Effect of Produced Sand Particles and Fines on Scale Inhibitor: A Review
Abstract
1. Introduction
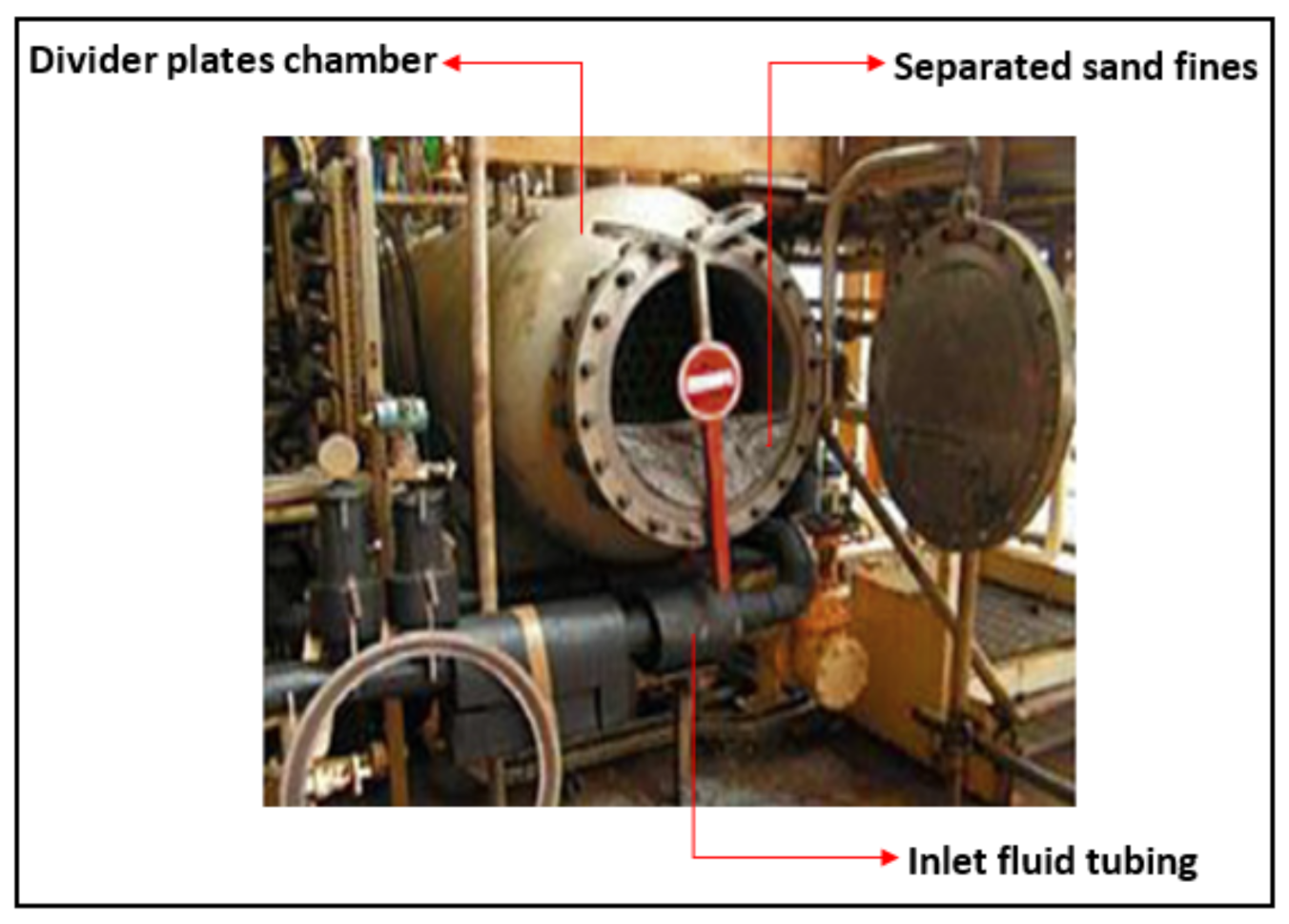
2. Fine Sand Production in Formations
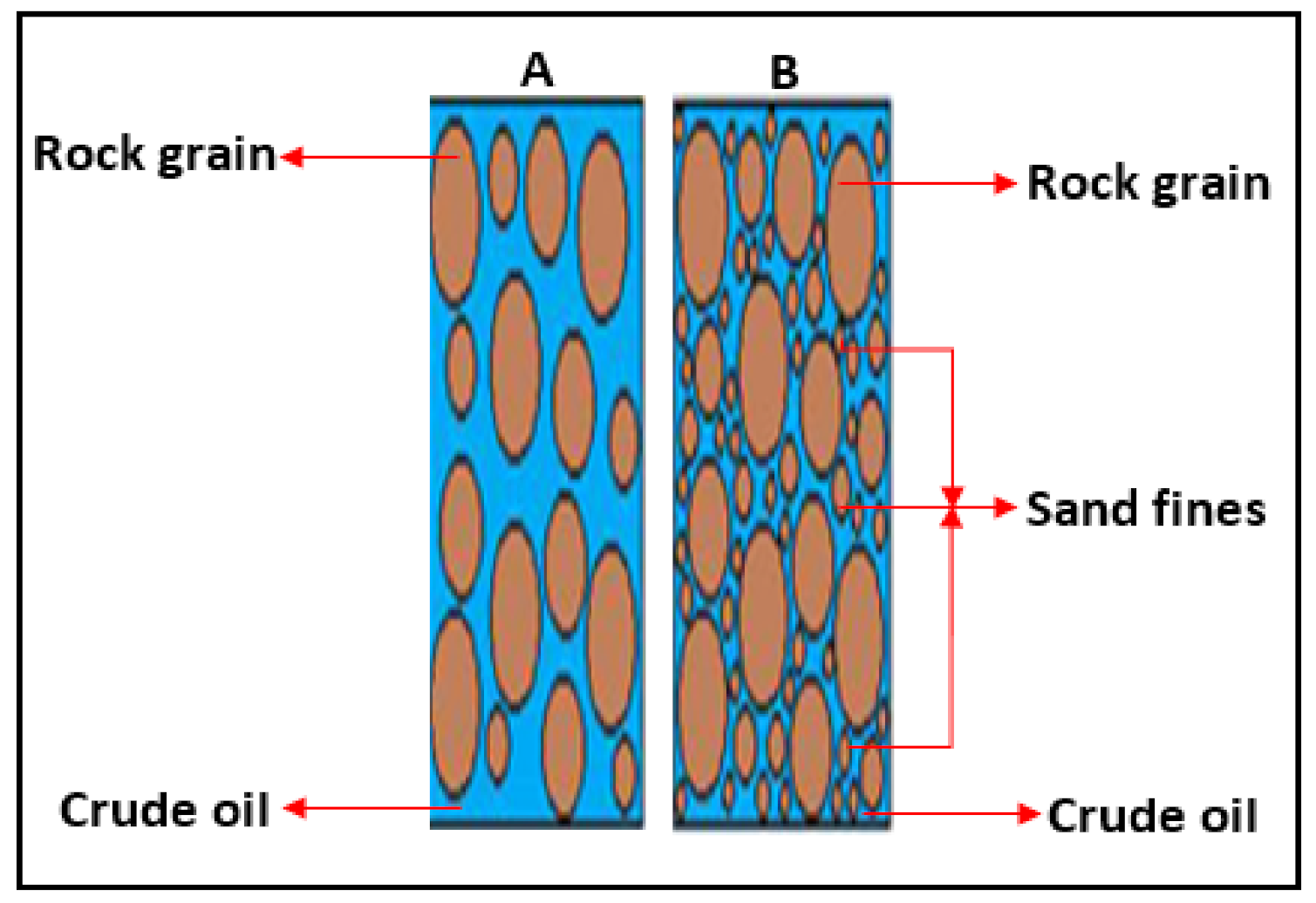
2.1. Migration of Colloidal Fines
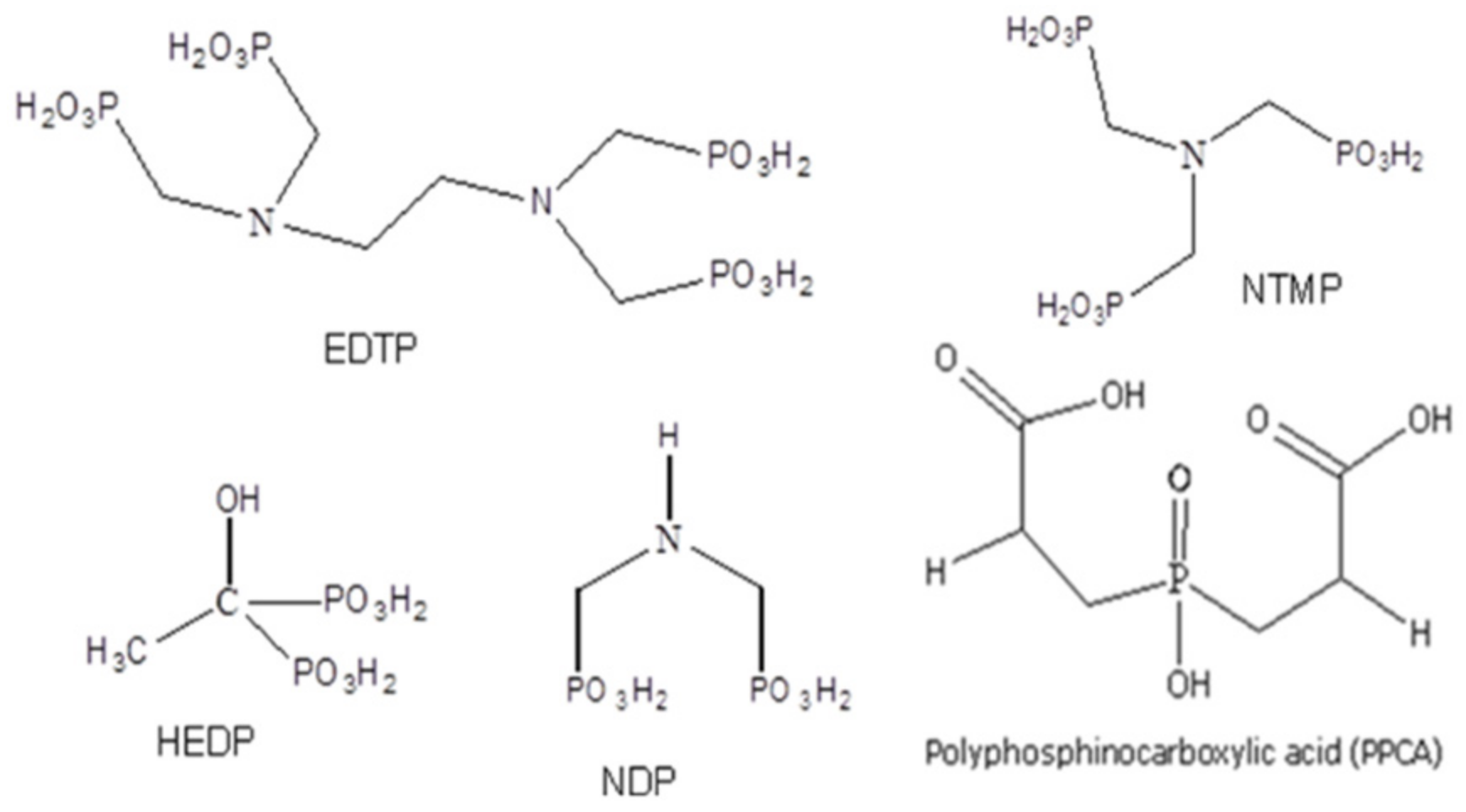
2.1.1. Scale Inhibitors
2.1.2. Effect of Formation Variables in Adsorbing Characteristics of Polymer on Adsorbent
2.1.3. The Effect of Molecular Weight on Scale Inhibitor Adsorption
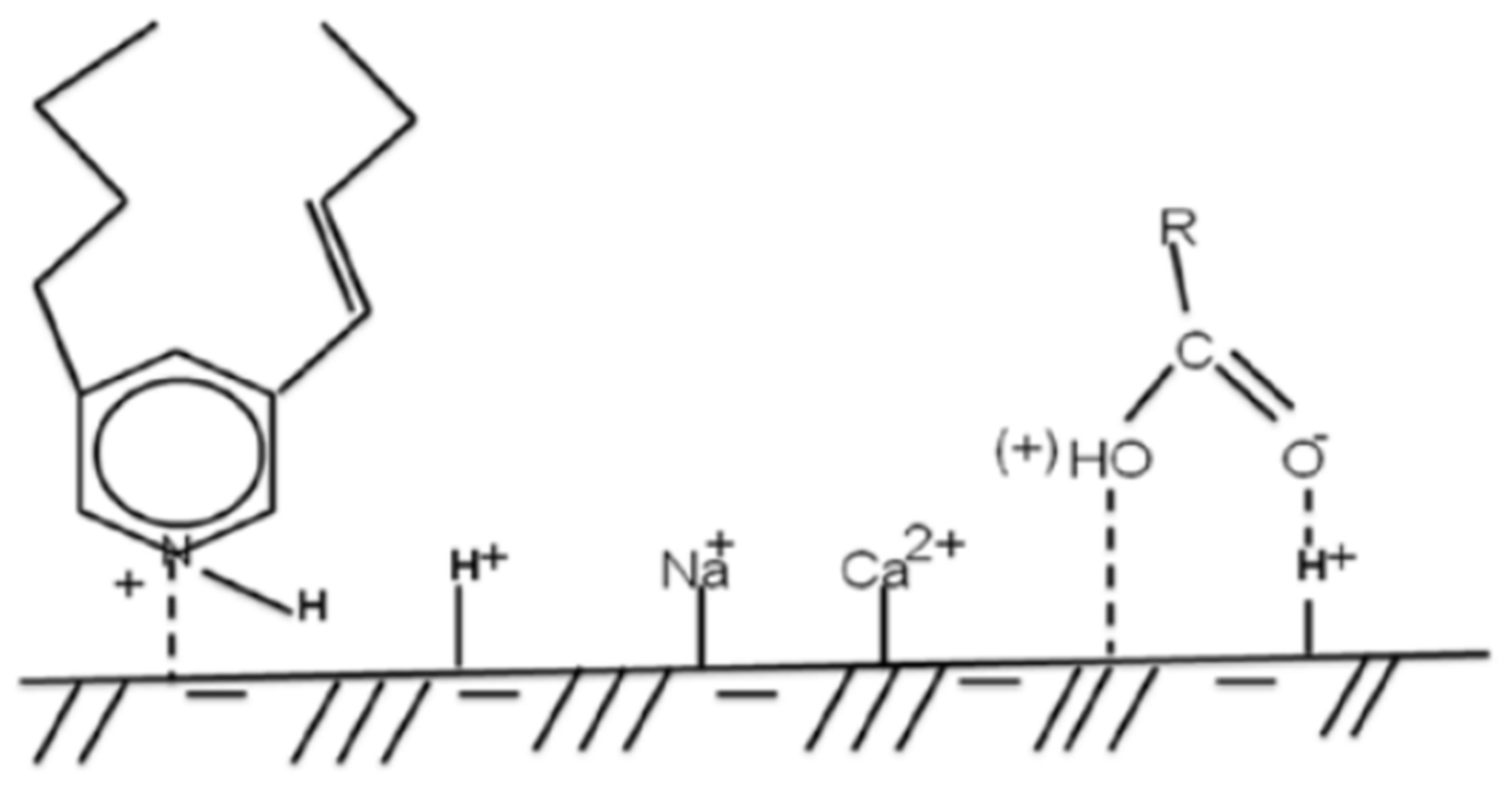
2.1.4. Functional Group Reactivity/Pathways to Scale Inhibitor Adsorption on a Given Sand Surface
2.1.5. Adsorption on Solid Surface
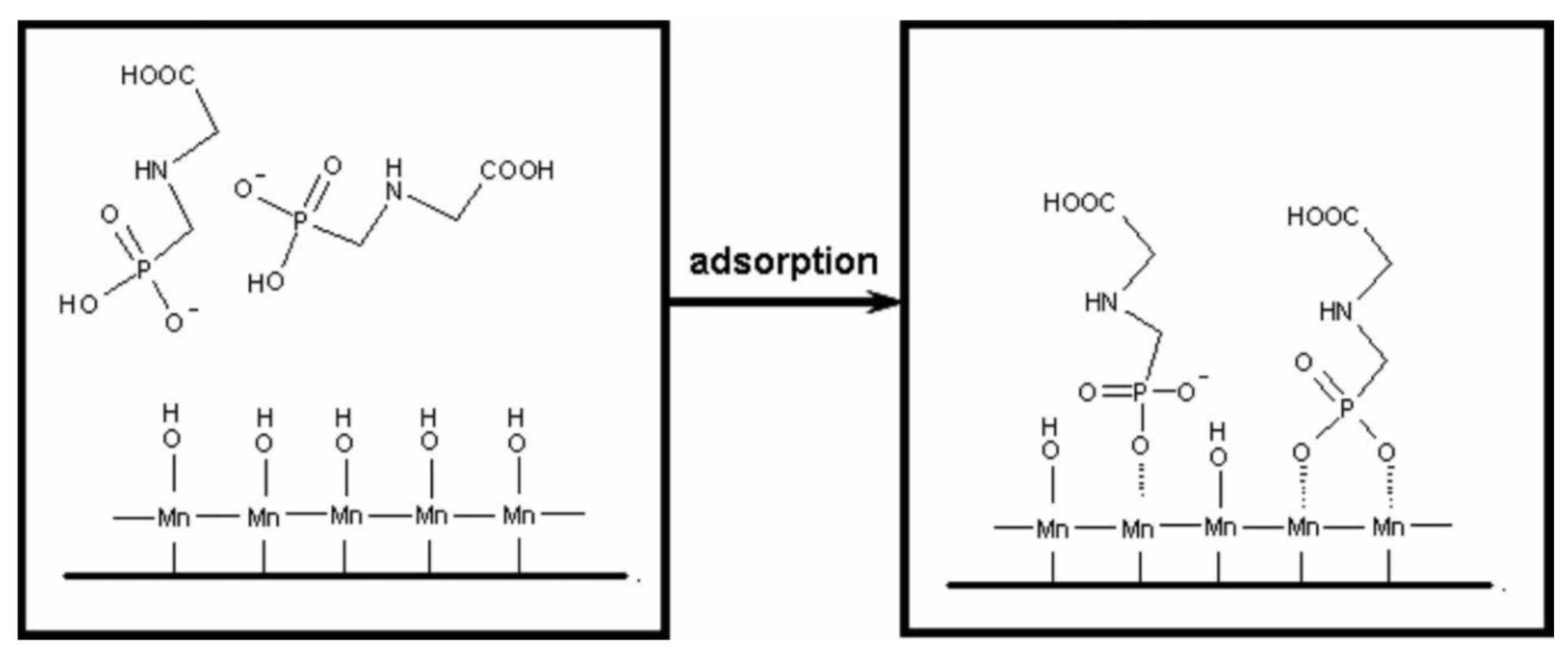
2.1.6. Chemical Adsorption of Scale Inhibitors onto Sand Surface
2.1.7. Physical Adsorption of Scale Inhibitors onto Sand Surface
Adsorption through Hydrogen Bonding Mechanism
2.1.8. Adsorption Isotherm
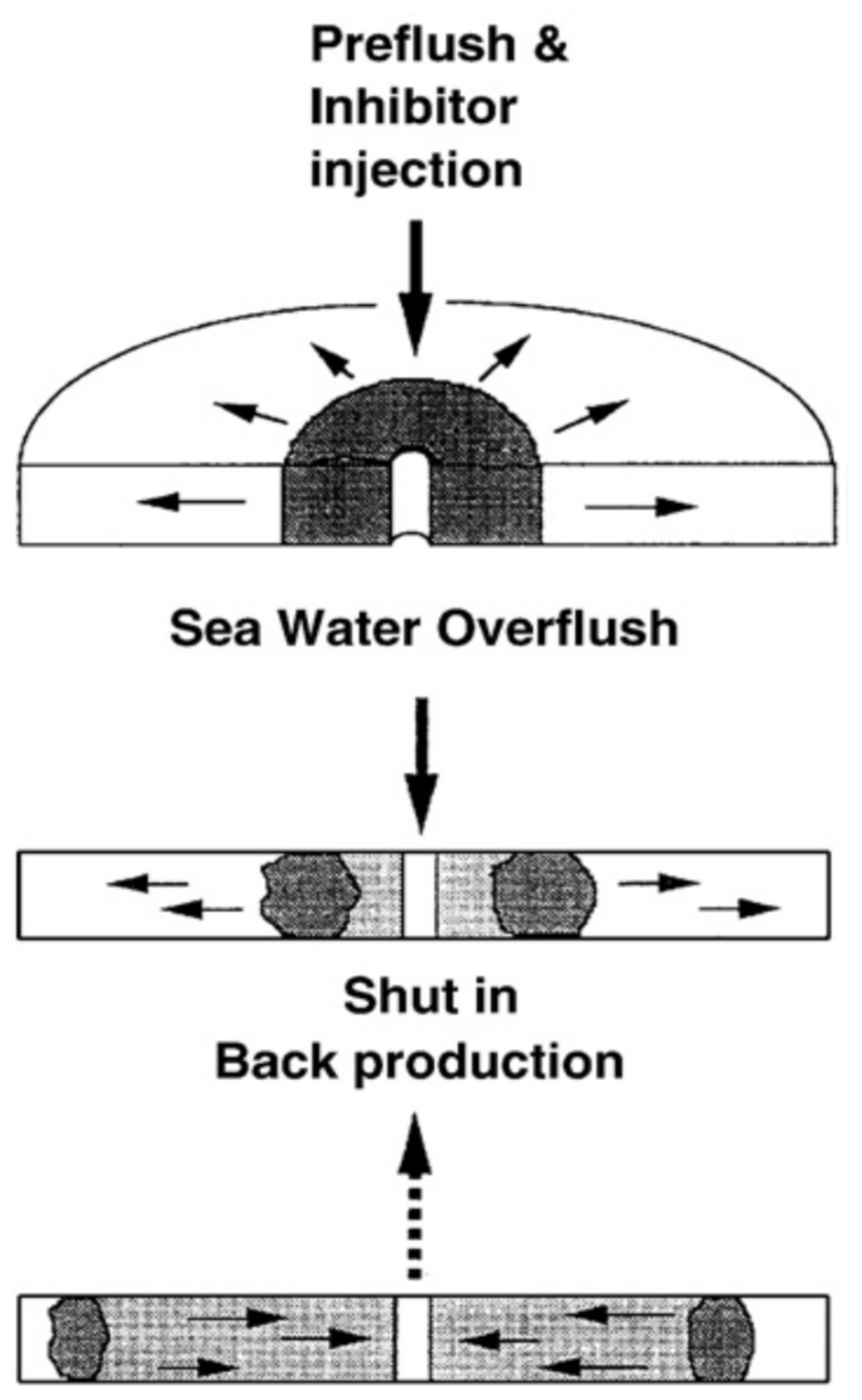
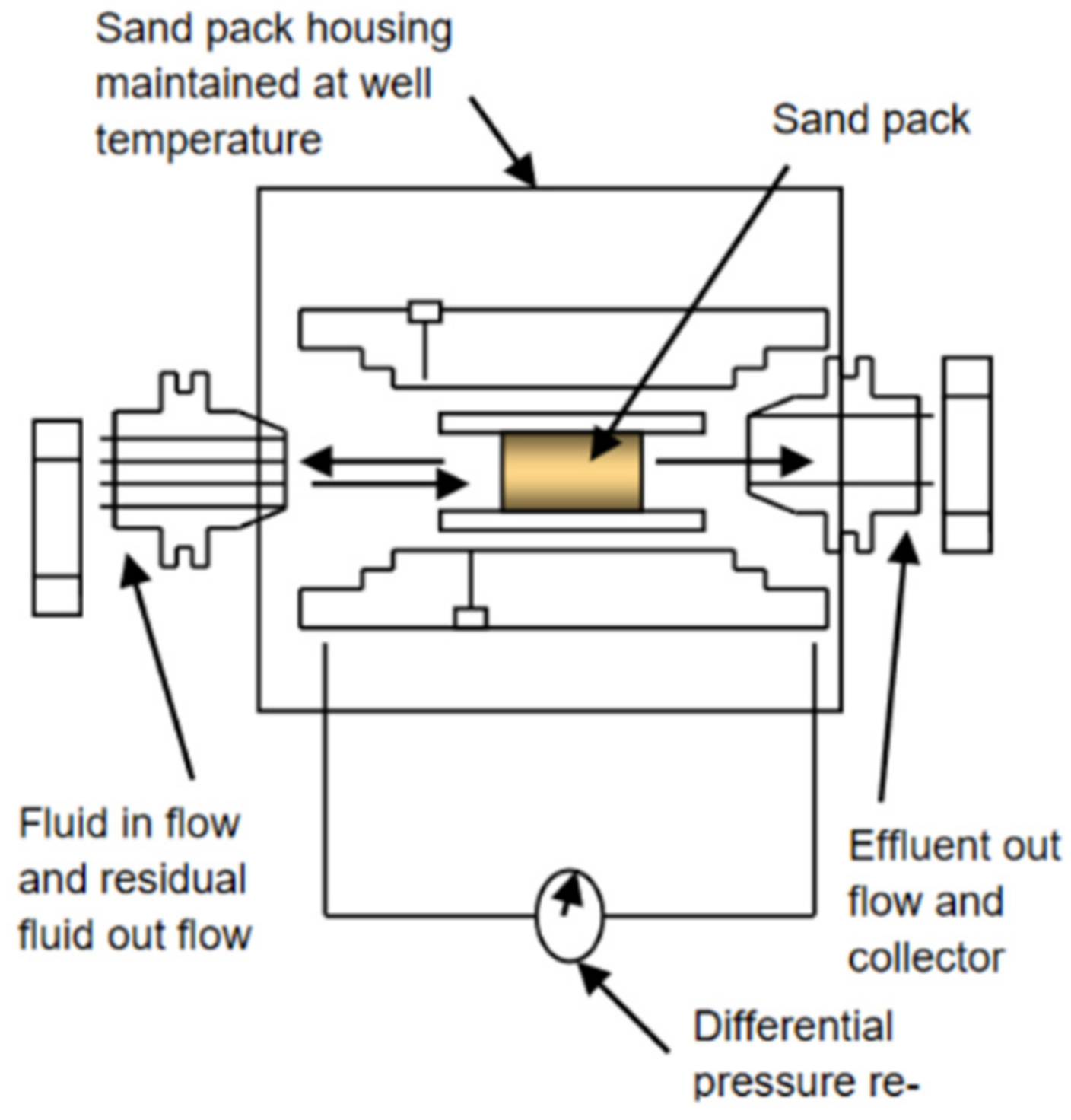
2.2. Theory and Techniques
3. Conclusions
Funding
Data Availability Statement
Conflicts of Interest
References
- Mahmuda, H.B.; Leonga, V.H.; Lestariono, Y. Sand production: A smart control framework for risk mitigation. Petroleum 2020, 6, 1–13. [Google Scholar] [CrossRef]
- Vincent, O.N.; Abiola, S.O.; Felix, O.O.; Ajienk, J.A. Sanding in Oil Well Reservoir Completions. In Proceedings of the Nigeria Annual International Conference and Exhibition, Lagos, Nigeria, 6–8 August 2012. [Google Scholar]
- Cook, J.M.; Bradford, I.D.R.; Plumb, R.A. A Study of the Physical Mechanisms of Sanding and Application to Sand Production Prediction. In Proceedings of the European Petroleum Conference, London, UK, 25 October 1994. [Google Scholar]
- Shafiq, M.U.; Mahmud, H.B. Sandstone matrix acidizing knowledge and future development. J. Petrol. Explor. Prod. Technol. 2017, 7, 1205–1216. [Google Scholar] [CrossRef]
- Salahi, A.; Dehghan, A.N.; Sheikhzakariaee, S.J. Sand production control mechanisms during oil well production and construction. Pet. Res. 2021, in press. [Google Scholar]
- Shchukin, E.D. The influence of Surface-Active Media on the Mechanical Properties of Materials. Adv. Colloid Interface Sci. 2006, 123–126, 33–34. [Google Scholar] [CrossRef] [PubMed]
- Traskin, V.Y. Rehbinder Effect in Tectonophysics. Izv. Phys. Solid Earth 2009, 45, 952–963. [Google Scholar] [CrossRef]
