A Brief Review on Gender Identification with Electrocardiography Data
Abstract
1. Introduction
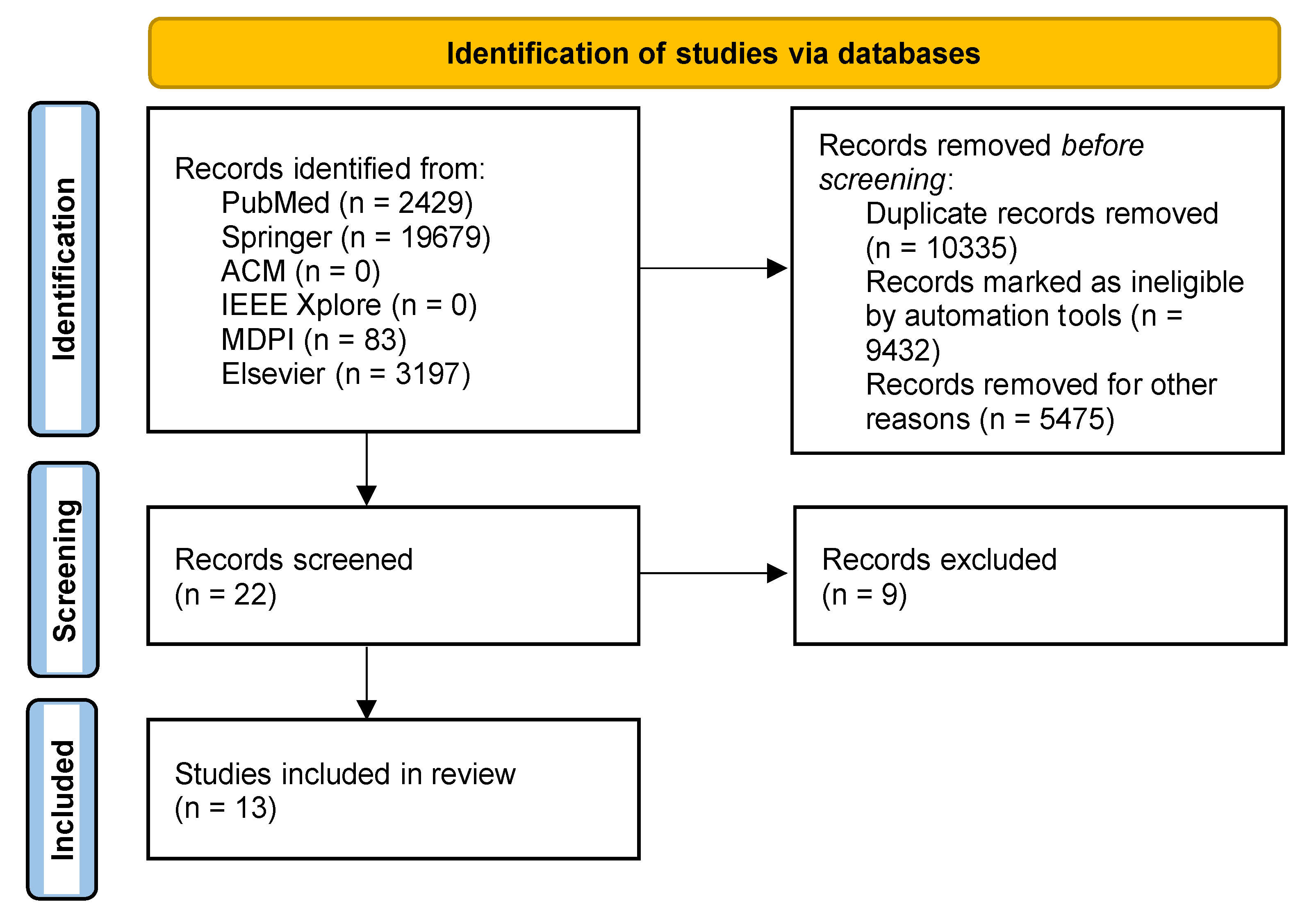
2. Materials and Methods
2.1. Research Questions
2.2. Inclusion Criteria
2.3. Search Strategy
2.4. Extraction of Study Characteristics
3. Results
4. Discussion and Conclusions
- (RQ1) Which methods can be used with ECG sensors for gender identification? The main methods used were multiple linear regression, the logistic regression model, classification and regression tree analysis, the linear regression model, spectrograms, scalograms, Rentrop classification, and attractor reconstruction;
- (RQ2) Which features can be extracted from the ECG sensors for gender identification? The features extracted from the ECG sensors that can be used for gender identification were mainly the RR interval, the degree of ST-segment elevation, the ST-segment depression, the maximum QRS interval, the P-duration, the P-amplitude, the P-area, the P-terminal negative force, the PR-interval, the QT/QTc, and the slope of the QT-RR;
- (RQ3) What are the benefits of using ECG sensors for gender identification? The benefits of using ECG sensors are the possibility of analyzing the differences in the ECG waves of different genders and using this to study and treat heart diseases. The different treatments can be adapted by the different characteristics related to gender, and different treatments can be standardized by gender to promote the automation of the prescription of different medicines.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Thoi, F.; Scherer, D.J.; Kaye, D.M.; Sanders, P.; Stokes, M.B. Methamphetamine-Associated Cardiomyopathy: Addressing the Clinical Challenges. Heart Lung Circ. 2022, 31, 616–622. [Google Scholar] [CrossRef] [PubMed]
- Kunal, S.; Madan, M.; Tarke, C.; Gautam, D.K.; Kinkar, J.S.; Gupta, K.; Agarwal, R.; Mittal, S.; Sharma, S.M. Emerging Spectrum of Post-COVID-19 Syndrome. Postgrad. Med. J. 2021, 98, 633–643. [Google Scholar] [CrossRef] [PubMed]
- Nayak, A.; Hicks, A.J.; Morris, A.A. Understanding the Complexity of Heart Failure Risk and Treatment in Black Patients. Circ. Heart Fail. 2020, 13, e007264. [Google Scholar] [CrossRef]
