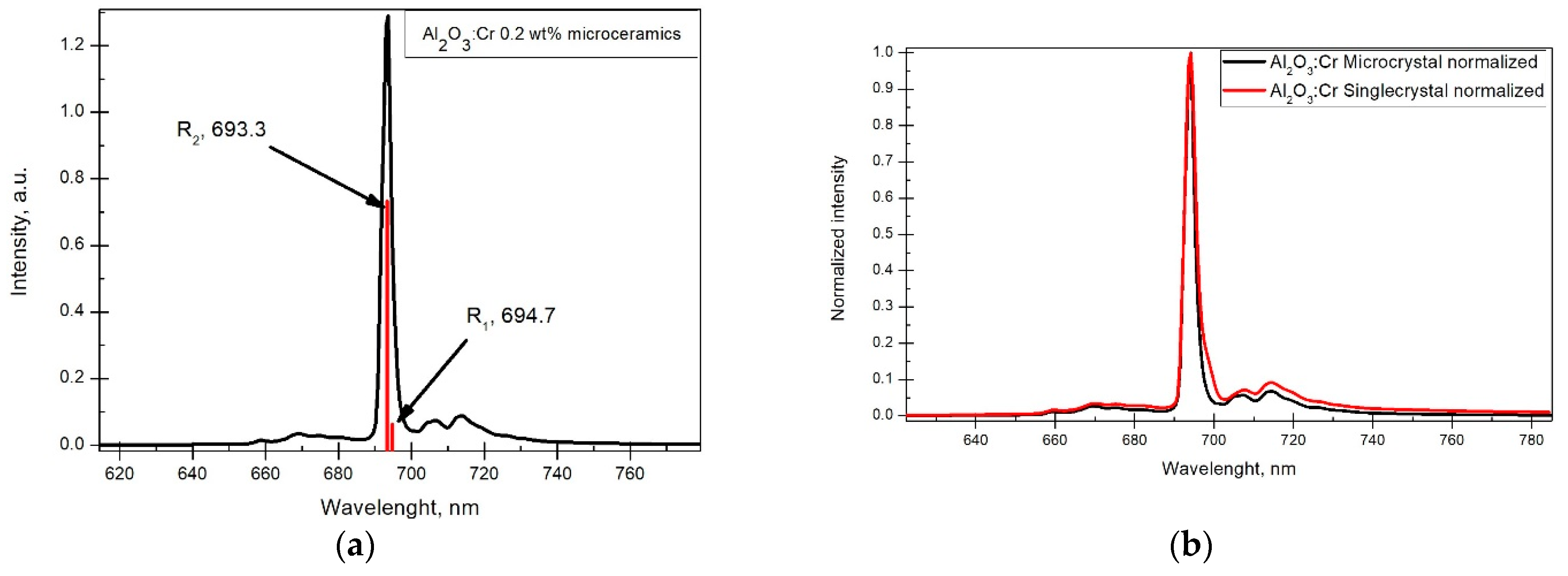
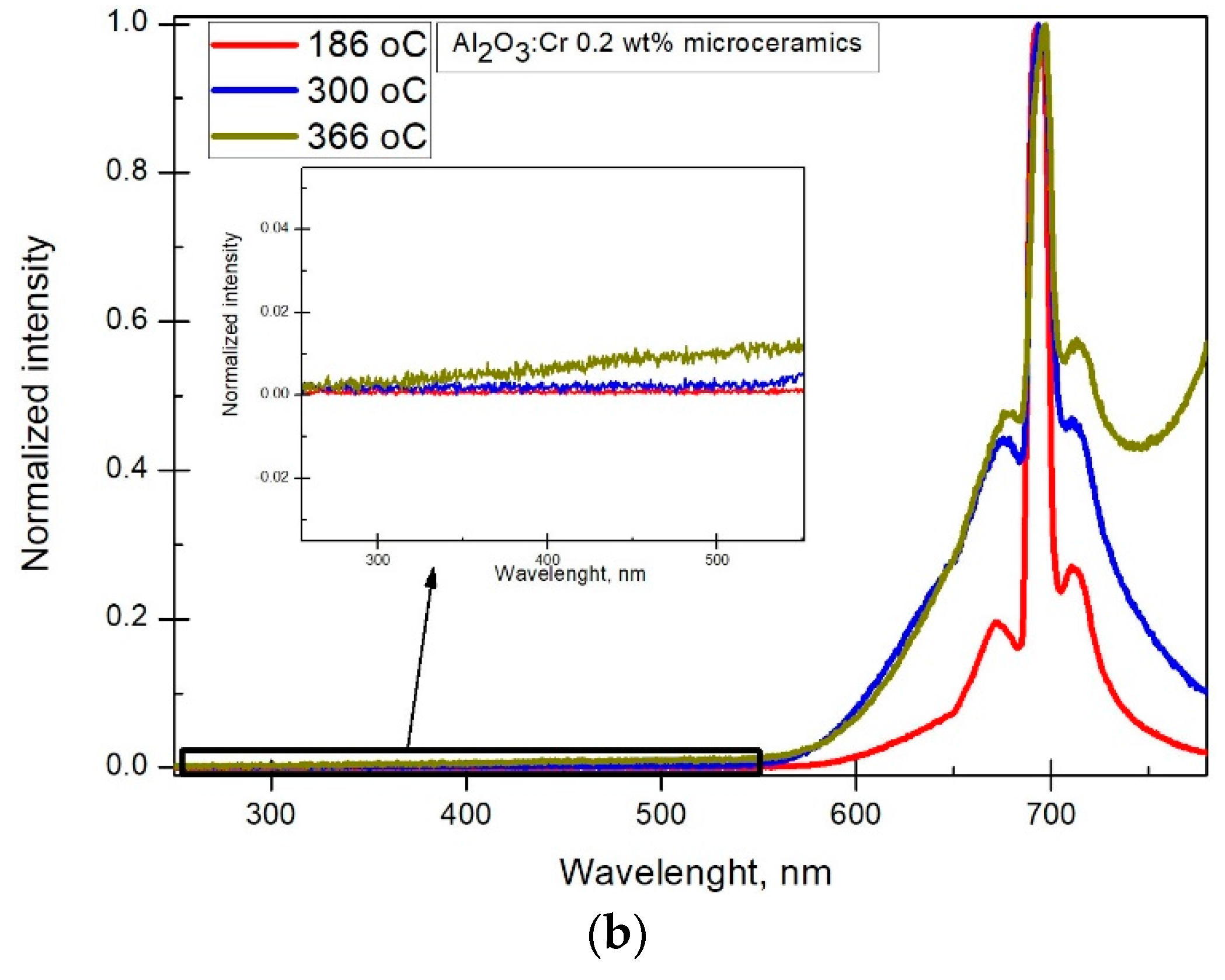
The X-ray luminescence spectra were registered for all microceramics samples. A ruby single crystal was used as a reference. The results can be seen in
Figure 4. Cr
3+ specific lines
,
and N were observed with the respective wavelengths being 694.7, 693.3 and 704 nm [
17].
and
arise from electron excitation from
to
and
state, where energies are about 2.2 and 3.0 eV above the ground state. Electrons relax to
state by generating phonons and afterward return to
by emitting photons with wavelengths 694.7 and 693.3 nm. The weaker N line around 704 nm arises from the interaction