Innovative Composites Based on Organic Modified Zirconium Phosphate and PEOT/PBT Copolymer
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Solvent
2.2. ZrP Synthesis
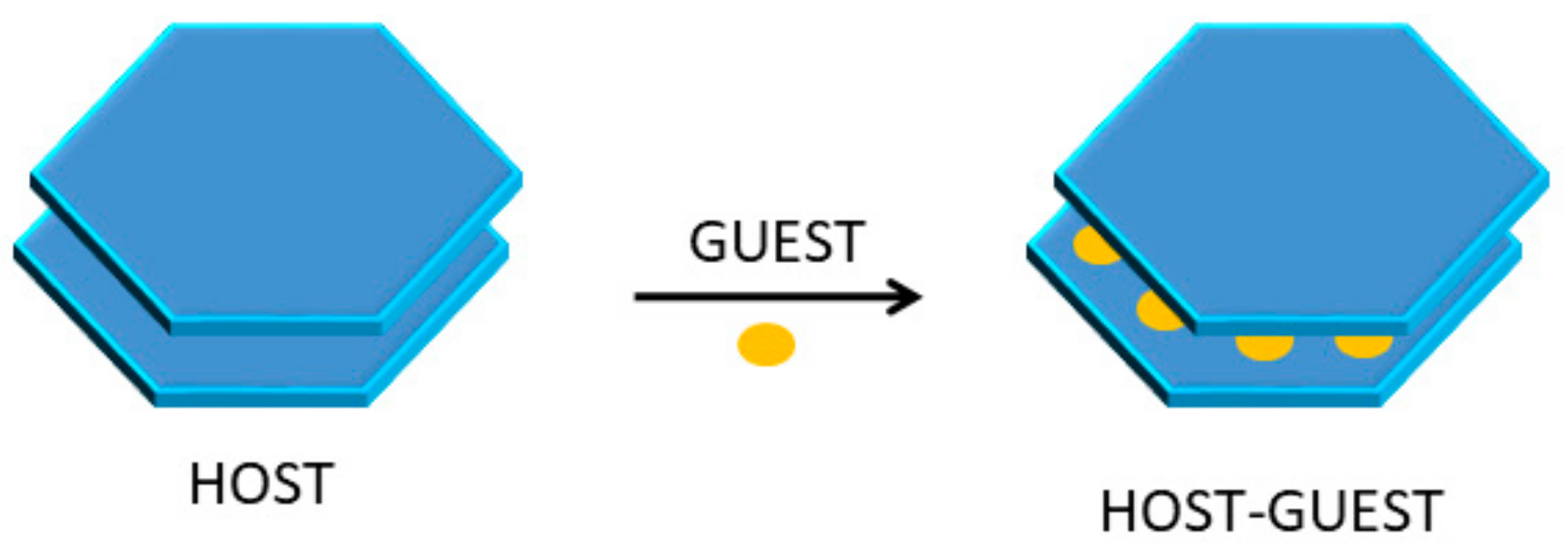
2.3. Intercalation of GTM in α-ZrP
2.4. Composite Production
2.5. Analytical Procedures and Instrumentation
3. Results and Discussion
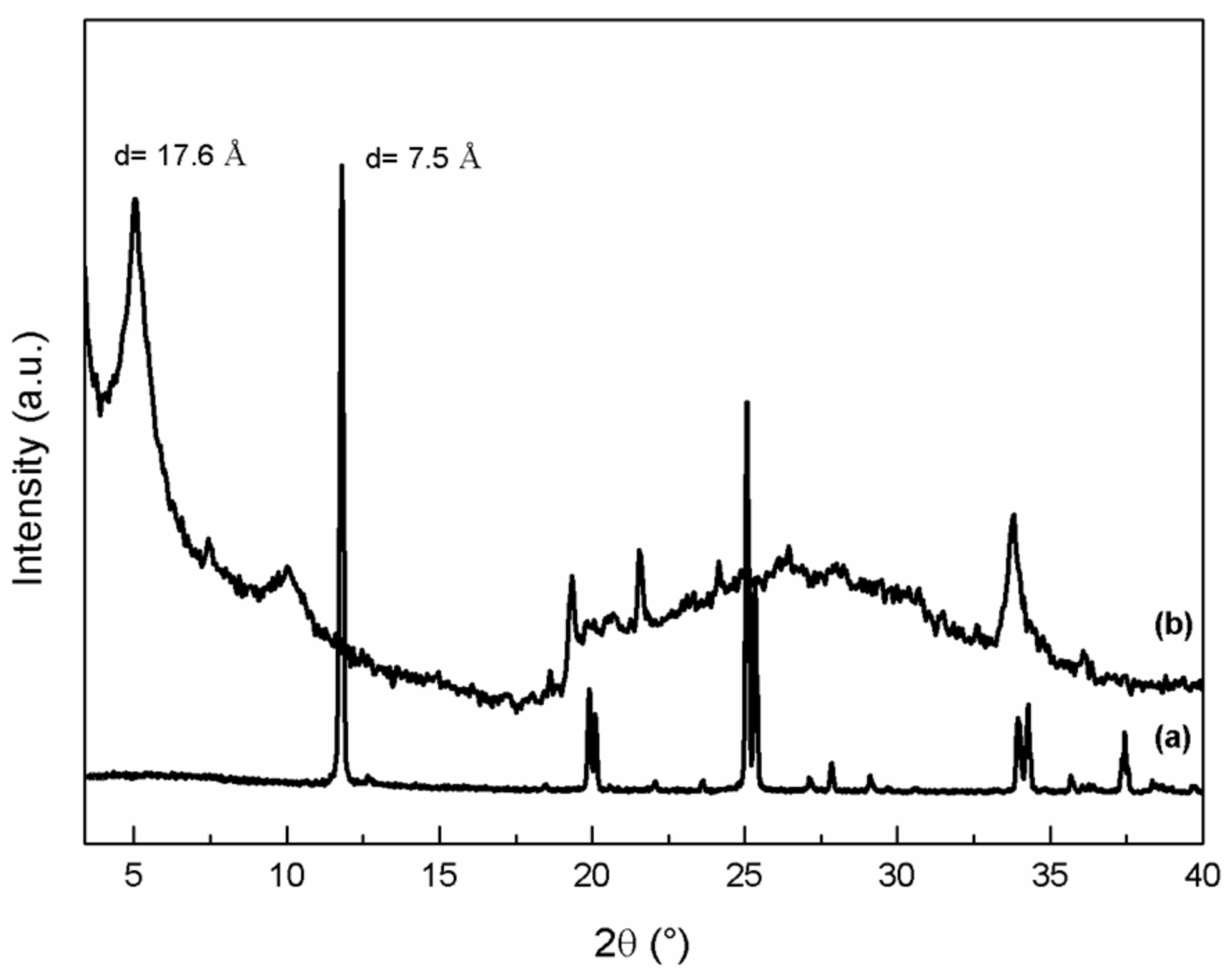
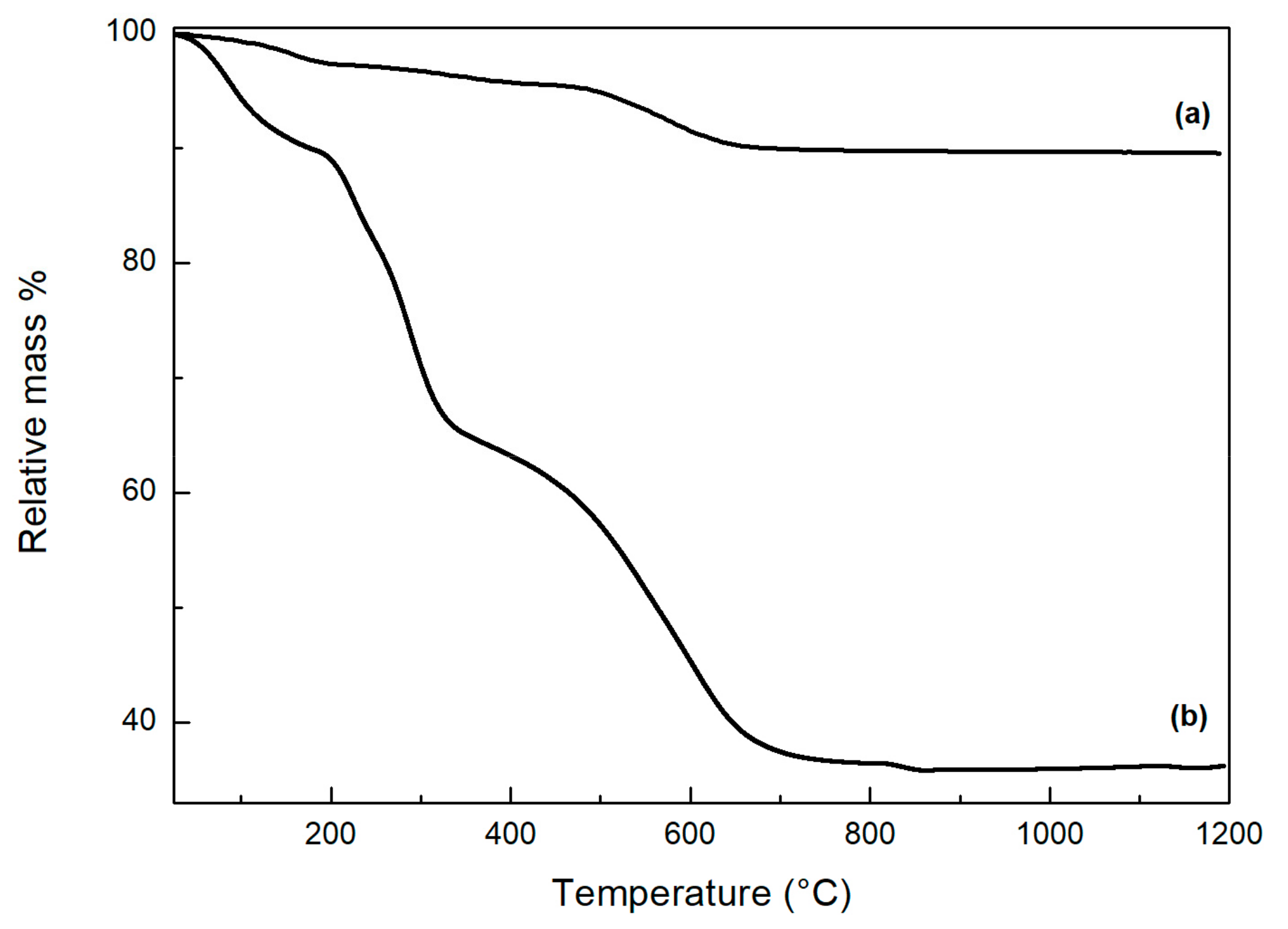
3.1. ZrP-GTM Characterization
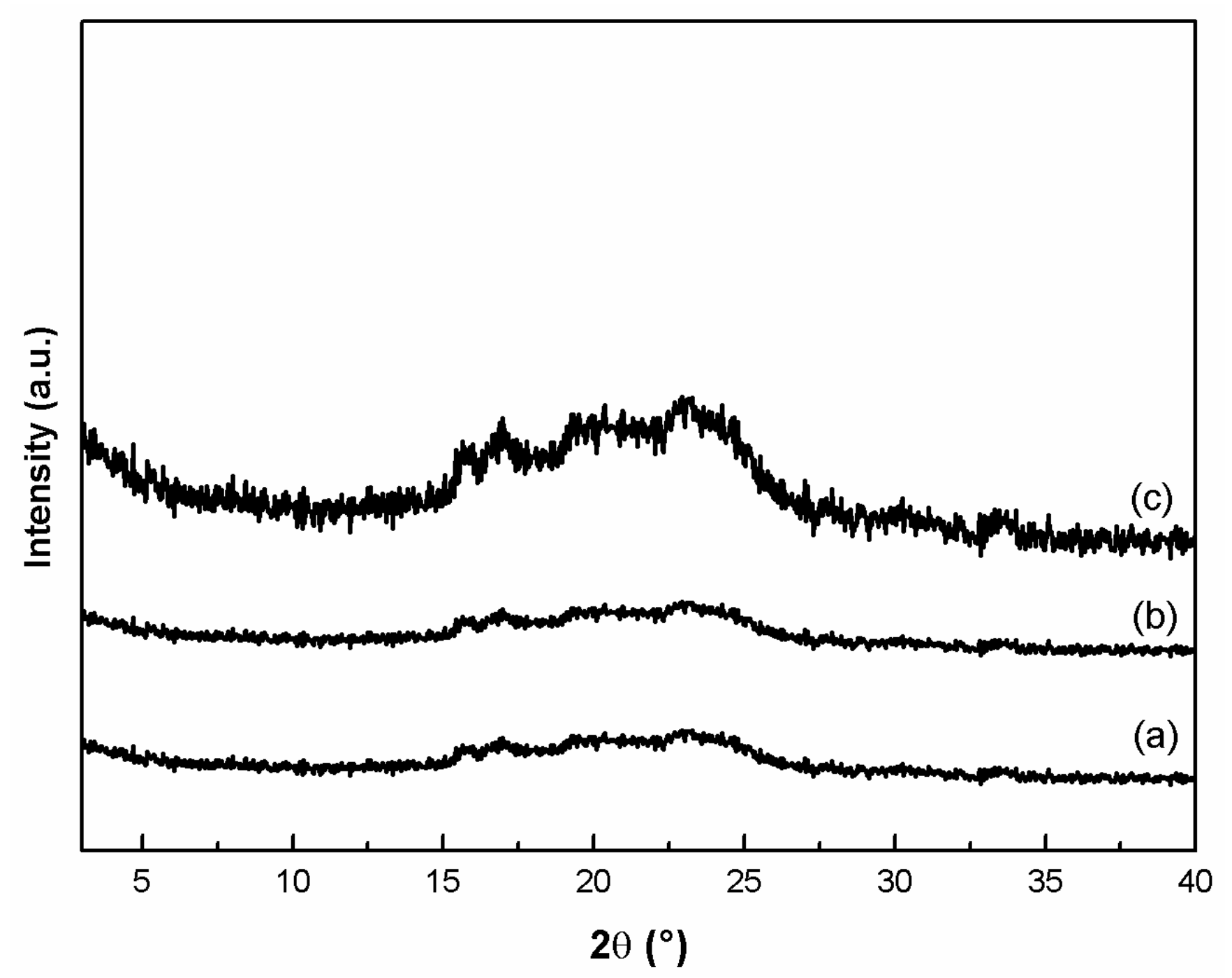
3.2. Composite Characterization
3.2.1. PBT/PEOT and ZrP-GTM Composites Characterization
3.2.2. Antimicrobial Activity
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Kumar, V.; Manju, T.; Thakur, K.; Gupta, R.K. Hybrid Polymer Composite Materials: Structure and Chemistry, 1st ed.; Woodhead Publishing: Cambridge, UK, 2017; pp. 157–171. ISBN 9780081007921. [Google Scholar]
- Kotal, M.; Bhowmick, A.K. Polymer nanocomposites from modified clays: Recent advances and challenges. Progr. Polym. Sci. 2015, 51, 127–187. [Google Scholar] [CrossRef]
- Casanas, B. Tailored Organic-Inorganic Materials, 1st ed.; John Wiley & Sons: Hoboken, NJ, USA, 2015; pp. 1–40. ISBN 978-1-118-77346-8. [Google Scholar]
- Clearfield, A.; Smith, G.D. Crystallography and structure of alpha-zirconium bis (monohydrogen orthophosphate) monohydrate. Inorg. Chem. 1969, 8, 431–436. [Google Scholar] [CrossRef]
