The Influence of Alkaline Pretreatment of Waste Nutshell for Use in Particulate Biocomposites
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Sample Preparation
2.3. Characterization
3. Results and Discussion
3.1. Crystallinity Index
3.2. FTIR Analysis
3.3. Simultaneous DTA/TGA Analysis
3.4. SEM Analysis
3.5. Surface Free Energy and Adhesion Parameters
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- He, K.; Zhang, J.; Zeng, Y. Knowledge domain and emerging trends of agricultural waste management in the field of social science: A scientometric review. Sci. Total Environ. 2019, 670, 236–244. [Google Scholar] [CrossRef]
- Sinka, M.; Korjakins, A.; Bajare, D.; Zimele, Z.; Sahmenko, G. Bio-based construction panels for low carbon development. Energy Procedia 2018, 147, 220–226. [Google Scholar] [CrossRef]
- Jannat, N.; Al-Mufti, R.L.; Hussein, A.; Abdullah, B.; Catgrave, A. Utilisation of nut shell wastes in brick, mortar and concrete: A review. Constr. Build. Mater. 2021, 293, 123546. [Google Scholar] [CrossRef]
- Demirbas, A. Calculation of higher heating values of biomass fuels. Fuel 1997, 76, 431–434. [Google Scholar] [CrossRef]
- Sujatha, A.; Balakrishnan, S.D. Properties of coconut shell aggregate concrete: A review. Adv. Civil Eng. 2020, 83, 759–769. [Google Scholar]
- Huang, G.; Lapsley, K. Almonds. In Integrated Processing Technologies for Food and Agricultural By-Products, 1st ed.; Pan, Z., Zhang, R., Zicari, S., Eds.; Academic Press: London, UK, 2019; pp. 373–390. [Google Scholar] [CrossRef]
- Demirbas, A. Fuel characteristics of olive husk and walnut, hazelnut, sunflower, and almond shells. Energy Sources 2002, 24, 215–221. [Google Scholar] [CrossRef]
- Yalchi, T. Determination of digestibility of almond hull in sheep. Afr. J. Biotechnol. 2010, 10, 3022–3026. [Google Scholar] [CrossRef]
- Aktas, T.; Thy, P.; Williams, R.B.; McCaffrey, Z.; Khatami, R.; Jenkins, B.M. Characterization of almond processing residues from the Central Valley of California for thermal conversion. Fuel Process. Technol. 2015, 140, 132–147. [Google Scholar] [CrossRef]
- Messaoudi, Y.; Smichi, N.; Bouachir, F.; Gargouri, M. Fractionation and biotransformation oflignocelluloses-based wastes for bioethanol, xylose and vanillin production. Waste Biomass Valoriz. 2017, 10, 357–367. [Google Scholar] [CrossRef]
- McCaffrey, Z.; Torres, L.; Flynn, S.; Cao, T.; Chiou, B.S.; Klamczynski, A.; Glenn, G.; Orts, M.J. Recycled polypropylene-polyethylene torrefied almond shell biocomposites. Ind. Crops Prod. 2018, 25, 425–432. [Google Scholar] [CrossRef]
- Al-Ajji, M.A.; Al-Ghouti, M.A. Novel insights into the nanoadsorption mechanisms of crystal violet using nano-hazelnut shell from aqueous solution. J. Water Process Eng. 2021, 44, 102354. [Google Scholar] [CrossRef]
- Contini, M.; Baccelloni, S.; Massantini, R.; Anelli, G. Extraction of natural antioxidants from hazelnut (Corylus avellana L.) shell and skin wastes by long maceration at room temperature. Food Chem. 2008, 110, 659–669. [Google Scholar] [CrossRef]
- Çam, A.S. Characterization of Clay Brick Materials Produced with Red Mud and Nut Shell Wastes for Building Applications. Master’s Thesis, Izmir Katip Çelebi University, Izmir, Turkey, 2017. Available online: http://hdl.handle.net/11469/683 (accessed on 17 July 2023).