- Prempeh, K.O.K.; Chequer, L.; Badalyan, A.; Bedrikovetsky, P. Effects of the Capillary-Entrapped Phase on Fines migration in Porous Media. J. Nat. Gas Sci. Eng. 2020, 73, 103047. [Google Scholar] [CrossRef]
- Merdhah, A.B.B.; Yassin, A.A.M. Study of Scale Formation in Oil Reservoir during Water Formation Injection—A review. Mar. Sci. Technol. Semin. 2007, 2047, 1–7. [Google Scholar]
- Hasson, D.; Shemer, H.; Sher, A. State of the Art of Friendly “Green” Scale Control Inhibitors: A Review Article. Ind. Eng. Chem. Res. 2011, 50, 7601–7607. [Google Scholar] [CrossRef]
- Voloshin, I.A.; Ragulin, V.V.; Yabayeva, E.N.; Diakonov, L.I.; Mackay, J.E. Scaling Problems in Western Siberia. In Proceedings of the SPE fifth International Symposium on Oilfield Scale, Aberdeen, UK, 29–30 January 2003; p. l-8. [Google Scholar]
- Jordan, M.M.; Johnston, C.J.; Sutherland, L. Impact of Reservoir Temperature on Scale Inhibitor Retention-The Challenge of Ultra-Low Temperature Sandstone Reservoirs. In Proceedings of the SPE International Oilfield Scale Conference and Exhibition, Aberdeen, UK, 11–12 May 2016. [Google Scholar]
- Farooqui, N.M.; Sorbie, K.S. The Use of PPCA in Scale-Inhibitor Precipitation Squeezes: Solubility, Inhibition Efficiency, and Molecular-Weight Effects. Society of Petroleum Engineers. In Proceedings of the SPE International Oilfield Scale Conference and Exhibition, Aberdeen, UK, 14–15 May 2014. [Google Scholar]
- Stamatakis, E.; Chatzichristos, C.; Sagen, J.; Stubos, A.K.; Palyvos, I.; Muller, J.; Stokkan, J.-A. An Integrated Radiotracer Approach for the Laboratory Evaluation of Scale Inhibitors Performance in Geological Environments. Chem. Eng. Sci. 2006, 61, 7057–7067. [Google Scholar] [CrossRef]
- Andrei, M.; Malandrino, A. Comparative Coreflood Studies for Precipitation and Adsorption Squeeze with PPCA as the Scales Inhibitor. Pet. Sci. Technol. 2003, 21, 1295–1315. [Google Scholar] [CrossRef]
- Oluyemi, G.F. Chemical Inhibitor Adsorption and Desorption Characteristics of Common Gravel Pack Sands under Static and Dynamic Conditions. Spec. Top. Rev. Porous Media Int. J. 2013, 4, 159–169. [Google Scholar] [CrossRef]
- Veloso, C.B.; Silva, A.N.A.; Watanabe, T.T.T.G.; Paes, J.F.B.C.; De Luna, F.M.T.; Cavalcante, C.L., Jr. Scale Inhibitor Adsorption Studies in Rock Sandstone Type. Adsorption 2014, 20, 977–985. [Google Scholar] [CrossRef]
- Bezemer, C.; Bauer, K.A. Prevention of Carbonate Scale Deposition: A Well-Packing Technique with Controlled Solubility Phosphates. J. Pet. Technol. 1969, 21, 505–514. [Google Scholar] [CrossRef]
