Fourier-Feature Neural Surrogate for Hemodynamic Field Reconstruction in Stenotic and Bifurcating Flows
Abstract
1. Introduction
2. Materials and Methods
2.1. AI Surrogate Modeling
2.2. CFD Calculations
2.3. Surrogate Model-Based Recovery of Flow Field
3. Results and Discussion
3.1. CFD Results
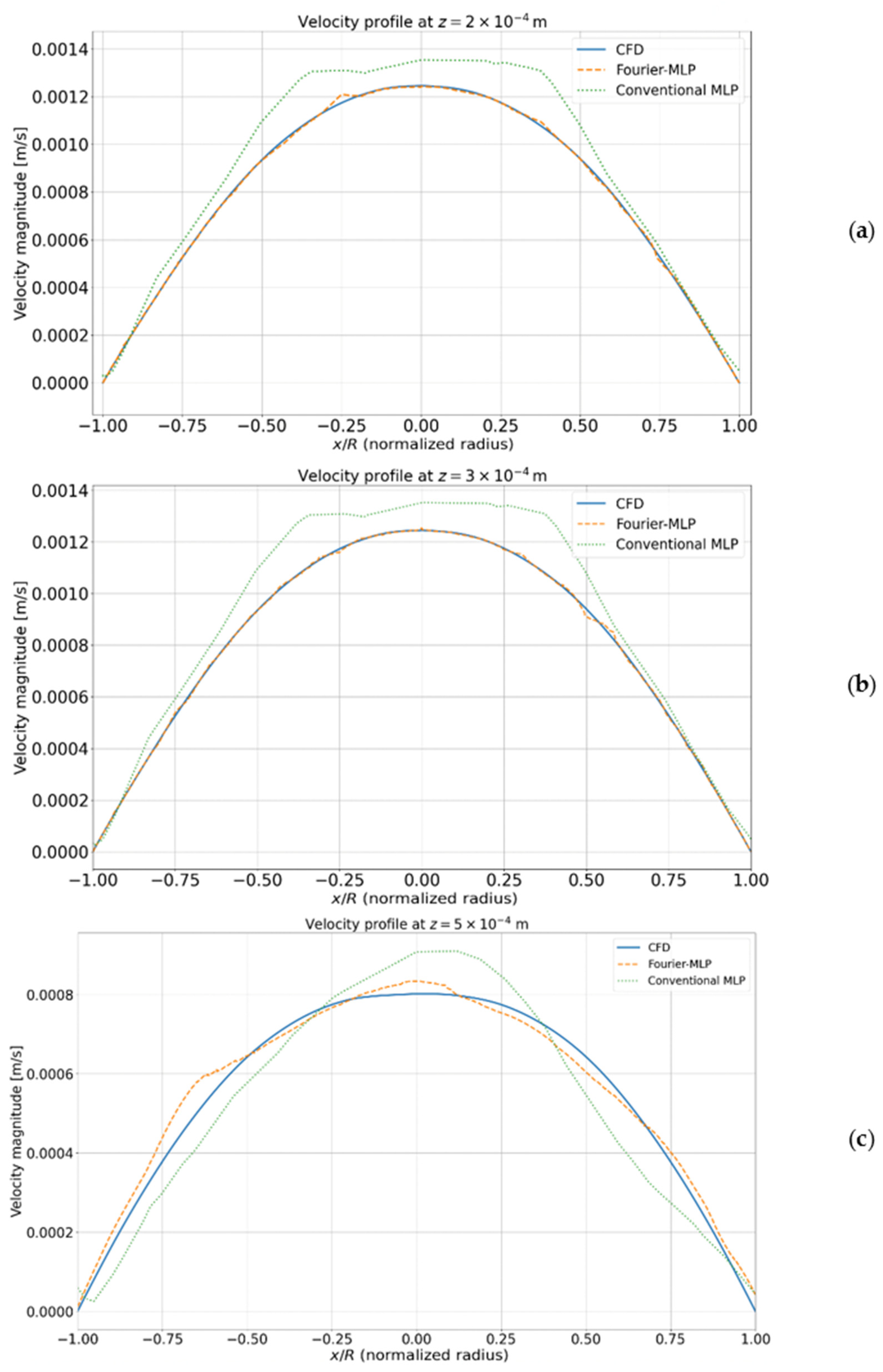
3.2. Results of Surrogate Models
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Taylor, C.A.; Figueroa, C.A. Patient-specific modeling of cardiovascular mechanics. Annu. Rev. Biomed. Eng. 2009, 11, 109–134. [Google Scholar] [CrossRef]
- Morris, P.D.; Narracott, A.; von Tengg-Kobligk, H.; Silva Soto, D.A.; Hsiao, S.; Lungu, A.; Evans, P.; Bressloff, N.W.; Lawford, P.V.; Hose, D.R.; et al. Computational fluid dynamics modelling in cardiovascular medicine. Heart 2016, 102, 18–28. [Google Scholar] [CrossRef]
- Prakash, S.; Ethier, C.R. Requirements for mesh resolution in 3D computational hemodynamics. J. Biomech. Eng. 2001, 123, 134–144. [Google Scholar] [CrossRef]
- Vignon-Clementel, I.E.; Figueroa, C.A.; Jansen, K.E.; Taylor, C.A. Outflow boundary conditions for 3D simulations of non-periodic blood flow and pressure fields in deformable arteries. Comput. Methods Biomech. Biomed. Eng. 2010, 13, 625–640. [Google Scholar] [CrossRef] [PubMed]
- Taylor, C.A.; Petersen, K.; Xiao, N.; Sinclair, M.; Bai, Y.; Lynch, S.R.; UpdePac, A.; Schaap, M. Patient-specific modeling of blood flow in the coronary arteries: A state-of-the-art review. Comput. Methods Appl. Mech. Eng. 2023, 417, 116414. [Google Scholar] [CrossRef]
- Szajer, J.; Ho-Shon, K. A comparison of 4D flow MRI-derived wall shear stress with computational fluid dynamics in intracranial aneurysms. J. Magn. Reson. Imaging 2018, 48, 1249–1258. [Google Scholar] [CrossRef]