- Demirbas, A.; Aslan, A. Effects of ground hazelnut shell, wood, and tea waste on the mechanical properties of cement. Cem. Concr. Res. 1998, 28, 1101–1104. [Google Scholar] [CrossRef]
- Brleković, F.; Fiolić, T.; Šipušić, J. Sustainable insulating composite from almond shell. In Proceedings of the 2nd International Conference Construction Materials for a Sustainable Future, CoMS 2020/21, Bled, Slovenia, 20–21 April 2021; Šajna, A., Legat, A., Jordan, S., Horvat, P., Kemperle, E., Dolenec, S., Ljubešek, M., Michelizza, M., Eds.; Slovenian National Building and Civil Engineering Institute: Ljubljana, Slovenia, 2020; pp. 32–39. [Google Scholar]
- Abdelmouleh, M.; Boufi, S.; Belgacem, M.N.; Dufresne, A. Short natural-fibre reinforced polyethylene and natural rubber composites: Effect of silane coupling agents and fibres loading. Compos. Sci. Technol. 2007, 67, 1627–1639. [Google Scholar] [CrossRef]
- Bychkov, A.L.; Podgorbunskikh, E.M.; Ryabchikova, E.I.; Lomovsky, O.I. The role of mechanical action in the process of the thermomechanical isolation of lignin. Cellulose 2018, 25, 338–347. [Google Scholar] [CrossRef]
- Perez-Rodriguez, N.; Garcia-Bernet, D.; Dominguez, J.M. Faster methane production after sequential extrusion and enzymatic hydrolysis of vine trimming shoots. Environ. Chem. Lett. 2017, 16, 295–299. [Google Scholar] [CrossRef]
- Contreras-Hernández, M.G.; Ochoa-Martínez, L.A.; Rutiaga-Quiñones, J.G.; Rocha-Guzmán, N.E.; Lara-Ceniceros, T.E.; Contreras-Esquivel, J.C.; Barragán, L.P.; Rutiaga-Quiñones, O.M. Effect of ultrasound pre-treatment on the physicochemical composition of Agave durangensis leaves and potential enzyme production. Bioresour. Technol. 2018, 249, 439–446. [Google Scholar] [CrossRef]
- Kumar, A.K.; Sharma, S. Recent updates on different methods of pretreatment of lignocellulosic feedstocks: A review. Bioresour. Bioprocess. 2017, 4, 7. [Google Scholar] [CrossRef]
- Hasan, A.; Rabbi, M.S.; Billah, M.M.D. Making the lignocellulosic fibers chemically compatible for composite: A comprehensive review. Clean. Mater. 2022, 4, 100078. [Google Scholar] [CrossRef]
- Jedruejczyk, M.; Soszka, E.; Czapnik, M.; Ruppert, A.M.; Grams, J. Physical and chemical pretreatment of lignocellulosic biomass. In Second and Third Generation of Feedstocks: The Evolution of Biofuels, 1st ed.; Basile, A., Dalena, F., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 143–196. [Google Scholar]
- Kriven, W.M. 5.9 Geopolymer-based composites. In Comprehensive Composite Materials II; Beaumont, P.W.R., Zweben, C.H., Eds.; Academic Press: Oxford, UK, 2018; Volume 5, pp. 269–280. [Google Scholar]
- Davidovits, J. Geopolymer Chemistry and Applications, 5th ed.; Institut Geopolymere: Saint-Quentin, France, 2020; pp. 3–338. [Google Scholar]
- Alawi, A.; Milad, A.; Barbieri, D.; Alosta, M.; Alaneme, G.U.; Imran Latif, Q.B.A. Eco-Friendly geopolymer composites prepared from agro-industrial wastes: A state-of-the-art review. CivilEng 2023, 4, 433–453. [Google Scholar] [CrossRef]
- Mahmood, A.; Noman, M.T.; Pechočiaková, M.; Amor, N.; Petru, M.; Abdelkader, M.; Militký, J.; Sozcu, S.; Hassan, S.Z.U. Geopolymers and fiber-reinforced concrete composites in civil engineering. Polymers 2021, 13, 2099. [Google Scholar] [CrossRef]
- Fityk Curve Fitting and Data Analysis. Available online: https://fityk.nieto.pl/ (accessed on 25 February 2023).
