Application of Bio-Based Wrinkled Surfaces as Cell Culture Scaffolds
Abstract
:1. Introduction
2. Results and Discussion
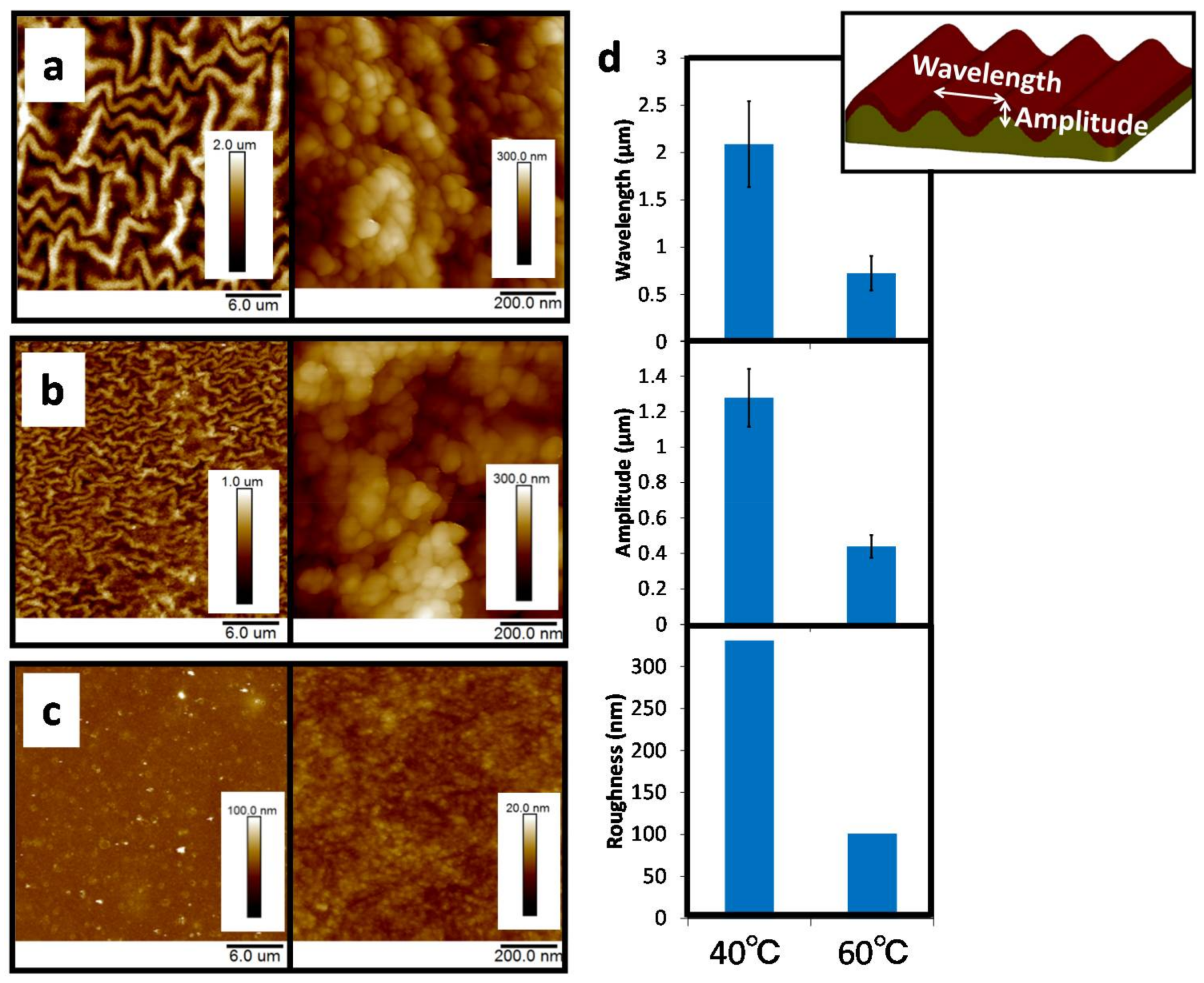
2.1. AFM Analysis of the Wrinkle-CS Films in Air and PBS
2.2. 3T3 Cell Culture on the Wrinkle-CS Film
2.3. 3T3 Biocompatibility of the Wrinkle-CS Film
3. Conclusions
4. Materials and Methods
4.1. Materials
4.2. Instrumentation
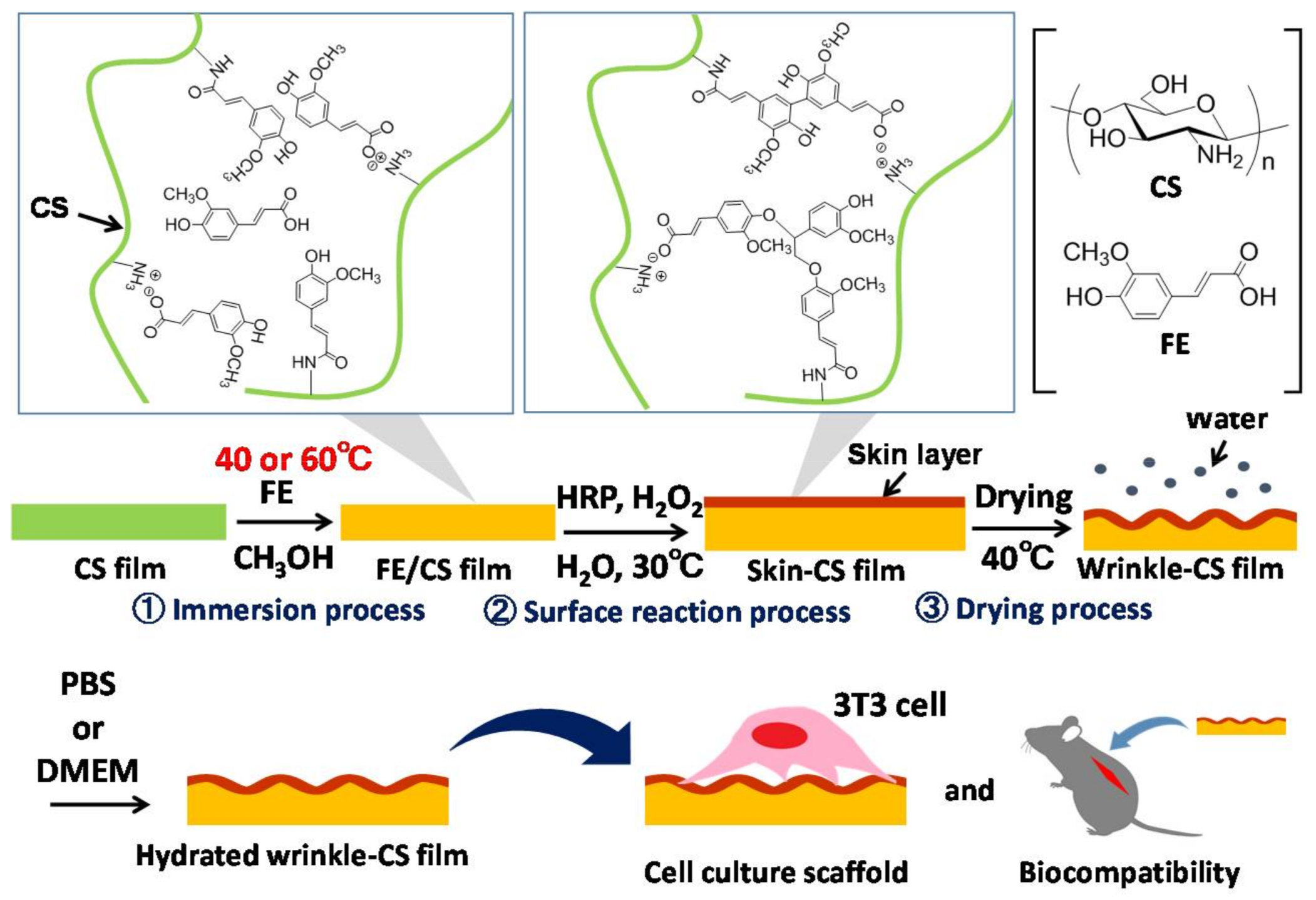
4.3. Preparation of the CS Film
4.4. Surface Wrinkling of Films
4.5. AFM and Force Curve Measurement
4.6. In Vitro Cell Culture
4.7. Animals
4.8. In Vivo Evaluation
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| CS | Chitosan |
| HRP | Horseradish peroxidase |
| PBS | Phosphate buffered saline |
| AFM | Atomic force microscopy |
| FE | Ferulic acid |
References
- Drury, J.L.; Mooney, D.J. Hydrogels for tissue engineering: Scaffold design variables and applications. Biomaterials 2003, 24, 4337–4351. [Google Scholar] [CrossRef]
- Lutolf, M.P.; Hubbell, J.A. Synthetic biomaterials as instructive extracellular microenvironments for morphogenesis in tissue engineering. Nat. Biotechnol. 2005, 23, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Metavarayuth, K.; Sitasuwan, P.; Zhao, X.; Lin, Y.; Wang, Q. Influence of Surface Topographical Cues on the Differentiation of Mesenchymal Stem Cells in Vitro. ACS Biomater. Sci. Eng. 2016, 2, 142–151. [Google Scholar] [CrossRef]
- Uto, K.; Tsui, J.H.; DeForest, C.A.; Kim, D.H. Dynamically tunable cell culture platforms for tissue engineering and mechanobiology. Prog. Polym. Sci. 2017, 65, 53–82. [Google Scholar] [CrossRef] [PubMed]
- Genzer, J.; Groenewold, J. Soft matter with hard skin: From skin wrinkles to templating and material characterization. Soft Matter 2006, 2, 310–323. [Google Scholar] [CrossRef]