- Saxena, V.; Diaz, A.; Hussain, M.D.; Clearfield, A.; Batteas, J.D. Zirconium phosphate nanoplatelets: A biocompatible nanomaterial for drug delivery to cancer. Nanoscale 2013, 5, 2328–2336. [Google Scholar] [CrossRef] [PubMed]
- Díaz, A.; González, M.L.; Pérez, R.J. Direct intercalation of cisplatin into zirconium phosphate nanoplatelets for potential cancer nanotherapy. Nanoscale 2013, 5, 11456–11463. [Google Scholar] [CrossRef] [PubMed]
- González-Villegas, J.; Kan, Y.B.; Vladimir, I.B.; García-Vargas, A.; Martínez, M.; Clearfield, A.; Colón, J.L. Poly(ethylene glycol)-modified zirconium phosphate nanoplatelets for improved doxorubicin delivery. Inorg. Chim. Acta 2017, 468, 270–279. [Google Scholar] [CrossRef]
- Li, R.; Liu, T.; Wang, K. Hyaluronic acid-modified zirconium phosphate nanoparticles for potential lung cancer therapy. Biomed. Tech. 2016, 62, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Safari, M.; Kamari, Y.; Ghiaci, M.; Sadeghi-Aliabadi, H.; Mirian, M. Synthesis and characterization of insulin/zirconium phosphate@TiO2 hybrid composites for enhanced oral insulin delivery applications. Drug Dev. Ind. Pharm. 2017, 43, 862–870. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, A.J.; Nazar, L.F. Intercalation Chemistry, Encyclopedia of Inorganic Chemistry; John Wiley and Sons Ltd.: New York, NY, USA, 2006. [Google Scholar]
- Scatto, M.; Sisani, M. Active polymer nanocomposites: Application in thermoplastic polymers. AIP Conf. Proc. 2016, 1779. [Google Scholar] [CrossRef]
