ICS/Ultra LABA in the Treatment of Obstructive Airway Diseases: A Consensus of Indian Experts
Abstract
Highlights
- FF/Vi is an effective choice for patients who are uncontrolled on their current Asthma treatment and also for controlled Asthma patients.
- SABA or ICS/SABA can be a viable reliever option when personalizing treatment for appropriate sets of patients with FF/Vi.
- FF/Vi offers a definitive advantage in terms of treatment adherence and compliance benefit as compared to the conventional ICS/LABAs.
- Adding LAMA to FF/Vi (100/25 μg) in GOLD group D and B COPD patients can be an optimized strategy.
Abstract
1. Background
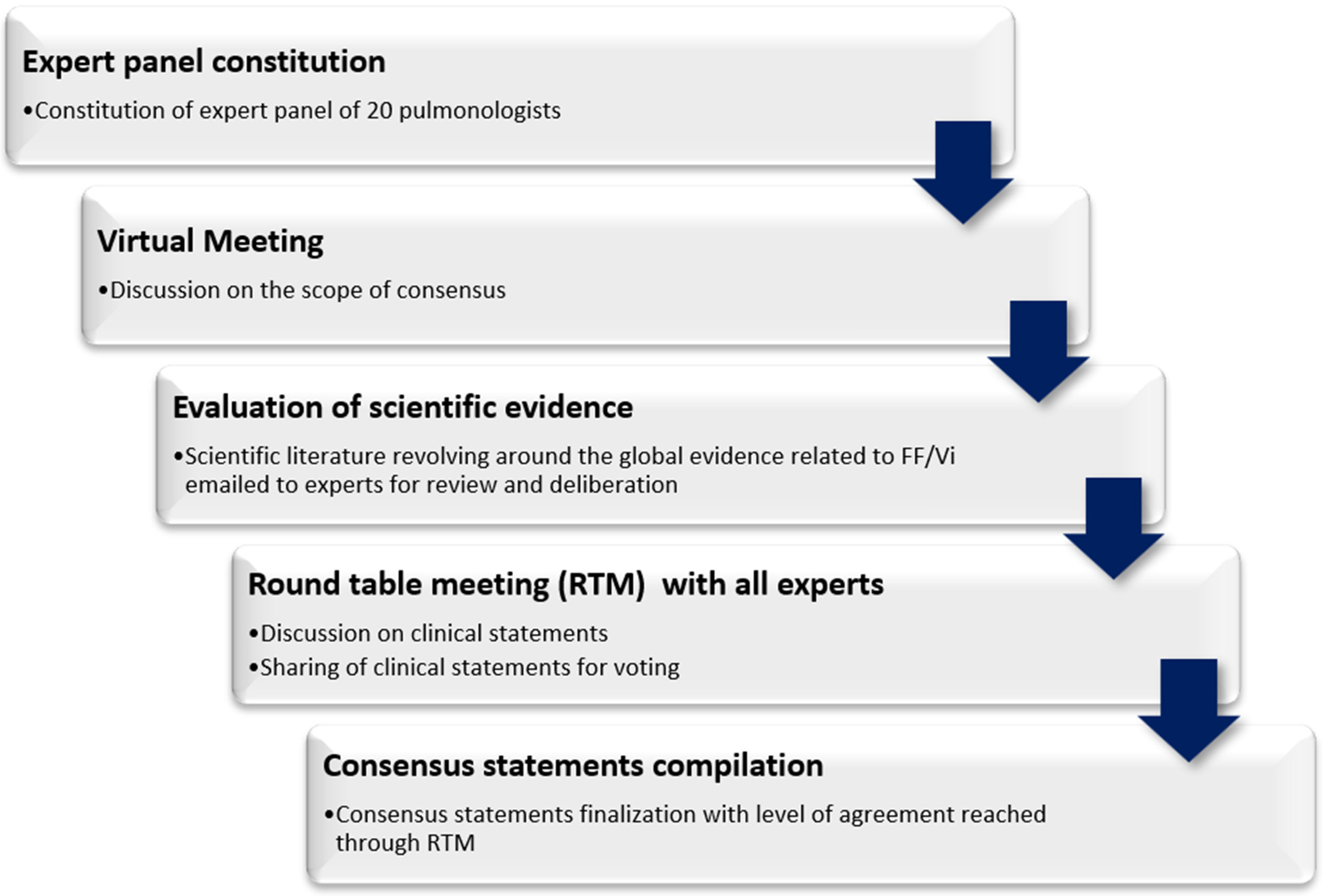
2. Methodology
3. Results and Discussion
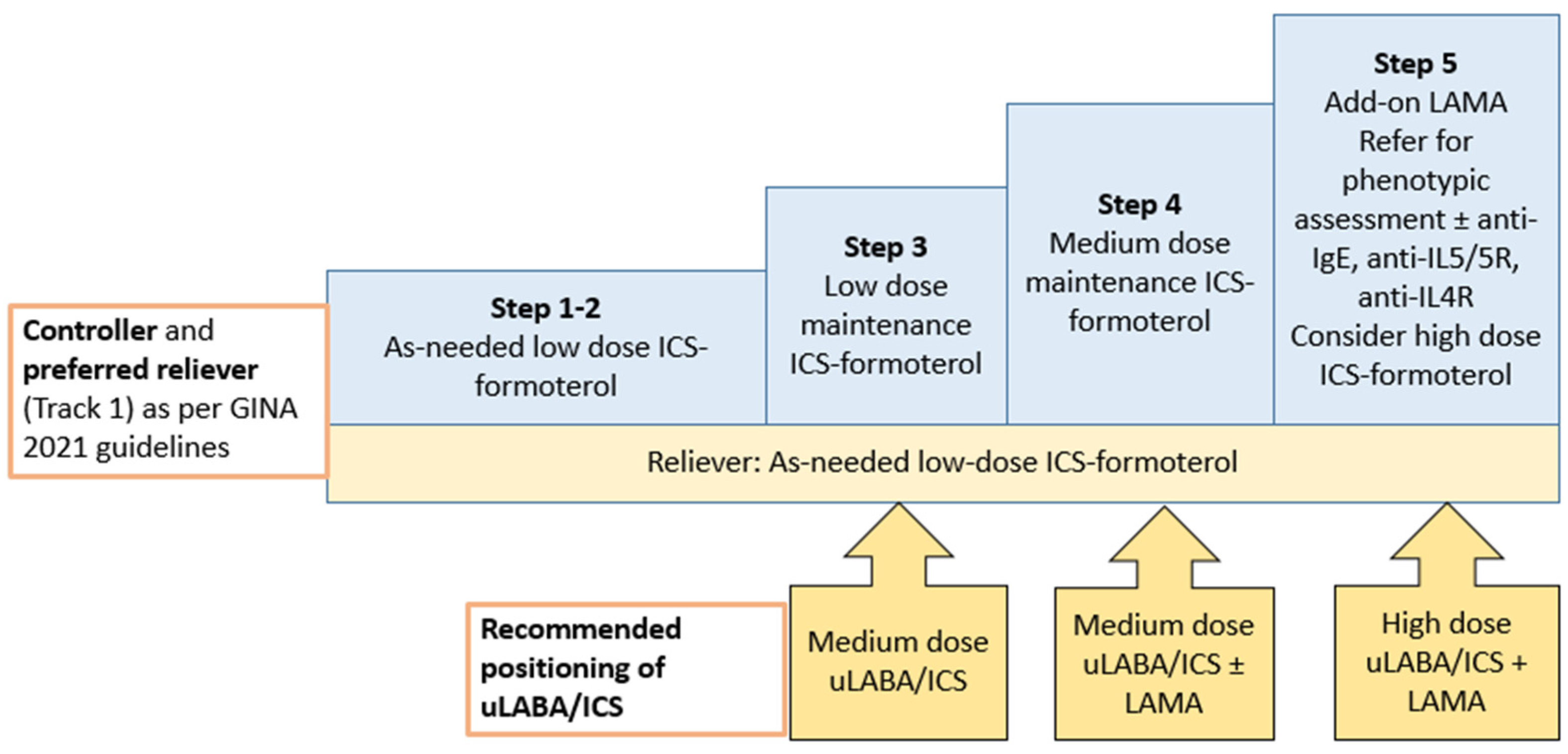
3.1. Asthma
3.1.1. Comparison of FF/Vi to Current Gold Standard (BUD/FOR) in Asthma
- Rescue-medication use is a key indicator of Asthma control. When compared with BUD/FOR, FF/Vi was associated with a 10% reduction in the use of rescue medication (SABA) in real-world settings [14].
- When measured using the Asthma Quality of Life Questionnaire (AQLQ), 27% more patients improved their quality of life versus conventional ICS/LABAs in everyday practice [29].
- For uncontrolled asthma patients (Step 3-4) on BUD/FOR → Switch to FF/Vi (IA)
- Stable Asthma (Step 3–4) on BUD/FOR → Shared decision making with patient on choice of treatment (IIC)
3.1.2. Reliever Use with FF/Vi in Asthma Patients
- For treatment naïve patients, SABA must be used as a reliever with FF/Vi (IA)
