Modern Analogue Approach Applied to High-Resolution Varved Sediments—A Synthesis for Lake Montcortès (Central Pyrenees)
Abstract
:1. Introduction
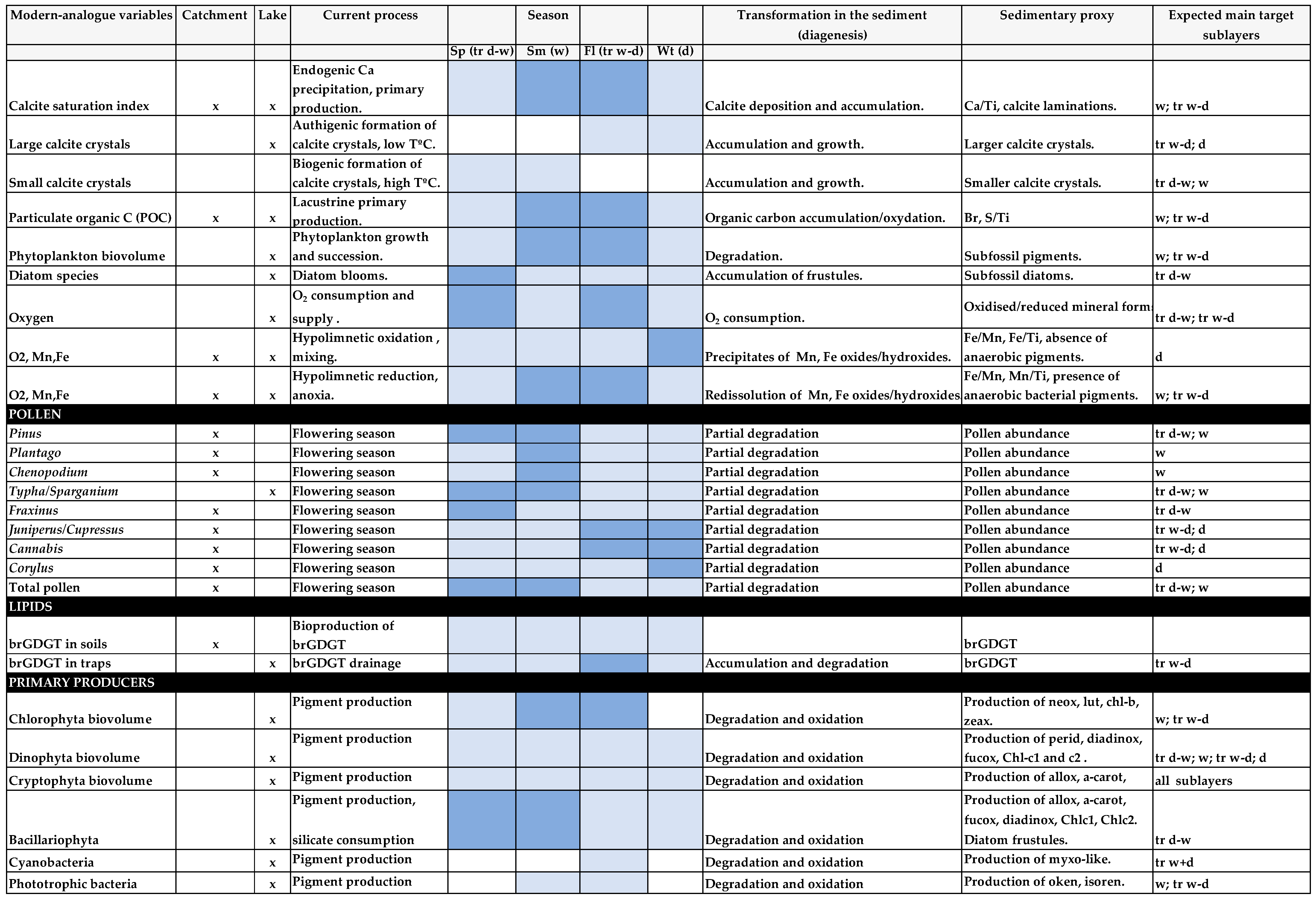
- (1)
- Relationships between calcite precipitation, environmental variables and primary producers and their influences on the thickness and patterns of calcareous varve sublayers were examined as high-resolution paleoclimate indicators [18].
- (2)
- Seasonal pollen sedimentation was compared with local meteorological variables to determine whether contemporary seasonal deposition is consistent with the two layers of the varve pattern archived in the sediments [19].
- (3)
- Contemporary shifts in dissolved O2, Fe, Mn and anaerobic phototrophic bacteria markers were assessed, in order to investigate whether mixing and oxygenation dynamics Lake Montcortès have been the same over the last five centuries [20].
- (4)
- Distribution of branched glycerol dialkyl glycerol tetraethers (GDGTs) in settling particulate matter and catchment soils was analysed in order to determine whether branched glycerol dialkyl glycerol tetraethers (brGDGTs) are seasonally biased in environments where annual brGDGTs production may not be constant [21].