- Itu, L.M.; Rapaka, S.; Passerini, T.; Georgescu, B.; Schwemmer, C.; Schoebinger, M.; Rohkohl, C.; Comaniciu, D.; Suciu, C. A machine-learning approach for computation of fractional flow reserve from coronary CT. Med. Image Anal. 2016, 36, 306–317. [Google Scholar] [CrossRef]
- Kissas, G.; Yang, Y.; Hwuang, E.; Witschey, W.R.; Detre, J.A.; Perdikaris, P. Machine learning in cardiovascular flows modeling: Predicting arterial blood pressure from non-invasive 4D flow MRI. Front. Phys. 2020, 8, 54. [Google Scholar] [CrossRef]
- Guo, X.; Li, W.; Iorio, F. Convolutional neural networks for steady flow approximation. In Proceedings of the 22nd ACM SIGKDD International Conference on Knowledge Discovery and Data Mining—KDD ’16, San Francisco, CA, USA, 13–17 August 2016; pp. 481–490. [Google Scholar] [CrossRef]
- Pfaff, T.; Fortunato, M.; Sanchez-Gonzalez, A.; Battaglia, P.W. Learning Mesh-Based Simulation with Graph Networks. arXiv 2020, arXiv:2010.03409. [Google Scholar] [CrossRef]
- Raissi, M.; Perdikaris, P.; Karniadakis, G.E. Physics-informed neural networks: A deep learning framework for solving forward and inverse problems involving nonlinear PDEs. J. Comput. Phys. 2019, 378, 686–707. [Google Scholar] [CrossRef]
- Li, Z.; Kovachki, N.; Azizzadenesheli, K.; Liu, B.; Bhattacharya, K.; Stuart, A.; Anandkumar, A. Fourier Neural Operator for parametric partial differential equations. In Proceedings of the ICLR 2021—The Ninth International Conference on Learning Representations, Virtual, 3–7 May 2021; Available online: https://openreview.net/forum?id=c8P9NQVtmnO (accessed on 15 November 2025).