- Costa, F.R.; Saphiannikova, M.; Wagenknecht, U.; Heinrich, G. Wax Crystal Control Nanocomposites Stimuli-Responsive Polymers; Springer: Berlin/Heidelberg, Germany, 2007; pp. 101–168. ISBN 978-3-540-75500-5. [Google Scholar]
- Duan, Y.-Y.; Jia, J.; Wang, S.-H.; Yan, W.; Jin, L.; Wang, Z.-Y. Preparation of antimicrobial poly(ϵ-caprolactone) electrospun nanofibers containing silver-loaded zirconium phosphate nanoparticles. J. Appl. Polym. Sci. 2007, 106, 1208–1214. [Google Scholar] [CrossRef]
- Costantino, U.; Bugatti, V.; Gorrasi, G.; Montanari, F.; Nocchetti, M.; Tammaro, L.; Vittoria, V. New Polymeric Composites Based on Poly(ϵ-caprolactone) and Layered Double Hydroxides Containing Antimicrobial Species. ACS Appl. Mater. Interfaces 2009, 1, 668–677. [Google Scholar] [CrossRef] [PubMed]
- Ambrogi, V.; Pietrella, D.; Nocchetti, M.; Casagrande, S.; Moretti, V.; De Marco, S.; Ricci, M. Montmorillonite–chitosan–chlorhexidine composite films with antibiofilm activity and improved cytotoxicity for wound dressing. J. Colloid Interface Sci. 2017, 491, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Merchán, M.; Ouk, T.S.; Kubát, P.; Lang, K.; Coelho, C.; Verney, V.; Commereuc, S.; Leroux, F.; Sol, V.; Taviot-Guého, C. Photostability and photobactericidal properties of porphyrin-layered double hydroxide–polyurethane composite films. Mater. Chem. B 2013, 1, 2139–2146. [Google Scholar] [CrossRef]
- Donnadio, A.; Ambrogi, V.; Pietrella, D.; Pica, M.; Sorrentino, G.; Casciola, M. Carboxymethylcellulose films containing chlorhexidine–zirconium phosphate nanoparticles: Antibiofilm activity and cytotoxicity. RSC Adv. 2016, 6, 46249–46257. [Google Scholar] [CrossRef]
