1. Introduction
The cleaning and sterilization of medical devices are imperative processes to guarantee patient safety, ensure the functionality of devices, and comply with regulatory mandates [
1,
2]. Proper cleaning removes organic and inorganic contaminants such as blood, tissue, and bodily fluids, while sterilization eliminates all forms of microbial life, including bacteria, viruses, and spores [
3,
4]. These procedures are crucial in preventing healthcare-associated infections (HAIs), which can lead to serious health complications or even death [
5]. Furthermore, it is imperative to implement regular and meticulous cleaning procedures to ensure the preservation of mechanical integrity and optimal performance of medical devices. This contributes to extending their lifespan and mitigating the risk of malfunction during utilization. In order to circumvent the potential for legal ramifications and to ensure the continuity of accreditation, it is imperative to adhere to the regulatory standards established by entities such as the FDA or ISO [
6]. In addition, using clean and sterile instruments reflects a healthcare facility’s commitment to quality care and helps preserve its reputation and patient trust, especially when dealing with reusable instruments that pose a high risk of cross-contamination if not properly handled [
7,
8].
Silicone plays a vital role in the medical device industry due to its unique combination of biocompatibility, flexibility, durability, and chemical stability. Medical-grade silicone is a material of choice in a wide variety of medical applications due to its non-reactivity with body tissues and fluids. This characteristic minimizes the risk of allergic reactions or adverse immune responses, making it a preferred material in medical devices such as catheters, implants, tubing, wound dressings, and seals [
9,
10]. Its flexibility allows for comfort and adaptability in both short- and long-term applications inside the body, while its resistance to extreme temperatures and chemical degradation ensures consistent performance in demanding clinical environments [
11,
12].
Given the direct contact of silicone-based medical devices with internal organs, bloodstreams, or open wounds, the importance of proper cleaning and sterilization is paramount [
13,
14]. The presence of contaminants on these devices has the potential to introduce harmful pathogens, which can result in infections or complications. Despite its ability to withstand various sterilization methods, including autoclaving, ethylene oxide (ETO), and gamma radiation, it is imperative that silicone undergo thorough cleaning to remove biological residues prior to sterilization [
7,
15]. Effective cleaning ensures that the sterilization process is successful, maintains the integrity and functionality of the silicone device, and meets regulatory standards. Altogether, the combination of silicone’s properties and rigorous cleaning and sterilization processes ensures that these devices are safe, reliable, and suitable for critical medical applications [
16,
17].
Medical-grade silicone is renowned for its exceptional resistance to heat, chemicals, and aging. However, it is imperative to note that improper or repeated cleaning and sterilization processes have the potential to compromise the integrity of silicone-based medical devices [
18]. High-temperature sterilization methods, such as autoclaving, may lead to gradual loss of elasticity, surface cracking, or material fatigue, especially after many cycles. Harsh chemical disinfectants or aggressive cleaning agents can degrade the silicone surface, causing discoloration, swelling, or erosion [
19]. Furthermore, radiation-based sterilization methods, such as gamma or electron beam, have been observed to induce alterations in the polymer structure, thereby affecting its mechanical properties or clarity. Despite the aforementioned risks, silicone continues to be a preferred material in medical applications due to its inherent stability and durability when exposed to a broad spectrum of sterilization methods, encompassing steam, ethylene oxide (ETO), and low-temperature gas plasma [
20].