- Pustak, A.; Denac, M.; Leskovac, M.; Švab, I.; Musil, V.; Šmit, I. Structure and morphology of silica-reinforced polypropylene composites modified with m-EPR copolymers. J. Polym. Res. 2016, 23, 37. [Google Scholar] [CrossRef]
- Chen, H.; Yu, Y.; Zhong, T.; Wu, Y. Effect of alkali treatment on microstructure and mechanical properties of individual bamboo fibers. Cellulose 2016, 24, 333–347. [Google Scholar] [CrossRef]
- Sun, Q.; Foston, M.; Sawada, D.; Pingali, S.V.; O’Neill, H.M.; Li, H.; Wyman, C.E.; Langan, P.; Pu, Y.; Ragauskas, A.J. Comparison of changes in cellulose ultrastructure during different pretreatments of poplar. Cellulose 2014, 21, 2419–2431. [Google Scholar] [CrossRef]
- Ferreira, S.R.; Silva, F.D.A.; Lima, P.R.L.; Toledo Filho, R.D. Effect of fiber treatments on the sisal fiber properties and fiber-matrix bond in cement based systems. Constr. Build. Mater. 2015, 101, 730–740. [Google Scholar] [CrossRef]
- Sanchez-Echeverri, L.A.; Medina-Perilla, J.A.; Ganjian, E. Nonconventional Ca(OH)2 Treatment of Bamboo for the Reinforcement of Cement Composites. Materials 2020, 13, 1892. [Google Scholar] [CrossRef]
- Le Troedec, M.; Sedan, D.; Peyratout, C.; Bonnet, J.P.; Smith, A.; Guinebretiere, R.; Gloaguen, V.; Krausz, P. Influence of various chemical treatments on the composition and structure of hemp fibres. Compos. Part A Appl. Sci. Manuf. 2008, 39, 514–522. [Google Scholar] [CrossRef]
