Development and Validation of an Ultrasensitive Procalcitonin Sandwich Immunoassay
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of Capture and Detection Antibody
2.2. Simoa Assay Setup and Preparation of Reagents
2.3. Simoa Assay Validation Procedure
2.4. Laboratory Methods
2.5. Human Blood and CSF Samples
3. Results and Discussion
3.1. Assay Development and Validation
3.2. Correlation with Clinical Analyzer (B·R·A·H·M·S PCT-Sensitive Kryptor)
3.3. Baseline PCT Concentration in Healthy Volunteers
3.4. Meningits Samples
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Assicot, M.; Gendrel, D.; Carsin, H.; Raymond, J.; Guilbaud, J.; Bohuon, C. High serum procalcitonin concentrations in patients with sepsis and infection. Lancet 1993, 341, 515–518. [Google Scholar] [CrossRef]
- Becker, K.L.; Snider, R.; Nylen, E.S. Procalcitonin assay in systemic inflammation, infection, and sepsis: Clinical utility and limitations. Crit. Care Med. 2008, 36, 941–952. [Google Scholar] [CrossRef] [PubMed]
- Muller, B.; Becker, K.L.; Schächinger, H.; Rickenbacher, P.R.; Huber, P.R.; Zimmerli, W.; Ritz, R. Calcitonin precursors are reliable markers of sepsis in a medical intensive care unit. Crit. Care Med. 2000, 28, 977–983. [Google Scholar] [CrossRef] [PubMed]
- Schuetz, P.; Albrich, W.; Mueller, B. Procalcitonin for diagnosis of infection and guide to antibiotic decisions: Past, present and future. BMC Med. 2011, 9. [Google Scholar] [CrossRef] [PubMed]
- Russwurm, S.; Wiederhold, M.; Oberhoffer, M.; Stonans, I.; Zipfel, P.F.; Reinhart, K. Molecular aspects and natural source of procalcitonin. Clin. Chem. Lab. Med. 1999, 37, 789–797. [Google Scholar] [CrossRef] [PubMed]
- Sager, R.; Kutz, A.; Mueller, B.; Schuetz, P. Procalcitonin-guided diagnosis and antibiotic stewardship revisited. BMC Med. 2017, 15. [Google Scholar] [CrossRef] [PubMed]
- Schuetz, P.; Bretscher, C.; Bernasconi, L.; Mueller, B. Overview of procalcitonin assays and procalcitonin-guided protocols for the management of patients with infections and sepsis. Expert Rev. Mol. Diagn. 2017, 17, 593–601. [Google Scholar] [CrossRef] [PubMed]
- Rissin, D.M.; Fournier, D.R.; Piech, T.; Kan, C.W.; Campbell, T.G.; Song, L.; Chang, L.; Rivnak, A.J.; Patel, P.P.; Provuncher, G.K.; et al. Simultaneous detection of single molecules and singulated ensembles of molecules enables immunoassays with broad dynamic range. Anal. Chem. 2011, 83, 2279–2285. [Google Scholar] [CrossRef] [PubMed]
- Wilson, D.H.; Rissin, D.M.; Kan, C.W.; Fournier, D.R.; Piech, T.; Campbell, T.G.; Meyer, R.E.; Fishburn, M.W.; Cabrera, C.; Patel, P.P.; et al. The Simoa HD-1 Analyzer: A Novel Fully Automated Digital Immunoassay Analyzer with Single-Molecule Sensitivity and Multiplexing. J. Lab. Autom. 2016, 21, 533–547. [Google Scholar] [CrossRef] [PubMed]
- Rissin, D.M.; Kan, C.W.; Campbell, T.G.; Howes, S.C.; Fournier, D.R.; Song, L.; Piech, T.; Patel, P.P.; Chang, L.; Rivnak, A.J.; et al. Single-molecule enzyme-linked immunosorbent assay detects serum proteins at subfemtomolar concentrations. Nat. Biotechnol. 2010, 28, 595–599. [Google Scholar] [CrossRef] [PubMed]
- Chang, L.; Rissin, D.M.; Fournier, D.R.; Piech, T.; Patel, P.P.; Wilson, D.H.; Duffy, D.C. Single molecule enzyme-linked immunosorbent assays: Theoretical considerations. J. Immunol. Methods 2012, 378, 102–115. [Google Scholar] [CrossRef] [PubMed]
- Rivnak, A.J.; Rissin, D.M.; Kan, C.W.; Song, L.; Fishburn, M.W.; Piech, T.; Campbell, T.G.; DuPont, D.R.; Gardel, M.; Sullivan, S.; et al. A fully-automated, six-plex single molecule immunoassay for measuring cytokines in blood. J. Immunol. Methods 2015, 424, 20–27. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.W.; Devanarayan, V.; Barrett, Y.C.; Weiner, R.; Allinson, J.; Fountain, S.; Keller, S.; Weinryb, I.; Green, M.; Duan, L.; et al. Fit-for-purpose method development and validation for successful biomarker measurement. Pharm. Res. 2006, 23, 312–328. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.W.; Hall, M. Method validation of protein biomarkers in support of drug development or clinical diagnosis/prognosis. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2009, 877, 1259–1271. [Google Scholar] [CrossRef] [PubMed]
- Food and Drug Administration (FDA). Guidance for Industry: Bioanalytical Method Validation; US Department of Health and Human Services: Washington, DC, USA; Food and Drug Administration, Center for Drug Evaluation and Research: Silver Spring, MD, USA, 2013.