- Liu, B.; Zeng, L.; Mao, J.; Ren, Q. Simulation of Levulinic Acid Adsorption in Packed Beds Using Parallel Pore/Surface Diffusion Model. Chem Eng. Technol. 2010, 33, 1146–1152. [Google Scholar] [CrossRef]
- Selvaraju, N.; Pushpavanam, S. Adsorption Characteristics on Sand and Brick Beds. Chem. Eng. J. 2009, 147, 130–138. [Google Scholar] [CrossRef]
- Garmeh, G.; Johns, R.T.; Lake, L.W. Pore-Scale Simulation of Dispersion in Porous Media. In Proceedings of the SPE Annual Technical Conference and Exhibition, Anaheim, CA, USA, 11–14 November 2009; pp. 559–567. [Google Scholar]
- Kahrwad, M.; Sorbie, K.S.; Boak, L.S. Coupled Adoption/Precipitation of Scale Inhibitors; Experimental Results and Modelling. In Proceedings of the SPE International Oilfield Scale Conference, Aberdeen, UK, 28–29 May 2008. [Google Scholar]
- Tantayakom, V.; Fogler, H.S.; Chavadej, S. Study of Scale Inhibitor Reactions in Precipitation Squeeze Treatments. In Proceedings of the 2005 SPE International Symposium on Oilfield Chemistry, Houston, TX, USA, 2–4 February 2005. SPE 92771. [Google Scholar]
- Yan, F.; Zang, F.; Bhandari, N.; Liu, Y.; Wang, L.; Dai, Z.; Zhang, Z.; Bolanos, V.; Kan, A.; Tomson, M. Interaction between Scale Inhibitors and Shale and Sandstone Formations. Society of Petroleum Engineers. In Proceedings of the SPE International Symposium on Oilfield Chemistry, The Woodlands, TX, USA, 13–15 April 2015. [Google Scholar]
- Ibrahim, J.; Sorbie, K.S.; Boak, L.S. Coupled Adsorption/Precipitation Experiments: 2. In In Non-Equilibrium Sand Pack Treatments. In Proceedings of the SPE International Conference on Oilfield Scale, Aberdeen, UK, 30–31 May 2012. [Google Scholar]
- Sorbie, K.S.; Gdanski, R.D. A Complete Theory of Scale-Inhibitor transport and Adsorption/Desorption in Squeeze treatments. In Proceedings of the SPE International Symposium on Oilfield Scale, Aberdeen, UK, 11–12 May 2005. [Google Scholar]
- Jordan, M.M.; Sorbie, K.S.; Griffin, P.; Hennessey, S.; Hourston, K.E.; Waterhouse, P. Scale Inhibitor Adsorption/Desorption vs Precipitation: The Potential for Extending Squeeze Life While Minimising Formation Damage. In Proceedings of the European Formation Damage Conference, Hague, The Netherlands, 15–16 May 1995. [Google Scholar]
- Brown, J.M.; Shen, D.; Satya Gupta, D.V.; Taylor, G.; Self, R.W. Laboratory and Field Studies of Long-Term Release Rates for a Solid Scale. In Proceedings of the SPE International Symposium on Oilfield Chemistry, Woodlands, TX, USA, 11–13 April 2011. [Google Scholar]