- For previously BUD/FOR treated patients switched to FF/Vi as maintenance, ICS-SABA or BUD/FOR to be used as reliever (IIC)
3.1.3. Adding OD LAMA to FF/Vi or High Dose FF/Vi in Uncontrolled Severe Asthma as Optimized Strategy
- Adding LAMA to FF/Vi improved lung function in patients with moderate or severe Asthma uncontrolled on conventional ICS/LABA. There are higher odds of Asthma control when LAMA is added to FF/Vi. The mean annualized rate of exacerbations was found to be 0.31 for both FF/Vi/UMEC (100 and 200) and for FF/Vi (100 and 200), with a 2.6% reduction in rate with FF/Vi/UMEC compared with FF/Vi.
- ACQ-7 responders at 24 weeks were numerically higher (62%) with LAMA added to FF/Vi (100/25) as compared to higher dose of FF/Vi (200/25) when used alone (58%)
- Numerically greater improvements in clinic-trough FEV1 were observed with FF/UMEC/Vi 100/62.5/25 μg versus FF/Vi 200/25 μg across the baseline eosinophil and FeNO ranges.
- There is now substantial evidence on the efficacy and safety of LAMAs in uncontrolled Asthma notwithstanding treatment with ICS/LABA combinations. This regimen is recommended by GINA as an optimization step for patients with severe Asthma before any biologic or systemic corticosteroid treatment is initiated.
- Addition of LAMA to FF/Vi should be considered preferentially for patients with persistent airflow limitation and bronchodilator reversibility, independent of blood eosinophil and/or FENO levels (IIA)
- Step-up to high-dose ICS should be considered particularly in patients with increased eosinophil (>300 cells/μL) and/or FENO levels → FF/Vi (100/25) to FF/Vi (200/25) (IA)