- Amin, M.S.; Chiam, Y.K.; Varathan, K.D. Identification of Significant Features and Data Mining Techniques in Predicting Heart Disease. Telemat. Inform. 2019, 36, 82–93. [Google Scholar] [CrossRef]
- Almustafa, K.M. Prediction of Heart Disease and Classifiers’ Sensitivity Analysis. BMC Bioinform. 2020, 21, 278. [Google Scholar] [CrossRef] [PubMed]
- Ali, F.; El-Sappagh, S.; Islam, S.R.; Kwak, D.; Ali, A.; Imran, M.; Kwak, K.-S. A Smart Healthcare Monitoring System for Heart Disease Prediction Based on Ensemble Deep Learning and Feature Fusion. Inf. Fusion 2020, 63, 208–222. [Google Scholar] [CrossRef]
- Pylypchuk, R.; Wells, S.; Kerr, A.; Poppe, K.; Riddell, T.; Harwood, M.; Exeter, D.; Mehta, S.; Grey, C.; Wu, B.P. Cardiovascular Disease Risk Prediction Equations in 400,000 Primary Care Patients in New Zealand: A Derivation and Validation Study. Lancet 2018, 391, 1897–1907. [Google Scholar] [CrossRef]
- Sharma, A.; Badea, M.; Tiwari, S.; Marty, J.L. Wearable Biosensors: An Alternative and Practical Approach in Healthcare and Disease Monitoring. Molecules 2021, 26, 748. [Google Scholar] [CrossRef]
- Rao, G.H. Diagnosis of Early Risks, Management of Risks, and Reduction of Vascular Diseases; Jaypee Brothers Medical Publishers: New Delhi, India, 2018. [Google Scholar]
- Cheol Jeong, I.; Bychkov, D.; Searson, P.C. Wearable Devices for Precision Medicine and Health State Monitoring. IEEE Trans. Biomed. Eng. 2018, 66, 1242–1258. [Google Scholar] [CrossRef]
- Wang, P.; Hu, J. A Hybrid Model for EEG-Based Gender Recognition. Cogn. Neurodyn. 2019, 13, 541–554. [Google Scholar] [CrossRef]
- Hicks, S.A.; Isaksen, J.L.; Thambawita, V.; Ghouse, J.; Ahlberg, G.; Linneberg, A.; Grarup, N.; Strümke, I.; Ellervik, C.; Olesen, M.S. Explaining Deep Neural Networks for Knowledge Discovery in Electrocardiogram Analysis. Sci. Rep. 2021, 11, 10949. [Google Scholar] [CrossRef]
- Ramaraj, E. A Novel Deep Learning Based Gated Recurrent Unit with Extreme Learning Machine for Electrocardiogram (ECG) Signal Recognition. Biomed. Signal Process. Control 2021, 68, 102779. [Google Scholar]
- AlDuwaile, D.A.; Islam, M.S. Using Convolutional Neural Network and a Single Heartbeat for ECG Biometric Recognition. Entropy 2021, 23, 733. [Google Scholar] [CrossRef]