- MacRaild, M.; Sarrami-Foroushani, A.; Lassila, T.; Frangi, A.F. Accelerated simulation methodologies for computational vascular flow modelling. J. R. Soc. Interface 2024, 21, 20230565. [Google Scholar] [CrossRef]
- Lin, D.; Kenjereš, S. Towards fast and reliable estimations of 3D pressure, velocity and wall shear stress in aortic blood flow: CFD-based machine learning approach. Comput. Biol. Med. 2025, 191, 110137. [Google Scholar] [CrossRef]
- Du, P.; Zhu, X.; Wang, J.-X. Deep learning-based surrogate model for 3-D patient-specific computational fluid dynamics. arXiv 2022, arXiv:2204.08939. [Google Scholar] [CrossRef]
- Catalani, G.; Agarwal, S.; Bertrand, X.; Tost, F.; Bauerheim, M.; Morlier, J. Neural fields for rapid aircraft aerodynamics simulations. Sci. Rep. 2024, 14, 25496. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Jin, P.; Pang, G.; Zhang, Z.; Karniadakis, G.E. Learning nonlinear operators via DeepONet based on the universal approximation theorem of operators. Nat. Mach. Intell. 2021, 3, 218–229. [Google Scholar] [CrossRef]
- Lu, L.; Meng, X.; Cai, S.; Mao, Z.; Goswami, S.; Zhang, Z.; Karniadakis, G.E. A comprehensive and fair comparison of two neural operators (with practical extensions) based on FAIR data. Comput. Methods Appl. Mech. Eng. 2022, 393, 114778. [Google Scholar] [CrossRef]
- Azizzadenesheli, K.; Kovachki, N.; Li, Z.; Liu-Schiaffini, M.; Kossaifi, J.; Anandkumar, A. Neural operators for accelerating scientific simulations and design. Nat. Rev. Phys. 2024, 6, 320–328. [Google Scholar] [CrossRef]
- Benbarka, N.; Höfer, T.; Riaz, H.u.-M.; Zell, A. Seeing Implicit Neural Representations as Fourier Series. In Proceedings of the IEEE/CVF Winter Conference on Applications of Computer Vision (WACV), Waikoloa, HI, USA, 3–8 January 2022. [Google Scholar]
- Duvall, J.; Duraisamy, K. Discretization-independent surrogate modeling of physical fields around variable geometries using coordinate-based networks. Data-Centric Eng. 2025, 6, e5. [Google Scholar] [CrossRef]
- Aravanis, T.; Chrimatopoulos, G.; Xenos, M.; Tzirtzilakis, E.E. Forecasting two-dimensional channel flow using machine learning. Phys. Fluids 2024, 36, 103617. [Google Scholar] [CrossRef]
- Rahimi, A.; Recht, B. Random features for large-scale kernel machines. In Advances in Neural Information Processing Systems; NeurIPS: San Diego, CA, USA, 2007; Volume 20, Available online: https://people.eecs.berkeley.edu/~brecht/papers/07.rah.rec.nips.pdf (accessed on 3 December 2025).