3.1.4. Triple Therapy in Asthma—Open-Triple Versus Single-Inhaler-Triple Therapy?
- Poorly controlled symptomatic patients could be effectively treated with triple combination therapies including different strengths of an ICS according to the severity of previous exacerbations (IA)
- Physicians may exercise both open triple and SITT options in clinical practice as per the clinical need and patient preference (IC)
- Flexibility for dose titration, especially of ICS
- Flexibility for choice of components
- Split ICS/LABA & LAMA as am/pm dosing in select patients
- Optimal synergy benefit of giving LABA/LAMA/ICS together
- Better compliance due to less frequent dosing
- Reduced device related errors
3.2. COPD
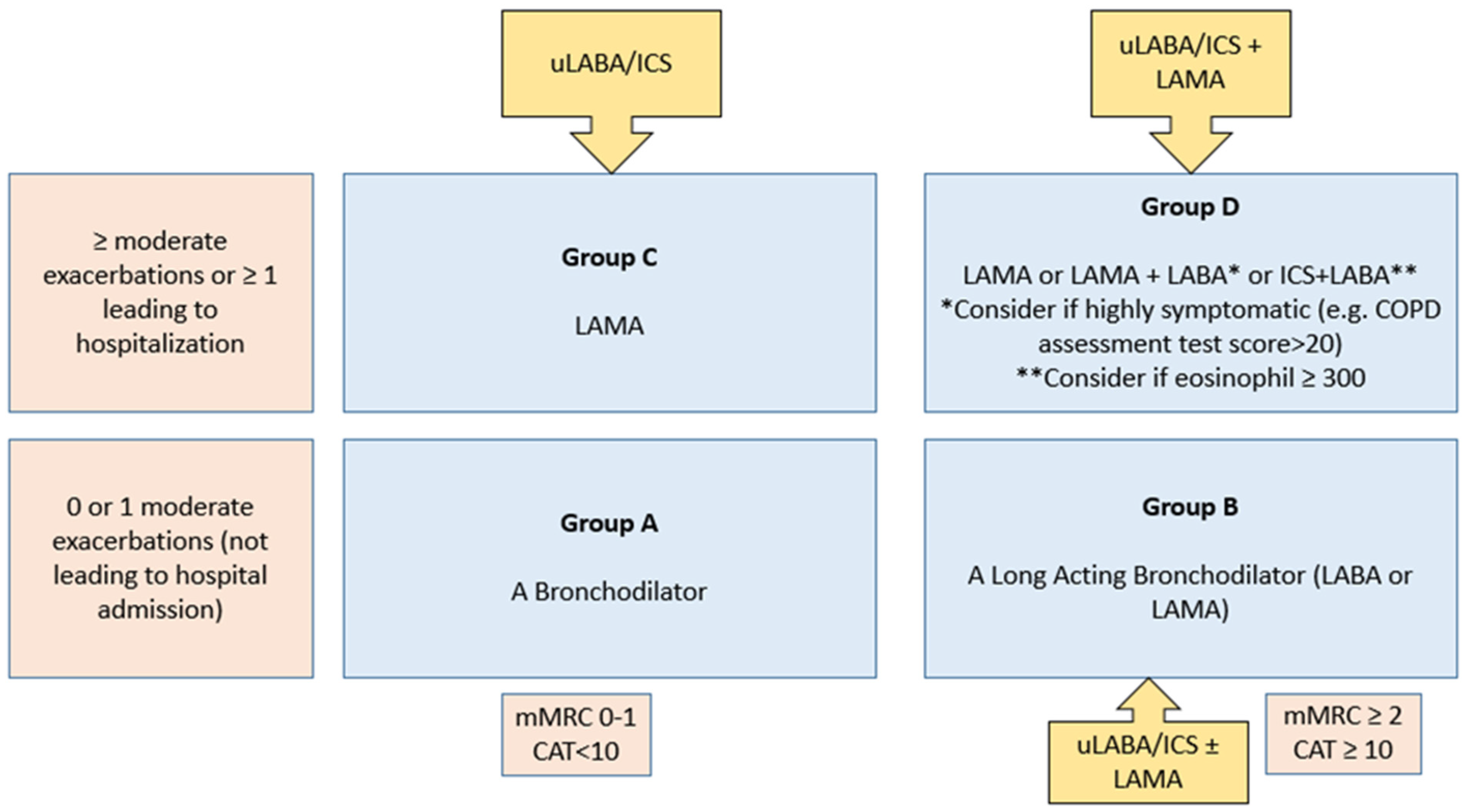
3.2.1. Profiling of the Patients for Use of FF/Vi in COPD
- ICS use based on clinical and biomarker profiling of COPD patient. FF/Vi has potential to use across clinical spectrum of COPD proposed by experts is as below and in Figure 3.
- GOLD Group D—FF/Vi can be used with add on LAMA (IA)
- GOLD Group B—FF/Vi can be used without or with add on LAMA driven by BEC, smoking status, ACO, pre-existing ICS therapy (IIA)
- GOLD Group C—FF/Vi alone can be used (IIB)
- Pre-existing ICS/LABA therapy—Switch to FF/Vi after patient profiling by shared decision making (IIC)
3.2.2. Pneumonia Risk with FF/Vi in COPD
- A real-world effectiveness study in COPD patients showed that there was no increase in the risk of pneumonia in FF/Vi group as compared to the usual care group [18].
- Another large clinical trial conducted in patients with moderate COPD and heightened cardiovascular disease risk showed that the incidence of pneumonia was comparable in the placebo, fluticasone furoate, vilanterol and FF/Vi groups [20].
- A recent study showed that the risk of pneumonia with fluticasone furoate 100 μg (RR = 1.39) was significantly lesser than the 200 μg dose (RR = 1.90). The same study also showed that fluticasone furoate 100 μg was safer in terms of pneumonia risk as compared to fluticasone propionate 500 μg (RR = 1.80) and 1000 μg (RR = 1.64) and was closer to 800 μg budesonide (RR = 1.26) [41].
- There is a clinical dilemma while diagnosing exacerbation vs pneumonia in COPD patients. Along with clinical signs, radiological diagnosis must be considered for distinctive diagnosis of pneumonia while ruling out exacerbation (IA)
- The choice of the ICS prescribed should not solely rely upon background incidence of pneumonia in COPD patients. An individualized risk benefit assessment should be performed considering risk of exacerbation (IA)
- There is a lesser risk of pneumonia with fluticasone furoate (100 µg) as compared to fluticasone propionate in COPD patients (IA)
3.2.3. Benefits of FF/Vi (ICS/uLABA) as Compared to Conventional ICS/LABA Combinations
- (a)
- Exacerbation reduction
- Real-world evidence suggests that FF/Vi provides a significant exacerbation benefit. A large-scale effectiveness study showed that the rate of moderate or severe exacerbations is significantly lower, by 8.4% in the FF/Vi group as compared to the usual care group. Patients switched from an ICS-containing regimen (FP/SAL or BUD/FOR) showed the greater benefit [18].
- Further evidence in the form of a clinical trial showed that FF/Vi with or without LAMA had better and consistent protection than LABA/LAMA group against moderate-to-severe exacerbations in patients with increasing baseline eosinophil count [19].
- A large observational study showed that FF/Vi was associated with a 14% lower risk of having a COPD-related moderate or severe exacerbation compared to BUD/FOR [21].
- As far as the single inhaler triple combination is concerned, rate ratio for moderate to severe exacerbations in FF/VI/UMEC group was comparable to that of BUD/FORM/Glycopyrronium (rate ratio = 0.99 in both the groups) [44].