- Pires, I.M.; Denysyuk, H.V.; Villasana, M.V.; Sá, J.; Lameski, P.; Chorbev, I.; Zdravevski, E.; Trajkovik, V.; Morgado, J.F.; Garcia, N.M. Mobile 5P-Medicine Approach for Cardiovascular Patients. Sensors 2021, 21, 6986. [Google Scholar] [CrossRef]
- Zdravevski, E.; Lameski, P.; Trajkovik, V.; Chorbev, I.; Goleva, R.; Pombo, N.; Garcia, N.M. Automation in Systematic, Scoping and Rapid Reviews by an NLP Toolkit: A Case Study in Enhanced Living Environments. In Enhanced Living Environments; Ganchev, I., Garcia, N.M., Dobre, C., Mavromoustakis, C.X., Goleva, R., Eds.; Lecture Notes in Computer Science; Springer International Publishing: Cham, Switzerland, 2019; Volume 11369, pp. 1–18. ISBN 978-3-030-10751-2. [Google Scholar]
- Król-Józaga, B. Atrial Fibrillation Detection Using Convolutional Neural Networks on 2-Dimensional Representation of ECG Signal. Biomed. Signal Process. Control 2022, 74, 103470. [Google Scholar] [CrossRef]
- Shehta, M.; Rayan, M.M.; Fahmy, N.A.; Onsy, A.; Bastawy, I. Global Longitudinal Strain Detects Subtle Left Ventricular Systolic Dysfunction in Duchenne Muscular Dystrophy Patients and Carriers. Egypt Heart J. 2021, 73, 91. [Google Scholar] [CrossRef]
- Jiang, J.; Deng, H.; Xue, Y.; Liao, H.; Wu, S. Detection of Left Atrial Enlargement Using a Convolutional Neural Network-Enabled Electrocardiogram. Front. Cardiovasc. Med. 2020, 7, 609976. [Google Scholar] [CrossRef]
- Kapolas, C.; Kosirog-Glowacki, J.; Barney, K.L.; Advincula, L.; Klein, L.M.; Bitran, J.D.; Bufalino, S.; Rodriguez, T.E. Risk Factors for the Development of Cardiac Arrhythmias during Hematopoietic Stem Cell Transplantation. Blood 2018, 132, 3387. [Google Scholar] [CrossRef]
- Song, C.; Fu, R.; Dou, K.; Yang, J.; Xu, H.; Gao, X.; Li, W.; Gao, G.; Zhao, Z.; Liu, J.; et al. The CAMI-Score: A Novel Tool Derived From CAMI Registry to Predict In-Hospital Death among Acute Myocardial Infarction Patients. Sci. Rep. 2018, 8, 9082. [Google Scholar] [CrossRef]
- Keller, K.; Beule, J.; Balzer, J.O.; Dippold, W. Right Bundle Branch Block and SIQIII-Type Patterns for Risk Stratification in Acute Pulmonary Embolism. J. Electrocardiol. 2016, 49, 512–518. [Google Scholar] [CrossRef]