- Tancik, M.; Srinivasan, P.P.; Mildenhall, B.; Fridovich-Keil, S.; Raghavan, N.; Singhal, U.; Ramamoorthi, R.; Barron, J.T.; Ng, R. Fourier features let networks learn high-frequency functions in low-dimensional domains. arXiv 2020, arXiv:2006.10739. [Google Scholar]
- Aravanis, T.; Papadopoulos, P.; Georgikos, D. Fourier Feature-Enhanced Neural Networks for Wind Turbine Power Modeling. Electricity 2025, 6, 70. [Google Scholar] [CrossRef]
- Vaswani, A.; Shazeer, N.; Parmar, N.; Uszkoreit, J.; Jones, L.; Gomez, A.N.; Kaiser, Ł.; Polosukhin, I. Attention is all you need. arXiv 2017, arXiv:1706.03762. [Google Scholar]
- Mildenhall, B.; Srinivasan, P.P.; Tancik, M.; Barron, J.T.; Ramamoorthi, R.; Ng, R. NeRF: Representing scenes as neural radiance fields for view synthesis. arXiv 2020, arXiv:2003.08934. [Google Scholar] [CrossRef]
- Lin, C.-H.; Ma, W.-C.; Torralba, A.; Lucey, S. BARF: Bundle-adjusting neural radiance fields. arXiv 2021, arXiv:2104.06405. [Google Scholar] [CrossRef]
- Barron, J.T.; Mildenhall, B.; Tancik, M.; Hedman, R.; Martin-Brualla, R.; Srinivasan, P.P. Mip-NeRF: A multiscale representation for anti-aliasing neural radiance fields. arXiv 2021, arXiv:2103.13415. [Google Scholar]
- Müller, T.; Evans, A.; Schied, C.; Keller, A. Instant Neural Graphics Primitives with a Multiresolution Hash Encoding. arXiv 2022, arXiv:2201.05989v2. [Google Scholar] [CrossRef]
- Sitzmann, V.; Martel, J.N.P.; Bergman, A.W.; Lindell, D.B.; Wetzstein, D. Implicit neural representations with periodic activation functions. arXiv 2020, arXiv:2006.09661v1. [Google Scholar] [CrossRef]
- Sherman, T.F. On connecting large vessels to small. The meaning of Murray’s law. J. Gen. Physiol. 1981, 78, 431–453. [Google Scholar] [CrossRef]
- Kassab, G.S. Scaling laws of vascular trees: Of form and function. Am. J. Physiol. Heart Circ. Physiol. 2006, 290, H894–H903. [Google Scholar] [CrossRef]
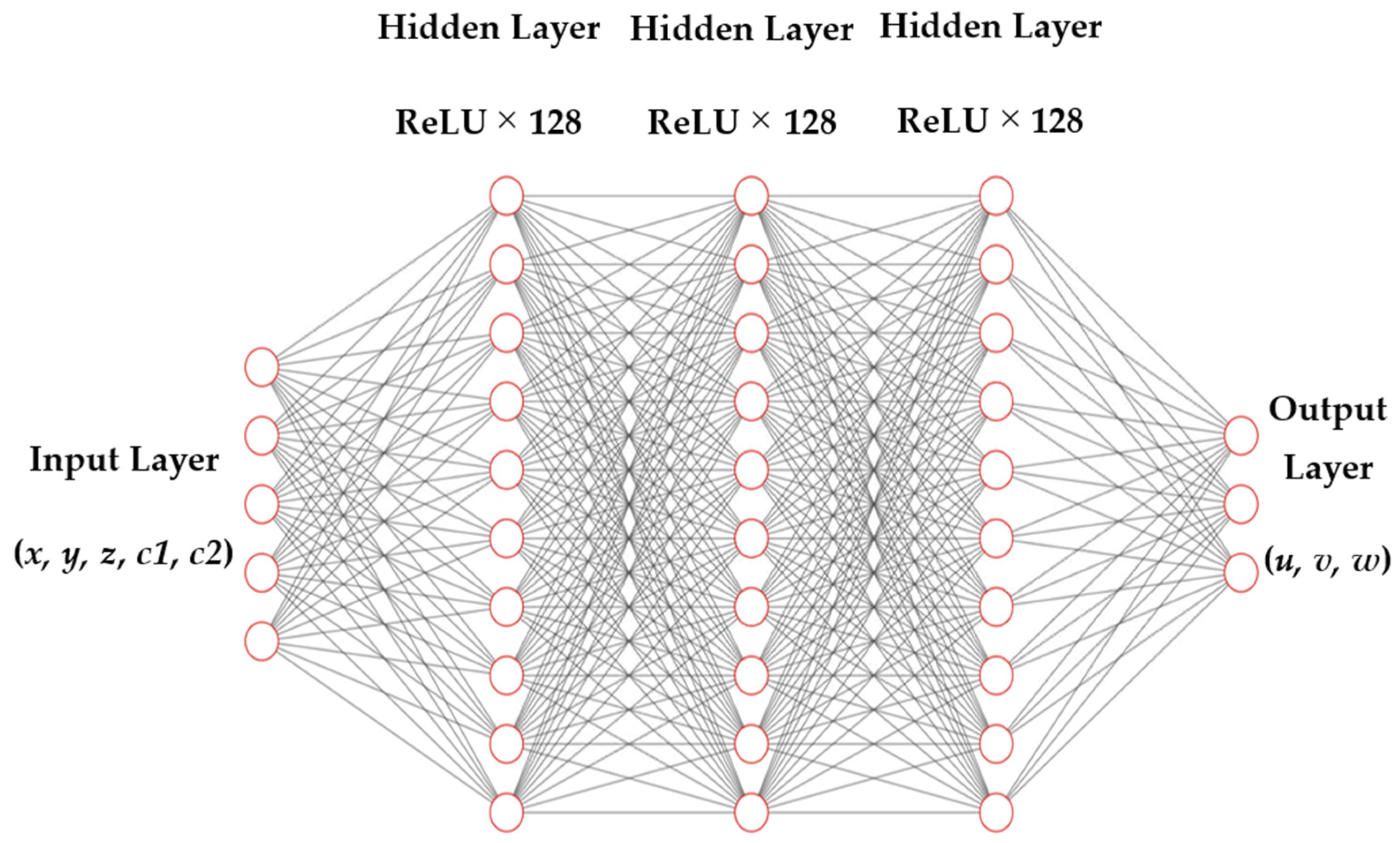











| Settings | Conventional MLP | Fourier MLP |
|---|---|---|
| Hidden layers × width | 3 × 128 | 3 × 128 |
| Activation (backbone) | ReLU | ReLU |
| Spectral embedding | - | RFF on (x, y, z) only |