- Oyeneyin, B. Integrated Sand Management for Effective Hydrocarbon Flow Assurance. In Development in Petroleum Science, 1st ed.; Dennis, S., Ed.; Elsevier: Amsterdam, The Netherlands, 2015; Volume 63, pp. 129–138. [Google Scholar]
- Berntsen, A.N. Scale Effect in Volumetric Sand Production. American Rock Mechanics Association. In Proceedings of the 46th US Rock Mechanics/Geomechanics Symposium, Chicago, IL, USA, 24–27 June 2012. [Google Scholar]
- Mcmahon, A.J.; Martin, J.W.; Harris, L. Effects of Sand and Interfacial Adsorption Loss on Corrosion Inhibitor Efficiency. In Proceedings of the NACE International-Corrosion 2005, Houston, TX, USA, 3–7 April 2005; pp. 1–23. [Google Scholar]
- Molnes, S.N.; Torrijos, I.P.; Strand, S.; Paso, K.G.; Syverud, K. Sandstone Injectivity and Salt Stability of Cellulose Nanocrystals (CNC) Dispersions—Premises for use of CNC in Enhanced Oil Recovery. Ind. Crop. Prod. 2016, 93, 152–160. [Google Scholar] [CrossRef]
- Cai, L.; Tong, M.; Wang, X.; Kim, H. Influence of Clay Particles on the Transport and Retention of Titanium Dioxide Nanoparticles in Quartz Sand. Environ. Sci. Technol. 2014, 48, 7323–7332. [Google Scholar] [CrossRef]
- Tusher, K.S.; Mehajan, S.P.; Khilar, K.C. Colloid-Associated Contaminant Transport in Porous Media: 1. Experimental Studies. AIChE J. 2002, 48, 2366–2374. [Google Scholar]
- Nisrine, S.; Azita, A.S.; Aziz, O.; Henri, B. Numerical Simulation of Retention and Release of Colloids in Porous Media at the Pore Scale. Colloids Surf. A Physicochem. Eng. Asp. 2013, 427, 33–40. [Google Scholar]
- Abadzic, D.S.; Ryan, N.J. Particle Release and Permeability Reduction in a Natural Zeolite (clinoptilolite) and Sand porous Medium. Environ. Sci. Technol. 2001, 35, 4502–4508. [Google Scholar] [CrossRef]
- Wang, S.; Chung, K.H.; Masliyah, J.H.; Gray, M.R. Deposition of Fine Particles in Packed Beds at Hydrotreating Conditions: Role of Surface Chemistry. Ind. Eng. Chem. Res. 1999, 38, 4878–4888. [Google Scholar] [CrossRef]
- Fielder, G.D. Scale Inhibitor Selection for a High Suspended Solids Produced Water Systems. In Proceedings of the International Conference on Corrosion, Houston, TX, USA, 26 March 2000. [Google Scholar]
- Chakraborty, A.; Saha, B.B.; Aristov, Y.I. Dynamic Behaviours of Adsorption Chiller: Effects of the Silica Gel Grain Size and Layers. Energy 2014, 78, 304–312. [Google Scholar] [CrossRef]