A study conducted by Hauser et al. [
19] evaluated the effectiveness of low-pressure gas plasma sterilization on silicone implant materials. The research demonstrated the efficacy of this method in effectively inactivating microorganisms without causing structural damage to the silicone. This finding suggests that the method is a viable sterilization technique for heat-sensitive materials. Santhaveesuk et al. [
21] investigated the impact of microwave disinfection on facial silicone prostheses over a simulated 1.5-year period. The findings indicated that microwave disinfection did not adversely affect the mechanical properties of the silicone materials tested, supporting its use as a safe disinfection method. Galante et al. [
13] explored the use of ozone gas as a sterilization method for silicone-based hydrogels. The results demonstrated that ozone sterilization effectively preserved the hydrogels’ properties, including transparency and mechanical strength. This suggests that ozone sterilization has the potential to serve as a promising alternative to conventional sterilization techniques. Wezgowiec et al. [
22] assessed the effects of various disinfection methods, including UVC radiation and ozone gas, on the material properties of silicone dental impressions. The study determined that these disinfection methods did not cause any substantial changes to the physical properties of silicone materials, suggesting their suitability for routine use. Gautriaud et al. [
14] examined how different sterilization methods affect silicone materials. The study revealed that gamma and electron-beam irradiation significantly altered the mechanical properties, particularly in peroxide-cured silicone, by increasing hardness and modulus while reducing tensile strength and elongation. Conversely, ethylene oxide (EtO) sterilization exhibited minimal impact on mechanical performance, with occasional improvements observed, particularly in liquid silicone rubber (LSR). The study recommends EtO as a safer sterilization method for preserving silicone integrity in medical applications.
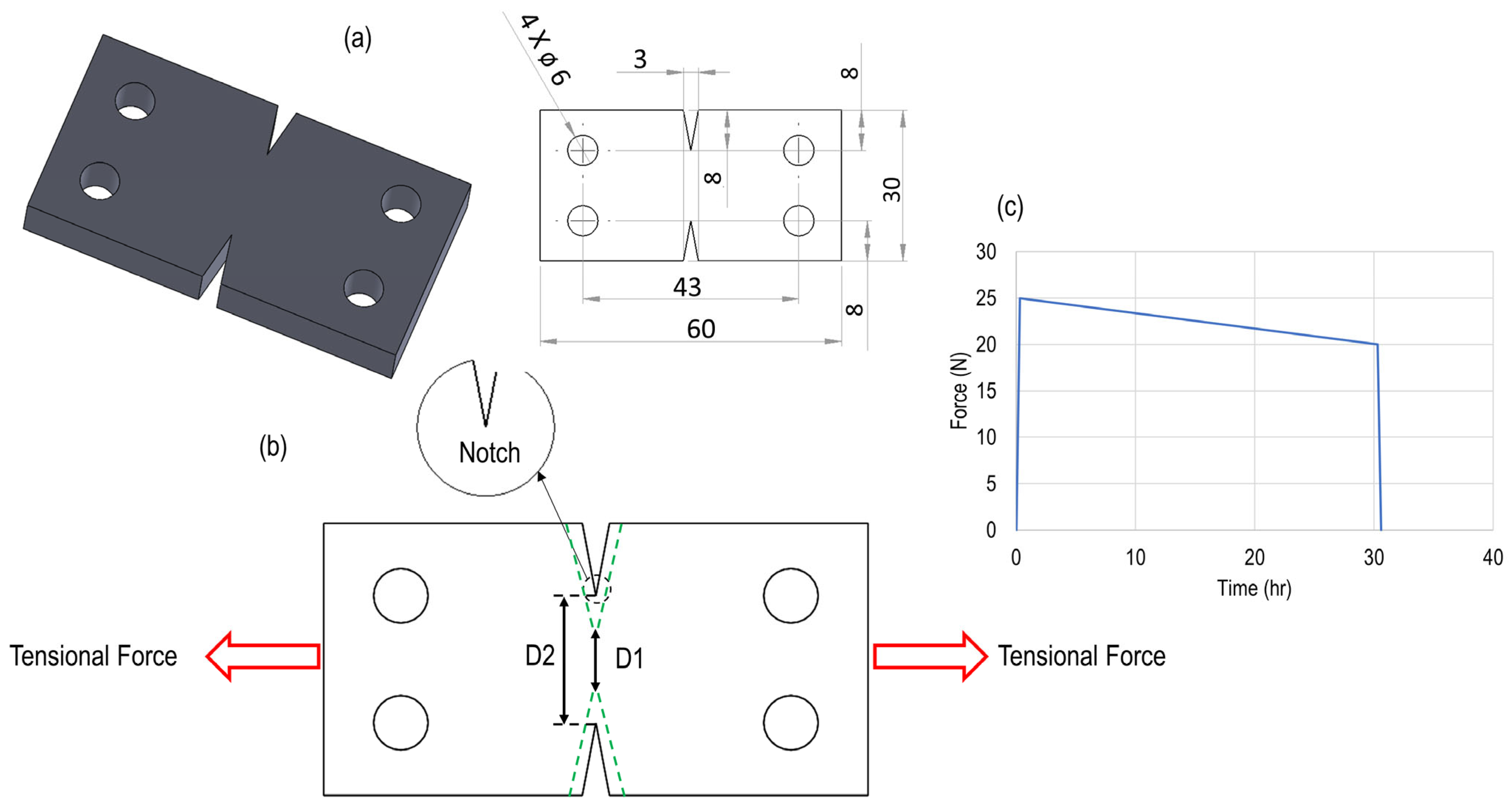
