Explainable Deep Learning for Neonatal Jaundice Classification Using Uncalibrated Smartphone Images
Abstract
1. Introduction
2. Critical Review of Existing Work
3. Materials and Methods
3.1. Dataset
3.2. Segmentation of Irregular Region of Interest
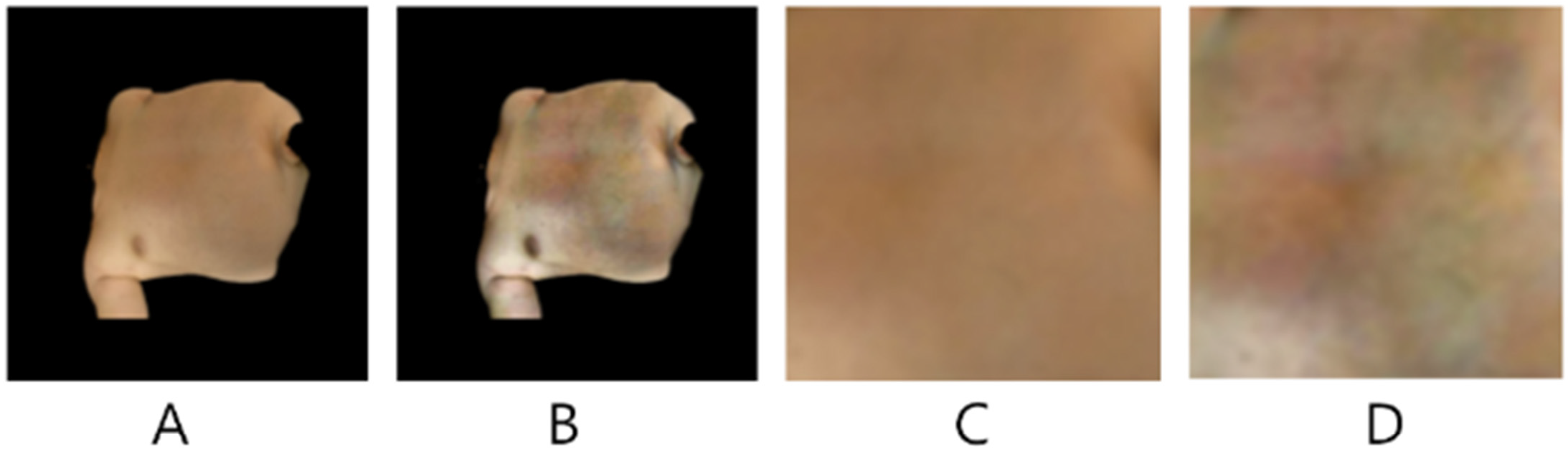
3.3. Contrast Enhancement
3.4. Image Resizing and Patch Extraction
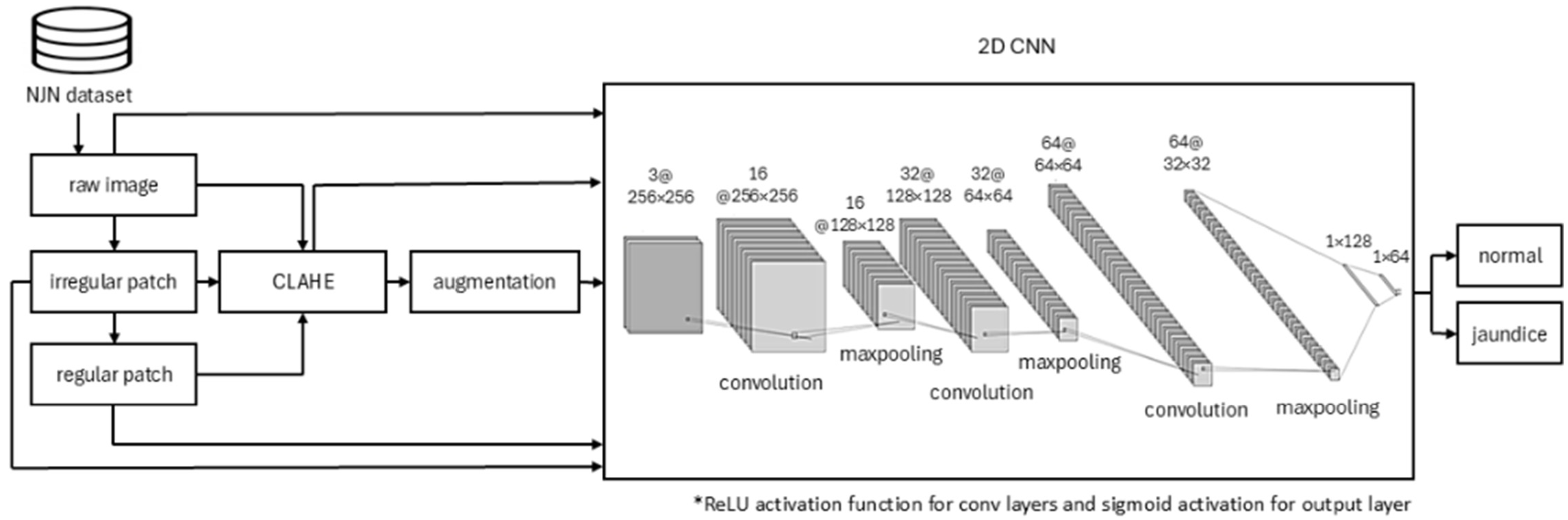
3.5. Binary Classifier 2D CNN Model
3.6. Data Augmentation
3.7. Performance Metrics
3.8. Code Availability
4. Results
4.1. Classification Performance
4.2. Grad-CAM Analysis
5. Discussion
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AUC | Area Under Curve |
| CLAHE | Contrast Limited Adaptive Histogram Equalization |
| CNN | Convolutional Neural Network |
| FN | False Negative |
| FP | False Positive |
| Grad-CAM | Gradient-weighted Class Activation Mapping |
| IR | Imbalance Ratio |
| k-NN | K Nearest Neighbours |
| LOOCV | Leave One Out Cross Validation |
| MAE | Mean Absolute Error |
| NJN | Normal and Jaundice Newborn (Dataset) |
| NICU | Neonatal Intensive Care Unit |
| PCA | Principal Components Analysis |
| ReLU | Rectified Linear Unit |
| ROI | Region of Interest |
| SAM | Segment Anything Model |
| SVM | Support Vector Machine |
| SVR | Support Vector Regression |
| TcB | Transcutaneous Bilirubin |
| TN | True Negative |
| TP | True Positive |
| TSB | Total Serum Bilirubin |
| XAI | Explainable AI |
Appendix A