- Europe, the Middle East and Africa (EMEA). Guideline on Bioanalytical Method Validation; European Medicines Agency: London, UK, 2011. [Google Scholar]
- Kremmer, E.; Meyer, K.; Grässer, F.A.; Flatley, A.; Kösters, M.; Luppa, P.B.; Krämer, P.M. A new strategy for the development of monoclonal antibodies for the determination of human procalcitonin in serum samples. Anal. Bioanal. Chem. 2012, 402, 989–995. [Google Scholar] [CrossRef] [PubMed]
- Passing, H.; Bablok. A new biometrical procedure for testing the equality of measurements from two different analytical methods. Application of linear regression procedures for method comparison studies in clinical chemistry, Part I. J. Clin. Chem. Clin. Biochem. 1983, 21, 709–720. [Google Scholar] [CrossRef] [PubMed]
- Shen, H.Y.; Gao, W.; Cheng, J.J.; Zhao, S.D.; Sun, Y.; Han, Z.J.; Hua, J. Direct comparison of the diagnostic accuracy between blood and cerebrospinal fluid procalcitonin levels in patients with meningitis. Clin. Biochem. 2015, 48, 1079–1082. [Google Scholar] [CrossRef] [PubMed]
- Wei, T.T.; Hu, Z.D.; Qin, B.D.; Ma, N.; Tang, Q.Q.; Wang, L.L.; Zhou, L.; Zhong, R.Q. Diagnostic Accuracy of Procalcitonin in Bacterial Meningitis Versus Nonbacterial Meningitis: A Systematic Review and Meta-Analysis. Medicine (Baltimore) 2016, 95, e3079. [Google Scholar] [CrossRef] [PubMed]
- Alons, I.M.; Verheul, R.J.; Kuipers, I.; Jellema, K.; Wermer, M.J.H.; Algra, A.; Ponjee, G. Procalcitonin in cerebrospinal fluid in meningitis: A prospective diagnostic study. Brain Behav. 2016, 6, e00545. [Google Scholar] [CrossRef] [PubMed]