- Wuyep, E.O.; Oluyemi, G.F.; Yates, K.; Akinsanya, A.R. Evaluation of Interaction between Oilfield Chemicals and Reservoir Rocks. Nat. Resour. Res. 2019, 29, 1239–1258. [Google Scholar] [CrossRef]
- Ghosh, B.; Kundu, S.; Senthilmurugan, B. A New Squeeze Scale Inhibitor for a Sandstone Reservoir with a Stimulation Effect. Pet. Sci. Technol. 2012, 30, 402–411. [Google Scholar] [CrossRef]
- Zeng, J.-P.; Wang, F.-H.; Gong, S.-D. Molecular Dynamics Simulation of the Interaction between Polyaspartic Acid and Calcium Carbonate. Mol. Simul. 2013, 39, 169–175. [Google Scholar] [CrossRef]
- Kelland, M.A. Production Chemicals for the Oil and Gas Industry; CRC Press: Boca Raton, FL, USA, 2009; pp. 53–91. [Google Scholar]
- Fleming, N.; Bourne, H.M.; Strachan, C.J.; Buckley, A.S. Development of an Ecofriendly Scale Inhibitor for Harsh Scaling Environments. In Proceedings of the SPE International Symposium on Oilfield Chemistry, Houston, TX, USA, 13–16 February 2001. [Google Scholar]
- Ketrane, R.; Saidani, B.; Gil, O.; Leleyter, L.; Baraud, F. Efficiency of Five Scale Inhibitors on Calcium Carbonate Precipitation from Hard Water: Effect of Temperature and Concentration. Desalination 2009, 249, 1397–1404. [Google Scholar] [CrossRef]
- Jonasson, R.G.; Rispler, K.; Wiwchar, B.; Gunter, W.D. Effect of Phosphonate Inhibitors on Calcite Nucleation kinetics as a Function of Temperature using Light Scattering in an Autoclave. Chem. Geol. 1996, 132, 215–225. [Google Scholar] [CrossRef]
- Li, F.; Wu, W.; Li, R.; Fu, X. Adsorption of Phosphate by Acid-Modified Fly Ash and Palygorskite in Aqueous Solution: Experimental and Modeling. Appl. Clay Sci. 2016, 132–133, 343–352. [Google Scholar] [CrossRef]
- Wylde, J.J. Scale Inhibition Application in Northern Alberta: A Case History of an Ultra-High Temperature Scale Inhibition Solution in Fire Tube Heaters. In Proceedings of the SPE International Conference on Oilfield Scale, Aberdeen, UK, 26–27 May 2010. [Google Scholar]
- Kumar, T.; Vishwanatham, S.; Kundu, S.S. A Laboratory Study on Pteroyl-l-Glutamic Acid as a Scale Prevention Inhibitor of Calcium Carbonate in Aqueous Solution of Synthetic Produced Water. J. Pet. Sci. Eng. 2010, 71, 1–7. [Google Scholar] [CrossRef]
- Jones, F.; Stanley, A.; Oliveira, A.; Rohl, A.L.; Reyhani, M.M.; Parkinson, G.M.; Ogden, M.I. The Role of Phosphonate Speciation on the Inhibition of Barium Sulfate Precipitation. J. Cryst. Growth 2003, 249, 584–593. [Google Scholar] [CrossRef][Green Version]