| SVM Models | #Vectors | TP | TN | FP | FN | Accuracy | Recall | Specificity | Precision | F1 Score |
|---|---|---|---|---|---|---|---|---|---|---|
| Mean RGB | 294 | 49 | 329 | 36 | 103 | 0.73 | 0.32 | 0.90 | 0.58 | 0.41 |
| Median RGB | 292 | 50 | 329 | 36 | 102 | 0.73 | 0.33 | 0.90 | 0.58 | 0.42 |
| Mean YCbCr | 292 | 54 | 329 | 36 | 98 | 0.74 | 0.36 | 0.90 | 0.60 | 0.45 |
| Median YCbCr | 292 | 46 | 331 | 34 | 106 | 0.73 | 0.30 | 0.91 | 0.57 | 0.40 |
| RF Models | #Trees | TP | TN | FP | FN | Accuracy | Recall | Specificity | Precision | F1 Score |
|---|---|---|---|---|---|---|---|---|---|---|
| Mean RGB | 100 | 69 | 317 | 48 | 83 | 0.75 | 0.45 | 0.87 | 0.59 | 0.51 |
| Median RGB | 100 | 73 | 307 | 58 | 79 | 0.74 | 0.48 | 0.84 | 0.56 | 0.52 |
| Mean YCbCr | 100 | 77 | 306 | 59 | 75 | 0.74 | 0.51 | 0.84 | 0.57 | 0.53 |
| Median YCbCr | 100 | 75 | 310 | 55 | 77 | 0.74 | 0.49 | 0.85 | 0.58 | 0.53 |

References
- Hazarika, C.J.; Borah, A.; Gogoi, P.; Ramchiary, S.S.; Daurai, B.; Gogoi, M.; Saikia, M.J. Development of non-invasive biosensors for neonatal jaundice detection: A review. Biosensors 2024, 14, 254. [Google Scholar] [CrossRef]
- Ansong-Assoku, B.; Shah, S.; Adnan, M.; Ankola, P. Neonatal jaundice. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2024. [Google Scholar]
- NHS Jaundice in Newborns, NHS, 2023. Available online: https://www.nhs.uk/conditions/jaundice-newborn/ (accessed on 2 July 2025).