- Kalita, H.; Pal, P.; Dhara, S.; Pathak, A. Fabrication and characterization of polyvinyl alcohol/metal (Ca, Mg, Ti) doped zirconium phosphate nanocomposite films for scaffold-guided tissue engineering application. Mat. Sci. Eng. C 2017, 71, 363–371. [Google Scholar] [CrossRef] [PubMed]
- Schmid, M.; Muller, K. Review on the Processing and Properties of Polymer Nanocomposites and Nanocoatings and Their Applications in the Packaging. Autom. Sol. Energy Fields 2017, 7, 74. [Google Scholar]
- Chen, L.; Sun, D.; Li, J.; Zhu, G. Exfoliation of layered zirconium phosphate nanoplatelets by melt compounding. Mater. Des. 2017, 122, 247–254. [Google Scholar] [CrossRef]
- Ai, J.; Chen, Y.; Jing, H.; Gao, X.; Liu, J.; Ma, K.; Suo, J.; Yu, S.; Song, S. Dynamic Release of Antibiotic Drug Gentamicin Sulfate from Novel Zirconium Phosphate Nano-Platelets. Adv. Mater. 2014, 6, 2603–2610. [Google Scholar] [CrossRef]
- Barron, V.; Merghani, K.; Shaw, G.; Coleman, C.M.; Hayes, J.S.; Ansboro, S.; Manian, A.; O’Malley, G.; Connolly, E.; Nandakumar, A.; et al. Evaluation of Cartilage Repair by Mesenchymal Stem Cells Seeded on a PEOT/PBT Scaffold in an Osteochondral Defect. Ann. Biomed. Eng. 2015, 43, 2069–2082. [Google Scholar] [CrossRef] [PubMed]