- Rahman, A.; Ulven, C.A.; Johnson, M.A.; Durant, C.; Hossain, K.G. Pretreatment of Wheat Bran for Suitable Reinforcement in Biocomposites. J. Renew. Mater. 2017, 5, 62–73. [Google Scholar] [CrossRef]
- Barreto, A.C.H.; Rosa, D.S.; Fechine, P.B.A.; Mazzetto, S.E. Properties of sisal fibers treated by alkali solution and their application into cardanol-based biocomposites. Compos. Part A Appl. Sci. Manuf. 2011, 42, 492–500. [Google Scholar] [CrossRef]
- Zhang, F.-D.; Xu, C.; Li, M.-Y.; Chen, X.-D. Identification of Dalbergia cochinchinensis (CITES Appendix II) from other three Dalbergia species using FT-IR and 2D correlation IR spectroscopy. Wood Sci. Technol. 2016, 50, 693–704. [Google Scholar] [CrossRef]
- Javier-Astete, R.; Jimenez-Davalos, J.; Zolla, G. Determination of hemicellulose, cellulose, holocellulose and lignin content using FTIR in Calycophyllum spruceanum (Benth.) K. Schum. and Guazuma crinita Lam. PLoS ONE 2021, 16, e0256559. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Liu, Y.; Hao, J.; Wang, W. Study of Almond Shell Characteristics. Materials 2018, 11, 1782. [Google Scholar] [CrossRef] [PubMed]


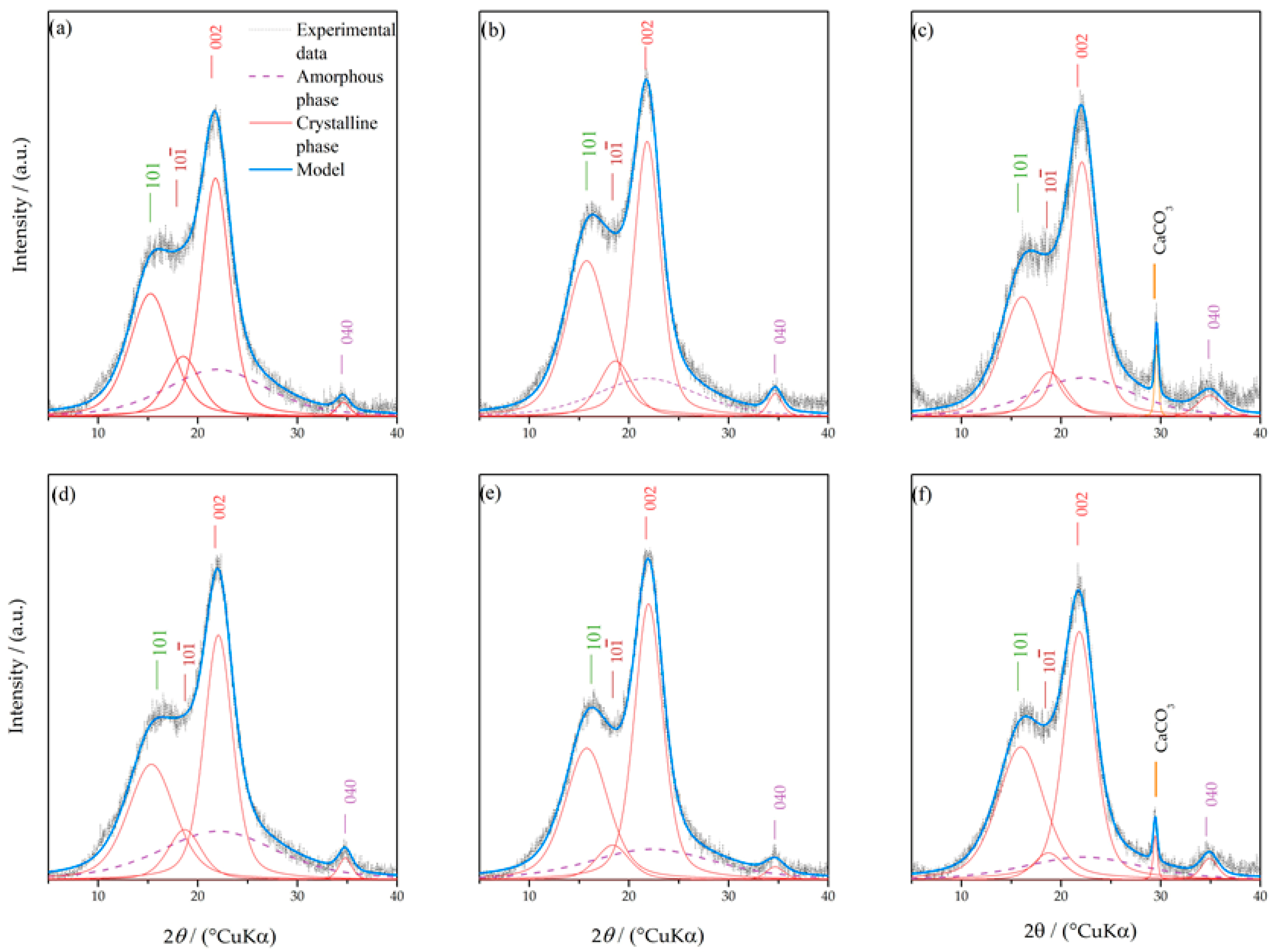
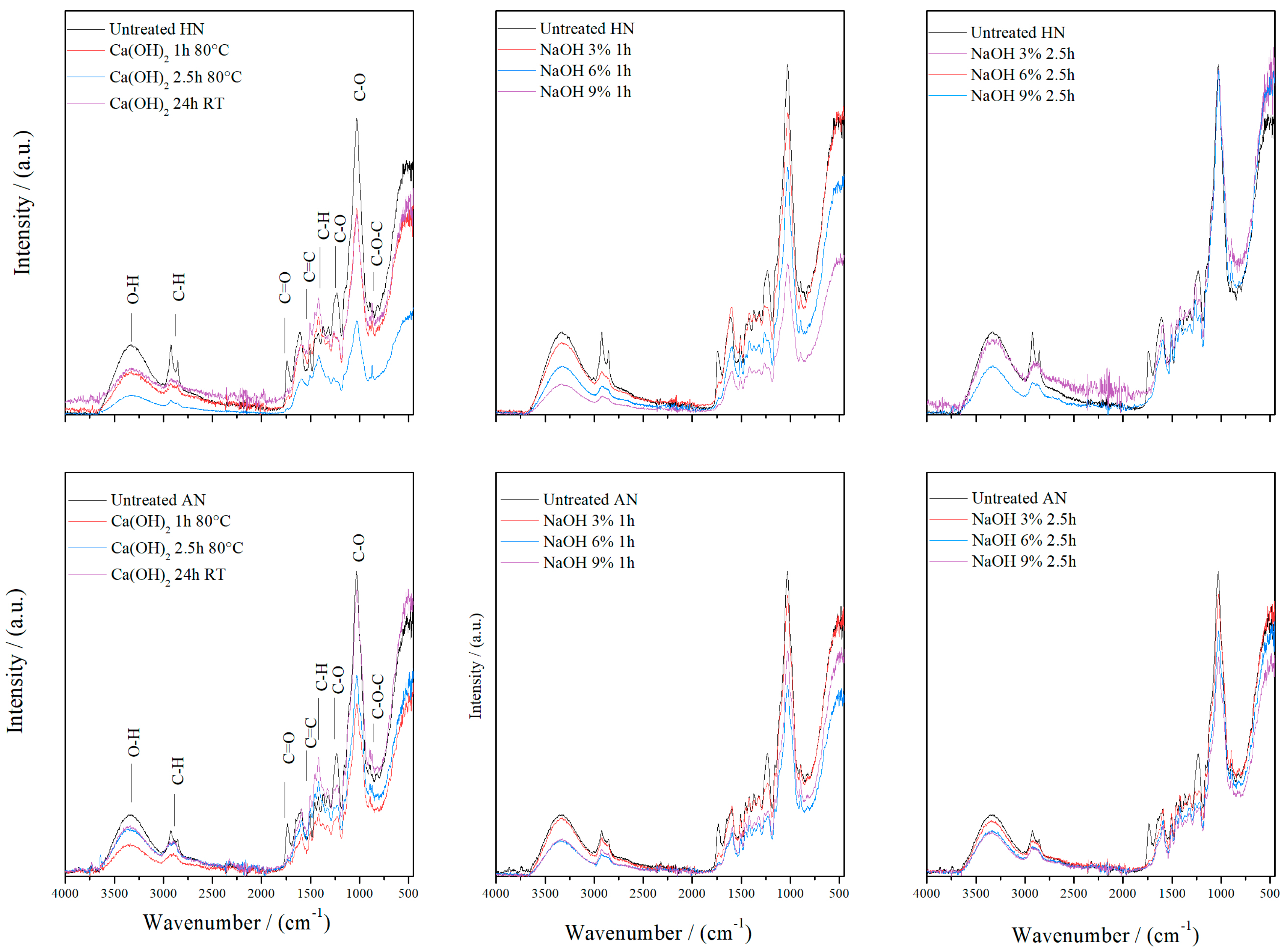
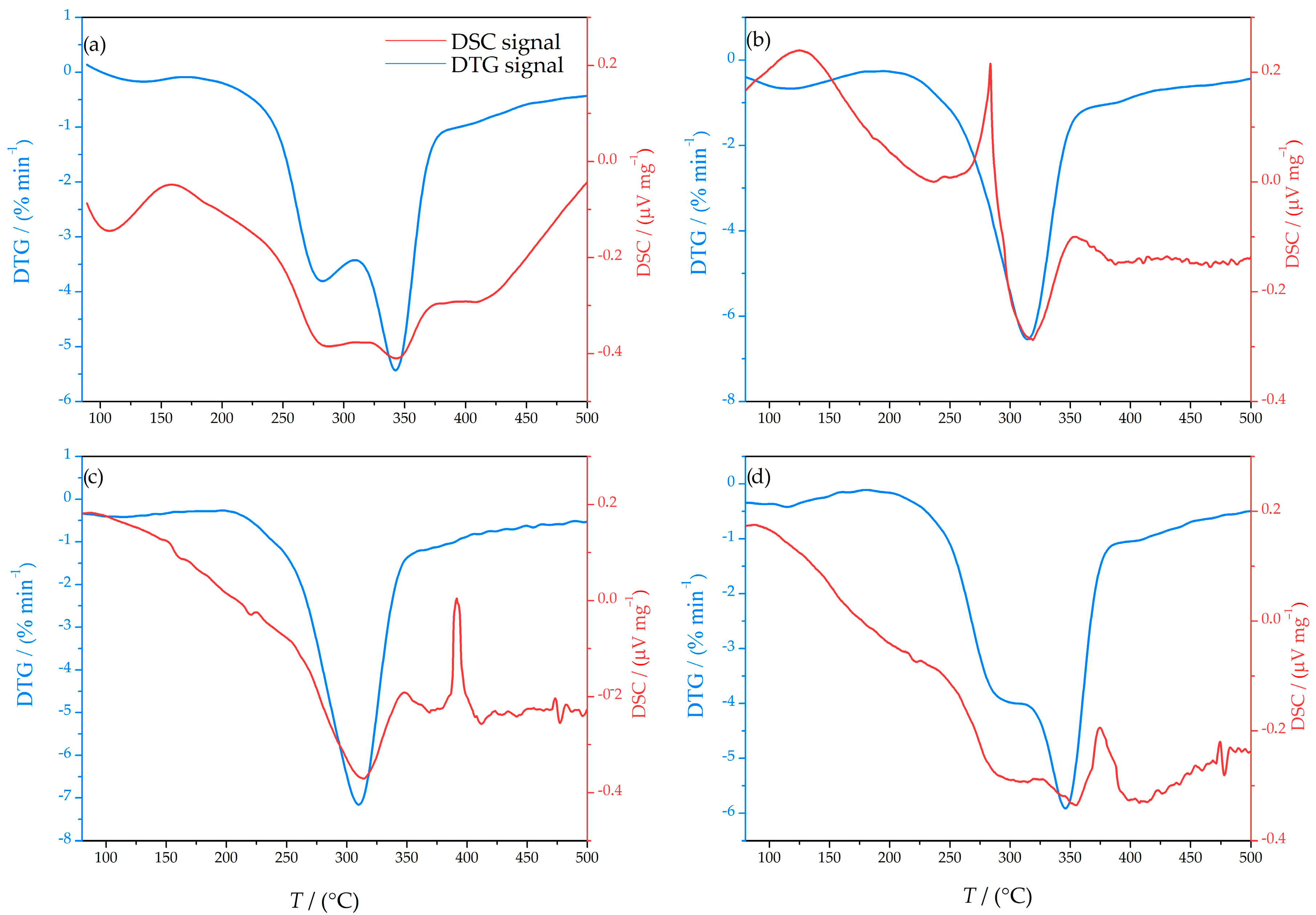
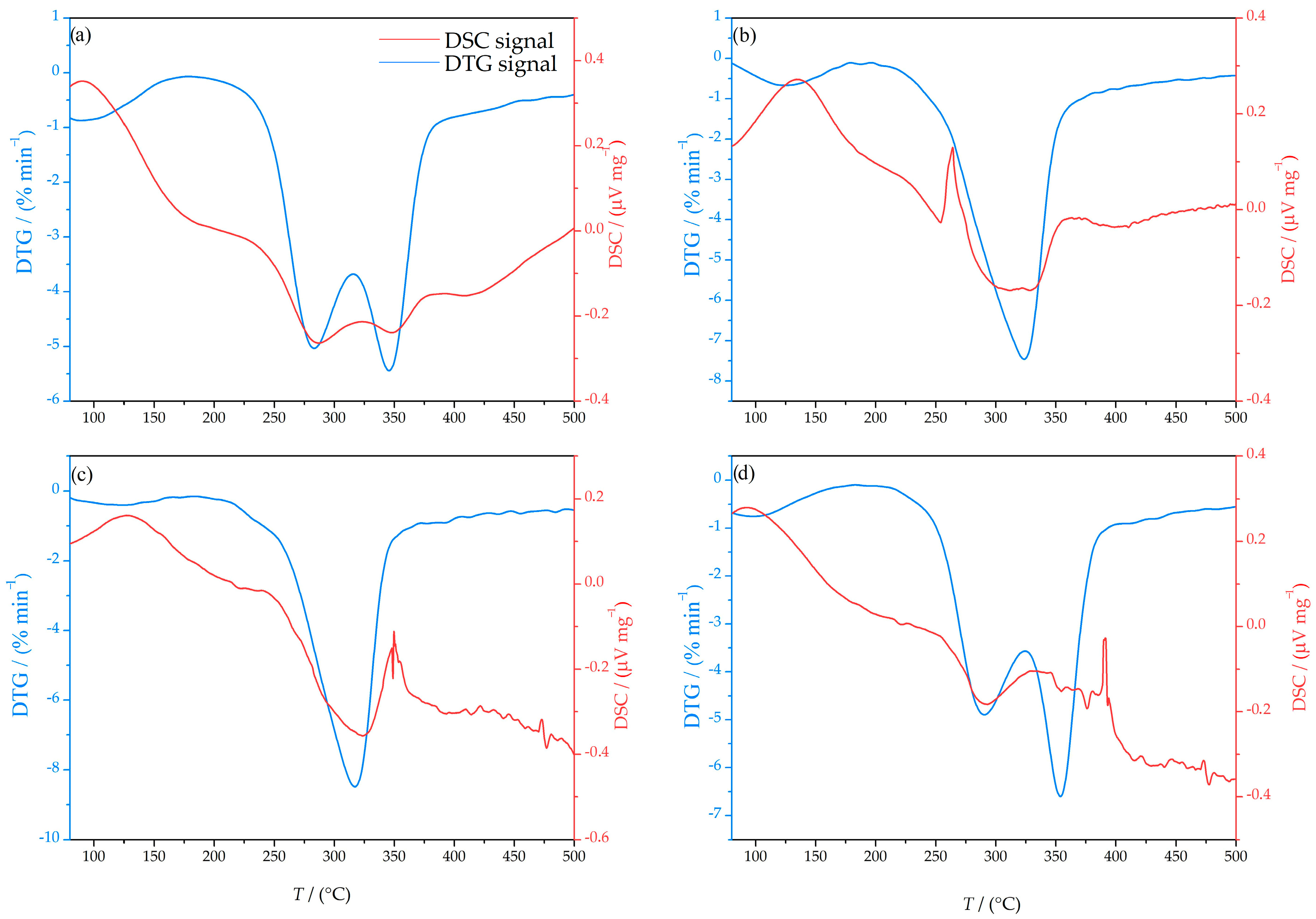