| Embedding dimension D Spectral bandwidth σ | - | D = 64 σ = 2.0 |
| Input to backbone | [(x, y, z), c] | [φ(x, y, z), c] |
| Loss | MSE on (u, v, w) | MSE on (u, v, w) |
| Optimized/WD | AdamW, 10−4 | AdamW, 10−4 |
| Learning rate | 10−3 cosine anneal | 10−3 cosine anneal |
| Batch size/epochs | 1024/100 | 1024/100 |
| Early stopping/selection | Lowest validation loss | Lowest validation loss |
| EMA of weights | On (0.999) | On (0.999) |
| Meshing Case | Total Number of Elements | Simulation Wall-Clock Time [min] |
|---|---|---|
| 1 | 619,719 | 8.2 |
| 2 | 1,239,438 | 14.7 |
| 3 | 2,478,876 | 140 |
| Stenosis Severity (%) | Number of Points |
|---|---|
| 0 | 226,949 |
| 10 | 231,564 |
| 20 | 236,812 |
| 30 | 241,098 |
| 40 | 245,366 |
| 50 | 248,882 |
| 60 | 252,331 |
| 70 | 255,806 |
| 80 | 260,064 |
| Total | 2,198,872 |
| Split | Number of Points |
|---|---|
| Total | 2,198,872 |
| Training | 1,364,354 |
| Validation | 341,089 |
| External Evaluation (Testing) | 493,429 |
| Model Variant | Time (min) |
|---|---|
| Conventional MLP | 15.16 |
| Fourier MLP | 53.00 |
| Metric | u-Component (x-Direction) | v-Component (y-Direction) | w-Component (z-Direction) |
|---|---|---|---|
| MAE (m/s) | Conventional MLP: 1.00 × 10−5 | Conventional MLP: 7.50 × 10−6 | Conventional MLP: 3.46 × 10−5 |
| Fourier MLP: 1.19 × 10−6 | Fourier MLP: 9.27 × 10−7 | Fourier MLP: 4.88 × 10−6 | |
| RMSE (m/s) | Conventional MLP: 2.06 × 10−5 | Conventional MLP: 1.75 × 10−5 | Conventional MLP: 8.40 × 10−5 |
| Fourier MLP: 3.53 × 10−6 | Fourier MLP: 3.12 × 10−6 | Fourier MLP: 1.52 × 10−5 | |
| R2 | Conventional MLP: 0.9530 | Conventional MLP: 0.9333 | Conventional MLP: 0.9586 |
| Fourier MLP: 0.9986 | Fourier MLP: 0.9979 | Fourier MLP: 0.9986 | |
| NRMSE (global): | Plain MLP: 0.1725 (17.25%) Fourier MLP: 0.0657 (6.57%) |
| Metric | u-Component (x-Direction) | v-Component (y-Direction) | w-Component (z-Direction) |
|---|---|---|---|
| MAE (m/s) | Conventional MLP: 7.28 × 10−6 | Conventional MLP: 5.56 × 10−6 | Conventional MLP: 3.03 × 10−5 |
| Fourier MLP: 2.37 × 10−6 | Fourier MLP: 1.85 × 10−7 | Fourier MLP: 1.15 × 10−5 | |