- Kelly, C.N.; Miller, A.T.; Hollister, S.J.; Guldberg, R.E.; Gall, K. Design and Structure–Function Characterization of 3D Printed Synthetic Porous Biomaterials for Tissue Engineering. Adv. Healthc. Mater. 2017. [Google Scholar] [CrossRef] [PubMed]
- Polyvation. Available online: http://www.polyvation.com/enabling-technologies/peotpbt-polymers/ (accessed on 31 March 2018).
- Du, Y.; Deng, F.; Jiang, X.; Ji, H.; Yu, D.; Wang, W.; Sun, B.; Zhu, M. Preparation and performance of lipophilic α-zirconium phosphate with high thermal stability and its application in thermal-plastic polymers. Prog. Nat. Sci. Mater. Int. 2015, 25, 503–511. [Google Scholar] [CrossRef]
- Lino, A.S.; Mendes, L.C.; de França da Silva, D.; Malm, O. High density polyethylene and zirconium phosphate nanocomposites. Polímeros 2015, 25, 477–482. [Google Scholar] [CrossRef]
- De França da Silva, D.; Mendes, L.C.; Lino, A.S. Polyamide-6/Organo intercalated Lamellar Zirconium Phosphate Nanocomposites: Molecular Mobility, Crystallography and Thermo-Mechanical Evaluation. J. Nanosci. Nanotechnol. 2017, 17, 3042–3050. [Google Scholar]
- Saengmee-Anupharb, S.; Thaweboon, S.; Amornsakchai, T.; Thaweboon, B.; Dechkunakorn, S.; Suddhasthira, T.; Srikhirin, T. The Preparation and Antimicrobial Effect of AgZrP/Nylon 6,10 Fibers Used as Dental Hygiene Materials. J. Appl. Polym. Sci. 2012, 124, 4016–4024. [Google Scholar] [CrossRef]
- Xiang, H.; Li, L.; Chen, W.; Yu, S.; Sun, B.; Zhu, M. Flame retardancy of polyamide 6 hybrid fibers: Combined effects of α-zirconium phosphate and ammonium sulfamate. Prog. Nat. Sci. Mater. Int. 2017, 27, 369–373. [Google Scholar] [CrossRef]