- (b)
- Lung-function improvement
- A clinical trial in COPD patients showed that the rate of lung-function decline decreased by 8 mL/year in the FF/Vi group as compared to the placebo group (p = 0.019) [20].
- Evidence also shows that weighted mean (wm) FEV1 (mean 130 mL) is greater and time to 100 mL improvement shorter (median 16 min) with FF/Vi as compared to FP/SAL (weighted mean 108 mL, median 28 min) [47].
- A recent study showed that once-daily FF/Vi improved trough FEV1 by about 230 mL as compared to a placebo in COPD patients. However, other trials suggest a more modest increase (100–130 mL) in FEV1 [48].
- (c)
- Symptom control
- (d)
- Quality-of-life improvement
- IMPACT trial was the first large trial reporting mortality data with the use of triple therapy in the treatment of COPD. The trial showed that triple therapy offered significant mortality benefits as compared to LABA/LAMA (HR = 0.58). The trial also showed that dual therapy with FF/Vi provided a significant mortality benefit versus LABA/LAMA (HR = 0.61) [51].
- The SUMMIT trial was conducted in patients with moderate COPD and heightened cardiovascular disease risk showed that all-cause mortality with FF/Vi was 12.2% lower as compared to a placebo group but was not statistically significant (p = 0.137). However, the all-cause mortality was significantly lower in the FF/Vi group (HR = 0.76) among subjects with a Summit Score of 14 to 19. The SUMMIT trial is one of the largest randomized controlled COPD trials, with more than 16,000 patients [20].
- The results of recent network meta-analysis provides high quality evidence that both MD ICS/LABA/LAMA and MD ICS/LABA FDCs were significantly effective in reducing on-treatment all-cause of death, whereas only MD ICS/LABA/LAMA FDC significantly prevented adjudicated cardiovascular mortality. Indeed, the protective effect against mortality was related to the dose of the ICS in the FDC. A total of ~125 COPD patients had to be treated for one year with an MD ICS-containing combination to prevent one death compared with LABA/LAMA FDC. Given the importance of the outcome “mortality”, the specific context of COPD characterized by a high prevalence, and the safety profile of MD ICS-containing FDCs, an NNT of 125 seems to be more than acceptable and of clinically relevant magnitude [52].
- (e)
- Safety benefits
- (f)
- Adherence and compliance benefit
- The exacerbation and symptom control benefit showed by FF/Vi in the real world studies outweighs the benefit from usual ICS/LABAs (IIA)
- FF/Vi to be used over usual ICS/LABAs in patients with cardiovascular morbidity because of its beneficial effects on the cardiovascular system (IB)
- FF/Vi offers a definitive advantage in terms of adherence and compliance benefit as compared to the other ICS/LABAs (IA)
- Since COPD is a chronic disease and needs long term treatment, experts suggested that once daily dosing would be the greatest advantage offered by FF/Vi (IC)
3.2.4. Triple Therapy in COPD—Open-Triple Versus Single-Inhaler-Triple Therapy?
- Add LAMA to FF/VI (100/25 μg) in group D and B COPD patients as an optimized strategy (IIA)However, the de-escalation strategy needs to be defined while considering an improvement in clinical symptoms.
- Open triple therapy to be a good strategy in unstable COPD (post exacerbation requiring hospitalization) patients as it allows to titrate the dose of ICS, it also gives flexibility to prescribe ICS/LABA in the morning and LAMA in the evening in those where exacerbation is predominant over dyspnea (IC)
- (a)
- International guidelines (GINA/GOLD) do not lay much importance and do not differentiate ICS/uLABAs from conventional ICS/LABAs.
- (b)
- Clinical studies comparing SMART versus ICS/uLABAs in Asthma patients are lacking.
- (c)
- Possibility of using FF/Vi as a SMART strategy considering its quicker onset of action needs to be evaluated.
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Coleman, C.I.; Limone, B.; Sobieraj, D.M.; Lee, S.; Roberts, M.S.; Kaur, R.; Alam, T. Dosing Frequency and Medication Adherence in Chronic Disease. JMCP 2012, 18, 527–539. [Google Scholar] [CrossRef] [PubMed]
- Restrepo, R. Medication adherence issues in patients treated for COPD. COPD 2008, 3, 371–384. [Google Scholar] [CrossRef]
- Slack, R.J.; Barrett, V.J.; Morrison, V.S.; Sturton, R.G.; Emmons, A.J.; Ford, A.J.; Knowles, R.G. In Vitro Pharmacological Characterization of Vilanterol, a Novel Long-Acting β2-Adrenoceptor Agonist with 24-Hour Duration of Action. J. Pharm. Exp. 2013, 344, 218–230. [Google Scholar] [CrossRef]
- Rogliani, P.; Calzetta, L.; Braido, F.; Cazzola, M.; Clini, E.; Pelaia, G.; Rossi, A.; Scichilone, N.; Di Marco, F. LABA/LAMA fixed-dose combinations in patients with COPD: A systematic review. COPD 2018, 13, 3115–3130. [Google Scholar] [CrossRef]
- Calzetta, L.; Rinaldi, B.; Cazzola, M.; Matera, M.G. Pharmacodynamic and pharmacokinetic assessment of fluticasone furoate + vilanterol for the treatment of asthma. Expert. Opin. Drug Metab. Toxicol. 2016, 12, 813–822. [Google Scholar] [CrossRef]
- Daley-Yates, P.; Brealey, N.; Thomas, S.; Austin, D.; Shabbir, S.; Harrison, T.; Singh, D.; Barnes, N. Therapeutic index of inhaled corticosteroids in asthma: A dose–response comparison on airway hyperresponsiveness and adrenal axis suppression. Br. J. Clin. Pharm. 2021, 87, 483–493. [Google Scholar] [CrossRef] [PubMed]
- Woodcock, A.; Bateman, E.D.; Busse, W.W.; Lötvall, J.; Snowise, N.G.; Forth, R.; Jacques, L.; Haumann, B.; Bleecker, E.R. Efficacy in asthma of once-daily treatment with fluticasone furoate: A randomized, placebo-controlled trial. Respir Res. 2011, 12, 132. [Google Scholar] [CrossRef]
- Breo Ellipta. Highlights of Prescribing Information 2019. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/204275s017lbl.pdf (accessed on 10 March 2021).