of two closely located Cr ions, and the broader emission line around 715 nm is assigned to Cr ion cluster interaction [
17].
3.5. Concentration Optimization
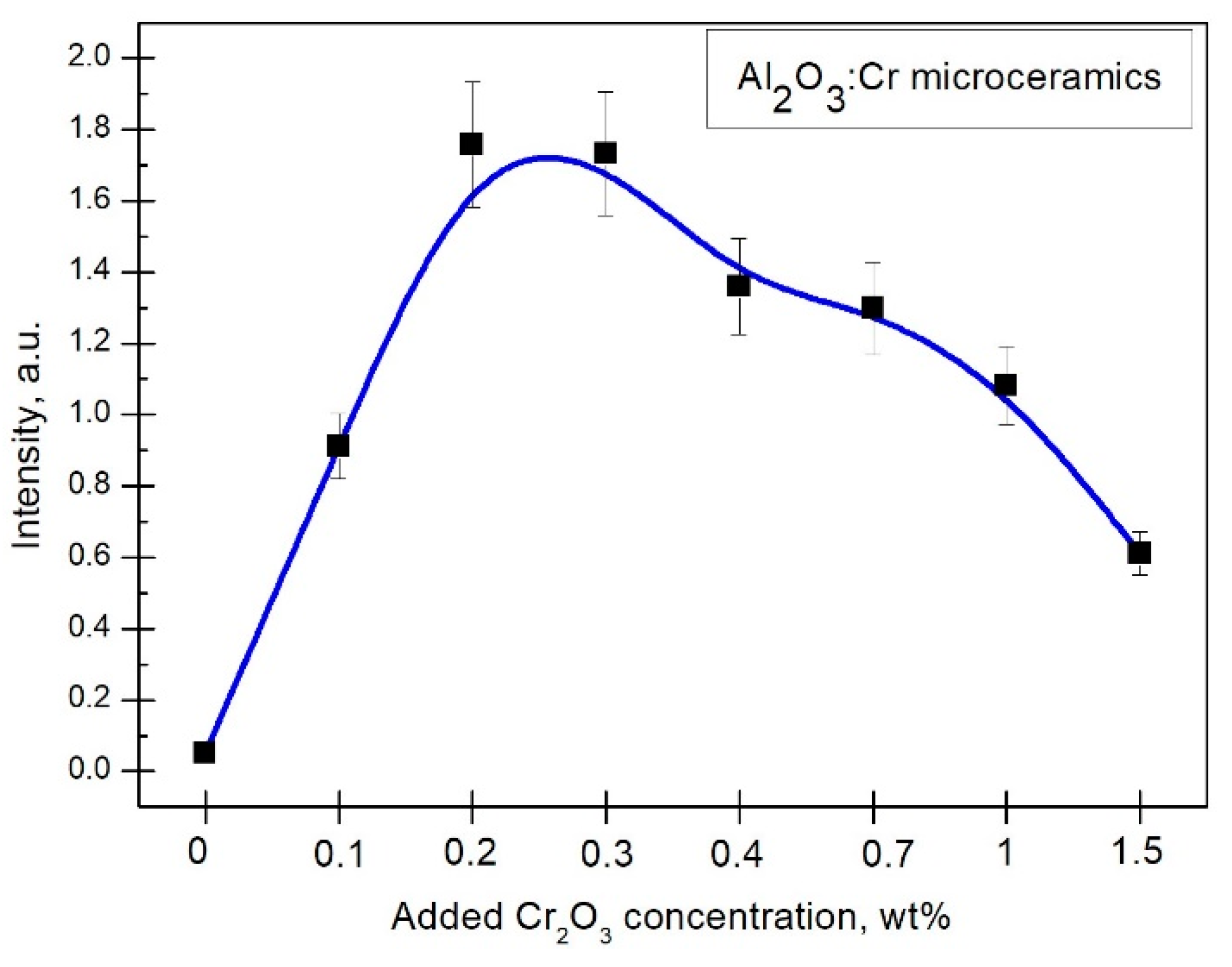
By analyzing the relation between the added
concentration and the luminescence intensity (
Figure 5), a conclusion was drawn that the optimal Cr concentration could be found in the range of 0.2 to 0.3 wt%. To get a more accurate optimal
concentration for dosimetric purposes, the integral intensity was determined. Error bars in
Figure 5 represent the measurement uncertainty that arises from our optical systems, which is estimated to be 10% of the total intensity.