- Ionov, L. Biomimetic 3D self-assembling biomicroconstructs by spontaneous deformation of thin polymer films. J. Mater. Chem. 2012, 22, 19366–19375. [Google Scholar] [CrossRef]
- Ohzono, T.; Suzuki, K.; Yamaguchi, T.; Fukuda, N. Tunable Optical Diffuser Based on Deformable Wrinkles. Adv. Opt. Mater. 2013, 1, 374–380. [Google Scholar] [CrossRef]
- Lee, S.G.; Kim, H.; Choi, H.H.; Bong, H.; Park, Y.D.; Lee, W.H.; Cho, K. Evaporation-Induced Self-Alignment and Transfer of Semiconductor Nanowires by Wrinkled Elastomeric Templates. Adv. Mater. 2013, 25, 2162–2166. [Google Scholar] [CrossRef] [PubMed]
- Stenberg, H.; Matikainen, A.; Daniel, S.; Nuutinen, T.; Stenberg, P.; Honkanen, S.; Pakkanen, T.; Vahimaa, P.; Suvanto, M. Self-organized Polymer Wrinkles: A Lithography-free Pathway for Surface-enhanced Raman Scattering (SERS) Substrates. Macromol. Mater. Eng. 2015, 300, 386–390. [Google Scholar] [CrossRef]
- Li, Y.Y.; Dai, S.X.; John, J.; Carter, K.R. Superhydrophobic Surfaces from Hierarchically Structured Wrinkled Polymers. ACS Appl. Mater. Interfaces 2013, 5, 11066–11073. [Google Scholar] [CrossRef] [PubMed]
- Davis, C.S.; Martina, D.; Creton, C.; Lindner, A.; Crosby, A.J. Enhanced Adhesion of Elastic Materials to Small-Scale Wrinkles. Langmuir 2012, 28, 14899–14908. [Google Scholar] [CrossRef] [PubMed]
- Aufan, M.R.; Sumi, Y.; Kim, S.; Lee, J.Y. Facile Synthesis of Conductive Polypyrrole Wrinkle Topographies on Polydimethylsiloxane via a Swelling-Deswelling Process and Their Potential Uses in Tissue Engineering. ACS Appl. Mater. Interfaces 2015, 7, 23454–23463. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.; Lieu, D.K.; Freschauf, L.; Lew, V.; Sharma, H.; Wang, J.X.; Nguyen, D.; Karakikes, I.; Hajjar, R.J.; Gopinathan, A.; et al. Shrink-Film Configurable Multiscale Wrinkles for Functional Alignment of Human Embryonic Stem Cells and their Cardiac Derivatives. Adv. Mater. 2011, 23, 5785–5791. [Google Scholar] [CrossRef]