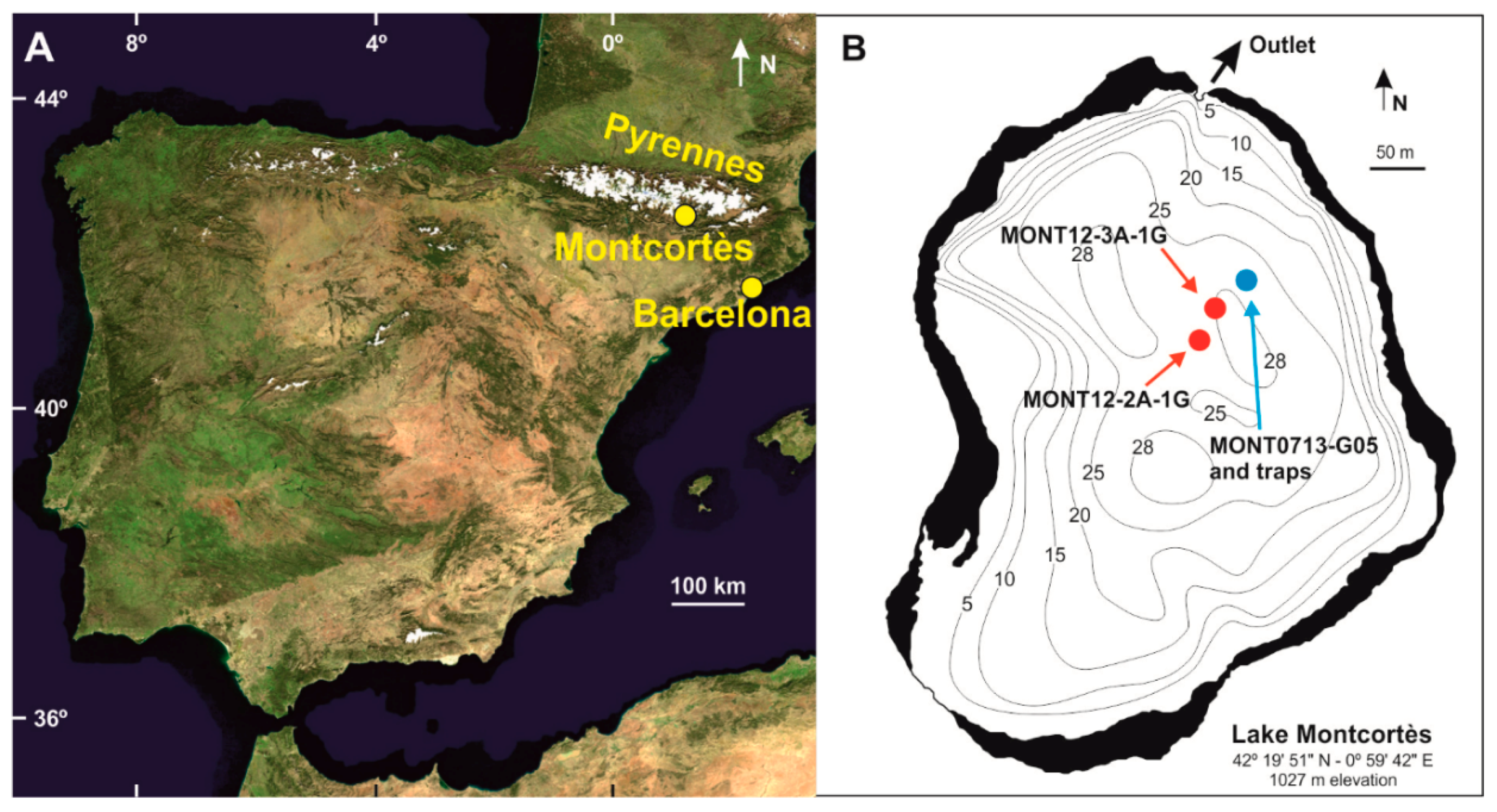
2. Environmental Settings
3. Materials and Methods
3.1. Monitoring and Coring
3.2. Laboratory Analyses
4. Results
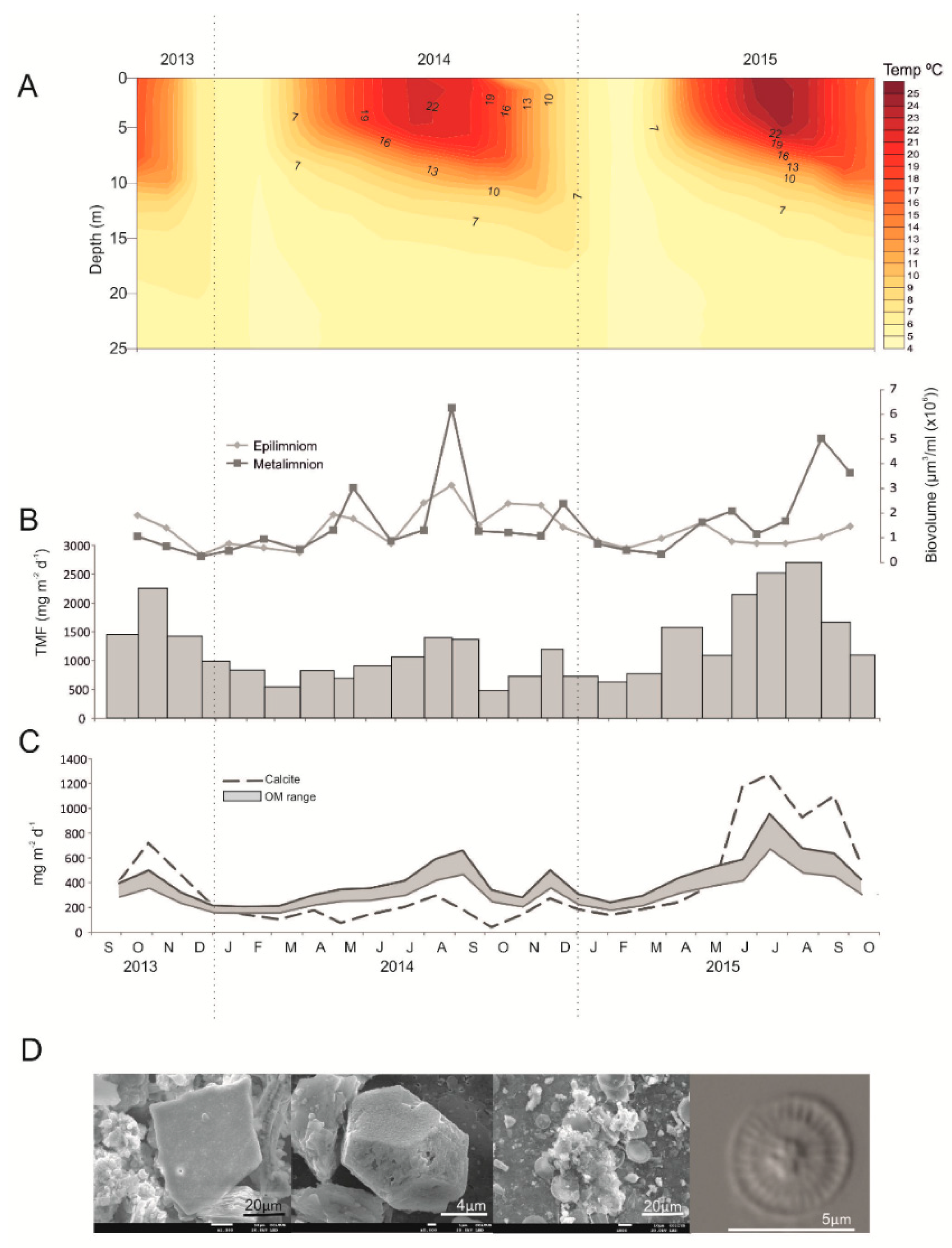
4.1. Biogeochemical Processes Involved in Varve Formation: Where, When, and How?
4.2. Is Contemporary Seasonal Deposition of Pollen Consistent with a Two-Layer Varve Model?
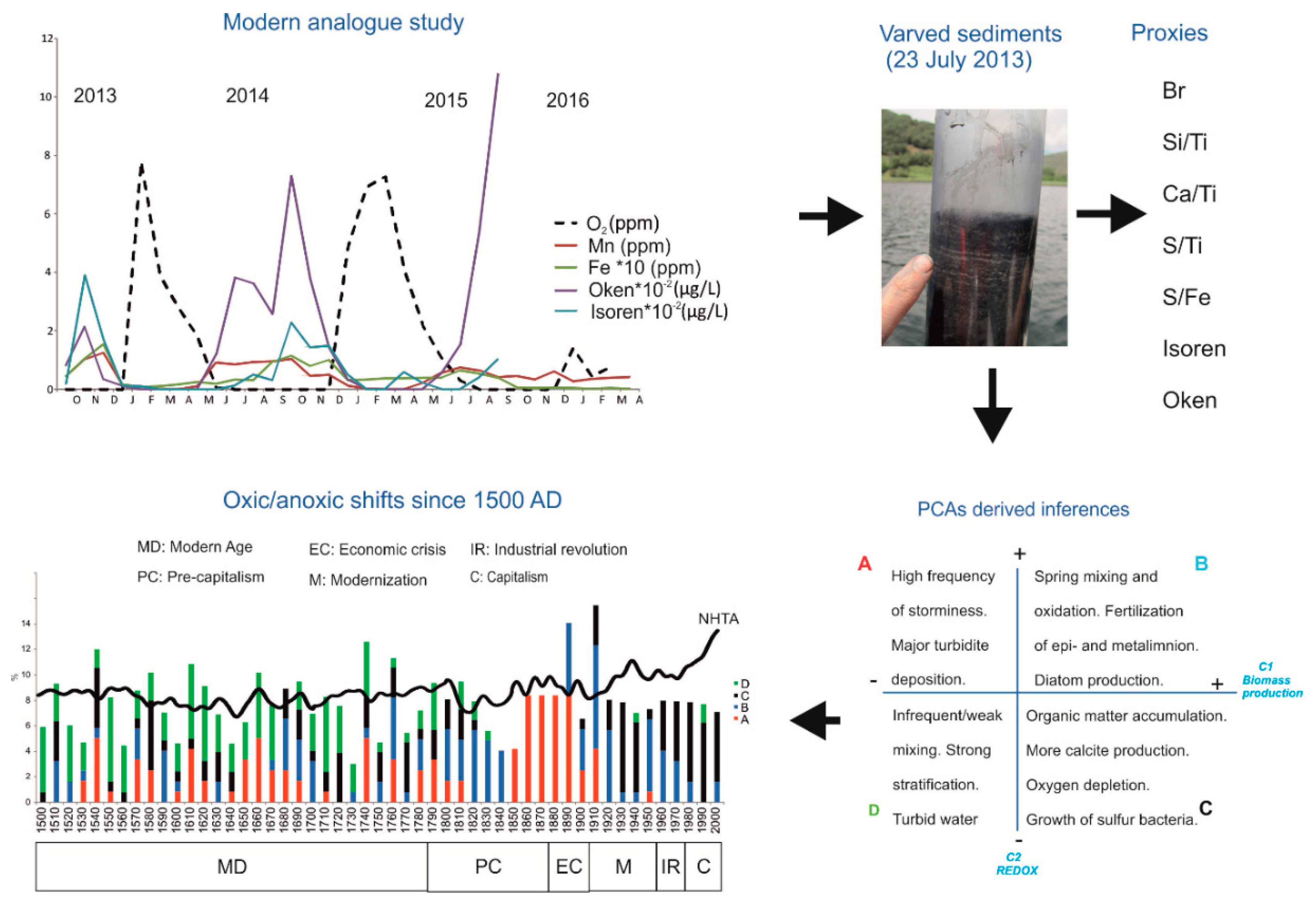
4.3. Revisiting Meromictic Lake Montcortès: Has the Mixing Regime Changed?
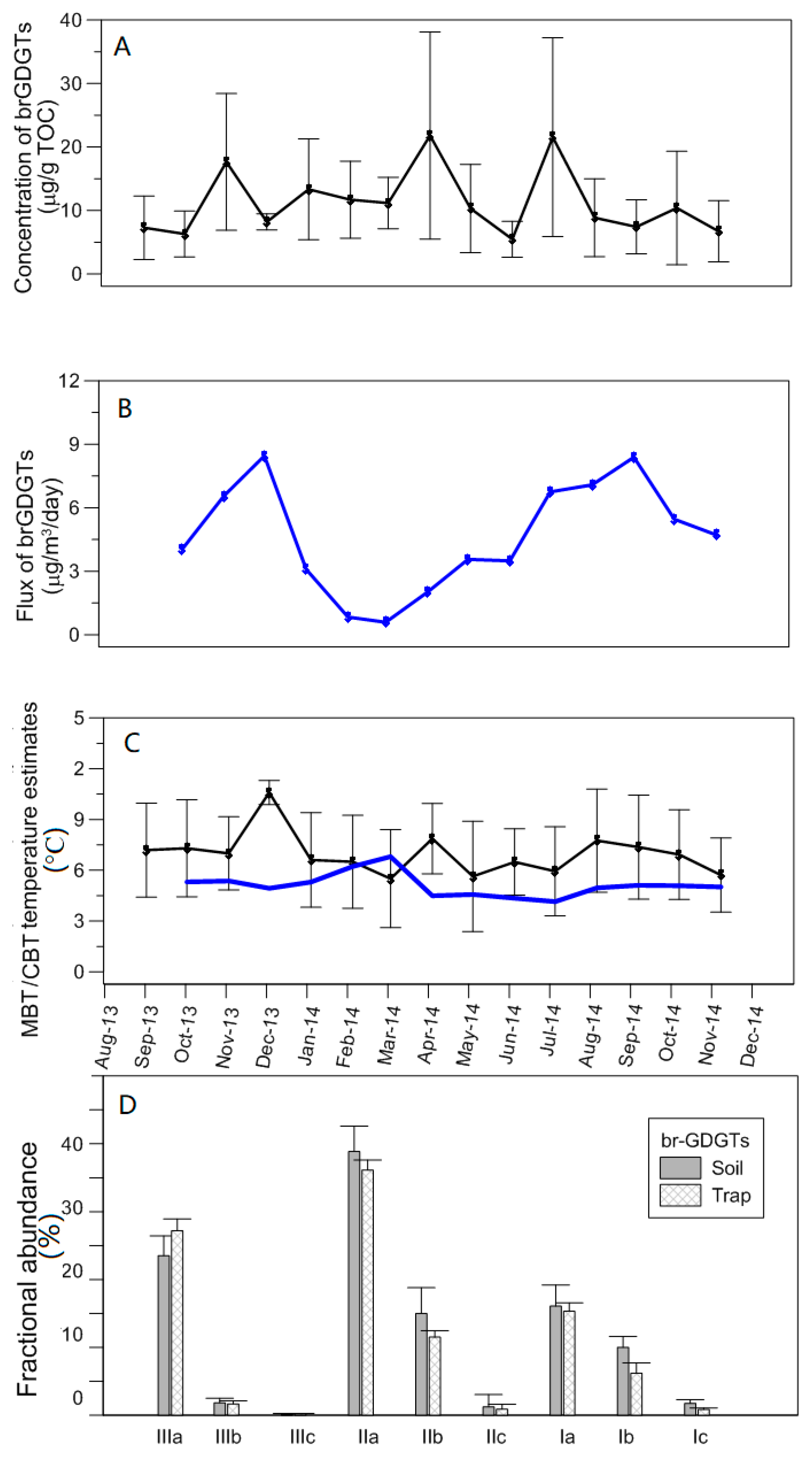
4.4. Are GDGTs Promising Indicators of Seasonal Temperature Shifts?
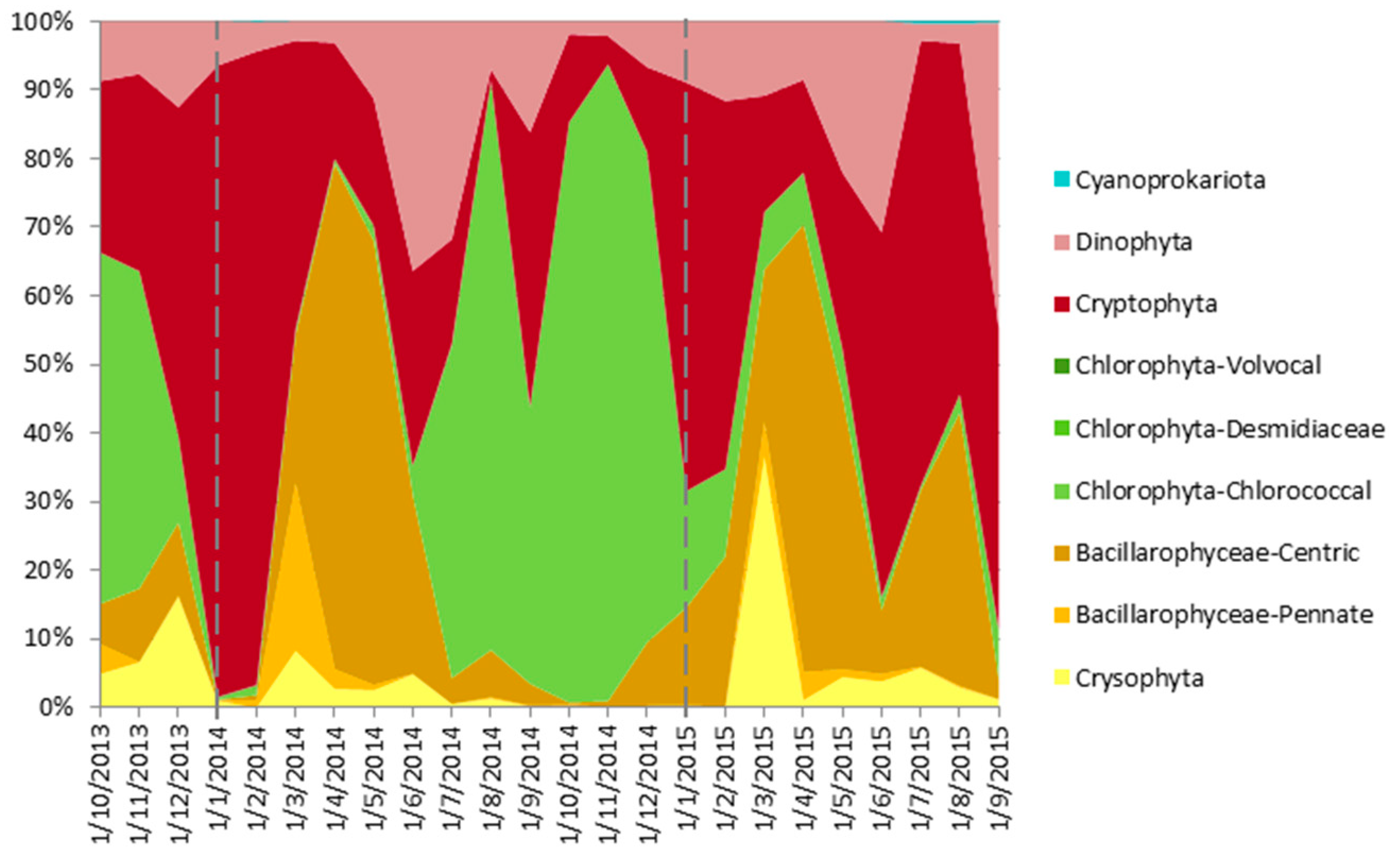
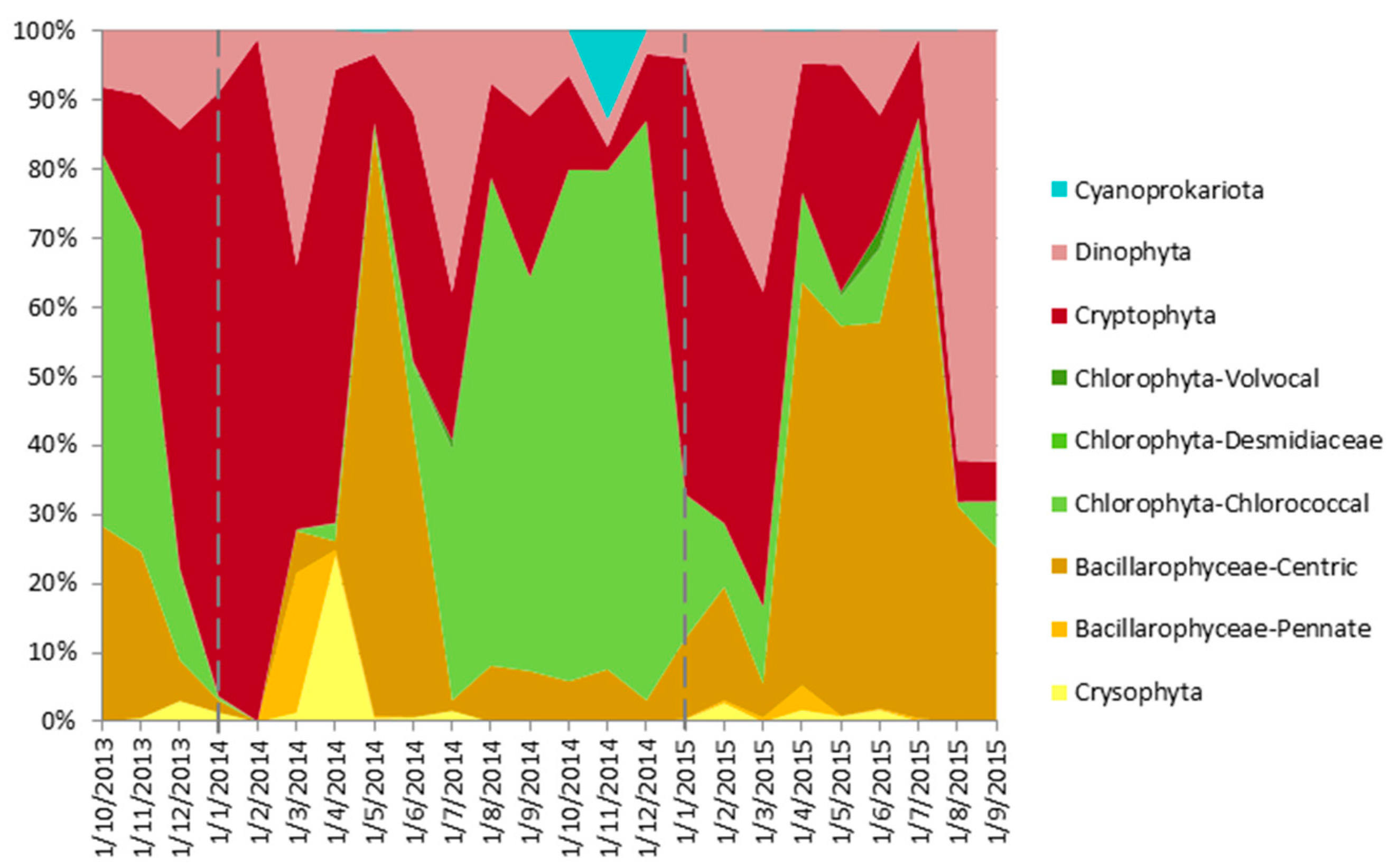
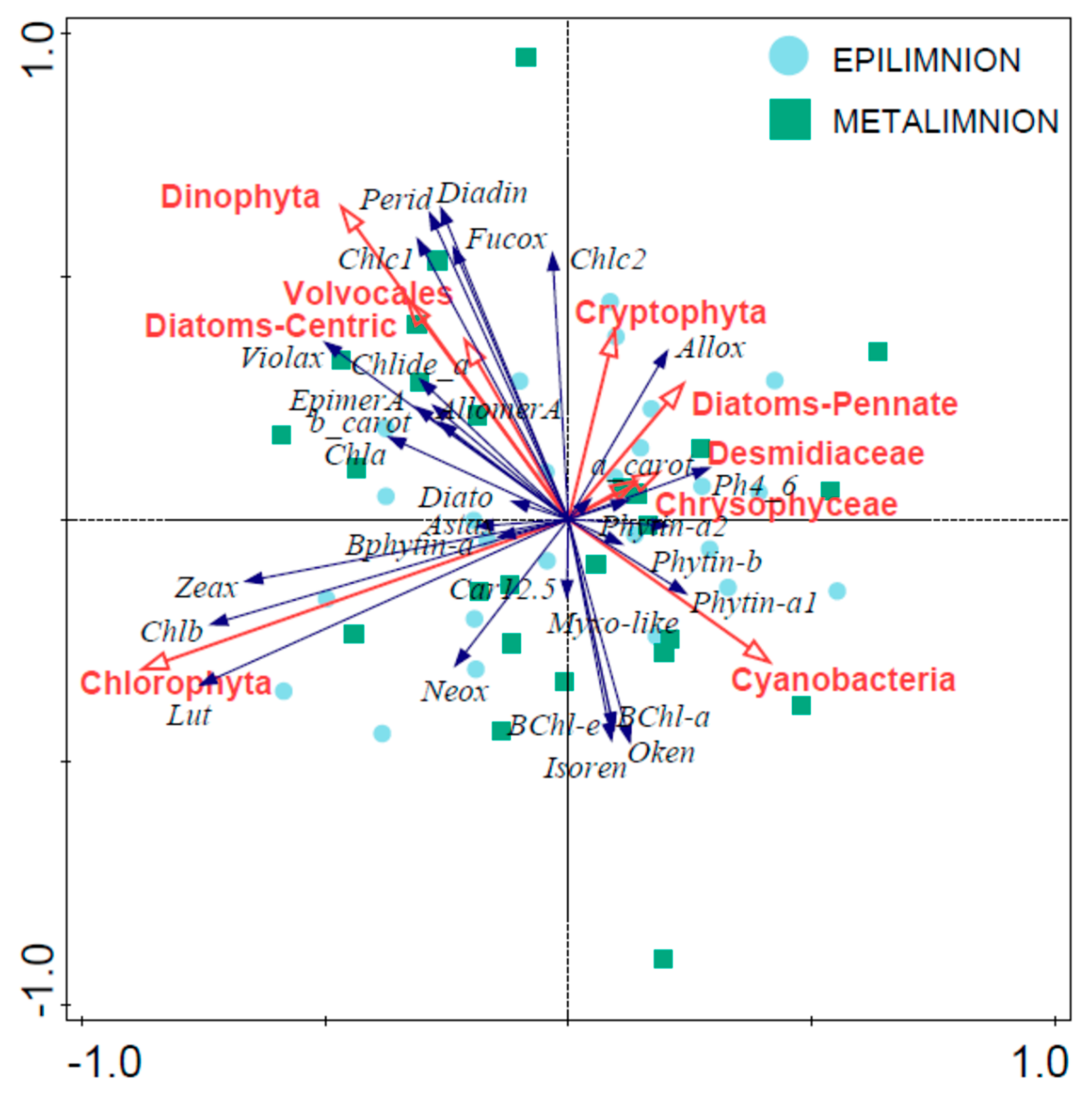
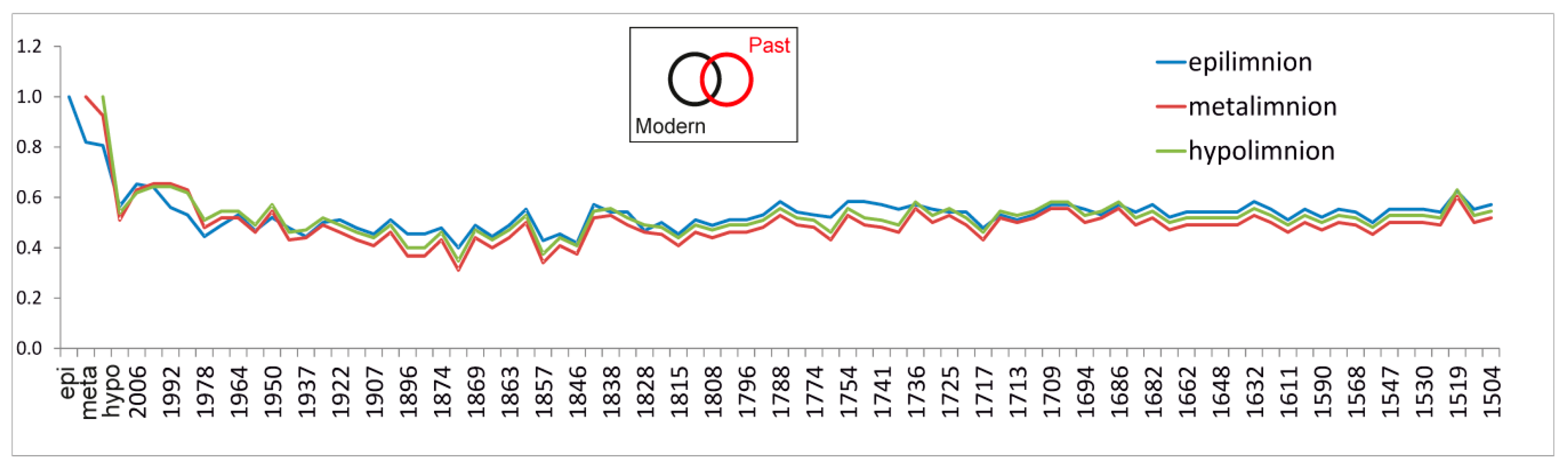
4.5. From Contemporary Phytoplankton to Subfossil Pigments, What Can We Learn about Community Changes?
5. Discussion, Conclusions and Guidelines for Future Research
- i.
- Calcite: There is evidence that a temperature increase enhances the amount of calcite precipitation and flux to the sediment. This relationship may be an important factor, when considering the role of calcite as a sink of atmospheric CO2 in the context of global warming