- Yang, Q.; Liu, Y.; Gu, A.; Ding, J.; Shen, Z. Investigation of Calcium Carbonate Scaling Inhibition and Scale Morphology by AFM. J. Colloid Interface Sci. 2001, 240, 608–621. [Google Scholar] [CrossRef]
- Hao, C.; Qin, L.; Yan, Q.; Qiu, Z.; Jianping, Z. Preparation and Adsorption Performance of MnO2/PAC Composite towards Aqueous Glyphosate. Environ. Technol. 2012, 33, 2049–2056. [Google Scholar]
- Saldungaray, P.M.; Caretta, F.; Sofyan, M. Formation Fines Stabilization using Surface Adsorption Polymerization: Field Application of an Emerging Technology. In Proceedings of the SPE Asia Pacific Oil and Gas Conference and Exhibition, Jakarta, Indonesia, 17–19 April 2001. [Google Scholar]
- Jordana, R.; Howe, R.F.; Guan, H. Retention and Release of Scale Inhibitors in Different Clay Minerals. In Proceedings of the SPE International Conference and Exhibition on Oilfield Scale, Aberdeen, UK, 30–31 May 2012. [Google Scholar]
- Mason, B.T.; Kan, A.T.; Fu, G.; Shen, D.; El Din, H.A.N.; Al-Saaaiari, H.; Al Thubaiti, M. Mechanistic Understanding of Rock/Phosphonate Interactions and the Effect of Metal Ions on Inhibitor Retention. In Proceedings of the 2006 SPE International Symposium on Oilfield Scale, Aberdeen, UK, 31 May–1 June 2008; pp. 325–336. [Google Scholar]
- Dantas, T.L.P.; Luna, F.M.T.; Silva, I.J., Jr.; De Azevedo, D.C.S.; Grande, C.A.; Rodrigues, A.E.; Moreira, R.F.P.M. Modelling of the Fixed-Bed Adsorption of Carbon Dioxide and a Carbon Dioxide-Nitrogen Mixture on Zeolite 13X. Braz. J. Chem. Eng. 2011, 28, 533–544. [Google Scholar] [CrossRef]
- Zaitoun, A.; Kohler, N. The Role of Adsorption in Polymer Propagation through Reservoir Rocks. In Proceedings of the SPE International Symposium on Oilfield Chemistry, San Antonio, TX, USA, 4–6 February 1987. [Google Scholar]
- Kosmulski, M. pH-Dependent Surface Charging and Points of Zero Charge: III. Update. J. Colloid Interface Sci. 2006, 298, 730–741. [Google Scholar] [CrossRef]
- Kosmulski, M. The pH-Dependent Surface Charging and Points of Zero Charge: V. Update. J. Colloid Interface Sci. 2011, 353, 1–15. [Google Scholar] [CrossRef]
- Flood, C.; Cosgrove, T.; Espidel, Y.; Howell, I.; Revel, P. Sodium Polyacrylate Adsorption onto Anionic and Cationic Silica in the Presence of Salts. Langmuir 2007, 23, 6191–6197. [Google Scholar] [CrossRef]