- Greco, F.; Fujie, T.; Ricotti, L.; Taccola, S.; Mazzolai, B.; Mattoli, V. Microwrinkled Conducting Polymer Interface for Anisotropic Multicellular Alignment. ACS Appl. Mater. Interfaces 2013, 5, 573–584. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.J.; Li, X.Y.; Ma, H.C.; Guan, Y.; Zhang, Y.J. One-step synthesis of PHEMA hydrogel films capable of generating highly ordered wrinkling patterns. Polymer 2017, 110, 114–123. [Google Scholar] [CrossRef]
- Guvendiren, M.; Burdick, J.A. The control of stem cell morphology and differentiation by hydrogel surface wrinkles. Biomaterials 2010, 31, 6511–6518. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.Q.; Gu, J.J.; Zhao, Y.N.; Guan, Y.; Zhu, X.X.; Zhang, Y.J. Hydrogel Thin Film with Swelling-Induced Wrinkling Patterns for High-Throughput Generation of Multicellular Spheroids. Biomacromolecules 2014, 15, 3306–3312. [Google Scholar] [CrossRef] [PubMed]
- Izawa, H. Preparation of biobased wrinkled surfaces via lignification-mimetic reactions and drying: A new approach for developing surface wrinkling. Polym. J. 2017, 49, 759–765. [Google Scholar] [CrossRef]
- Izawa, H.; Dote, Y.; Okuda, N.; Sumita, M.; Ifuku, S.; Morimoto, M.; Saimoto, H. Wood-mimetic skins prepared using horseradish peroxidase catalysis to induce surface wrinkling of chitosan film upon drying. Carbohydr. Polym. 2017, 173, 519–525. [Google Scholar] [CrossRef] [PubMed]
- Izawa, H.; Okuda, N.; Ifuku, S.; Morimoto, M.; Saimoto, H.; Rojas, O.J. Bio-based Wrinkled Surfaces Harnessed from Biological Design Principles of Wood and Peroxidase Activity. ChemSusChem 2015, 8, 3892–3896. [Google Scholar] [CrossRef] [PubMed]