3.6. TL Measurments
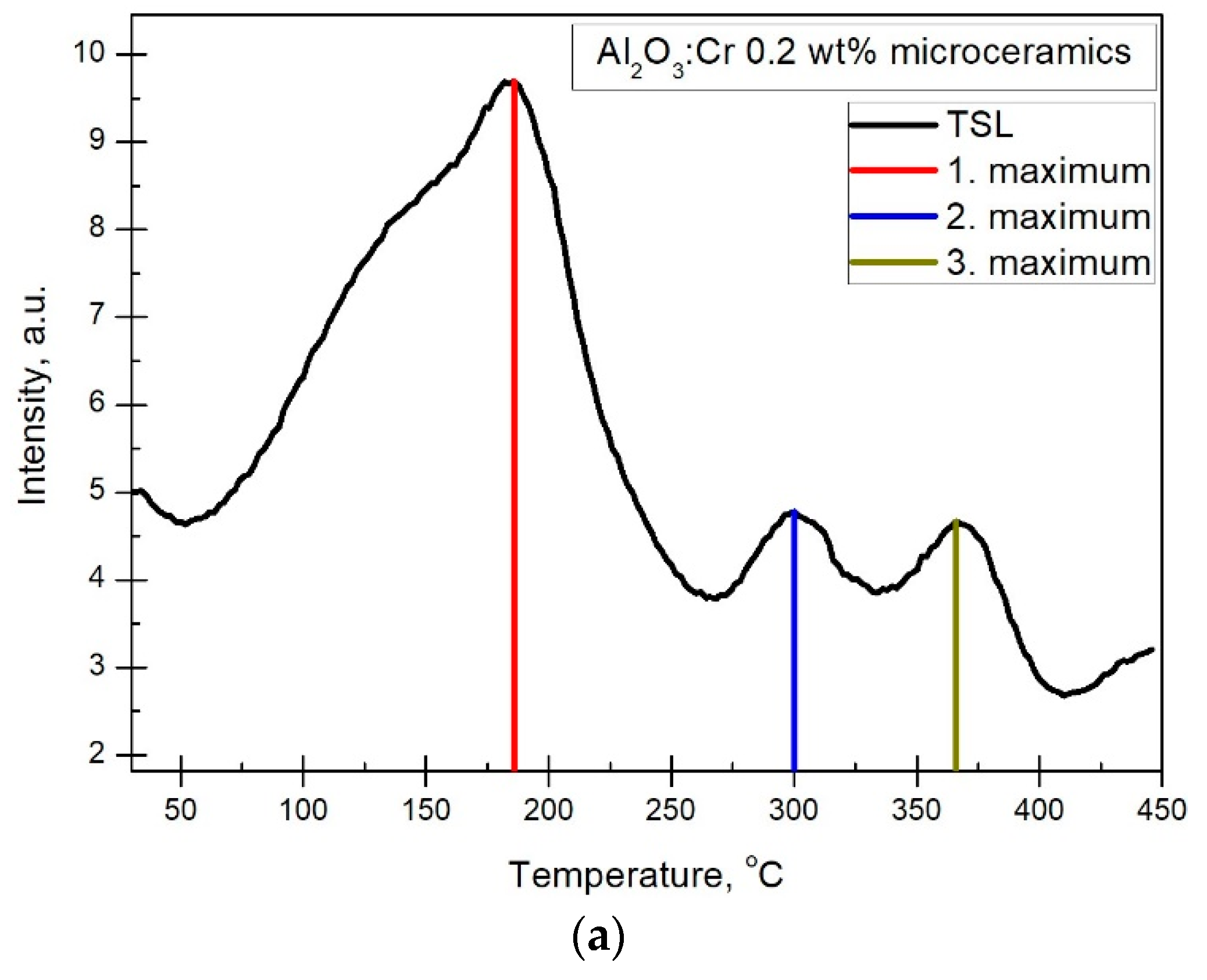
TL measurements were conducted from 20 °C to 450 °C. To determine the optimal concentration, a constant irradiation time of 30 min and a 10 min delay time after irradiation was used to avoid registering sample afterglow. An example is shown in
Figure 6. As the experimental setup allows the measuring of both the temperature and wavelength dependence of luminescence simultaneously, the analysis of both TL and spectral distribution in TL peaks is presented. The spectra for the main TL peaks are presented in
Figure 6b. The glow peak around 120 °C was not studied due to difficulties determining the exact maximum of the peak. All observed glow peaks arise from charge traps with different depths. Spectra in
Figure 6b show that the main contributors to thermostimulated luminescence are Cr
3+ ions. Two broad lines on each side of the previously mentioned R lines (
Figure 4) were observed, and their intensities relative to the R line increased at higher temperatures. In other studies, these are mentioned as vibrational sidebands of R lines [
18].