- Alotaibi, M.B.; Azmy, R.; El Din, H.A.N. A Comprehensive EOR Study Using Low Salinity Water in Sandstone Reservoirs. In Proceedings of the SPE Improved Oil Recovery Symposium, Tulsa, OK, USA, 24–28 April 2010. [Google Scholar]
- Vazquez, O.; Thanasutives, P.; Eliasson, C.; Fleming, N.; Mackay, E. Modelling the Application of Scale-Inhibitor-Squeeze Retention-Enhancing Additives. SPE Prod. Oper. 2011, 26, 270–277. [Google Scholar]
- Strand, S.; Puntervold, T.; Austad, T. Water Based EOR from Clastic Oil Reservoirs by Wettability Alteration: A review of chemical aspects. J. Pet. Sci. Eng. 2016, 146, 1079–1091. [Google Scholar] [CrossRef]
- Zhang, R.; Somasundaran, P. Advances in adsorption of surfactants and their mixtures at solid/solution interfaces. Adv. Colloid Interface Sci. 2006, 123–126, 213–229. [Google Scholar] [CrossRef]
- Kan, A.T.; Fu, G.; Tomson, M.B.; Al-Thubaiti, M.; Xiao, A.J. Factors Affecting Scale Inhibitor Retention in Carbonate-Rich Formation during Squeeze Treatment. SPE J. 2004, 9, 280–289. [Google Scholar] [CrossRef]
- Tomson, M.B.; Fu, G.; Watson, M.A.; Kan, A.T. Mechanisms of Mineral Scale Inhibition. SPE Prod. Facil. 2003, 192–199. [Google Scholar] [CrossRef]
- Gupta, S.S.; Bhattacharyya, K.G. Adsorption of Metal Ions by Clays and Inorganic Solids. RSC Adv. 2014, 4, 28537–28586. [Google Scholar] [CrossRef]
- Sorbie, K.S.; Yuan, M.D.; Chen, P.; Todd, A.C.; Wat, R.M.S. The Effect of pH, Calcium, and Temperature on the Adsorption of Phosphonate Inhibitor onto Consolidated and Crushed Sandstone. In Proceedings of the 68th Annual Technical Conference and Exhibition of the SPE, Houston, TX, USA, 3–4 October 1993. [Google Scholar]
- Dove, P.M.; Craven, C.M. Surface charge density on silica in alkali and alkaline earth chloride electrolyte solutions. Geochim. Cosmochim. Acta 2005, 69, 4963–4970. [Google Scholar] [CrossRef]
- Buyuktimkin, T.; Wurster, D.E. The Influence of the Adsorption of Metoclopramide on the Surface Ionization of Fumed Silica. Int. J. Pharm. 2015, 478, 164–171. [Google Scholar] [CrossRef]
- Gupta, S.S.; Bhattacharyya, K.G. Kinetics of Adsorption of Metal Ions on Inorganic Materials: A review. Adv. Colloid Interface Sci. 2011, 162, 39–58. [Google Scholar] [CrossRef]
- Ivan, D.; Pinerez, T.; Austad, T.; Strand, S.; Puntervoid, T.; Wrobel, S. Linking Low Salinity EOR Effects in Sandstone to pH, Mineral Properties and Water Composition. In Proceedings of the SPE Improved Oil Recovery Conference, Tulsa, OK, USA, 11–13 April 2016; pp. 1–17. [Google Scholar]