and needs further examination. On the other hand, if the size of endogenic calcite crystals varies seasonally, inferences at the sub-annual scale may be derived from calcite crystals contained in sublayers of varves.
- ii.
- These hypotheses encourage the execution of further modern analogue studies. Hence, three improvements are suggested: (1) increase the frequency and duration of sampling, in order to better document shifts in calcite precipitation as related to the current warming trend; (2) install a meteorological station in the lake’s catchment, in order to capture local temperature and other meteorological variables that condition water temperature; (3) perform a detailed survey of calcite crystal features retrieved from seasonal traps and from varve sublayers to precisely determine their origin and diagnostic characteristics.
- iii.
- Pollen: Two advantages of pollen studies are (1) the ability of identifying and characterizing seasonal layers even in the absence of varves and (2) the possibility of recording interannual variability and associated meteorological drivers.
- iv.
- Oxygen: Even though our approach cannot provide estimations of past oxygen concentrations, it is a useful tool to connect and compare the dynamics of past and present oxic and anoxic events with annual resolution. However, if past oxygen concentrations are to be inferred, a more specific modern analogue study is required. Perhaps monthly and multiannual monitoring of the evolution of hypolimnetic oxygen concentrations and a concurrently examination of the content of Fe and Mn oxides in surface sediment samples, catches mixing and meromictic years. It should then be possible to establish appropriate transfer functions to estimate dissolved oxygen from most related proxies. If successful, truly long-term oxygen time series linking estimated and instrumental data could be established.
- v.
- Cyanoprokariota assemblages lack a modern analogue, since they are nearly absent today, whereas they were abundant, diverse and pervasive until, at least, the 1970s [54]. This observation is surprising because, to our knowledge, there have been few changes in land use or lake exploitation that could have affected Lake Montcortès’ trophic conditions. In contrast, we expect Cyanoprokariota to play a prominent role during the coming warmer years, when Lake Montcortès becomes more intensely stratified. Cyanobacteria marker pigments (myxoxanthophyll and zeaxanthin) have been detected in the water column during monitoring, the lack of correspondence with microscopical counts may be due to the low cell size of species involved, but this will need further studies. Therefore, considering the known deleterious effects of Cyanobacteria on aquatic ecosystems and human health, it would be advisable to continue present-day monitoring. The results would aid in obtaining a long-term picture of cyanobacterial successions and importance and allow deriving future scenarios, in the context of global warming.
- vi.
- brGDGT production in the soils of the lake catchment show no seasonality. However, in sediment trap particles, the brGDGT flux presents a clear seasonality. The seasonal variations of flux rates confirm transport variations from the catchment rather than in situ production of brGDGT in the lacustrine environment. The MBT/CBT temperature estimates indicate that brGDGT signatures in the sediment traps show a mixed signature and originate from surrounding soils. These results suggest that any inferences made from the high-resolution record must be made with caution.