- Mitra, S.; Rennie, J. Neonatal jaundice: Aetiology, diagnosis and treatment. Br. J. Hosp. Med. 2017, 78, 699–704. [Google Scholar] [CrossRef]
- Naghavi, M.; Abajobir, A.A.; Abbafati, C.; Abbas, K.M.; Abd-Allah, F.; Abera, S.F.; Aboyans, V.; Adetokunboh, O.; Afshin, A.; Agrawal, A.; et al. Global, regional, and national age-sex specific mortality for 264 causes of death, 1980–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1151–1210. [Google Scholar] [CrossRef]
- Olusanya, B.O.; Kaplan, M.; Hansen, T.W. Neonatal hyperbilirubinaemia: A global perspective. Lancet Child Adolesc. Health 2018, 2, 610–620. [Google Scholar] [CrossRef]
- Olusanya, B.O.; Teeple, S.; Kassebaum, N.J. The contribution of neonatal jaundice to global child mortality: Findings from the GBD 2016 Study. Pediatrics 2018, 141, e20171471. [Google Scholar] [CrossRef] [PubMed]
- Ayalew, T.; Molla, A.; Kefale, B.; Alene, T.D.; Abebe, G.K.; Ngusie, H.S.; Zemariam, A.B. Factors associated with neonatal jaundice among neonates admitted at referral hospitals in northeast Ethiopia: A facility-based unmatched case-control study. BMC Pregnancy Childbirth 2024, 24, 150. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, A.; Anderson, J.; Bond, M.; Schwarz, R.; Carns, J.; Mtenthaonga, P.; Kumwenda, W.; Johnston, R.; Miros, R.; Dube, Q.; et al. A low-cost bilirubin measurement tool for neonatal jaundice monitoring at the point-of-care: A comparison of BiliDx with a standard laboratory bilirubinometer and transcutaneous bilirubinometer. Lancet Glob. Health 2021, 9, S23. [Google Scholar] [CrossRef]
- Ercan, Ş.; Özgün, G. The accuracy of transcutaneous bilirubinometer measurements to identify hyperbilirubinemia in an outpatient newborn population. Clin. Biochem. 2018, 55, 69–74. [Google Scholar] [CrossRef]
- Okwundu, C.I.; Olowoyeye, A.; Uthman, O.A.; Smith, J.; Wiysonge, C.S.; Bhutani, V.K.; Fiander, M.; Gautham, K.S. Transcutaneous bilirubinometry versus total serum bilirubin measurement for newborns. Cochrane Database Syst. Rev. 2023, 5, CD012660. [Google Scholar] [CrossRef]
- Kirillov, A.; Mintun, E.; Ravi, N.; Mao, H.; Silvester, C.; Dollár, P.; He, K. Segment Anything. In Proceedings of the IEEE/CVF International Conference on Computer Vision (ICCV), Paris, France, 2–3 October 2023; pp. 4015–4026. [Google Scholar] [CrossRef]
- Selvaraju, R.R.; Cogswell, M.; Das, A.; Vedantam, R.; Parikh, D.; Batra, D. Grad-cam: Visual explanations from deep networks via gradient-based localization. In Proceedings of the IEEE International Conference on Computer Vision, Venice, Italy, 22–29 October 2017; pp. 618–626. [Google Scholar] [CrossRef]
- Aune, A.; Vartdal, G.; Bergseng, H.; Randeberg, L.L.; Darj, E. Bilirubin estimates from smartphone images of newborn infants’ skin correlated highly to serum bilirubin levels. Acta Paediatr. 2020, 109, 2532–2538. [Google Scholar] [CrossRef]