The primary objective of this research is to evaluate the effects of high-cycle (1000 cycles) cleaning, disinfection, and sterilization processes on the mechanical and microstructural properties of silicone materials used in medical instruments. The scientific contribution of this work lies in its comprehensive and quantitative investigation of how repeated cleaning, disinfection, and sterilization cycles affect the mechanical and surface properties of medical-grade silicone, including both silicone and silicone–steel samples. While sterilization is essential for infection control, its long-term impact on the performance and durability of silicone-based components in reusable medical devices remains underexplored. Three different types of samples were prepared for this purpose: silicone samples, silicone–steel samples, and notched silicone samples. Each sample type was designated for specific analyses, including hardness testing, surface characterization, silicone–steel interface evaluation, and mechanical property assessment. To investigate the impact of repeated cleaning and sterilization, the samples were divided into three groups, each subjected to 200, 500, and 1000 cycles, respectively, to assess changes in their properties over time. A processing cycle consists of mechanical cleaning and disinfection, followed by sterilization using the steam sterilization process.
3. Results and Discussion
3.1. Surface Contact Angle
In the domain of medical instrumentation, the contact angle assumes significant importance in the context of regulating the interaction between fluids and surfaces. A low contact angle is indicative of a hydrophilic surface, which promotes fluid spread and improves biocompatibility, making it ideal for use in implants and diagnostic tools. The presence of a high contact angle is indicative of hydrophobicity, a property that is advantageous in reducing fluid retention in devices such as catheters. By modifying the contact angle through the application of coatings or surface treatments, the performance, cleanliness, and safety of medical instruments can be considerably improved.