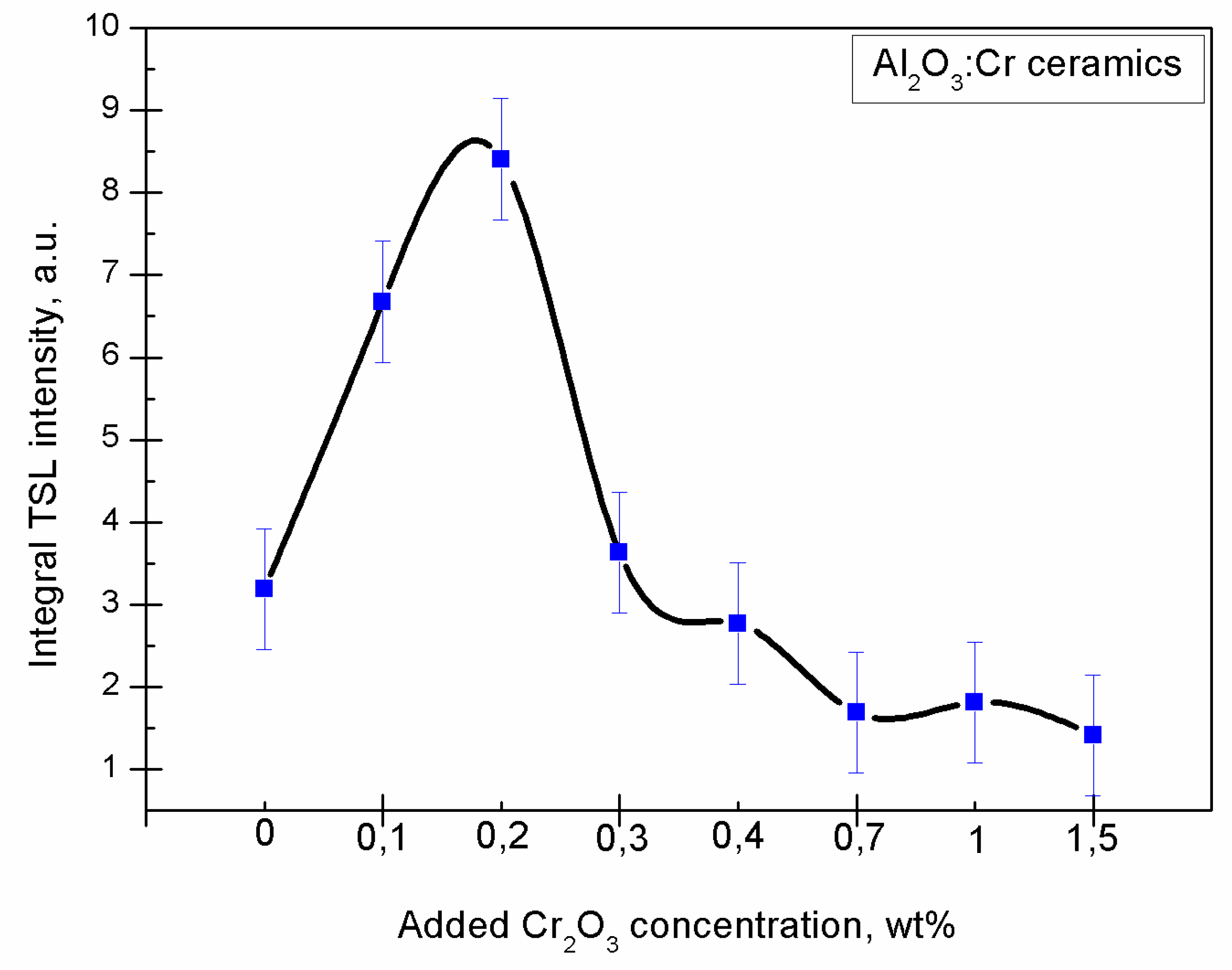
The relation between integral TL intensity and added
concentration can be seen in
Figure 7. A conclusion can be made that the optimal
concentration is around 0.2 wt%. Error bars represent standard deviation between 3 consecutive measurements and the black line connecting points serves for visual clarity only.
Both rise and fall in TL intensity can be explained by an increase in
concentration. A rise could be due to an increase in luminescence or recombination centers in the crystal lattice. The steep fall could be explained by an effect known as concentration quenching, where photon absorption rises more quickly than emission as the
concentration increases [
19].