| Fraction (mm) | >2.5 | 2.5–1.25 | 1.25–0.8 | <0.8 |
| Hazelnut WN (wt%) | 4.35 | 69.90 | 11.18 | 13.77 |
| Almond WN (wt%) | 6.7 | 68.21 | 12.05 | 12.18 |
| Untreated | Ca(OH)2 | NaOH | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Concentration (%) | - | 12.5 | 3 | 6 | 9 | |||||
| Temperature (°C) | - | 80 | 25 | 80 | ||||||
| Duration (h) | - | 1 | 2.5 | 24 | 1 | 2.5 | 1 | 2.5 | 1 | 2.5 |
| Hazelnut CI (%) | 55 | 66 | 71 | 66 | 60 | 67 | 63 | 65 | 77 | 61 |
| Almond CI (%) | 51 | 63 | 68 | 66 | 63 | 61 | 65 | 63 | 65 | 68 |
| OWRK | γd, mJ m−2 | γp, mJ m−2 | γ, mJ m−2 | |||||
|---|---|---|---|---|---|---|---|---|
| Hazelnut | Almond | Hazelnut | Almond | Hazelnut | Almond | |||
| Untreated nutshell | 25.0 | 36.7 | 5.2 | 13.8 | 30.2 | 50.5 | ||
| Ca(OH)2 | 1 h, 80 °C | 30.1 | 29.8 | 0.3 | 2.1 | 30.4 | 31.9 | |
| 2.5 h, 80 °C | 33.5 | 39.9 | 0.1 | 3.4 | 33.6 | 43.3 | ||
| 24 h, RT | 32.6 | 38.7 | 0.4 | 1.6 | 33 | 40.3 | ||
| NaOH | 3% | 1 h, 80 °C | 23.5 | 36.4 | 4.8 | 1.8 | 28.3 | 38.2 |
| 2.5 h, 80 °C | 25.8 | 35.9 | 6.0 | 3.4 | 31.8 | 39.3 | ||
| 6% | 1 h, 80 °C | 18.0 | 29.1 | 6.5 | 3.9 | 24.5 | 33.0 | |
| 2.5 h, 80 °C | 30.7 | 38.4 | 9.2 | 4.6 | 39.9 | 43.0 | ||
| 9% | 1 h, 80 °C | 13.4 | 22.4 | 7.3 | 6.6 | 20.7 | 29.0 | |
| 2.5 h, 80 °C | 32.3 | 34.4 | 8.8 | 1.7 | 41.1 | 36.1 | ||
| Geopolymer Sample | OWRK | ||
|---|---|---|---|