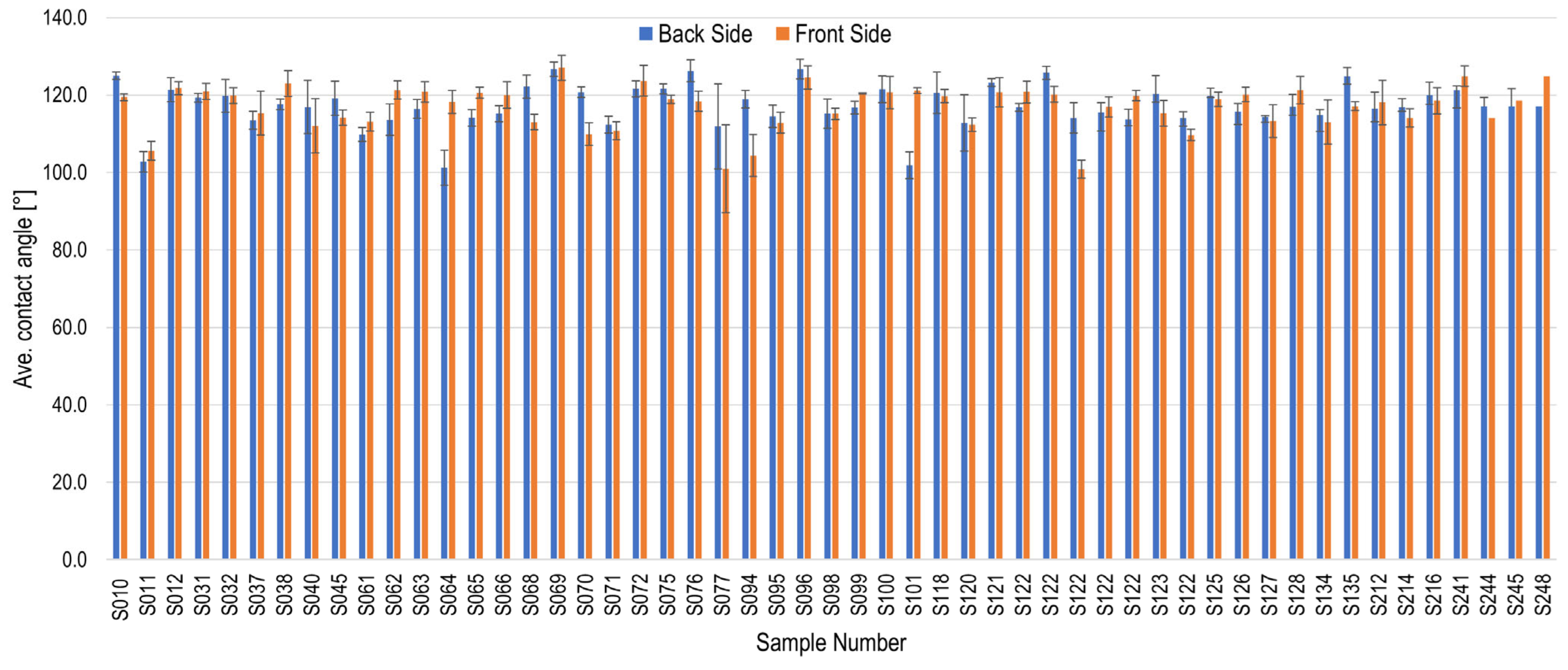
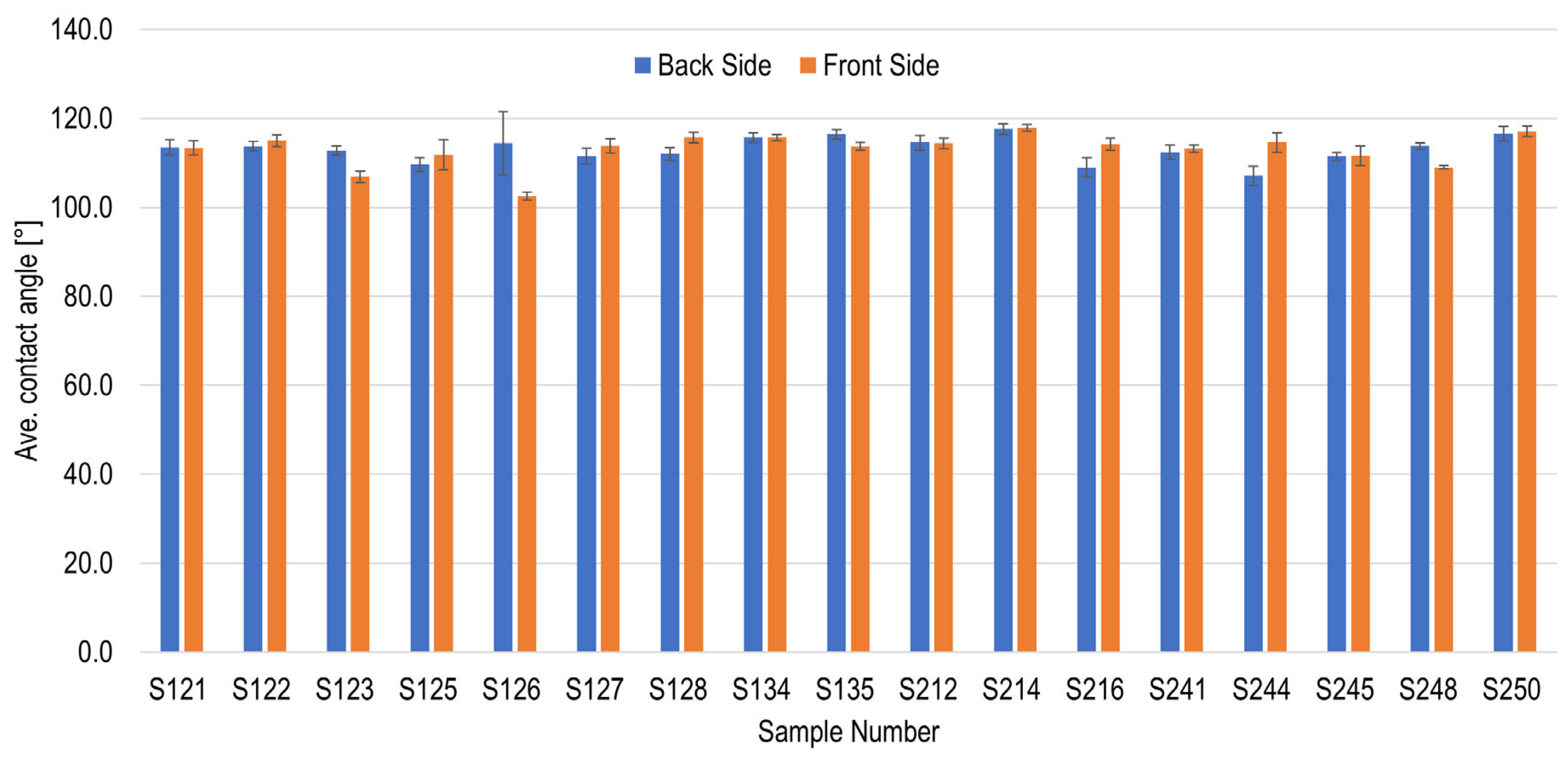
Figure 5 indicates the initial contact angle results for the silicone samples prior to starting the cleaning, disinfection, and sterilization process. The measured contact angles ranged from 110 to 120° for the majority of the samples. The surface exhibited a high degree of hydrophobicity, which was anticipated given the use of silicone rubber. The standard deviation was predominantly less than 5°, indicating that the measured values exhibited minimal variability and can thus be regarded as reliable. However, the contact angle of some samples was found to be significantly smaller, with an average measurement ranging from 100 to 110°. Furthermore, an elevated standard deviation was observed in the individual samples, indicating significant variations in their measured values. This phenomenon could be attributed to the presence of a partially heterogeneous sample surface, which was discernible to the naked eye. In such instances, it becomes imperative to meticulously examine the manufacturing process or to consult with the manufacturer of the silicone rubber, as deemed necessary, in order to enhance the manufacturing process.