- Clearfield, A.; Stynes, J.A. The preparation of crystalline zirconium phosphate and some observations on its ion exchange behaviour. J. Inorg. Nucl. Chem. 1964, 26, 117–129. [Google Scholar] [CrossRef]
- Casciola, M.; Alberti, G.; Donnadio, A.; Pica, M.; Marmottini, F.; Bottino, A.; Piaggio, P. Gels of zirconium phosphate in organic solvents and their use for the preparation of polymeric nanocomposites. J. Mater. Chem. 2005, 15, 4262–4267. [Google Scholar] [CrossRef]
- Chuong, M.C.; Chin, J.; Han, J.W.; Kim, E.; Alhomayin, W.; Al Dosary, F.; Rizg, W.; Moukhachen, O.; Williams, D.A. High performance liquid chromatography of gentamicin sulfate reference standards and injection USP. Int. J. Pharm. Anal. 2013, 4, 25–29. [Google Scholar]
- Albertsson, J.; Oskarsson, A.; Tellgren, R.; Thomas, J.O. Inorganic ion exchangers. 10. A neutron powder diffraction study of the hydrogen bond geometry in alpha-zirconium bis (monohydrogen orthophosphate) monohydrate. A model for the ion exchange. J. Phys. Chem. 1977, 16, 1574–1578. [Google Scholar] [CrossRef]
- Sabu, T.; Kuruvilla, J.; Malhotra, S.K.; Goda, K.; Sreekala, M.S. Polymer Composites, Volume 3, Biocomposites; Wiley Inc.: New York, NY, USA, 2013; p. 2109. ISBN 978-3-527-32980-9. [Google Scholar]