- Konstantinidis, T.; Cassimos, D.; Gioka, T.; Tsigalou, C.; Parasidis, T.; Alexandropoulou, L.; Nikolaidis, C.; Kampouromiti, G.; Constantinidis, T.; Chatzimichael, A.; et al. Can Procalcitonin in Cerebrospinal Fluid be a Diagnostic Tool for Meningitis? J. Clin. Lab. Anal. 2015, 29, 169–174. [Google Scholar] [CrossRef]
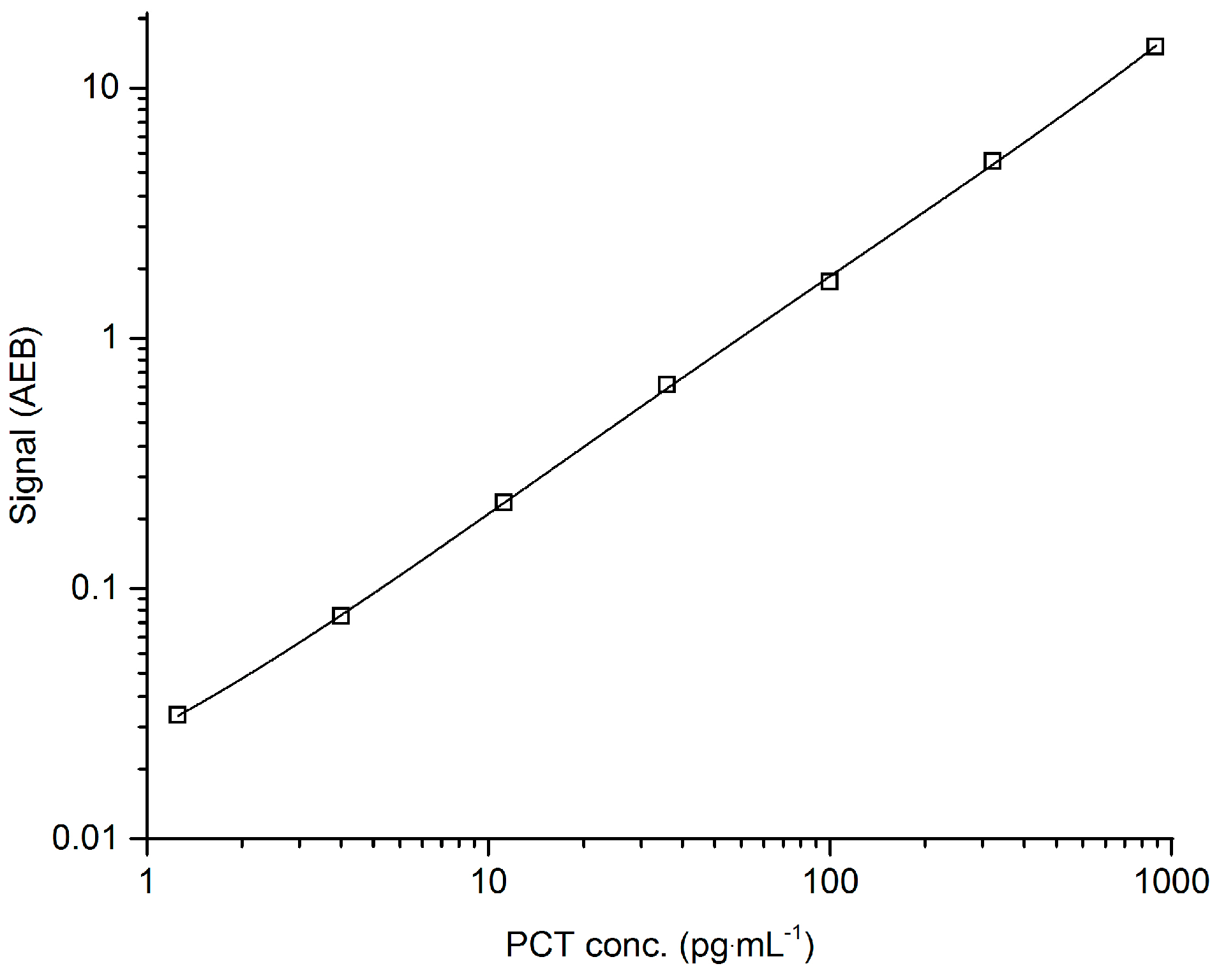
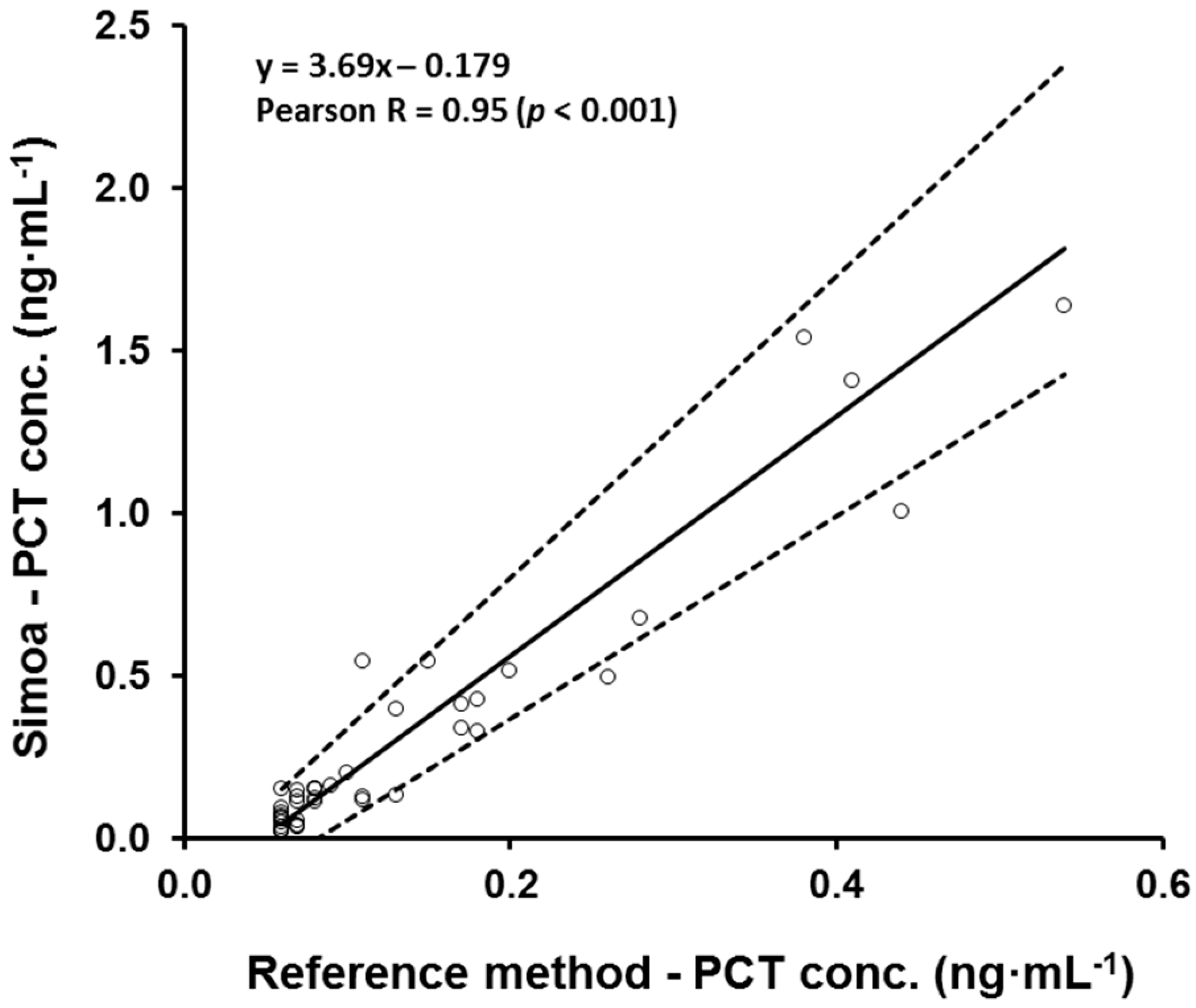
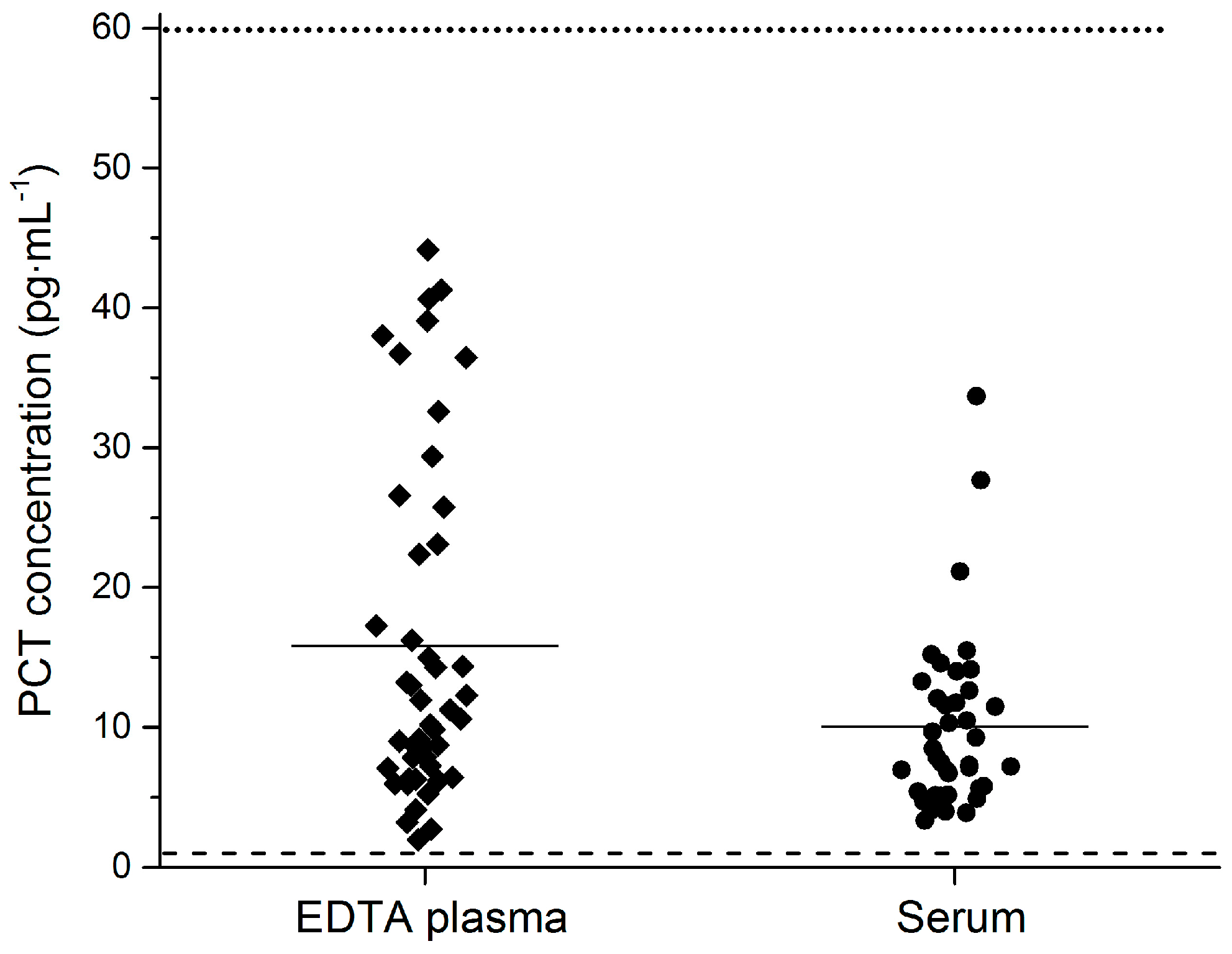
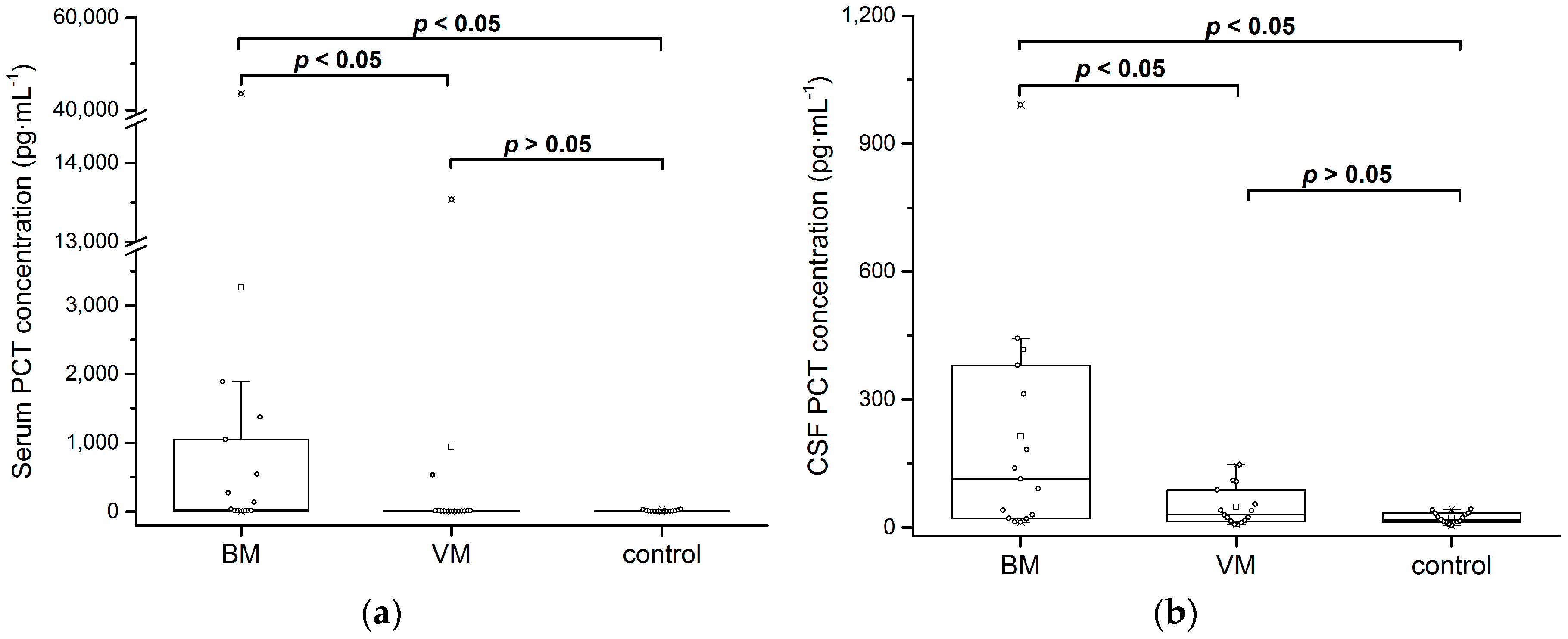
| Validation Parameter | Acceptance Criteria | Results | Conclusion |
|---|---|---|---|
| Calibration model | 80–120% recovery CV < 20% | four-parameteric logistics (1/Y2) 99–101% recovery CV < 20% | Complies |
| ULoQ | 80–120% recovery CV < 20% | 900 pg·mL−1 102% recovery CV 5.0% | Complies |