- vii.
- From a methodological point of view, cylindrical traps with seasonal or quarterly recovery yield coherent results in all cases, i.e., total suspended solids, calcite crystals, pollen and spores and particles for brGDGT extractions. However, it is recommended that additional samples of surface sediments at the end of sampling years should be taken, to assess post-depositional transformations of these proxies. In our case, surface sediment samples would have provided complementary information, such as total fluxes to the sediment and subsequent accumulation, interactions at the sediment-water interface and early diagenesis. For example, in the case of calcite precipitation as related to varve formation, initial data of organic matter content may be needed to estimate the degree of sediment compaction with time [63] and for a full understanding of structure and thickness of varve sublayers at any depth. In the end, all this information allows a better connection and comparison between contemporary proxies and their modified version after being buried in the sediment, making the modern analogue technique more powerful.
- viii.
- Sometimes, modern analogue studies performed at a seasonal or annual frequency cannot be fully exploited for the interpretation of sedimentary records, because of a mismatch in resolution between present-day and past samples. This mismatch occurs when a relatively large weight or volume of sediment is needed to extract enough material for proxy analyses, because contiguous varves are too thin and/or must be joined into one sample. In doing so, the resolution of the samples decreases. With time and targeted effort, this disadvantage will hopefully be overcome with the advent of new technologies. In the field of marker pigments, hyperspectral image spectroscopy is progressing quickly and offers a non-destructive and inexpensive approach that permits high resolution. Hyperspectral image spectroscopy is being used in lake sediments, e.g., for concentrations of sedimentary bacteriopheophytin “a”, based upon diagnostic spectral properties with high spatial and temporal resolution [64]. A good match of the resolutions of modern and past studies is indispensable to successfully connect both and to better understand the evolution of ecosystems with time.
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gentner, D. Structure-mapping: A theoretical framework for analogy. Cogn. Sci. 1983, 7, 155–170. [Google Scholar] [CrossRef]
- Gentner, D. The mechanisms of analogical learning. In Similarity and Analogical Reasoning; Vosniadou, S., Ortony, A., Eds.; Cambridge University Press: London, UK, 1989; pp. 199–224. [Google Scholar]
- Hesse, M.B. Models and Analogies in Science; University of Notre Dame Press: Notre Dame, IN, USA, 1970; p. 184. [Google Scholar]
- Markman, A.B. Constraints on analogical inference. Cogn. Sci. 1997, 21, 373–418. [Google Scholar] [CrossRef]
- Delcourt, H.R.; Delcourt, P. The Paleoecological Perspective. In Quaternary Ecology: A Paleoecological Perspective; Chapman Hall: London, UK, 1991; p. 241. [Google Scholar]
- Jackson, S.T.; Williams, J.P. Modern Analogs in Quaternary Paleoecology: Here Today, Gone Yesterday, Gone Tomorrow? Ann. Rev. Earth Planet. Sci. 2004, 32, 495–537. [Google Scholar] [CrossRef]
- Simpson, G.L. Analogue Methods in Palaeolimnology. In Tracking Environmental Change Using Lake Sediments; Birks, H.J.B., Lotter, A.F., Juggins, S., Smol, J.P., Eds.; Springer Science Business Media: London, UK, 2012; Volume 5, pp. 495–522. [Google Scholar]
- Legendre, P.; Birks, H.J.B. From classical to canonical ordination. In Tracking Environmental Change Using Lake Sediments; Smol, J.P., Birks, H.J.B., Last, W.M., Eds.; Kluwer: Dordrecht, The Netherlands, 2012; pp. 201–248. [Google Scholar]
- Weng, C.; Bush, M.B.; Silman, M.R. An analysis of modern pollen rain on an elevational gradient in southern Peru. J. Trop. Ecol. 2004, 20, 113–124. [Google Scholar] [CrossRef] [Green Version]
- Reuss, N.; Leavitt, P.R.; Hall, R.I.; Bigler, C.; Hammarlund, D. Development and application of sedimentary pigments for assessing effects of climatic and environmental changes on subarctic lakes in northern Sweden. J. Paleolimnol. 2010, 43, 149–169. [Google Scholar] [CrossRef]
- Sachs, H.; Webb, T.; Clark, D.R. Paleoecological transfer funcions. Ann. Rev. Earth. Planet. Sci. 1977, 5, 159–178. [Google Scholar] [CrossRef]
- Massaferro, J.; Larocque-Tobler, I. Using a newly developed chironomid transfer function for reconstructing mean annual air temperature at Lake Potrok Aike, Patagonia, Argentina. Ecol. Ind. 2013, 24, 201–210. [Google Scholar] [CrossRef]
- Pla, S.; Catalan, J. Chrysophyte cysts from lake sediments reveal the submillennial winter/spring climate variability in the northwestern Mediterranean region throughout the Holocene. Clim. Dyn. 2005, 24, 263–278. [Google Scholar] [CrossRef]
- Wittkop, C.; Teranes, J.; Lubenow, B.; Dean, W.E. Carbon-and oxygen-stable isotopic signatures of methanogenesis, temperature, and water column stratification in Holocene siderite varves. Chem. Geol. 2014, 11, 153–166. [Google Scholar] [CrossRef]
- Zolitschka, B.; Francus, P.; Ojala, A.E.; Schimmelmann, A. Varves in lake sediments–A review. Quat. Sci. Rev. 2015, 117, 1–41. [Google Scholar] [CrossRef]
- Rull, V. Time continuum and true long-term ecology: From theory to practice. Front. Ecol. Evol. 2014, 2, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Brauer, A. Annually laminated lake sediments and their paleoclimatic relevance. In Climate in Historical Time: Towards a Synthesis of Holocene Proxy Data and Climate Models; Fisher, H., Ed.; Springer: Berlin/Heidelberg, Germany, 2004; pp. 108–128. [Google Scholar]
- Trapote, M.C.; Vegas-Vilarrúbia, T.; López, P.; Puche, E.; Gomà, J.; Buchaca, T.; Cañellas-Boltà, N.; Safont, E.; Corella, J.P.; Rull, V. Modern sedimentary analogues and integrated monitoring to understand varve formation in the Mediterranean Lake Montcortès (Central Pyrenees, Spain). Palaeogeogr. Palaeoclimatol. Palaeoecol. 2018, 496, 292–304. [Google Scholar] [CrossRef] [Green Version]
- Rull, V.; Trapote, M.C.; Safont, E.; Cañellas-Bolta, N.; Pérez-Zanón, N.; Sigró, J.; Buchaca, T.; Vegas-Vilarrúbia, T. Seasonal patterns of pollen sedimentation in Lake Montcortès (Central Pyrenees) and potential applications to high-resolution paleoecology: A 2-year pilot study. J. Paleolimnol. 2017, 57, 95–108. [Google Scholar] [CrossRef] [Green Version]