- Relvar Ellipta. Summary of Product Characteristics. 2013. Available online: https://www.ema.europa.eu/en/documents/product-information/relvar-ellipta-epar-product-information_en.pdf (accessed on 11 March 2021).
- Form CT-23; Grant of Permission to Manufacture and Market FDC of Fluticaosne Furoate + Vilanterol Trifenatate eq. to Vilanterol Dry Powder for Inhalation in Capsule. Government of India; Central Drugs Standard Control Organisation: New Delhi, India, 2022.
- Kerwin, E.M.; Scott-Wilson, C.; Sanford, L.; Rennard, S.; Agusti, A.; Barnes, N.; Crim, C. A randomised trial of fluticasone furoate/vilanterol (50/25 μg; 100/25 μg) on lung function in COPD. Respir. Med. 2013, 107, 560–569. [Google Scholar] [CrossRef]
- Martinez, F.J.; Boscia, J.; Feldman, G.; Scott-Wilson, C.; Kilbride, S.; Fabbri, L.; Crim, C.; Calverley, P.M.A. Fluticasone furoate/vilanterol (100/25; 200/25 μg) improves lung function in COPD: A randomised trial. Respir. Med. 2013, 107, 550–559. [Google Scholar] [CrossRef]
- Furuhashi, K.; Fujisawa, T.; Hashimoto, D.; Kamiya, Y.; Yasui, H.; Karayama, M.; Suzuki, Y.; Hozumi, H.; Enomoto, N.; Nakamura, Y.; et al. Once-daily fluticasone furoate/vilanterol combination versus twice-daily budesonide/formoterol combination in the treatment of controlled stable asthma: A randomized crossover trial. JAA 2019, 12, 253–261. [Google Scholar] [CrossRef]
- Averell, C.M.; Laliberté, F.; Germain, G.; Duh, M.S.; Lima, R.; Mahendran, M.; Slade, D.J. Symptom control in patients with asthma using inhaled corticosteroids/long-acting β2-agonists (fluticasone furoate/vilanterol or budesonide/formoterol) in the US: A retrospective matched cohort study. J. Asthma 2021, 59, 1805–1818. [Google Scholar] [CrossRef] [PubMed]
- Global Strategy for Asthma Prevention Report 2021. Available online: https://ginasthma.org/gina-reports/ (accessed on 19 October 2021).
- Global Initiative for Chronic Obstructive Lung Disease COPD Guidelines 2021. Available online: https://goldcopd.org (accessed on 18 October 2021).
- Knuuti, J.; Wijns, W.; Saraste, A.; Capodanno, D.; Barbato, E.; Funck-Brentano, C.; Prescott, E.; Storey, R.F.; Deaton, C.; Cuisset, T.; et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes. Eur. Heart J. 2020, 41, 407–477. [Google Scholar] [CrossRef] [PubMed]
- Vestbo, J.; Leather, D.; Diar Bakerly, N.; New, J.; Gibson, J.M.; McCorkindale, S.; Collier, S.; Crawford, J.; Frith, L.; Harvey, C.; et al. Effectiveness of Fluticasone Furoate–Vilanterol for COPD in Clinical Practice. N. Engl. J. Med. 2016, 375, 1253–1260. [Google Scholar] [CrossRef]
- Lipson, D.A.; Barnhart, F.; Brealey, N.; Brooks, J.; Criner, G.J.; Day, N.C.; Dransfield, M.T.; Halpin, D.M.G.; Han, M.K.; Jones, C.E.; et al. Once-Daily Single-Inhaler Triple versus Dual Therapy in Patients with COPD. N. Engl. J. Med. 2018, 378, 1671–1680. [Google Scholar] [CrossRef]
- Vestbo, J.; Anderson, J.A.; Brook, R.D.; Calverley, P.M.A.; Celli, B.R.; Crim, C.; Martinez, F.; Yates, J.; Newby, D.E. Fluticasone furoate and vilanterol and survival in chronic obstructive pulmonary disease with heightened cardiovascular risk (SUMMIT): A double-blind randomised controlled trial. Lancet 2016, 387, 1817–1826. [Google Scholar] [CrossRef]
- Stanford, R.H.; Parker, E.D.; Reinsch, T.K.; Buikema, A.R.; Blauer-Peterson, C. Assessment of COPD-related outcomes in patients initiating a once daily or twice daily ICS/LABA. Respir. Med. 2019, 150, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Woodcock, A.; Vestbo, J.; Bakerly, N.D.; New, J.; Gibson, J.M.; McCorkindale, S.; Jones, R.; Collier, S.; Lay-Flurrie, J.; Frith, L.; et al. Effectiveness of fluticasone furoate plus vilanterol on asthma control in clinical practice: An open-label, parallel group, randomised controlled trial. Lancet 2017, 390, 2247–2255. [Google Scholar] [CrossRef]
- Lee, L.A.; Bailes, Z.; Barnes, N.; Boulet, L.-P.; Edwards, D.; Fowler, A.; Hanania, N.A.; Kerstjens, H.A.M.; Kerwin, E.; Nathan, R.; et al. Efficacy and safety of once-daily single-inhaler triple therapy (FF/UMEC/VI) versus FF/VI in patients with inadequately controlled asthma (CAPTAIN): A double-blind, randomised, phase 3A trial. Lancet Respir. Med. 2021, 9, 69–84. [Google Scholar] [CrossRef]
- Chapman, K.R.; Barnes, N.C.; Greening, A.P.; Jones, P.W.; Pedersen, S. Single maintenance and reliever therapy (SMART) of asthma: A critical appraisal. Thorax 2010, 65, 747–752. [Google Scholar] [CrossRef]