- Izawa, H.; Okuda, N.; Moriyama, A.; Miyazaki, Y.; Ifuku, S.; Morimoto, M.; Saimoto, H. Biobased Wrinkled Surfaces Induced by Wood Mimetic Skins upon Drying: Effect of Mechanical Properties on Wrinkle Morphology. Langmuir 2016, 32, 12799–12804. [Google Scholar] [CrossRef] [PubMed]
- Thakur, V.K.; Thakur, M.K. Recent Advances in Graft Copolymerization and Applications of Chitosan: A Review. ACS Sustain. Chem. Eng. 2014, 2, 2637–2652. [Google Scholar] [CrossRef]
- Tian, M.; Tan, H.; Li, H.; You, C. Molecular weight dependence of structure and properties of chitosan oligomers. Rsc. Adv. 2015, 5, 69445–69452. [Google Scholar] [CrossRef]
- Li, M.G.; Joung, D.H.; Hughes, B.; Waldman, S.D.; Kozinski, J.A.; Hwang, D.K. Wrinkling Non-Spherical Particles and Its Application in Cell Attachment Promotion. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Izumi, R.; Komada, S.; Ochi, K.; Karasawa, L.; Osaki, T.; Murahata, Y.; Tsuka, T.; Imagawa, T.; Itoh, N.; Okamoto, Y.; et al. Favorable effects of superficially deacetylated chitin nanofibrils on the wound healing process. Carbohydr. Polym. 2015, 123, 461–467. [Google Scholar] [CrossRef] [PubMed]
- Minami, S.; Okamoto, Y.; Saimoto, H.; Shigemasa, Y. Effects of Chitin and Its Derivatives on Wound-Healing Acceleration Mechanisms. J. Chitin Chitosan Sci. 2014, 2, 163–178. [Google Scholar] [CrossRef]
- Azuma, K.; Ifuku, S.; Osaki, T.; Okamoto, Y.; Minami, S. Preparation and biomedical applications of chitin and chitosan nanofibers. J. Biomed. Nanotechnol. 2014, 10, 2891–2920. [Google Scholar] [CrossRef] [PubMed]
- Beanes, S.R.; Dang, C.; Soo, C.; Ting, K. Skin repair and scar formation: The central role of TGF-beta. Exp. Rev. Mol. Med. 2003, 5, 1–22. [Google Scholar] [CrossRef]