- Munkholm, S.B.; Krøgholt, T.; Ebbesen, F.; Szecsi, P.B.; Kristensen, S.R. The smartphone camera as a potential method for transcutaneous bilirubin measurement. PLoS ONE 2018, 13, e0197938. [Google Scholar] [CrossRef]
- Outlaw, F.; Nixon, M.; Odeyemi, O.; MacDonald, L.W.; Meek, J.; Leung, T.S. Smartphone screening for neonatal jaundice via ambient-subtracted sclera chromaticity. PLoS ONE 2020, 15, e0216970. [Google Scholar] [CrossRef]
- Hardalaç, F.; Aydin, M.; Kutbay, U.; Ayturan, K.; AKYEL, A.; Çağlar, A.; Hai, B.; Mert, F. Classification of neonatal jaundice in mobile application with noninvasive imageprocessing methods. Turk. J. Electr. Eng. Comput. Sci. 2021, 29, 2116–2126. [Google Scholar] [CrossRef]
- Porter, M.L.; Dennis, B.L. Hyperbilirubinemia in the term newborn. Am. Fam. Physician 2002, 65, 599–606. [Google Scholar]
- De Greef, L.; Goel, M.; Seo, M.J.; Larson, E.C.; Stout, J.W.; Taylor, J.A.; Patel, S.N. BiliCam: Using mobile phones to monitor newborn jaundice. In Proceedings of the 2014 ACM International Joint Conference on Pervasive and Ubiquitous Computing, Washington, DC, USA, 13–17 September 2014; ACM: New York, NY, USA, 2014; pp. 331–342. [Google Scholar] [CrossRef]
- Sobel, I.; Feldman, G. A 3 × 3 isotropic gradient operator for image processing. Talk Stanf. Artif. Proj. 1968, 1968, 271–272. [Google Scholar]
- Taylor, J.A.; Stout, J.W.; de Greef, L.; Goel, M.; Patel, S.; Chung, E.K.; Koduri, A.; McMahon, S.; Dickerson, J.; Simpson, E.A.; et al. Use of a smartphone app to assess neonatal jaundice. Pediatrics 2017, 140, e20170312. [Google Scholar] [CrossRef] [PubMed]
- Rong, Z.H.; Luo, F.; Ma, L.Y.; Chen, L.; Wu, L.; Liu, W.; Du, L.Z.; Luo, X.P. Evaluation of an automatic image-based screening technique for neonatal hyperbilirubinemia. Chin. J. Pediatr. 2016, 54, 597–600. [Google Scholar] [CrossRef]
- Lingaldinna, S.; Konda, K.C.; Bapanpally, N.; Alimelu, M.; Singh, H.; Ramaraju, M. Validity of bilirubin measured by Biliscan (smartphone application) in neonatal jaundice: An observational study. J. Nepal Paediatr. Soc. 2021, 41, 93–98. [Google Scholar] [CrossRef]
- Ngeow, A.J.H.; Moosa, A.S.; Tan, M.G.; Zou, L.; Goh, M.M.R.; Lim, G.H.; Tagamolila, V.; Ereno, I.; Durnford, J.R.; Cheung, S.K.H.; et al. Development and Validation of a Smartphone Application for Neonatal Jaundice Screening. JAMA Netw. Open 2024, 7, e2450260. [Google Scholar] [CrossRef]
- Aydın, M.; Hardalaç, F.; Ural, B.; Karap, S. Neonatal jaundice detection system. J. Med. Syst. 2016, 40, 166. [Google Scholar] [CrossRef]
- Juliastuti, E.; Nadhira, V.; Satwika, Y.W.; Aziz, N.A.; Zahra, N. Risk zone estimation of newborn jaundice based on skin color image analysis. In Proceedings of the 6th International Conference on Instrumentation, Control, and Automation (ICA), Bandung, Indonesia, 31 July–2 August 2019; pp. 176–181. [Google Scholar] [CrossRef]