- Lipworth, B.; Chan, R.; Kuo, C.R. Anti-inflammatory reliever therapy for asthma. Ann. Allergy Asthma Immunol. 2020, 124, 13–15. [Google Scholar] [CrossRef]
- Chapman, K.R.; An, L.; Bosnic-Anticevich, S.; Campomanes, C.M.; Espinosa, J.; Jain, P.; Lavoie, K.L.; Li, J.; Butta, A.K. Asthma patients’ and physicians’ perspectives on the burden and management of asthma. Respir Med. 2021, 186, 106524. [Google Scholar] [CrossRef] [PubMed]
- Bianco, A.; Contoli, M.; Di Marco, F.; Saverio Mennini, F.; Papi, A. Participants of the regional meetings As-needed anti-inflammatory reliever therapy for asthma management: Evidence and practical considerations. Clin. Exp. Allergy 2021, 51, 873–882. [Google Scholar] [CrossRef] [PubMed]
- Bateman, E.D.; O’Byrne, P.M.; Busse, W.W.; Lötvall, J.; Bleecker, E.R.; Andersen, L.; Jacques, L.; Frith, L.; Lim, J.; Woodcock, A. Once-daily fluticasone furoate (FF)/vilanterol reduces risk of severe exacerbations in asthma versus FF alone. Thorax 2014, 69, 312–319. [Google Scholar] [CrossRef] [PubMed]
- Svedsater, H.; Jones, R.; Bosanquet, N.; Jacques, L.; Lay-Flurrie, J.; Leather, D.A.; Vestbo, J.; Collier, S.; Woodcock, A. Patient-reported outcomes with initiation of fluticasone furoate/vilanterol versus continuing usual care in the Asthma Salford Lung Study. Respir. Med. 2018, 141, 198–206. [Google Scholar] [CrossRef]
- Fletcher, M.; Hiles, D. Continuing discrepancy between patient perception of asthma control and real-world symptoms: A quantitative online survey of 1083 adults with asthma from the UK. Prim. Care Respir. J. 2013, 22, 431–438. [Google Scholar] [CrossRef]
- Langley, R.J.; Dryden, C.; Westwood, J.; Anderson, E.; Thompson, A.; Urquhart, D. Once daily combined inhaled steroid and ultra long-acting bronchodilator prescribing in pediatric asthma: A dual Center retrospective cohort study. J. Asthma 2021, 58, 512–513. [Google Scholar] [CrossRef]
- Bernstein, D.I.; Bateman, E.D.; Woodcock, A.; Toler, W.T.; Forth, R.; Jacques, L.; Nunn, C.; O’Byrne, P.M. Fluticasone furoate (FF)/vilanterol (100/25 mcg or 200/25 mcg) or FF (100 mcg) in persistent asthma. J. Asthma 2015, 52, 1073–1083. [Google Scholar] [CrossRef]
- Braithwaite, I.; Williams, M.; Power, S.; Pilcher, J.; Weatherall, M.; Baines, A.; Moynihan, J.; Kempsford, R.; Beasley, R. Randomised, double-blind, placebo-controlled, cross-over single dose study of the bronchodilator duration of action of combination fluticasone furoate/vilanterol inhaler in adult asthma. Respir. Med. 2016, 119, 115–121. [Google Scholar] [CrossRef][Green Version]
- Bardsley, G.; Daley-Yates, P.; Baines, A.; Kempsford, R.; Williams, M.; Mallon, T.; Braithwaite, I.; Riddell, K.; Joshi, S.; Bareille, P.; et al. Anti-inflammatory duration of action of fluticasone furoate/vilanterol trifenatate in asthma: A cross-over randomised controlled trial. Respir Res. 2018, 19, 133. [Google Scholar] [CrossRef]
- Papi, A.; Fabbri, L.M.; Kerstjens, H.A.M.; Rogliani, P.; Watz, H.; Singh, D. Inhaled long-acting muscarinic antagonists in asthma—A narrative review. Eur. J. Intern. Med. 2021, 85, 14–22. [Google Scholar] [CrossRef]
- Rogliani, P.; Ritondo, B.L.; Calzetta, L. Triple therapy in uncontrolled asthma: A network meta-analysis of phase III studies. Eur. Respir. J. 2021, 58, 2004233. [Google Scholar] [CrossRef] [PubMed]
- Adcock, I.M.; Bhatt, S.P.; Balkissoon, R.; Wise, R.A. The Use of Inhaled Corticosteroids for Patients with COPD Who Continue to Smoke Cigarettes: An Evaluation of Current Practice. Am. J. Med. 2022, 135, 302–312. [Google Scholar] [CrossRef]
- Hartley, B.F.; Barnes, N.C.; Lettis, S.; Compton, C.H.; Papi, A.; Jones, P. Risk factors for exacerbations and pneumonia in patients with chronic obstructive pulmonary disease: A pooled analysis. Respir. Res. 2020, 21, 5. [Google Scholar] [CrossRef]
- Trethewey, S.P.; Hurst, J.R.; Turner, A.M. Pneumonia in exacerbations of COPD: What is the clinical significance? ERJ Open Res. 2020, 6, 00282–02019. [Google Scholar] [CrossRef] [PubMed]
- EMA Completes Review of Inhaled Corticosteroids for Chronic Obstructive Pulmonary Disease 2016. Available online: https://www.ema.europa.eu/en/news/ema-completes-review-inhaled-corticosteroids-chronic-obstructive-pulmonary-disease (accessed on 31 December 2021).