- Vegas-Vilarrúbia, T.; Corella, J.P.; Pérez-Zanón, N.; Buchaca, T.; Trapote, M.C.; López, P.; Sigró, J.; Rull, V. Historical shifts in oxygenation regime as recorded in the laminated sediments of lake Montcortès (Central Pyrenees) support hypoxia as a continental-scale phenomenon. Sci. Tot. Environ. 2018, 612, 1577–1592. [Google Scholar] [CrossRef] [PubMed]
- Cao, M.; Rueda, G.; Rivas-Ruiz, P.; Trapote, M.C.; Henriksen, M.; Vegas-Vilarrúbia, T.; Rosell-Melé, A. Branched GDGT variability in sediments and soils from catchments with marked temperature seasonality. Org. Geochem. 2018, 122, 98–114. [Google Scholar] [CrossRef]
- Corella, J.P.; Moreno, A.; Morellón, M.; Rull, V.; Giralt, S.; Rico, M.T.; Pérez-Sanz, A.; Valero-Garcés, B.L. Climate and human impact on a meromictic lake during the last 6000 years (Montcortès Lake, Central Pyrenees, Spain). J. Paleolimnol. 2011, 46, 351–367. [Google Scholar] [CrossRef] [Green Version]
- Rosell, J. Geological Map of Spain and Report. Scale 1:50,000, Tremp Sheet (252); Instituto Tecnológico Geográfico de España (IGME): Madrid, Spain, 1994. [Google Scholar]
- Mercadé, A.; Vigo, J.; Rull, V.; Vegas-Villarrúbia, T.; Garcés, S.; Lara, A.; Cañellas-Boltà, N. Vegetation and landscape around Lake Montcortès (Catalan pre-Pyrenees) as a tool for palaeoecological studies of lake sediments. Collect. Bot. 2013, 32, 87–101. [Google Scholar] [CrossRef] [Green Version]
- Corella, J.P.; Benito, G.; Wilhelm, B.; Montoya, E.; Rull, V.; Vegas-Vilarrúbia, T.; Valero-Garcés, B.L. A millennium-long perspective of flood-related seasonal sediment yield in Mediterranean watersheds. Glob. Plan. Sci. 2019, 177, 127–140. [Google Scholar] [CrossRef]
- Corella, J.P.; Brauer, A.; Mangili, C.; Rull, V.; Vegas-Vilarrúbia, T.; Morellón, M.; Valero-Garcés, B.L. The 1.5-ka varved record of Lake Montcortès (Southern Pyrenees, NE Spain). Quat. Res. 2012, 78, 323–332. [Google Scholar] [CrossRef]
- Corella, J.P.; Benito, G.; Rodríguez-Lloveras, X.; Brauer, A.; Valero-Garcés, B.L. Annually resolved lake record of extreme hydro-meteorological events since AD 1347 in NE Iberian Peninsula. Quat. Sci. Rev. 2014, 93, 77–90. [Google Scholar] [CrossRef] [Green Version]
- Pierrot, D.; Lewis, E.; Wallace, D.W.R. MS Excel Program Developed for CO2 System Calculations, ORNL/CDIAC-105; Carbon Dioxide Information Analysis Center, Oak Ridge National Laboratory, US Department of Energy: Oak Ridge, TN, USA, 2006.
- Battarbee, R.W.; Jones, V.; Flower, R.J.; Cameron, N.G.; Bennion, H.; Carvalho, L.; Juggins, S. Diatoms. In Tracking Environmental Change Using Lake Sediments; Smol, J.P., Birks, H.J.B., Last, W.M., Eds.; Kluwer: Dordrecht, The Netherlands, 2001; Volume 3, pp. 155–202. [Google Scholar]
- Krammer, K.; Lange-Bertalot, H. Bacillariophyceae. In Süßwasserflora von Mitteleuropa, Bänder 2,3,4/5; Gustav FischerVerlag: Stuttgart, Germany, 2000. [Google Scholar]
- Lange-Bertalot, H. Diatoms of the Europe Inland Waters and Comparable Habitats; Gantner: Ruggell, Germany, 2001. [Google Scholar]
- Bennett, K.D.; Willis, K.J. Pollen. In Tracking Environmental Change Using Lake Sediments; Smol, J.P., Birks, H.J.B., Last, W.M., Eds.; Kluwer: Dordrecht, The Netherlands, 2002; pp. 5–30. [Google Scholar]
- Rull, V.; Vegas-Vilarrúbia, T. Preliminary report on a mid-19th century Cannabis pollen peak in NE Spain: Historical context and potential chronological significance. Holocene 2014, 24, 1378–1383. [Google Scholar] [CrossRef]
- Rull, V.; Vegas-Vilarrúbia, T. Crops and weeds from the Estany de Montcortès catchment, Central Pyrenees, during the last millennium: A comparison of palynological and historical records. Veg. Hist. Archaeobot. 2015, 24, 699–710. [Google Scholar] [CrossRef]
- Rull, V.; González-Sampériz, P.; Corella, J.P.; Morellón, M.; Giralt, S. Vegetation changes in the southern Pyrenean flank during the last millennium inrelation to climate and human activities: The Montcortès lacustrine record. J. Paleolimnol. 2011, 46, 387–404. [Google Scholar] [CrossRef] [Green Version]
- Buchaca, T.; Catalan, J. Factors influencing the variability of pigments in the surface sediments of mountain lakes. Fresh. Biol. 2007, 52, 1365–1379. [Google Scholar] [CrossRef]
- Buchaca, T. Pigments Indicadors: Estudi del Senyal en Estanys dels Pirineus i de la Seva Aplicació en Paleolimnologia; Institut d’Estudis Catalans: Catalans, Spain, 2009. [Google Scholar]
- Leemann, A.; Niessen, F. Varve formation and climatic record in an Alpine proglacial lake: Calibrating annually-laminated sediments against hydrological and meteorological data. Holocene 1994, 4, 1–8. [Google Scholar] [CrossRef]
- Mann, M.E.; Bradley, R.S.; Hughes, M.H. Northern Hemisphere Temperatures during the Past Millennium’ Inferences, Uncertainties, and Limitations. Geophs. Res. Lett. 1999, 26, 759–762. [Google Scholar] [CrossRef] [Green Version]
- Bolòs, O.; Vigo, J.; Masalles, R.M.; Ninot, J.M. Flora Manual dels Països Catalans; Editorial Pòrtic: Barcelona, Spain, 2000. [Google Scholar]
- Trapote, M.C.; Rull, V.; Giralt, S.; Corella, J.P.; Montoya, E.; Vegas-Vilarrúbia, T. High-resolution (sub-decadal) pollen analysis of varved sediments from Lake Montcortès (southern Pyrenean flank): A fine-tuned record of landscape dynamics and human impact during the last 500 years. Rev. Palaeobot. Palynol. 2018, 259, 207–222. [Google Scholar] [CrossRef] [Green Version]
- Hakala, A. Meromixis as a part of lake evolution; observations and a revisedclassification of true meromictic lakes in Finland. Boreal Environ. Res. 2004, 9, 37–53. [Google Scholar]
- Jellison, R.; Romero, J.; Melack, J.M. The onset of meromixis during restoration of Mono Lake, California: Unintended consequences of reducingwater diversions. Limnol. Oceanogr. 1998, 43, 706–711. [Google Scholar] [CrossRef]
- Jenny, J.P.; Francus, P.; Normandeau, A.; Lapointe, F.; Perga, M.E.; Ojala, A.; Schimmelmann, A.; Zolitschka, B. Global spread of hypoxia in freshwater ecosystems during the last three centuries is caused by rising local human pressure. Glob. Chang. Biol. 2016, 22, 1481–1489. [Google Scholar] [CrossRef]