- Hakimi Abu Bakar, A.; Hassan, N.M.; Zakaria, A.; Halim, K.A.A.; Halim, A.A.A. Jaundice (hyperbilirubinemia) detection and prediction system using color card technique. In Proceedings of the IEEE 13th International Colloquium on Signal Processing & Its Applications (CSPA), Penang, Malaysia, 10–12 March 2017; pp. 208–213. [Google Scholar] [CrossRef]
- Fitzpatrick, T.B. The validity and practicality of sun-reactive skin types I through VI. Arch. Dermatol. 1988, 124, 869–871. [Google Scholar] [CrossRef] [PubMed]
- Hashim, W.; Al-Naji, A.; Al-Rayahi, I.A.; Oudah, M. Computer vision for jaundice detection in neonates using graphic user interface. IOP Conf. Ser. Mater. Sci. Eng. 2021, 1105, 012076. [Google Scholar] [CrossRef]
- Hashim, W.; Al-Naji, A.; Al-Rayahi, I.A.; Alkhaled, M.; Chahl, J. Neonatal jaundice detection using a computer vision system. Designs 2021, 5, 63. [Google Scholar] [CrossRef]
- Dissaneevate, S.; Wongsirichot, T.; Siriwat, P.; Jintanapanya, N.; Boonyakarn, U.; Janjindamai, W.; Thatrimontrichai, A.; Maneenil, G. A mobile computer-aided diagnosis of neonatal hyperbilirubinemia using digital image processing and machine learning techniques. Int. J. Innov. Res. Sci. Stud. 2022, 5, 10–17. [Google Scholar] [CrossRef]
- Mansor, M.N.; Yaacob, S.; Muthusamy, H.; Nisha, S. PCA-based feature extraction and k-NN algorithm for early jaundice detection. Int. J. Soft Comput. Softw. Eng. 2011, 1, 25–29. [Google Scholar]
- Nihila, S.; Rajalakshmi, T.; Panda, S.S.; Lhazay, N.; Giri, G.D. Detection of jaundice in neonates using artificial intelligence. In Soft Computing: Theories and Applications: Proceedings of SoCTA 2020; Springer Nature: Singapore, 2021; Volume 2, pp. 431–443. [Google Scholar] [CrossRef]
- Mansor, M.; Hariharan, M.; Basah, S.; Yaacob, S. New newborn jaundice monitoring scheme based on combination of pre-processing and color detection method. Neurocomputing 2013, 120, 258–261. [Google Scholar] [CrossRef]
- Nayagi, B.S.; Angel, S.T. Diagnosis of neonatal hyperbilirubinemia using CNN model along with color card techniques. Biomed. Signal Process. Control 2023, 85, 104746. [Google Scholar] [CrossRef]
- Kalbande, D.; Majumdar, A.; Dorik, P.; Prajapati, P.; Deshpande, S. Deep learning approach for early diagnosis of jaundice. In International Conference on Innovative Computing and Communications; Lecture Notes in Networks and Systems; Gupta, D., Khanna, A., Hassanien, A.E., Anand, S., Jaiswal, A., Eds.; Springer Nature: Singapore, 2023; Volume 492, pp. 387–395. [Google Scholar] [CrossRef]
- He, K.; Zhang, X.; Ren, S.; Sun, J. Deep residual learning for image recognition. In Proceedings of the IEEE Conference on Computer Vision and Pattern Recognition, Las Vegas, NV, USA, 27–30 June 2016; pp. 770–778. [Google Scholar] [CrossRef]
- He, K.; Gkioxari, G.; Dollár, P.; Girshick, R. Mask R-CNN. In Proceedings of the IEEE International Conference on Computer Vision, Venice, Italy, 22–29 October 2017; pp. 2961–2969. [Google Scholar] [CrossRef]