| γld. mJ m−2 | γlp. mJ m−2 | γl. mJ m−2 | |
| MNa40C | 38.0 | 24.4 | 62.4 |
| MNaRT | 36.2 | 31.5 | 67.7 |
| MKRT | 38.5 | 37.7 | 76.2 |
| FANaRT | 11.2 | 63.7 | 74.9 |
| FAKRT | 20.3 | 52.6 | 72.9 |
| Hazelnut Nutshell | MNa40C | MNaRT | MKRT | FANaRT | FAKRT | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| γ12 | W12 | S12 | γ12 | W12 | S12 | γ12 | W12 | S12 | γ12 | W12 | S12 | γ12 | W12 | S12 | |||
| Untreated nutshell | 8.4 | 84.2 | 28.4 | 12.1 | 85.7 | 25.3 | 16.3 | 90.0 | 27.0 | 34.4 | 70.7 | 10.3 | 24.9 | 78.1 | 17.7 | ||
| Ca(OH)2 | 1 h, 80 °C | 16.7 | 73.1 | 12.4 | 25.8 | 72.2 | 11.5 | 31.6 | 75.0 | 14.2 | 58.6 | 56.7 | −14.1 | 45.7 | 57.5 | −3.3 | |
| 2.5 h, 80 °C | 21.4 | 74.7 | 7.5 | 27.9 | 73.3 | 6.1 | 33.9 | 76.0 | 8.7 | 63.3 | 45.2 | −22.0 | 49.5 | 56.9 | −10.2 | ||
| 24 h, RT | 19.0 | 76.5 | 10.5 | 25.1 | 75.5 | 9.5 | 30.8 | 78.4 | 12.4 | 58.8 | 49.1 | −16.9 | 45.6 | 60.3 | −5.7 | ||
| NaOH | 3% | 1 h, 80 °C | 9.3 | 81.4 | 24.9 | 13.1 | 82.8 | 26.3 | 17.4 | 87.0 | 30.5 | 35.0 | 68.2 | 11.6 | 25.7 | 75.4 | 18.8 |
| 2.5 h, 80 °C | 7.3 | 87.0 | 23.2 | 10.8 | 88.7 | 25.0 | 14.8 | 93.2 | 29.5 | 32.7 | 74.1 | 10.3 | 23.3 | 81.4 | 17.7 | ||
| 6% | 1 h, 80 °C | 9.4 | 77.5 | 28.5 | 12.5 | 79.6 | 30.6 | 16.8 | 83.9 | 34.9 | 29.7 | 69.7 | 20.7 | 22.2 | 75.1 | 26.1 | |
| 2.5 h, 80 °C | 4.0 | 98.3 | 18.5 | 6.9 | 100.7 | 20.9 | 10.0 | 106.0 | 26.2 | 28.4 | 86.4 | 6.6 | 18.8 | 93.9 | 14.1 | ||