- Song, Z.; Borgwardt, L.; Høiby, N.; Wu, H.; Sørensen, T.S.; Borgwardt, A. Prosthesis Infections after Orthopedic Joint Replacement: The Possible Role of Bacterial Biofilms. Orthoped. Rev. 2013, 5, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Yadav, S.; Vasanthan, N.; Sekosan, G. A study of antimicrobial property of textile fabric treated with modified dendrimers. J. Appl. Polym. Sci. 2010, 115, 716–722. [Google Scholar] [CrossRef]
- Available online: https://www.drugs.com/monograph/gentamicin-sulfate.html (accessed on 31 March 2018).



© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bastianini, M.; Scatto, M.; Sisani, M.; Scopece, P.; Patelli, A.; Petracci, A. Innovative Composites Based on Organic Modified Zirconium Phosphate and PEOT/PBT Copolymer. J. Compos. Sci. 2018, 2, 31. https://doi.org/10.3390/jcs2020031
Bastianini M, Scatto M, Sisani M, Scopece P, Patelli A, Petracci A. Innovative Composites Based on Organic Modified Zirconium Phosphate and PEOT/PBT Copolymer. Journal of Composites Science. 2018; 2(2):31. https://doi.org/10.3390/jcs2020031
Chicago/Turabian StyleBastianini, Maria, Marco Scatto, Michele Sisani, Paolo Scopece, Alessandro Patelli, and Annarita Petracci. 2018. "Innovative Composites Based on Organic Modified Zirconium Phosphate and PEOT/PBT Copolymer" Journal of Composites Science 2, no. 2: 31. https://doi.org/10.3390/jcs2020031
APA StyleBastianini, M., Scatto, M., Sisani, M., Scopece, P., Patelli, A., & Petracci, A. (2018). Innovative Composites Based on Organic Modified Zirconium Phosphate and PEOT/PBT Copolymer. Journal of Composites Science, 2(2), 31. https://doi.org/10.3390/jcs2020031