- Zhang, Q.; Li, S.; Zhou, W.; Yang, X.; Li, J.; Cao, J. Risk of Pneumonia with Different Inhaled Corticosteroids in COPD Patients: A Meta-Analysis. COPD J. Chronic. Obstr. Pulm. Dis. 2020, 17, 462–469. [Google Scholar] [CrossRef]
- Qureshi, H.; Sharafkhaneh, A.; Hanania, N.A. Chronic obstructive pulmonary disease exacerbations: Latest evidence and clinical implications. Ther. Adv. Chronic. Dis. 2014, 5, 212–227. [Google Scholar] [CrossRef]
- Kerkhof, M.; Voorham, J.; Dorinsky, P.; Cabrera, C.; Darken, P.; Kocks, J.W.; Sadatsafavi, M.; Sin, D.D.; Carter, V.; Price, D.B. The Long-Term Burden of COPD Exacerbations During Maintenance Therapy and Lung Function Decline. COPD 2020, 15, 1909–1918. [Google Scholar] [CrossRef]
- Ferguson, G.T.; Darken, P.; Ballal, S.; Siddiqui, M.K.; Singh, B.; Attri, S.; Holmgren, U.; de Nigris, E. Efficacy of Budesonide/Glycopyrronium/Formoterol Fumarate Metered Dose Inhaler (BGF MDI) Versus Other Inhaled Corticosteroid/Long-Acting Muscarinic Antagonist/Long-Acting β2-Agonist (ICS/LAMA/LABA) Triple Combinations in COPD: A Systematic Literature Review and Network Meta-analysis. Adv. Ther. 2020, 37, 2956–2975. [Google Scholar]
- Kakavas, S.; Kotsiou, O.S.; Perlikos, F.; Mermiri, M.; Mavrovounis, G.; Gourgoulianis, K.; Pantazopoulos, I. Pulmonary function testing in COPD: Looking beyond the curtain of FEV1. NPJ Prim. Care Respir. Med. 2021, 31, 23. [Google Scholar] [CrossRef]
- Corlateanu, A.; Botnaru, V.; Covantev, S.; Dumitru, S.; Siafakas, N. Predicting Health-Related Quality of Life in Patients with Chronic Obstructive Pulmonary Disease: The Impact of Age. Respiration 2016, 92, 229–234. [Google Scholar] [CrossRef]
- Agusti, A.; de Teresa, L.; De Backer, W.; Zvarich, M.T.; Locantore, N.; Barnes, N.; Bourbeau, J.; Crim, C. A comparison of the efficacy and safety of once-daily fluticasone furoate/vilanterol with twice-daily fluticasone propionate/salmeterol in moderate to very severe COPD. Eur. Respir. J. 2014, 43, 763–772. [Google Scholar] [CrossRef] [PubMed]
- Bollmeier, S.G.; Prosser, T.R. Combination of Fluticasone Furoate and Vilanterol for the Treatment of Chronic Obstructive Pulmonary Disease. Ann. Pharm. 2014, 48, 250–257. [Google Scholar] [CrossRef] [PubMed]
- Feliz-Rodriguez, D.; Zudaire, S.; Carpio, C.; Martínez, E.; Gómez-Mendieta, A.; Santiago, A.; Alvarez-Sala, R.; García-Río, F. Evolution of the COPD Assessment Test Score during Chronic Obstructive Pulmonary Disease Exacerbations: Determinants and Prognostic Value. Can. Respir. J. 2013, 20, e92–e97. [Google Scholar] [CrossRef]
- Stynes, G.; Svedsater, H.; Wex, J.; Lettis, S.; Leather, D.; Castelnuovo, E.; Detry, M.; Berry, S. Once-daily fluticasone furoate/vilanterol 100/25 mcg versus twice daily combination therapies in COPD—Mixed treatment comparisons of clinical efficacy. Respir. Res. 2015, 16, 25. [Google Scholar] [CrossRef][Green Version]
- Andreas, S.; Taube, C. Inhaled therapy reduces COPD mortality. ERJ Open Res. 2020, 6, 00634–02020. [Google Scholar] [CrossRef] [PubMed]
- Calzetta, L.; Ritondo, B.L.; de Marco, P.; Cazzola, M.; Rogliani, P. Evaluating triple ICS/LABA/LAMA therapies for COPD patients: A network meta-analysis of ETHOS, KRONOS, IMPACT, and TRILOGY studies. Expert. Rev. Respir. Med. 2021, 15, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Sin, D.D. Pharmacotherapy for Mortality Reduction in Chronic Obstructive Pulmonary Disease. Proc. Am. Thorac. Soc. 2006, 3, 624–629. [Google Scholar] [CrossRef]
- Stone, I.S.; Barnes, N.C.; James, W.-Y.; Midwinter, D.; Boubertakh, R.; Follows, R.; John, L.; Petersen, S.E. Lung Deflation and Cardiovascular Structure and Function in Chronic Obstructive Pulmonary Disease. A Randomized Controlled Trial. Am. J. Respir. Crit. Care Med. 2016, 193, 717–726. [Google Scholar] [CrossRef]
- López-Campos, J.L.; Quintana Gallego, E.; Carrasco Hernández, L. Status of and strategies for improving adherence to COPD treatment. COPD 2019, 14, 1503–1515. [Google Scholar] [CrossRef]
- Rogliani, P.; Ora, J.; Puxeddu, E.; Matera, M.G.; Cazzola, M. Adherence to COPD treatment: Myth and reality. Respir. Med. 2017, 129, 117–123. [Google Scholar] [CrossRef]
- Lipson, D.A.; Crim, C.; Criner, G.J.; Day, N.C.; Dransfield, M.T.; Halpin, D.M.G.; Han, M.K.; Jones, C.E.; Kilbride, S.; Lange, P.; et al. Reduction in All-Cause Mortality with Fluticasone Furoate/Umeclidinium/Vilanterol in Patients with Chronic Obstructive Pulmonary Disease. Am. J. Respir Crit Care Med. 2020, 201, 1508–1516. [Google Scholar] [CrossRef] [PubMed]
- Bremner, P.R.; Birk, R.; Brealey, N.; Ismaila, A.S.; Zhu, C.-Q.; Lipson, D.A. Single-inhaler fluticasone furoate/umeclidinium/vilanterol versus fluticasone furoate/vilanterol plus umeclidinium using two inhalers for chronic obstructive pulmonary disease: A randomized non-inferiority study. Respir. Res. 2018, 19, 19. [Google Scholar] [CrossRef] [PubMed]
- Salvi, S.; Balki, A.; Krishnamurthy, S.; Panchal, S.; Patil, S.; Kodgule, R.; Khandagale, H.; Pendse, A.; Wu, W.; Rangwala, S.; et al. Efficacy and safety of single-inhaler triple therapy of glycopyrronium, formoterol and fluticasone in patients with COPD: A double-blind, randomised controlled trial. ERJ Open Res. 2021, 7, 00255–02021. [Google Scholar] [CrossRef] [PubMed]