- Elliot, J.A. Is the future blue-green? A review of the current model predictions of how climate change could affect pelagic freshwater cyanobacteria. Water Res. 2014, 46, 1364–1371. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jankowski, T.; Livingstone, D.M.; Bührer, H.; Forster, R.; Niederhauser, P. Consequences of the 2003 European heat wave for lake temperature profiles, thermal stability, and hypolimnetic oxygen depletion: Implications for a warmer world. Limnol. Oceanogr. 2006, 51, 815–819. [Google Scholar] [CrossRef] [Green Version]
- Rempfer, J.; Livingstone, D.M.; Blodau, C.; Forster, R.; Niederhauser, P.; Kipfer, R. The effect of the exceptionally mild European winter of 2006–2007 on temperature and oxygen profiles in lakes in Switzerland: A foretaste of the future? Limnol. Oceanogr. 2010, 55, 2170–2180. [Google Scholar] [CrossRef]
- Leavitt, P.R. A review of factors that regulate carotenoid and chlorophyll deposition and fossil pigment abundance. J. Paleolimnol. 1993, 9, 109–127. [Google Scholar] [CrossRef]
- Villanueva, J.; Grimalt, J.O.; De Wit, R.; Keely, B.J.; Maxwell, J.R. Sources and transformations of chlorophylls and carotenoids in a monomictic sulphate-rich karstic lake environment. Org. Geochem. 1994, 22, 739–757. [Google Scholar] [CrossRef]
- Hodgson, D.A.; Wright, S.W.; Tyler, P.A.; Davies, N. Analysis of fossil pigments from algae and bacteria in meromictic Lake Fidler, Tasmania, and its application to lake management. J. Paleolimnol. 1998, 19, 1–22. [Google Scholar] [CrossRef]
- North, R.P.; North, R.L.; Livingstone, D.M.; Köster, O.; Kipfer, R. Long-term changes in hypoxia and soluble reactive phosphorus in the hypolimnion of a large temperate lake: Consequences of a climate regime shift. Glob. Chang. Biol. 2014, 20, 811–823. [Google Scholar] [CrossRef]
- Moreno, A.; Giralt, S.; Valero-Garcés, B.; Sáez, A.; Bao, R.; Prego, R.; Pueyo, J.J.; González-Sampériz, P.; Taberner, C. A 14kyr record of the tropical Andes: The Lago Chungará sequence (18° S, northern Chilean Altiplano). Quat. Int. 2007, 161, 4–21. [Google Scholar] [CrossRef]
- Davies, S.J.; Lamb, H.F.; Roberts, S.J. Micro-XRF core scanning in Palaeolimnology:recent developments. In Micro-XRF Studies of Sediment Cores: Applications of a Non-destructive Tool for the Environmental Sciences; Croudace, I.W., Rothwell, R.G., Eds.; Springer: Dordrecht, The Netherlands, 2015; pp. 189–226. [Google Scholar]
- Camps, J.; Gonzalvo, I.; Güell, J.; López, P.; Tejero, A.; Toldrà, X.; Vallespinós, F.; Vicens, M. El lago de Montcortès, descripción de un ciclo anual. Oecologia. Aquat. 1976, 2, 99–110. [Google Scholar]
- Weijers, J.W.H.; Schouten, S.; van den Donker, J.C.; Hopmans, E.C.; Sinninghe Damsté, J.S. Environmental controls on bacterial tetraether membrane lipid distribution in soils. Geoch. Cosmoch. Acta 2007, 71, 703–713. [Google Scholar] [CrossRef]
- Dang, X.; Yang, H.; Naafs, B.D.A.; Pancost, R.D. Direct evidence of moisture control on the methylation of branched glycerol dialkyl glycerol tetraethers in semi-arid and arid soils. Geoch. Cosmoch. Acta. 2016, 189, 24–36. [Google Scholar] [CrossRef] [Green Version]
- Weijers, J.W.H.; Bernhardt, B.; Peterse, F.; Werne, J.P.; Dungait, J.A.J.; Schouten, S.; Sinninghe Damsté, J.S. Absence of seasonal patterns in MBT-CBT indices in mid-latitude soils. Geoch. Cosmoch. Acta 2011, 75, 3179–3190. [Google Scholar] [CrossRef] [Green Version]
- Bonk, A.; Tylmann, W.; Amann, B.; Enters, D.; Grosjean, M. Modern limnology and varve-formation processes in lake Zabińskie, Northeastern Poland: Comprehensive process studies as a key to understand the sediment record. J. Limnol. 2015, 74, 358–370. [Google Scholar] [CrossRef] [Green Version]
- Miracle, M.R.; Camacho, A.; Juliâ, R.; Vicente, E. Sinking processes and their effect on the sedimentary record in the meromictic Lake La Cruz (Spain). Verh. Int. Verein. Limnol. 2000, 27, 1209–1213. [Google Scholar] [CrossRef]
- Romero, L.; Camacho, A.; Vicente, E.; Miracle, M.R. Sedimentation patterns of photosynthetic bacteria based on pigment markers in meromictic Lake La Cruz (Spain): Paleolimnological implications. J. Paleolimnol. 2006, 35, 167–177. [Google Scholar] [CrossRef]
- Rodrigo, M.A.; Vicente, E.; Miracle, M.R. Short-term calcite precipitation in the karstic meromictic Lake La Cruz (Cuenca, Spain). Verh. Int. Ver. Limnol. 1993, 25, 711–719. [Google Scholar] [CrossRef]
- Rodrigo, M.A.; Vicente, E.; Miracle, M.R. The meromictic Lake La Cruz (Central Spain): Patterns of stratification. Aquat. Sci. 2001, 63, 406–416. [Google Scholar] [CrossRef]
- Maier, D.; Rydberg, J.; Bigler, C.; Renberg, I. Compaction of recent varved lake sediments. GFF 2013, 135, 231–236. [Google Scholar] [CrossRef]
- Butz, C.; Grosjean, M.; Fischer, D.; Wunderle, S.; Tylmann, W.; Rei, B. Hyperspectral imaging spectroscopy: A promising method for the biogeochemical analysis of lake sediments. J. Appl. Remote Sens. 2015, 9, 1–20. [Google Scholar] [CrossRef] [Green Version]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vegas-Vilarrúbia, T.; Rull, V.; Trapote, M.d.C.; Cao, M.; Rosell-Melé, A.; Buchaca, T.; Gomà, J.; López, P.; Sigró, J.; Safont, E.; et al. Modern Analogue Approach Applied to High-Resolution Varved Sediments—A Synthesis for Lake Montcortès (Central Pyrenees). Quaternary 2020, 3, 1. https://doi.org/10.3390/quat3010001
Vegas-Vilarrúbia T, Rull V, Trapote MdC, Cao M, Rosell-Melé A, Buchaca T, Gomà J, López P, Sigró J, Safont E, et al. Modern Analogue Approach Applied to High-Resolution Varved Sediments—A Synthesis for Lake Montcortès (Central Pyrenees). Quaternary. 2020; 3(1):1. https://doi.org/10.3390/quat3010001
Chicago/Turabian StyleVegas-Vilarrúbia, Teresa, Valentí Rull, Maria del Carmen Trapote, Min Cao, Antoni Rosell-Melé, Teresa Buchaca, Joan Gomà, Pilar López, Javier Sigró, Elisabet Safont, and et al. 2020. "Modern Analogue Approach Applied to High-Resolution Varved Sediments—A Synthesis for Lake Montcortès (Central Pyrenees)" Quaternary 3, no. 1: 1. https://doi.org/10.3390/quat3010001
APA StyleVegas-Vilarrúbia, T., Rull, V., Trapote, M. d. C., Cao, M., Rosell-Melé, A., Buchaca, T., Gomà, J., López, P., Sigró, J., Safont, E., Cañellas, N., Garcés-Pastor, S., Giralt, S., Corella, J. P., & Pérez-Zanón, N. (2020). Modern Analogue Approach Applied to High-Resolution Varved Sediments—A Synthesis for Lake Montcortès (Central Pyrenees). Quaternary, 3(1), 1. https://doi.org/10.3390/quat3010001






