New Developments in the Synthesis of EMICORON
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Instrumentation
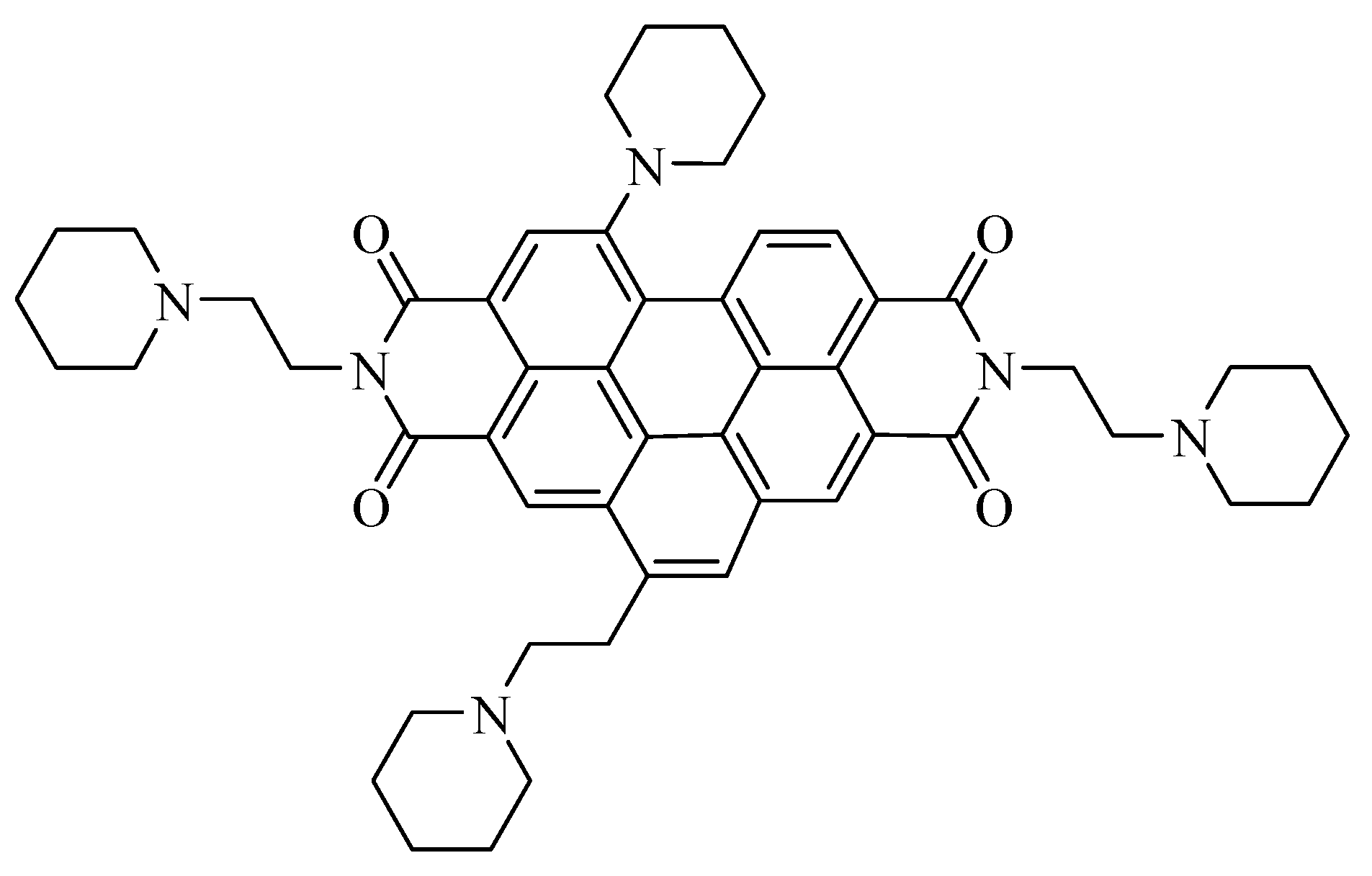
2.3. Synthesis
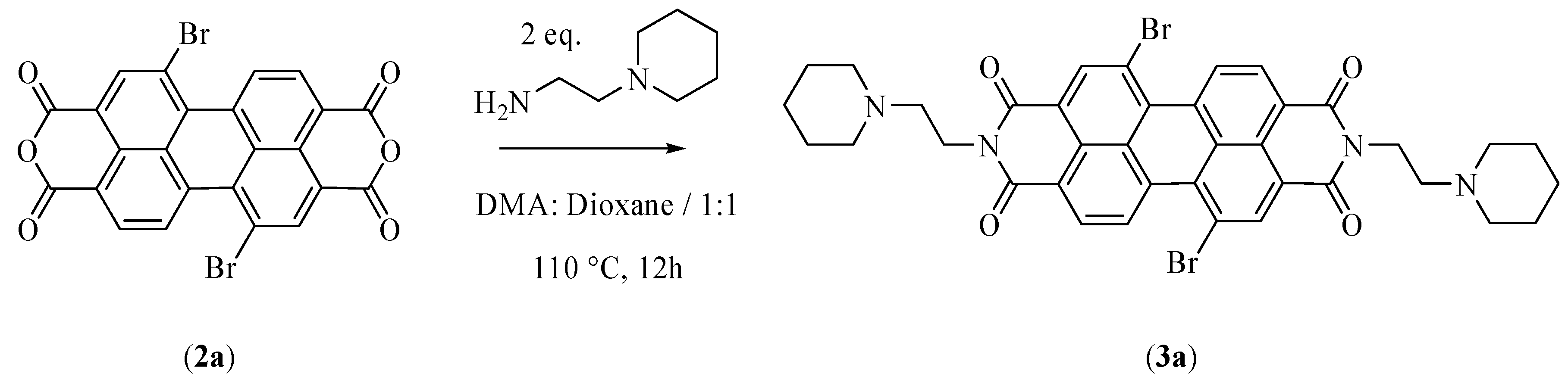
2.3.1. Synthesis of N,N′-bis[2-(1-piperidino)-ethyl]-1,7-dibromoperylene-3,4:9,10-tetracarboxylic diimide (PIPER-Br)
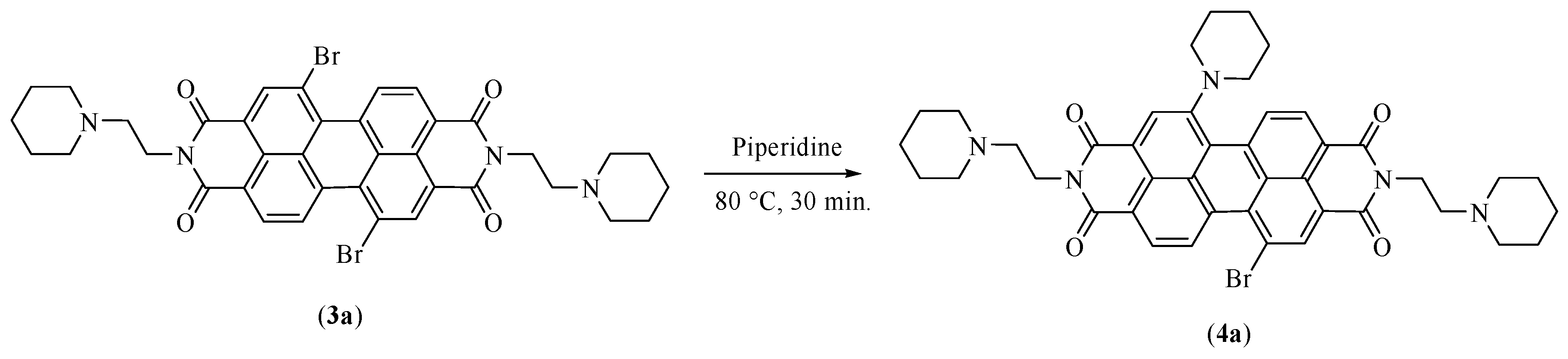
2.3.2. Synthesis of N,N′-bis[2-(1-piperidino)-ethyl]-1-(1-piperidinyl)-7-bromoperylene-3,4:9,10-tetracarboxylic diimide (PIP-PIPER-Br)
2.4. NMR Data of All the Synthetic Compounds
3. Results and Discussions
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Franceschin, M. G-quadruplex DNA structures and organic chemistry: More than one connection. Eur. J. Org. Chem. 2009, 14, 2225–2238. [Google Scholar] [CrossRef]
- D’Ambrosio, D.; Reichenbach, P.; Micheli, E.; Alvino, A.; Franceschin, M.; Savino, M.; Lingner, J. Specific binding of telomeric G-quadruplexes by hydrosoluble perylene derivatives inhibits repeat addition processivity of human telomerase. Biochimie 2012, 94, 854–863. [Google Scholar] [CrossRef] [PubMed]
- Biroccio, A.; Porru, M.; Rizzo, A.; Salvati, E.; D’Angelo, C.; Orlandi, A.; Passeri, D.; Franceschin, M.; Stevens, M.; Gilson, E.; et al. DNA damage persistence as determinant of tumor sensitivity to the combination of Topo I inhibitors and telomere-targeting agents. Clin. Cancer Res. 2011, 17, 2227–2236. [Google Scholar] [CrossRef] [PubMed]