| 9% | 1 h, 80 °C | 11.3 | 71.8 | 30.5 | 14.0 | 74.3 | 33.0 | 18.3 | 78.6 | 37.2 | 27.4 | 68.2 | 26.8 | 21.4 | 72.1 | 30.8 | |
| 2.5 h, 80 °C | 4.1 | 99.4 | 17.2 | 7.0 | 101.7 | 19.5 | 10.3 | 107.0 | 24.8 | 29.7 | 86.4 | 4.2 | 19.7 | 94.2 | 12.0 | ||
| Almond Nutshell | MNa40C | MNaRT | MKRT | FANaRT | FAKRT | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| γ12 | W12 | S12 | γ12 | W12 | S12 | γ12 | W12 | S12 | γ12 | W12 | S12 | γ12 | W12 | S12 | |||
| Untreated nutshell | 1.5 | 111.4 | 10.5 | 3.6 | 114.5 | 13.6 | 5.9 | 120.8 | 19.8 | 24.6 | 100.8 | −0.2 | 14.9 | 108.4 | 7.5 | ||
| Ca(OH)2 | 1 h, 80 °C | 12.7 | 81.7 | 17.9 | 17.6 | 81.9 | 18.1 | 22.5 | 85.5 | 21.8 | 46.1 | 60.7 | −3.1 | 34.5 | 70.1 | 6.4 | |
| 2.5 h, 80 °C | 9.5 | 96.2 | 9.6 | 14.1 | 96.8 | 10.4 | 18.3 | 101.2 | 14.5 | 45.1 | 73.0 | −13.6 | 32.3 | 83.8 | −2.8 | ||
| 24 h, RT | 13.4 | 89.3 | 8.8 | 18.8 | 89.2 | 8.6 | 23.6 | 92.2 | 12.3 | 52.0 | 63.2 | −17.4 | 38.6 | 74.6 | −6.0 | ||
| NaOH | 3% | 1 h, 80 °C | 13.0 | 87.7 | 11.5 | 18.3 | 87.6 | 11.1 | 23.0 | 91.4 | 14.9 | 50.2 | 62.9 | −13.5 | 37.2 | 73.8 | −2.6 |
| 2.5 h, 80 °C | 9.6 | 92.2 | 13.5 | 14.1 | 92.9 | 14.1 | 18.4 | 97.4 | 18.4 | 43.5 | 70.8 | −7.9 | 31.3 | 80.8 | 2.1 | ||
| 6% | 1 h, 80 °C | 9.3 | 86.1 | 20.1 | 13.5 | 87.2 | 21.1 | 17.9 | 91.3 | 25.3 | 39.1 | 68.8 | 2.8 | 28.4 | 77.4 | 11.4 | |