3.7. Dosimetric Properties
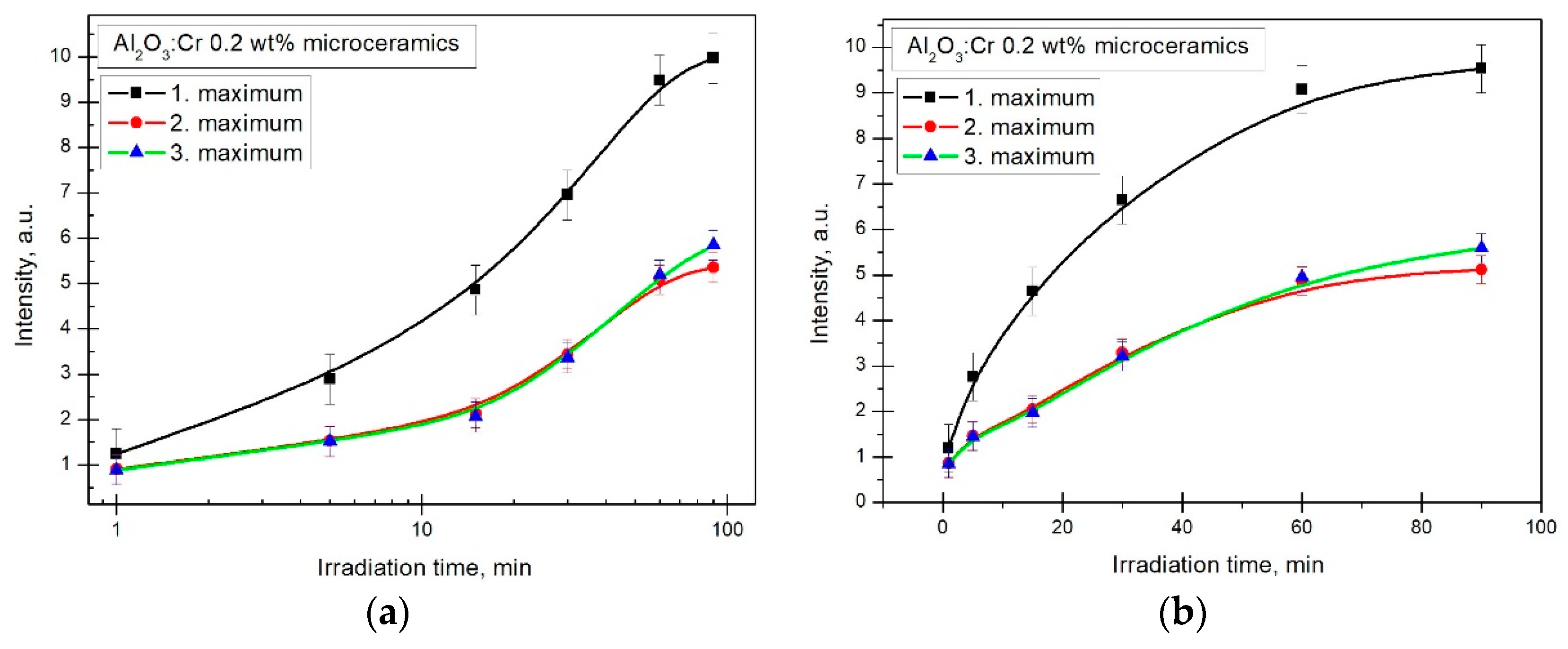
Based on the previous conclusions, the sample with 0.2 wt%
was investigated further. TL measurements were repeated in the same conditions: temperature range and the delay time before irradiation and actual measurements were held constant. The only varying parameter was the irradiation time. Results for all three TSL peaks can be seen in
Figure 8. To create the full dosimetric curve, six irradiation times were chosen: 1; 5; 15; 30; 60 and 90 min respectively [
20,
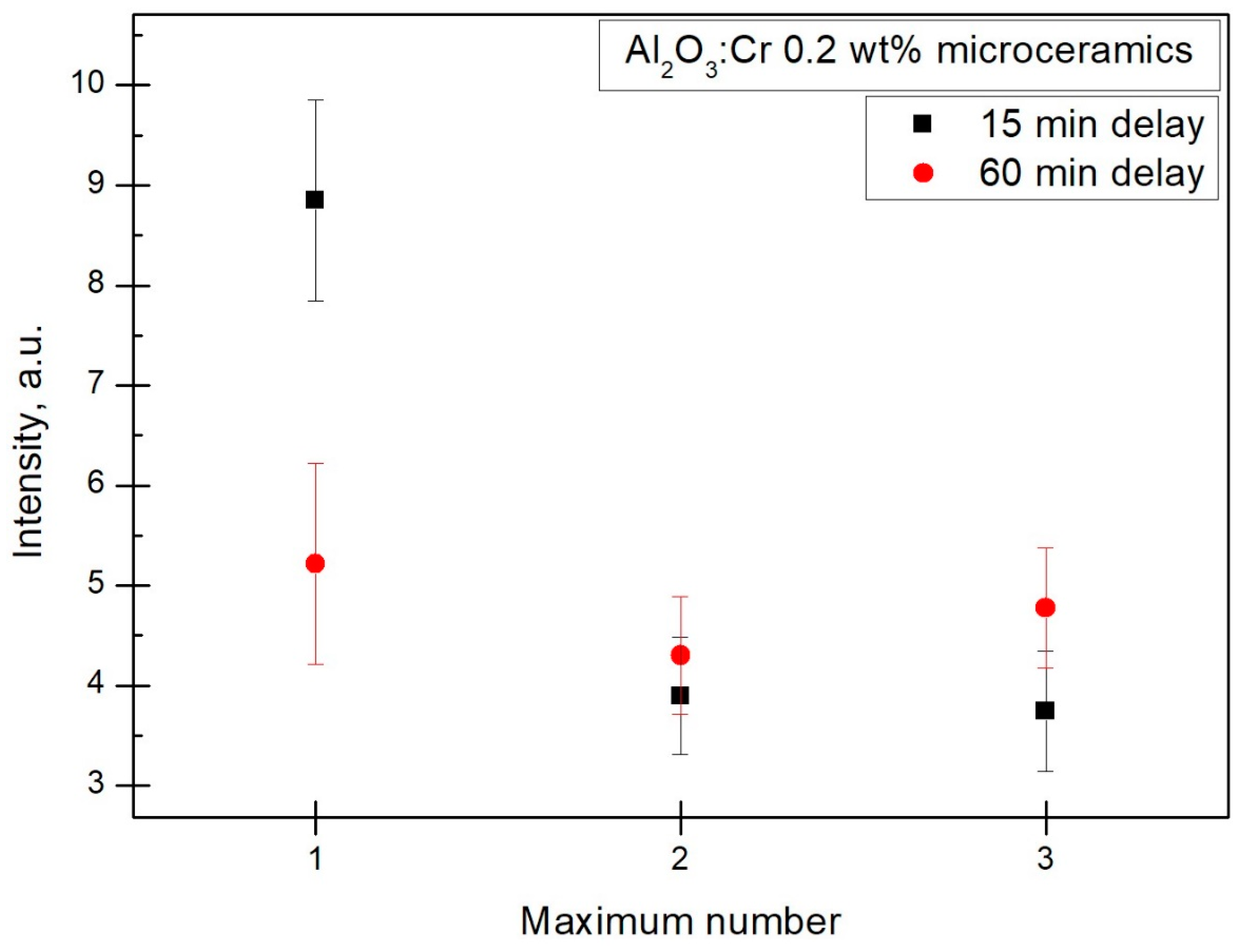
21]. To determine which maximum was the best for dosimetric applications, a separate measurement was made to check the impact of TL fading; this sample was irradiated for 15 min, and then a 60 min pause was taken before the TL readout. Results in
Figure 9 show that the first maximum cannot be reliably used for intended purposes, because it demonstrates essential fading. The 10% measurement uncertainty caused the second and third TSL peaks to appear to have a higher readout after the 60 min delay than it had after 15 min.
Relying on
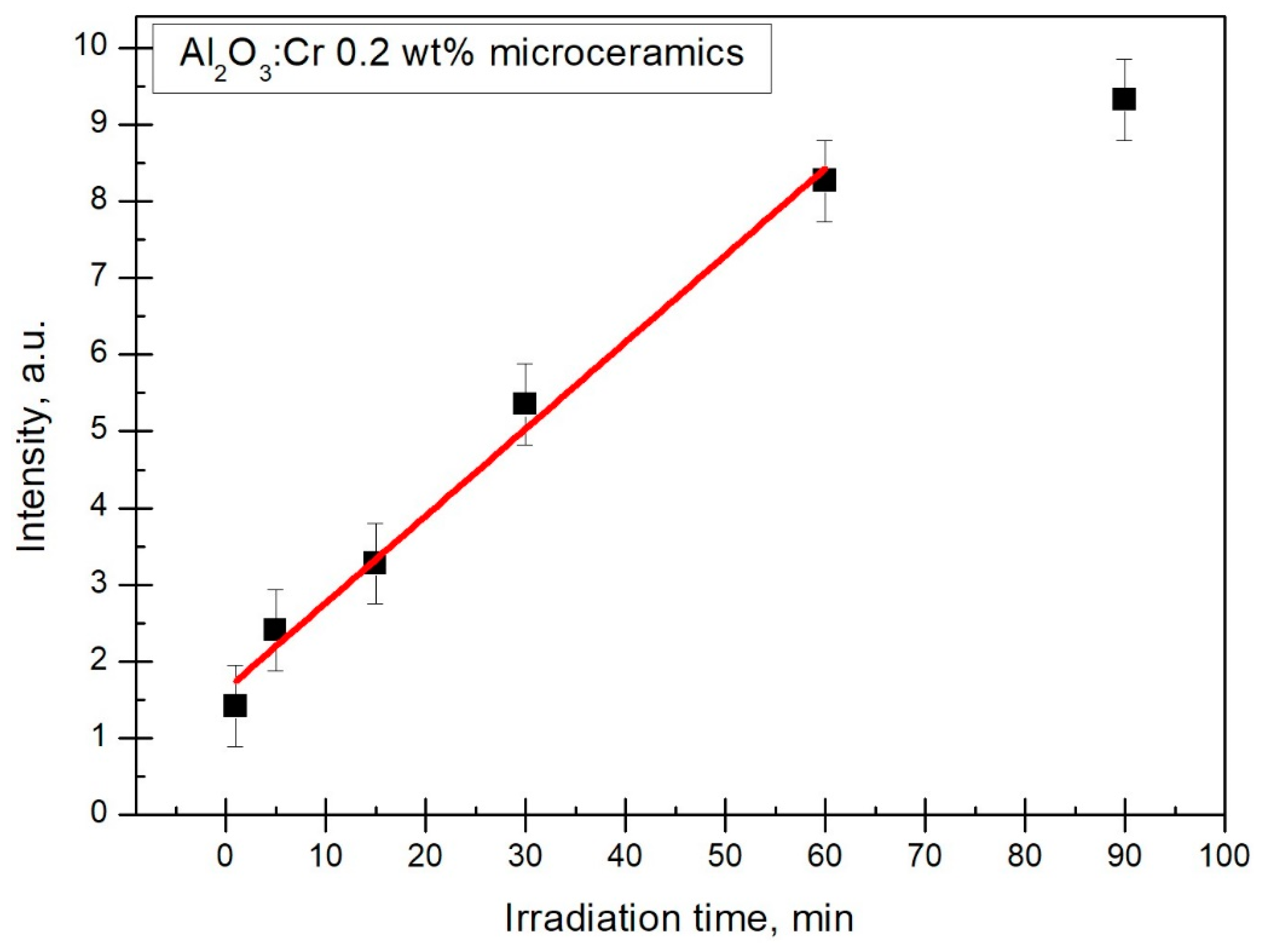
Figure 9, the linearity was determined only for the third peak. However, it is highly likely that the second peak would have the same linearity as the points almost overlap.