- Hutter, J.L.; Bechhoefer, J. Calibration of Atomic-Force Microscope Tips. Rev. Sci. Instrum. 1993, 64, 1868–1873. [Google Scholar] [CrossRef]
- Butt, H.J.; Cappella, B.; Kappl, M. Force measurements with the atomic force microscope: Technique, interpretation and applications. Surf. Sci. Rep. 2005, 59, 1–152. [Google Scholar] [CrossRef]
- Rehfeldt, F.; Brown, A.E.X.; Raab, M.; Cai, S.S.; Zajac, A.L.; Zemel, A.; Discher, D.E. Hyaluronic acid matrices show matrix stiffness in 2D and 3D dictates cytoskeletal order and myosin-II phosphorylation within stem cells. Integr. Biol. 2012, 4, 422–430. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, Y.; Azuma, K.; Izawa, H.; Morimoto, M.; Ochi, K.; Osaki, T.; Ito, N.; Okamoto, Y.; Saimoto, H.; Ifuku, S. Preparation and biocompatibility of a chitin nanofiber/gelatin composite film. Int. J. Biol. Macromol. 2017, 104 Pt B, 1882–1889. [Google Scholar] [CrossRef] [PubMed]




© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Izawa, H.; Okuda, N.; Yonemura, T.; Kuroda, K.; Ochi, K.; Ifuku, S.; Morimoto, M.; Saimoto, H.; Noda, M.; Azuma, K.; et al. Application of Bio-Based Wrinkled Surfaces as Cell Culture Scaffolds. Colloids Interfaces 2018, 2, 15. https://doi.org/10.3390/colloids2020015
Izawa H, Okuda N, Yonemura T, Kuroda K, Ochi K, Ifuku S, Morimoto M, Saimoto H, Noda M, Azuma K, et al. Application of Bio-Based Wrinkled Surfaces as Cell Culture Scaffolds. Colloids and Interfaces. 2018; 2(2):15. https://doi.org/10.3390/colloids2020015
Chicago/Turabian StyleIzawa, Hironori, Noriko Okuda, Tomoe Yonemura, Kohei Kuroda, Kosuke Ochi, Shinsuke Ifuku, Minoru Morimoto, Hiroyuki Saimoto, Mayuko Noda, Kazuo Azuma, and et al. 2018. "Application of Bio-Based Wrinkled Surfaces as Cell Culture Scaffolds" Colloids and Interfaces 2, no. 2: 15. https://doi.org/10.3390/colloids2020015