- Casagrande, V.; Salvati, E.; Alvino, A.; Bianco, A.; Ciammaichella, A.; D’Angelo, C.; Ginnari-Satriani, L.; Serrilli, A.M.; Iachettini, S.; Leonetti, C.; et al. N-cyclic bay-substituted perylene G-quadruplex ligands have selective antiproliferative effects on cancer cells and induce telomere damage. J. Med. Chem. 2011, 54, 1140–1156. [Google Scholar] [CrossRef] [PubMed]
- Franceschin, M.; Alvino, A.; Casagrande, V.; Mauriello, C.; Pascucci, E.; Savino, M.; Ortaggi, G.; Bianco, A. Specific interactions with intra-and intermolecular G-quadruplex DNA structures by hydrosolublecoronene derivatives: A new class of telomerase inhibitors. Bioorg. Med. Chem. 2007, 15, 1848–1858. [Google Scholar] [CrossRef] [PubMed]
- Franceschin, M.; Rizzo, A.; Casagrande, V.; Salvati, E.; Alvino, A.; Altieri, A.; Ciammaichella, A.; Iachettini, S.; Leonetti, C.; Ortaggi, G.; et al. Aromatic core extension in the series of N-cyclic bay-substituted perylene G-Quadruplex ligands: Increate telomere damage, antitumor activity, and strong selectivity for neoplastic over healthy cells. Chem. Med. Chem. 2012, 7, 2144–2154. [Google Scholar] [CrossRef] [PubMed]
- Franceschin, M.; Alvino, A.; Ortaggi, G.; Bianco, A. New hydrosoluble perylene and coronene derivatives. Tetrahedron Lett. 2004, 45, 9015–9020. [Google Scholar] [CrossRef]