Figure 10 shows that the third peak displays a linear response to irradiation time from 1 to 60 min. Points matched in a line with a reduced Chi–Sqr value of 0.34.
4. Discussion
Comparison with widely used carbon-doped alumina is rather difficult. For theoretical purposes, an estimation was made using RadPro Calculator 3.26. Inserting our measurement parameters yielded a dose rate of about 99 Gy min
−1. That means that
-doped alumina has a linear response from around 100 Gy to 5 kGy. Literature shows that carbon-doped alumina has a linear range from
to 1 Gy [
4]. This means that
-doped alumina could be applied as a high dose dosimeter, which could then be used in reactors or border control as an onboard equipment regulator.
For optimal dose response and sensitivity, the whole TL spectrum should be integrated when determining the ionizing radiation dose absorbed by the material, instead of only individual peak intensities. Integral value would provide maximal information without unnecessary loss of information, but a correction must be determined which considers fading rate, temperature at which the sample was stored and the amount of light the sample absorbed while in use as a passive dosimeter.
To determine the dose response, the irradiation time was varied. Longer irradiation leads to a higher absorbed dose by the sample, but also to the higher absolute impact of fading of the TL response. Divergence from linearity could be caused by fading under irradiation which was not taken into account during linearity analysis. Material could possess a wider linear response dose range than the one reported in this study, which could be used as a basis for further exploration of doped .
5. Conclusions
Study of dosimetric properties of -doped alumina microceramic shows that its TL response displayed a promising linearity in the performed measurement X-ray dose range, and the estimations imply that this material is suitable for detection of rather high doses of ionizing radiation (100 Gy to 5 kGy). The physical and chemical properties of the material, as well as its manufacturing cost, make it a good candidate for potential use as a dosimeter.
In future research, time dependence should also be studied over a considerably longer period. Single crystalline -doped alumina should also be explored in more detail. Cr-doped microceramic powder showed some features that are promising for dosimetry applications, however, the noticeable fading raises several concerns.
Author Contributions
E.E. carried out most of the measurements, performed data analysis, and is the main contributor to this article. A.Z. assisted in measurements and data interpretation. I.B. synthesized the samples and performed XRD measurements. K.L. and K.A. assisted in data analysis and proof-reading of the paper. K.S. and L.T. assisted in writing the paper and supervised the direction for the research.