- Valuckiene, Z.; Budrys, P.; Jurkevicius, R. Predicting Ischemic Mitral Regurgitation in Patients with Acute ST-Elevation Myocardial Infarction: Does Time to Reperfusion Really Matter and What Is the Role of Collateral Circulation? Int. J. Cardiol. 2016, 203, 667–671. [Google Scholar] [CrossRef] [PubMed]
- Dewhurst, M.J.; Di Marco, L.Y.; Dewhurst, F.; Adams, P.C.; Murray, A.; Orega, G.P.; Mwita, J.C.; Walker, R.W.; Langley, P. Electrocardiographic Reference Values for a Population of Older Adults in Sub-Saharan Africa: Tanzanian ECG Reference Values. Ann. Noninvasive Electrocardiol. 2014, 19, 34–42. [Google Scholar] [CrossRef]
- Miller, C.S.; Foley, J.D.; Floriano, P.N.; Christodoulides, N.; Ebersole, J.L.; Campbell, C.L.; Bailey, A.L.; Rose, B.G.; Kinane, D.F.; Novak, M.J.; et al. Utility of Salivary Biomarkers for Demonstrating Acute Myocardial Infarction. J. Dent. Res. 2014, 93, 72S–79S. [Google Scholar] [CrossRef] [PubMed]
- Couderc, J.; Xia, X.; Denjoy, I.; Extramiana, F.; Maison-Blanche, P.; Moss, A.J.; Zareba, W.; Lopes, C.M. Genotype- and Sex-Specific QT-RR Relationship in the Type-1 Long-QT Syndrome. JAHA 2012, 1, e000570. [Google Scholar] [CrossRef] [PubMed]
- Hussien, A.; Battah, A.; Ashraf, M.; El-Deen, T.Z. Electrocardiography as a Predictor of Left Main or Three-Vessel Disease in Patients with Non-ST Segment Elevation Acute Coronary Syndrome. Egypt. Heart J. 2011, 63, 103–107. [Google Scholar] [CrossRef][Green Version]
- Vetter, V.L.; Dugan, N.; Guo, R.; Mercer-Rosa, L.; Gleason, M.; Cohen, M.; Vogel, R.L.; Iyer, R. A Pilot Study of the Feasibility of Heart Screening for Sudden Cardiac Arrest in Healthy Children. Am. Heart J. 2011, 161, 1000–1006.e3. [Google Scholar] [CrossRef] [PubMed]
- Kronander, H.; Fischer-Colbrie, W.; Nowak, J.; Brodin, L.-Å.; Elmqvist, H. Diagnostic Performance and Partition Values of Exercise Electrocardiographic Variables in the Detection of Coronary Artery Disease—Improved Accuracy by Using ST/HR Hysteresis. Clin. Physiol. Funct. Imaging 2010, 30, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Pires, I.M.; Garcia, N.M.; Pires, I.; Pinto, R.; Silva, P. ECG Data Related to 30-s Seated and 30-s Standing for 5P-Medicine Project. Mendeley Data, 2022. Available online: https://data.mendeley.com/datasets/z4bbj9rcwd/1 (accessed on 10 July 2022).