- Althnian, A.; Almanea, N.; Aloboud, N. Neonatal jaundice diagnosis using a smartphone camera based on eye, skin, and fused features with transfer learning. Sensors 2021, 21, 7038. [Google Scholar] [CrossRef]
- Zhu, R.; Guo, Y.; Xue, J.H. Adjusting the imbalance ratio by the dimensionality of imbalanced data. Pattern Recognit. Lett. 2020, 133, 217–223. [Google Scholar] [CrossRef]
- Chowdhary, A.; Dutta, S.; Ghosh, R. Neonatal jaundice detection using colour detection method. Int. Adv. Res. J. Sci. Eng. Technol. 2017, 4, 197–203. Available online: https://iarjset.com/upload/2017/july-17/IARJSET%2033.pdf (accessed on 22 October 2025).
- Chakraborty, A.; Goud, S.; Shetty, V.; Bhattacharyya, B. Neonatal jaundice detection system using CNN algorithm and image processing. Int. J. Electr. Eng. Technol. 2020, 11, 248–264. Available online: https://ssrn.com/abstract=3636169 (accessed on 22 October 2025).
- Singla, R.; Singh, S. A framework for detection of jaundice in newborn babies using homomorphic filtering-based image processing. In Proceedings of the 2016 IEEE International Conference on Inventive Computation Technologies (ICICT), Coimbatore, India, 26–27 August 2016; Volume 3, pp. 1–5. [Google Scholar] [CrossRef]
- Aldabbagh, B.; Aziz, M.H. Neonatal jaundice severity detection from skin images using deep transfer learning techniques. J. Electron. Electromed. Eng. Med. Inform. 2024, 7, 92–104. [Google Scholar] [CrossRef]
- Simonyan, K.; Zisserman, A. Very deep convolutional networks for large-scale image recognition. In Proceedings of the 3rd International Conference on Learning Representations (ICLR 2015), San Diego, CA, USA, 7–9 May 2015. [Google Scholar] [CrossRef]
- Abdulrazzak, A.Y.; Mohammed, S.L.; Al-Naji, A.; Chahl, J. Real-time jaundice detection in neonates based on machine learning models. BioMedInformatics 2024, 4, 623–637. [Google Scholar] [CrossRef]
- Abdulrazzak, A.Y.; Mohammed, S.L.; Al-Naji, A. NJN: A Dataset for the Normal and Jaundiced Newborns. BioMedInformatics 2023, 3, 543–552. [Google Scholar] [CrossRef]
- Gupta, K.; Sharma, V.; Kathait, S.S. Smart screening: Non-invasive detection of severe neonatal jaundice using computer vision and deep learning. Int. J. Comput. Appl. 2024, 186, 35–43. [Google Scholar] [CrossRef]
- Makhloughi, F. Artificial intelligence-based non-invasive bilirubin prediction for neonatal jaundice using 1D convolutional neural network. Sci. Rep. 2025, 15, 11571. [Google Scholar] [CrossRef]
- Salami, F.O.; Muzammel, M.; Mourchid, Y.; Othmani, A. Artificial intelligence non-invasive methods for neonatal jaundice detection: A review. Artif. Intell. Med. 2025, 162, 103088. [Google Scholar] [CrossRef]
- Zuiderveld, K. Contrast limited adaptive histogram equalization. In Graphics Gems IV; Academic Press: Cambridge, MA, USA, 1994; pp. 474–485. [Google Scholar] [CrossRef]