| RMSE (m/s) | Conventional MLP: 1.24 × 10−5 | Conventional MLP: 1.06 × 10−5 | Conventional MLP: 5.30 × 10−5 |
| Fourier MLP: 4.54 × 10−6 | Fourier MLP: 3.73 × 10−6 | Fourier MLP: 2.03 × 10−5 | |
| R2 | Conventional MLP: 0.9785 | Conventional MLP: 0.9562 | Conventional MLP: 0.9769 |
| Fourier MLP: 0.9971 | Fourier MLP: 0.9946 | Fourier MLP: 0.9966 | |
| NRMSE (global) | Plain MLP: 0.2735 (27.35%) Fourier MLP: 0.0493 (4.93%) |
| z (m) | QCFD (m3/s) | QPlainMLP (m3/s) | Relative Error Plain-MLP (%) | QFourierMLP (m3/s) | Relative Error Fourier MLP (%) |
|---|---|---|---|---|---|
| 2 × 10−4 | 7.69 × 10−12 | 7.79 × 10−12 | 1.2 | 8.05 × 10−12 | 4.56 |
| 3 × 10−4 | 7.62 × 10−12 | 5.80 × 10−12 | 23.97 | 7.83 × 10−12 | 2.5 |
| 5 × 10−4 | 7.65 × 10−12 | 8.85 × 10−12 | 15.5 | 7.66 × 10−12 | 0.1 |
| z (m) | QCFD (m3/s) | QPlainMLP (m3/s) | Relative Error Plain-MLP (%) | QFourierMLP (m3/s) | Relative Error Fourier MLP (%) |
|---|---|---|---|---|---|
| 2 × 10−4 | 7.72 × 10−12 | 7.84 × 10−12 | 1.5 | 7.74 × 10−12 | 0.2 |
| 3 × 10−4 | 7.69 × 10−12 | 6.12 × 10−12 | 20.4 | 7.48 × 10−12 | 2.8 |
| 5 × 10−4 | 7.76 × 10−12 | 1.05 × 10−11 | 35.5 | 7.39 × 10−12 | 4.7 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Papadopoulos, P.N.; Burganos, V.N. Fourier-Feature Neural Surrogate for Hemodynamic Field Reconstruction in Stenotic and Bifurcating Flows. Mach. Learn. Knowl. Extr. 2026, 8, 59. https://doi.org/10.3390/make8030059
Papadopoulos PN, Burganos VN. Fourier-Feature Neural Surrogate for Hemodynamic Field Reconstruction in Stenotic and Bifurcating Flows. Machine Learning and Knowledge Extraction. 2026; 8(3):59. https://doi.org/10.3390/make8030059
Chicago/Turabian StylePapadopoulos, Polydoros N., and Vasilis N. Burganos. 2026. "Fourier-Feature Neural Surrogate for Hemodynamic Field Reconstruction in Stenotic and Bifurcating Flows" Machine Learning and Knowledge Extraction 8, no. 3: 59. https://doi.org/10.3390/make8030059
APA StylePapadopoulos, P. N., & Burganos, V. N. (2026). Fourier-Feature Neural Surrogate for Hemodynamic Field Reconstruction in Stenotic and Bifurcating Flows. Machine Learning and Knowledge Extraction, 8(3), 59. https://doi.org/10.3390/make8030059