| Study | Year of Publication | Location | Population | Purpose | Sensors Used | Type of Method | Diseases |
|---|---|---|---|---|---|---|---|
| Król-Józaga [17] | 2022 | Poland | 23 individuals | The study aimed to compare three two-dimensional representations. | Electrocardiogram | Statistical | Arrhythmia |
| Shehta et al. [18] | 2021 | Egypt | 53 individuals | The authors aimed to detect subtle cardiac changes in Duchenne muscular dystrophy patients with electrocardiography and echocardiography sensors. | Electrocardiogram Echocardiogram | Statistical | Duchenne muscular dystrophy |
| Jiang et al. [19] | 2020 | China | 3391 participants | The authors developed an artificial intelligence approach for the detection of left atrial enlargement. | Electrocardiogram | Machine Learning | Left atrial enlargement |
| Kapolas et al. [20] | 2018 | United States of America | 137 patients | The study aimed to determine risk factors for the development of CA in patients undergoing HSCT. | Electrocardiogram | Statistical | Arrhythmia Coronary artery disease |
| Song et al. [21] | 2018 | China | 23,417 patients | The study aimed to develop a risk model to predict in-hospital death among contemporary AMI patients as soon as possible after admission. | Electrocardiogram Echocardiogram | Machine Learning | Myocardial infarction |
| Keller et al. [22] | 2016 | Germany | 175 patients | The authors investigated the ECG alterations of the right bundle branch block and SIQIII-type patterns for risk stratification in acute PE. | Electrocardiogram | Statistical | Bundle branch block |
| Valuckiene et al. [23] | 2015 | Lithuania | 173 patients | The authors predicted ischemic mitral regurgitation in patients with acute ST-elevation myocardial infarction. | Angiogram Echocardiogram | Machine Learning | Coronary artery disease Myocardial infarction |
| Dewhurst et al. [24] | 2014 | United Kingdom | 2232 participants | The authors aimed to establish electrocardiographic reference values for a population likely to differ genetically and environmentally from others where reference values are established. | Electrocardiogram | Statistical | N/D |
| Miller et al. [25] | 2014 | United States of America | 197 individuals | The goal was to determine if salivary biomarkers could facilitate a screening diagnosis of acute myocardial infarction. | Electrocardiogram | Statistical | Myocardial infarction |
| Couderc et al. [26] | 2012 | France | 307 individuals | The study aimed at determining whether a harmful response to an increased heart rate leads to abnormal dynamic QT-RR profiles and may be responsible for the increased cardiac risk in these patients. | Electrocardiogram | Statistical | Congenital long-QT syndrome |
| Hussien et al. [27] | 2011 | Egypt | 300 patients | The authors aimed to analyze the ST-segment elevation and the maximal QRS duration and correlated the values to predict left main and three-vessel disease. | Electrocardiogram | Statistical | Acute coronary syndrome Myocardial infarction Unstable angina |
| Vetter et al. [28] | 2011 | United States of America | 400 participants | The study attempted to add an ECG to history and physical examination and to identify a methodology for a more extensive multicenter study. | Electrocardiogram Echocardiogram | Statistical | N/D |
| Kronander et al. [29] | 2010 | Sweden | 1876 patients | The study compared the measurements of ST-segment changes during exercise and early postexercise recovery in terms of diagnostic discrimination capacity and optimal partition values. | Angiogram Myocardial Scintigraphy | Statistical | Coronary artery disease |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bastos, E.S.; Duarte, R.P.; Marinho, F.A.; Rudenko, R.; Denysyuk, H.V.; Gonçalves, N.J.; Zdravevski, E.; Albuquerque, C.; Garcia, N.M.; Pires, I.M. A Brief Review on Gender Identification with Electrocardiography Data. Appl. Syst. Innov. 2022, 5, 81. https://doi.org/10.3390/asi5040081
Bastos ES, Duarte RP, Marinho FA, Rudenko R, Denysyuk HV, Gonçalves NJ, Zdravevski E, Albuquerque C, Garcia NM, Pires IM. A Brief Review on Gender Identification with Electrocardiography Data. Applied System Innovation. 2022; 5(4):81. https://doi.org/10.3390/asi5040081
Chicago/Turabian StyleBastos, Eduarda Sofia, Rui Pedro Duarte, Francisco Alexandre Marinho, Roman Rudenko, Hanna Vitaliyivna Denysyuk, Norberto Jorge Gonçalves, Eftim Zdravevski, Carlos Albuquerque, Nuno M. Garcia, and Ivan Miguel Pires. 2022. "A Brief Review on Gender Identification with Electrocardiography Data" Applied System Innovation 5, no. 4: 81. https://doi.org/10.3390/asi5040081
APA StyleBastos, E. S., Duarte, R. P., Marinho, F. A., Rudenko, R., Denysyuk, H. V., Gonçalves, N. J., Zdravevski, E., Albuquerque, C., Garcia, N. M., & Pires, I. M. (2022). A Brief Review on Gender Identification with Electrocardiography Data. Applied System Innovation, 5(4), 81. https://doi.org/10.3390/asi5040081