| LLoQ | 80–120% recovery CV < 20% | 1.23 pg·mL−1 101% recovery CV 15.3% | Complies |
| LoD (pg·mL−1) | zero calibrator +3× SD | 0.44 pg·mL−1 | |
| Intra-assay precision | mid-range: CV < 15% high-/low-range: CV < 20% | CV 13.5% CV 12.9–15.3% | Complies |
| Inter-assay precision | CV < 20% | CV 10.7–19.4% | Complies |
| Recovery | 80–120% recovery | 90–99% recovery | Complies |
| Dilutional linearity | 80–120% recovery | 82–117% recovery | Complies |
| Parallelism | 80–120% recovery | 91–115% recovery | Complies |
| Analyte stability | 80–120% recovery | 83–105% recovery | Complies |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carcamo Yañez, V.A.; Göpfert, J.C.; Otto, M.; Tumani, H.; Peter, A.; Joos, T.O. Development and Validation of an Ultrasensitive Procalcitonin Sandwich Immunoassay. High-Throughput 2017, 6, 18. https://doi.org/10.3390/ht6040018
Carcamo Yañez VA, Göpfert JC, Otto M, Tumani H, Peter A, Joos TO. Development and Validation of an Ultrasensitive Procalcitonin Sandwich Immunoassay. High-Throughput. 2017; 6(4):18. https://doi.org/10.3390/ht6040018
Chicago/Turabian StyleCarcamo Yañez, Viviana A., Jens C. Göpfert, Markus Otto, Hayrettin Tumani, Andreas Peter, and Thomas O. Joos. 2017. "Development and Validation of an Ultrasensitive Procalcitonin Sandwich Immunoassay" High-Throughput 6, no. 4: 18. https://doi.org/10.3390/ht6040018
APA StyleCarcamo Yañez, V. A., Göpfert, J. C., Otto, M., Tumani, H., Peter, A., & Joos, T. O. (2017). Development and Validation of an Ultrasensitive Procalcitonin Sandwich Immunoassay. High-Throughput, 6(4), 18. https://doi.org/10.3390/ht6040018