Funding
Financial support provided by Scientific Research of Luminescence Mechanisms and Dosimeter Properties in Prospective Nitrides and Oxides Using TL and OSL Methods LZP FLPP Nr. LZP-2018/1-0361 realized at the Institute of Solid State Physics, University of Latvia is greatly acknowledged.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Einstein, A.J.; Henzlova, M.J.; Rajagopalan, S. Estimating Risk of Cancer Associated with Radiation Exposure From 64-Slice. JAMA 2007, 298, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Cardis, E.; Gilbert, E.S.; Carpenter, L.; Howe, G.; Kato, I.; Armstrong, B.K.; Beral, V.; Cowper, G.; Douglas, A.; Fix, J.; et al. Effects of Low Doses and Low Dose Rates of External Ionizing Radiation: Cancer Mortality among Nuclear Industry Workers in Three Countries. Radiat. Res. 2006, 142, 117–132. [Google Scholar] [CrossRef]
- Duncan, J.R.; Lieber, M.R.; Adachi, N.; Wahl, R.L. DNA Repair After Exposure to Ionizing Radiation Is Not Error-Free. J. Nucl. Med. 2018, 59, 348. [Google Scholar] [CrossRef] [PubMed]
- Akselrod, M.S.; McKeever, S.W.S. A Radiation Dosimetry Method Using Pulsed Optically Stimulated Luminescence. Radiat. Prot. Dosim. 1999, 81, 167–176. [Google Scholar] [CrossRef]
- Yukihara, E.G.; McKeever, S.W.S. Optically stimulated luminescence (OSL) dosimetry in medicine. Phys. Med. Biol. 2008, 53, 351–379. [Google Scholar] [CrossRef] [PubMed]
- Yukihara, E.G.; Milliken, E.D.; Oliveira, L.C.; Orante-Barrón, V.R.; Jacobsohn, L.G.; Blair, M.W. Systematic development of new thermoluminescence and optically stimulated luminescence materials. J. Lumin. 2013, 133, 203–210. [Google Scholar] [CrossRef]
- Yanagisawa, S.; Shinsho, K.; Inoue, M.; Koba, Y.; Matsumoto, K.; Ushiba, H.; Andoh, T. Applicability of two-dimensional thermoluminescence slab dosimeter based on Al2O3: Cr for the quality assurance of robotic radiosurgery. J. Lumin. 2018, 30, 1591–1598. [Google Scholar]
- Akselrod, V.I.; Kortov, M.S.; Kravetsky, V.S.; Gotlib, D.J. Highly Sensitive Thermoluminescent Anion-Defective Alpha-Al2O3: C Single Crystal Detectors. Radiat. Prot. Dosim. 1990, 32, 15–20. [Google Scholar]
- Davis, K. Material Review: Alumina. Sch. Dr. Stud. Eur. Union J. 2010, 3, 109–115. [Google Scholar]
- Cunha, G.D.; Romão, L.P.C.; Macedo, Z.S. Production of alpha-alumina nanoparticles using aquatic humic substances. Powder Technol. 2014, 254, 344–351. [Google Scholar] [CrossRef]
- Lamouri, S.; Hamidouche, M.; Bouaouadja, N.; Belhouchet, H.; Garnier, V.; Fantozzi, G.; Trelkat, J.F. Control of the γ-alumina to α-alumina phase transformation for an optimized alumina densification. Boletín Soc. Española Cerámica Vidr. 2017, 56, 47–54. [Google Scholar] [CrossRef]
- Chandran, C.V.; Kirschhock, C.E.A.; Radhakrishnan, S.; Taulelle, F.; Martens, J.A.; Breynaert, E. Alumina: Discriminative analysis using 3D correlation of solid-state NMR parameters. Chem. Soc. Rev. 2019, 48, 134–156. [Google Scholar] [CrossRef] [PubMed]
- McKeever, S.W.S. Thermoluminescence of Solids; Cambridge University Press: Cambridge, UK, 1983. [Google Scholar]
- Laganovska, K.; Bite, I.; Zolotarjovs, A.; Smits, K.; Ofelt, J. Niobium enhanced europium ion luminescence in hafnia nanocrystals. J. Lumin. 2018, 203, 358–363. [Google Scholar] [CrossRef]
- Sadabadi, H.; Aftabtalab, A.; Zafarian, S.; Shaker, S.; Ahmadipour, M.; Rao, K.V. High purity Alpha Alumina nanoparticle: Synthesis and characterization. Int. J. Sci. Eng. Res. 2013, 4, 1593–1596. [Google Scholar]
- Chargui, F.; Hamidouche, M.; Belhouchet, H.; Jorand, Y.; Doufnoune, R.; Fantozzi, G. Mullite fabrication from natural kaolin and aluminium slag. Boletín Soc. Española Cerámica Vidr. 2018, 57, 169–177. [Google Scholar] [CrossRef]
- Bhardwaj, D.M.; Jain, D.C.; Rao, K.V.R.; Kumar, R.; Singh, F.; Gupta, R.P. Photoluminescence and atomic force microscopic studies on pre- and post-irradiated ruby with Ni6+ ion. Nucl. Instrum. Methods Phys. Res. Sect. B Beam Interact. Mater. At. 2004, 222, 533–537. [Google Scholar] [CrossRef]
- Trinkler, L.; Berzina, B.; Jakimovica, D.; Grabis, J.; Steins, I. UV-light induced luminescence processes in Al2O3 bulk and nanosize powders. Opt. Mater. 2010, 32, 789–795. [Google Scholar] [CrossRef]
- Harris, D.C. Quantitative Chemical Analysis, 9th ed.; W. H. Freeman: New York, NY, USA, 2015; Volume 407. [Google Scholar]
- Di Veroli, G.Y.; Fornari, C.; Goldlust, I.; Mills, G.; Koh, S.B.; Bramhall, J.L.; Richards, F.M.; Jodrell, D.I. An automated fitting procedure and software for dose-response curves with multiphasic features. Sci. Rep. 2015, 5, 14701. [Google Scholar] [CrossRef] [PubMed]
- Kalita, J.M.; Chithambo, M.L. The effect of annealing and beta irradiation on thermoluminescence spectra of α-Al2O3: C, Mg. J. Lumin. 2018, 196, 195–200. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).