- Glorot, X.; Bordes, A.; Bengio, Y. Deep sparse rectifier neural networks. In Proceedings of the 14th International Conference on Artificial Intelligence and Statistics (AISTATS), Fort Lauderdale, FL, USA, 11–13 April 2011; Volume 15. [Google Scholar]
- van der Linde, I. Why the confusion matrix fails as a model of knowledge. AI Soc. 2025. [Google Scholar] [CrossRef]
- Abebe, M.A.; Hardeberg, J.Y.; Vartdal, G. Smartphones’ skin colour reproduction analysis for neonatal jaundice detection. J. Imaging Sci. Technol. 2021, 34, 060407-1–060407-15. [Google Scholar] [CrossRef]



| A. Without Augmentation | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Input Type | CLAHE | TP | TN | FP | FN | Accuracy | Recall | Specificity | Precision | F1 Score |
| SAM Irregular 512 × 512 | N | 45 | 19 | 18 | 10 | 0.70 | 0.71 | 0.51 | 0.82 | 0.76 |
| Y | 39 | 27 | 9 | 17 | 0.72 | 0.81 | 0.75 | 0.70 | 0.75 | |
| SAM Patch 144 × 144 | N | 38 | 29 | 13 | 12 | 0.73 | 0.75 | 0.69 | 0.76 | 0.75 |
| Y | 39 | 33 | 7 | 13 | 0.78 | 0.85 | 0.83 | 0.75 | 0.80 | |
| Raw Image | N | 32 | 22 | 20 | 18 | 0.59 | 0.62 | 0.52 | 0.64 | 0.63 |
| Y | 33 | 25 | 21 | 13 | 0.63 | 0.61 | 0.54 | 0.72 | 0.66 | |
| B. With Augmentation | ||||||||||
| Input Type | CLAHE | TP | TN | FP | FN | Accuracy | Recall | Specificity | Precision | F1 Score |
| SAM Irregular 512 × 512 | N | 30 | 35 | 12 | 15 | 0.71 | 0.71 | 0.74 | 0.67 | 0.69 |
| Y | 32 | 34 | 10 | 16 | 0.72 | 0.76 | 0.77 | 0.67 | 0.71 | |
| SAM Patch 144 × 144 | N | 34 | 36 | 12 | 10 | 0.76 | 0.74 | 0.75 | 0.77 | 0.76 |
| Y | 36 | 37 | 9 | 10 | 0.79 | 0.80 | 0.81 | 0.78 | 0.79 | |
| Raw Image | N | 26 | 30 | 20 | 16 | 0.61 | 0.57 | 0.60 | 0.62 | 0.59 |
| Y | 30 | 29 | 16 | 17 | 0.64 | 0.65 | 0.64 | 0.64 | 0.65 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chakraborty, A.; Thota, Y.; Luca, C.; van der Linde, I. Explainable Deep Learning for Neonatal Jaundice Classification Using Uncalibrated Smartphone Images. Mach. Learn. Knowl. Extr. 2025, 7, 136. https://doi.org/10.3390/make7040136
Chakraborty A, Thota Y, Luca C, van der Linde I. Explainable Deep Learning for Neonatal Jaundice Classification Using Uncalibrated Smartphone Images. Machine Learning and Knowledge Extraction. 2025; 7(4):136. https://doi.org/10.3390/make7040136
Chicago/Turabian StyleChakraborty, Ashim, Yeshwanth Thota, Cristina Luca, and Ian van der Linde. 2025. "Explainable Deep Learning for Neonatal Jaundice Classification Using Uncalibrated Smartphone Images" Machine Learning and Knowledge Extraction 7, no. 4: 136. https://doi.org/10.3390/make7040136
APA StyleChakraborty, A., Thota, Y., Luca, C., & van der Linde, I. (2025). Explainable Deep Learning for Neonatal Jaundice Classification Using Uncalibrated Smartphone Images. Machine Learning and Knowledge Extraction, 7(4), 136. https://doi.org/10.3390/make7040136