- Dang, T.Q.C.; Chen, Z.; Nguyen, T.B.N.; Bae, W. Investigation of Isotherm Polymer Adsorption in Porous Media. Pet. Sci. Technol. 2014, 32, 1626–1640. [Google Scholar] [CrossRef]
- Dellostritto, M.J.; Kubicki, J.D.; Sofo, J.O. Effect of Ions on H-Bond Structure and Dynamics at the Quartz (101)–Water Interface. Langmuir 2016, 32, 11353–11365. [Google Scholar] [CrossRef]
- Jordan, M.M.; Sorbie, K.S.; Jiang, P.; Yuan, M.D.; Todd, A.C.; Taylor, K.; Hourston, K.E.; Ramstad, K. Mineralogical Control on Inhibitor Adsorption/Desorption in Brent Group Sandstone and their Importance in Predicting and Extending Field Squeeze Lifetimes. In Proceedings of the European Production Operations Conference and Exhibition, Aberdeen, UK, 15–17 March 1994. [Google Scholar]
- Bassioni, G. Mechanistic aspects on the influence of inorganic anion adsorption on oilfield scale inhibition by citrate. J. Pet. Sci. Eng. 2010, 70, 298–301. [Google Scholar] [CrossRef]
- Boels, L.; Keesman, K.J.; Witkamp, G. Adsorption of Phosphonate Antiscalant from Reverse Osmosis Membrane Concentrate onto Granular Ferric Hydroxide. Environ. Sci. Technol. 2012, 46, 9638–9645. [Google Scholar] [CrossRef]
- Baraka-Lokmane, S.; Sorbie, K. Effect of pH and scale inhibitor concentration on phosphonate–carbonate interaction. J. Pet. Sci. Eng. 2010, 70, 10–27. [Google Scholar] [CrossRef]
- Li, D.; Ren, S.; Xu, Y.; Rui, H. Experimental Study on Kinetic Behaviors of Natural Gas Hydrate Production via Continuous Simulated seawater Injection. Energy Fuels 2019, 33, 8222–8230. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anyanwu, U.C.; Oluyemi, G.F. Effect of Produced Sand Particles and Fines on Scale Inhibitor: A Review. Colloids Interfaces 2021, 5, 35. https://doi.org/10.3390/colloids5030035
Anyanwu UC, Oluyemi GF. Effect of Produced Sand Particles and Fines on Scale Inhibitor: A Review. Colloids and Interfaces. 2021; 5(3):35. https://doi.org/10.3390/colloids5030035
Chicago/Turabian StyleAnyanwu, Uche C., and Gbenga F. Oluyemi. 2021. "Effect of Produced Sand Particles and Fines on Scale Inhibitor: A Review" Colloids and Interfaces 5, no. 3: 35. https://doi.org/10.3390/colloids5030035
APA StyleAnyanwu, U. C., & Oluyemi, G. F. (2021). Effect of Produced Sand Particles and Fines on Scale Inhibitor: A Review. Colloids and Interfaces, 5(3), 35. https://doi.org/10.3390/colloids5030035