- Sengupta, S.; Dubey, R.K.; Hoek, R.W.M.; Van Eeden, S.P.P.; Gunbaş, D.D.; Grozema, F.C.; Sudholter, E.J.; Jager, W.F. Synthesis of Regioisomerically Pure 1,7-Dibromoperylene-3,4,9,10-tetracarboxylic Acid Derivatives. J. Org. Chem. 2014, 79, 6655–6662. [Google Scholar] [CrossRef] [PubMed]
- Porru, M.; Artuso, S.; Salvati, E.; Bianco, A.; Franceschin, M.; Diodoro, M.G.; Passeri, D.; Orlandi, A.; Savorani, F.; D’Incalci, M.; et al. Targeting G-Quadruplex DNA structures by EMICORON has a strong antitumor efficacy against advanced models of Human Colon Cancer. Mol. Cancer Ther. 2015, 14, 2541–2551. [Google Scholar] [CrossRef] [PubMed]
- Micheli, E.; Altieri, A.; Cianni, L.; Cingolani, C.; Iachettini, S.; Bianco, A.; Leonetti, C.; Cacchione, S.; Biroccio, A.; Franceschin, M.; et al. Perylene and coronene derivatives binding to G-rich promoter oncogene sequences efficiently reduce their expression in cancer cells. Biochimie 2016, 125, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Porru, M.; Zizza, P.; Franceschin, M.; Leonetti, C.; Biroccio, A. EMICORON: A multi-targeting G4 ligand with a promising preclinical profile. BBA Gen. Subj. 2017, 1861, 1362–1370. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Wasielewski, M.R. 3,4:9,10-Perylenebis(dicarboximide) chromophores that function as both electron donors and acceptors. Tetrahedron Lett. 1999, 40, 7047–7050. [Google Scholar] [CrossRef]