- Cazzola, M.; Rogliani, P.; Matera, M.G. Ultra-LABAs for the treatment of asthma. Respir. Med. 2019, 156, 47–52. [Google Scholar] [CrossRef] [PubMed]
| LABA/uLABA | Functional Selectivity (β2:β1) | Onset of Action (Minutes) |
|---|---|---|
| Formoterol | 130 | 5.9 |
| Salmeterol | 1595 | 13.7 |
| Indacaterol | 12.5 | 10.9 |
| Vilanterol | 2400 | 3.45 min |
| Summary: Vilanterol has a greater functional selectivity and quicker onset of action as compared to other LABAs/uLABA | ||
| Asthma |
|
| COPD |
|
| Class of Recommendation | Consensus Response | |
|---|---|---|
| I | Evidence and/or general agreement that a given treatment or procedure is beneficial, useful, effective | Agreement (It is recommended or is indicated) |
| II | Conflicting evidence and/or a divergence of opinion about the usefulness/efficacy of the given treatment or procedure | Conditional agreement (May be considered) |
| III | Evidence or general agreement that the given treatment or procedure is not useful/effective, and in some cases may be harmful | Disagreement (It is not recommended) |
| Level of evidence | ||
| A | Data derived from multiple randomized clinical trials or meta-analysis | |
| B | Data derived from a single randomized clinical trial or large non-randomized studies | |
| C | Consensus of opinion of the experts and/or small studies, retrospective studies, registries | |
| Indication (Year of Approval) | ||
|---|---|---|
| Regulator | FF/Vi (100/25 μg) | FF/Vi (200/25 μg) |
| US FDA | Asthma (≥18 years) (2015) and COPD (2013) | Asthma (≥18 years); 2015 |
| EMA | Asthma (≥12 years) and COPD; 2013 | Asthma (≥12 years); 2013 |
| TGA | Asthma (≥12 years) and COPD; 2014 | Asthma (≥12 years); 2014 |
| PMDA | Asthma (≥12 years) (2013) and COPD (2016) | Asthma (≥12 years); 2013 |
| DCGI, India | Asthma (≥12 years) (2017) * and COPD (2022) | Asthma (≥12 years); 2022 |
| Summary: FF/Vi has been recently approved and marketed for Asthma and COPD in India, while it has been marketed in countries such as US, Europe, Australia, Japan since 2013. | ||
| Clinical Trial | Trial Design and Endpoints—Primary/Secondary | Patient Population | Treatment and Duration | Key Findings |
|---|---|---|---|---|
| COPD | ||||
| SLS COPD [18] |
|
| FF/Vi (100/25 μg) DPI (n = 1291) versus Usual-care (UC) group (n = 1309) |
|
| IMPACT [19] |
|
| Triple therapy fluticasone furoate-vilanterol-umeclidinium (FF/Vi/UMEC); n = 4151 versus umeclidinium–vilanterol (UMEC/Vi); n = 2070 versus FF/Vi; n = 4134) |
|
| SUMMIT [20] |
|
| FF/Vi (n = 4121) versus FF (n = 4135) versus Vi (n = 4118) versus placebo (n = 4111) |
|
| Large observational study [21] |
|
| FF/Vi versus BUD/FOR (n = 4513 in each group) |
|
| Asthma | ||||
| SLS Asthma [22] |
score, both at week 52 |
| FF/Vi (100/25 μg) or FF/Vi (200/25 μg) (n = 2114) versus UC group (n = 2119) |
|
| CAPTAIN [23] |
|
| FF/Vi 100/25 μg group (n = 407) FF/Vi 200/25 μg group (n = 406) FF/UMEC/Vi 100/31·25/25 μg group (n = 405) FF/UMEC/Vi 100/62·5/25 μg group (n = 406) FF/UMEC/Vi 200/31·25/25 μg group (n = 404) FF/UMEC/Vi 200/62.5/25 μg group (n = 408) | Addition of UMEC 62·5 μg to FF/Vi 100/25 μg and FF/Vi 200/25 μg caused least squares mean change from baseline of 110 mL for FF/UMEC/Vi 100/62.5/25 μg and of 92 mL for FF/UMEC/Vi 200/62.5/25 μg in clinic trough FEV1 at week 24 ACQ-7 responder rate was comparable between FF/Vi (100/25) versus FF/Vi (200/25) |
| Crossover study [13] |
and FeNO at week 8. The incidence of Asthma exacerbation and adherence barrier questionnaire (Ask-12 survey) were also evaluated after 8 weeks. |
| FF/Vi DPI (100/25 μg) 1 puff once-daily versus BUD/FOR DPI treatment (160/4.5 μg) 2 puffs twice-daily |
|
| Summary: The efficacy and safety of FF/Vi in OAD has been established in several large-scale clinical trials including first pragmatic RCT for both Asthma and COPD. FF/Vi in COPD: Reduced rate of exacerbations and rate of decline of FEV1 in Group B/D patients, improved COPD-related health status. FF/Vi in Asthma: Improved Asthma control and quality of life, reduced rescue medication use and better adherence | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dhar, R.; Talwar, D.; James, P.; Mishra, A.; Vachaparambil, J.; Patil, S.; Khatri, N.; Bhagat, S.; Barkate, H. ICS/Ultra LABA in the Treatment of Obstructive Airway Diseases: A Consensus of Indian Experts. Adv. Respir. Med. 2022, 90, 407-424. https://doi.org/10.3390/arm90050051
Dhar R, Talwar D, James P, Mishra A, Vachaparambil J, Patil S, Khatri N, Bhagat S, Barkate H. ICS/Ultra LABA in the Treatment of Obstructive Airway Diseases: A Consensus of Indian Experts. Advances in Respiratory Medicine. 2022; 90(5):407-424. https://doi.org/10.3390/arm90050051
Chicago/Turabian StyleDhar, Raja, Deepak Talwar, Prince James, Ashwini Mishra, Judo Vachaparambil, Saiprasad Patil, Nishtha Khatri, Sagar Bhagat, and Hanmant Barkate. 2022. "ICS/Ultra LABA in the Treatment of Obstructive Airway Diseases: A Consensus of Indian Experts" Advances in Respiratory Medicine 90, no. 5: 407-424. https://doi.org/10.3390/arm90050051
APA StyleDhar, R., Talwar, D., James, P., Mishra, A., Vachaparambil, J., Patil, S., Khatri, N., Bhagat, S., & Barkate, H. (2022). ICS/Ultra LABA in the Treatment of Obstructive Airway Diseases: A Consensus of Indian Experts. Advances in Respiratory Medicine, 90(5), 407-424. https://doi.org/10.3390/arm90050051