| 2.5 h, 80 °C | 7.8 | 97.7 | 11.7 | 12.0 | 98.6 | 12.7 | 15.8 | 103.1 | 17.4 | 40.9 | 77.0 | −9.0 | 28.8 | 87.0 | 1.09 | ||
| 9% | 1 h, 80 °C | 7.7 | 83.7 | 25.8 | 10.9 | 85.7 | 27.8 | 14.9 | 90.2 | 32.3 | 30.4 | 73.4 | 15.6 | 21.9 | 79.9 | 22.0 | |
| 2.5 h, 80 °C | 13.3 | 85.1 | 13.0 | 18.6 | 85.1 | 13.0 | 23.5 | 88.7 | 16.6 | 49.9 | 61.1 | −11.0 | 37.2 | 71.7 | −0.5 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brleković, F.; Mužina, K.; Kurajica, S. The Influence of Alkaline Pretreatment of Waste Nutshell for Use in Particulate Biocomposites. J. Compos. Sci. 2024, 8, 26. https://doi.org/10.3390/jcs8010026
Brleković F, Mužina K, Kurajica S. The Influence of Alkaline Pretreatment of Waste Nutshell for Use in Particulate Biocomposites. Journal of Composites Science. 2024; 8(1):26. https://doi.org/10.3390/jcs8010026
Chicago/Turabian StyleBrleković, Filip, Katarina Mužina, and Stanislav Kurajica. 2024. "The Influence of Alkaline Pretreatment of Waste Nutshell for Use in Particulate Biocomposites" Journal of Composites Science 8, no. 1: 26. https://doi.org/10.3390/jcs8010026
APA StyleBrleković, F., Mužina, K., & Kurajica, S. (2024). The Influence of Alkaline Pretreatment of Waste Nutshell for Use in Particulate Biocomposites. Journal of Composites Science, 8(1), 26. https://doi.org/10.3390/jcs8010026