- Ahrens, M.J.; Tauber, M.J.; Wasielewski, M.R. Bis(n-octylamino)perylene-3,4:9,10-bis(dicarboximide)s and their radical cations: Synthesis, electrochemistry, and ENDOR Spectroscopy. J. Org. Chem. 2006, 71, 2107–2114. [Google Scholar] [CrossRef] [PubMed]
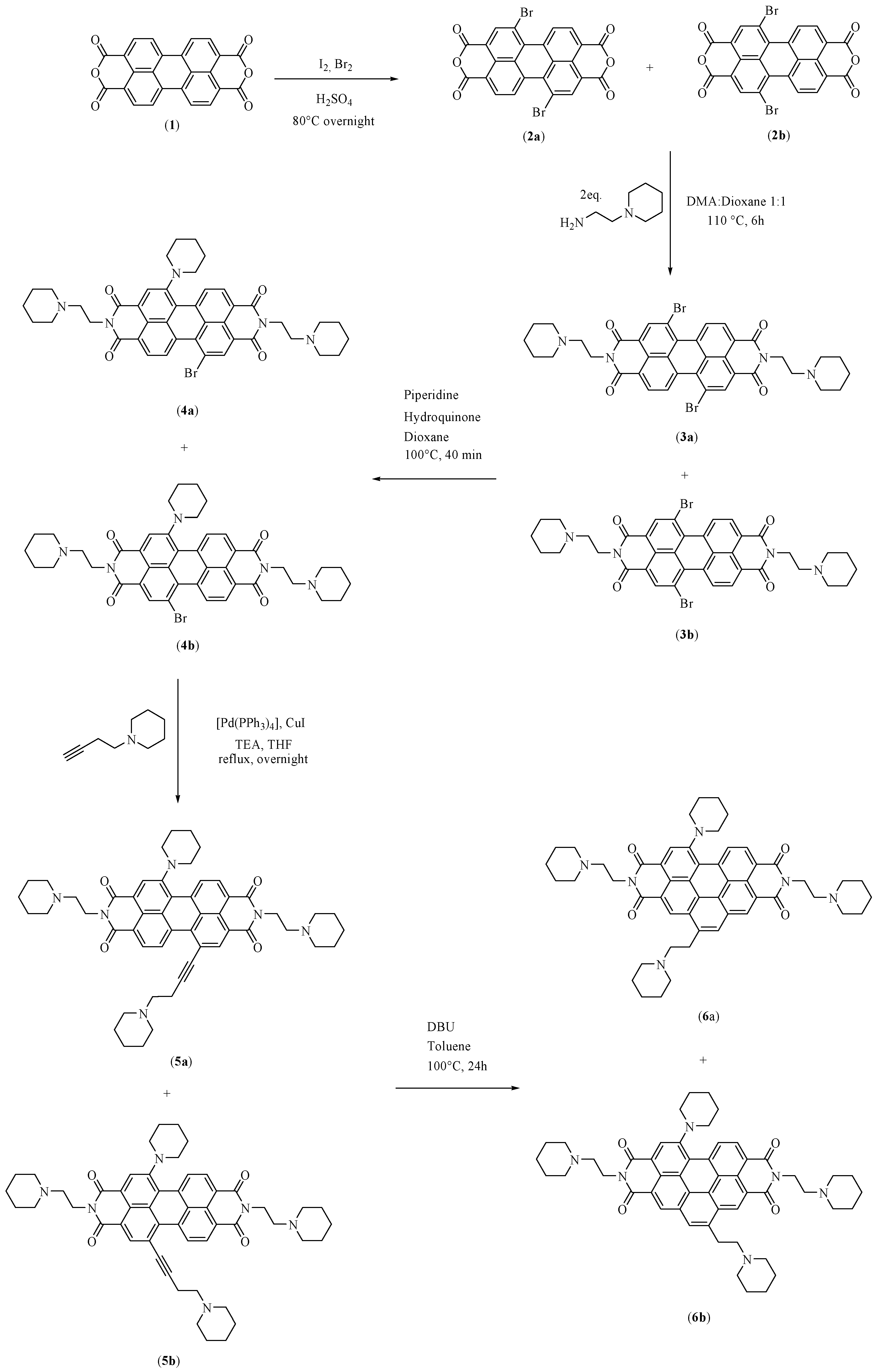
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pitorri, M.; Franceschin, M.; Serafini, I.; Ciccòla, A.; Frezza, C.; Bianco, A. New Developments in the Synthesis of EMICORON. High-Throughput 2018, 7, 22. https://doi.org/10.3390/ht7030022
Pitorri M, Franceschin M, Serafini I, Ciccòla A, Frezza C, Bianco A. New Developments in the Synthesis of EMICORON. High-Throughput. 2018; 7(3):22. https://doi.org/10.3390/ht7030022
Chicago/Turabian StylePitorri, Massimo, Marco Franceschin, Ilaria Serafini, Alessandro Ciccòla, Claudio Frezza, and Armandodoriano Bianco. 2018. "New Developments in the Synthesis of EMICORON" High-Throughput 7, no. 3: 22. https://doi.org/10.3390/ht7030022
APA StylePitorri, M., Franceschin, M., Serafini, I., Ciccòla, A., Frezza, C., & Bianco, A. (2018). New Developments in the Synthesis of EMICORON. High-Throughput, 7(3), 22. https://doi.org/10.3390/ht7030022