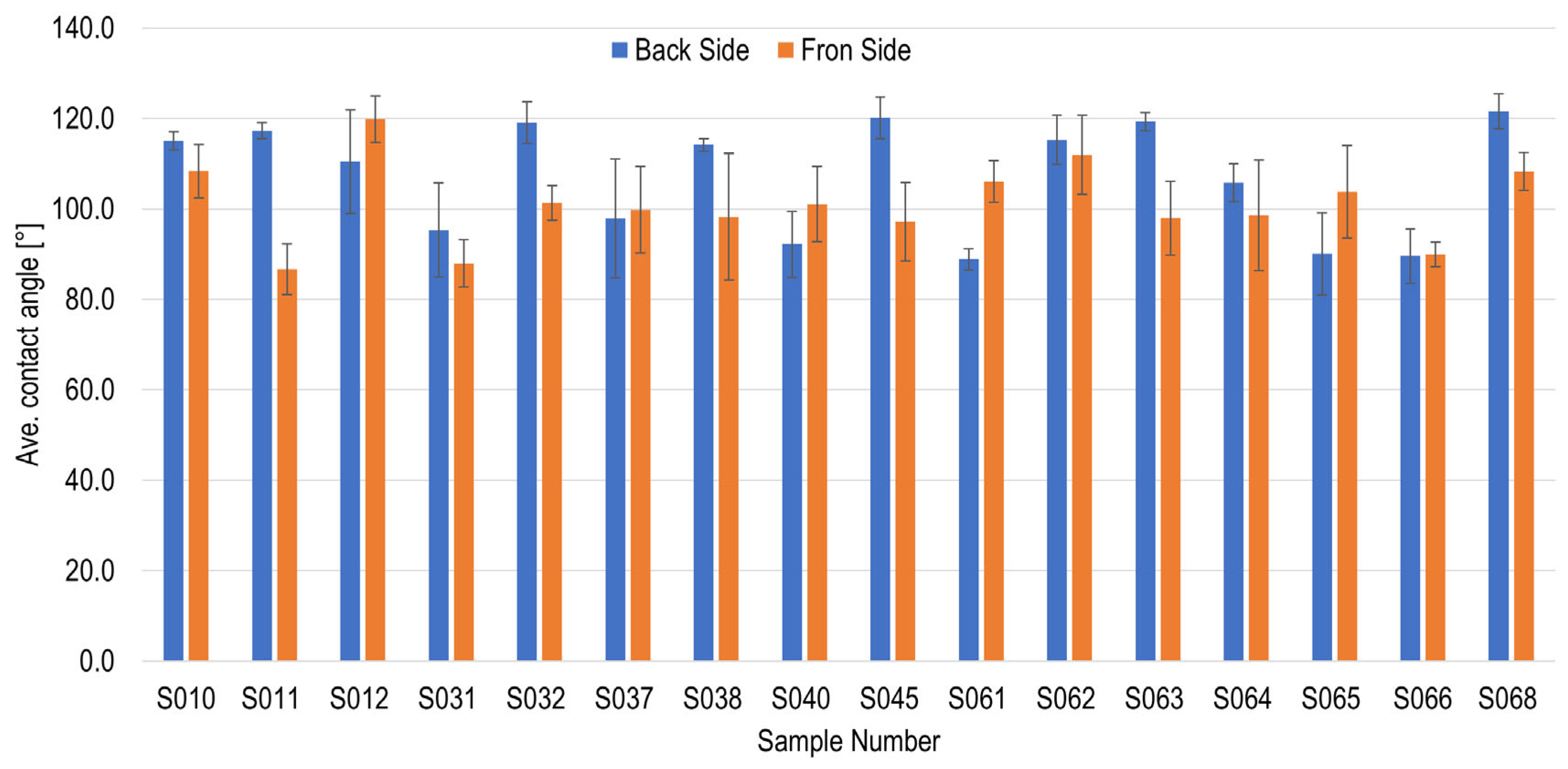
Figure 6 shows the contact angle measurements of the silicone samples after 200 cycles of cleaning, disinfection, and sterilization. The average contact angle decreased to (104.0 ± 10.9)°, indicating a reduction in surface hydrophobicity by up to 12% compared to the initial value of (117.1 ± 5.7)°. This decrease, along with the increased variation, is primarily attributed to surface roughening caused by repeated cycling. The resulting partial erosion altered the originally smooth surface, leading to a more uneven and worn texture. These changes in surface topography affect how the liquid droplet interacts with the material, thereby influencing the contact angle.
Figure 7 presents the surface characterization of the samples before and after 200 cycles, revealing significant surface wear in certain areas. This observation further supports the previously discussed explanation for the decrease in the contact angle.
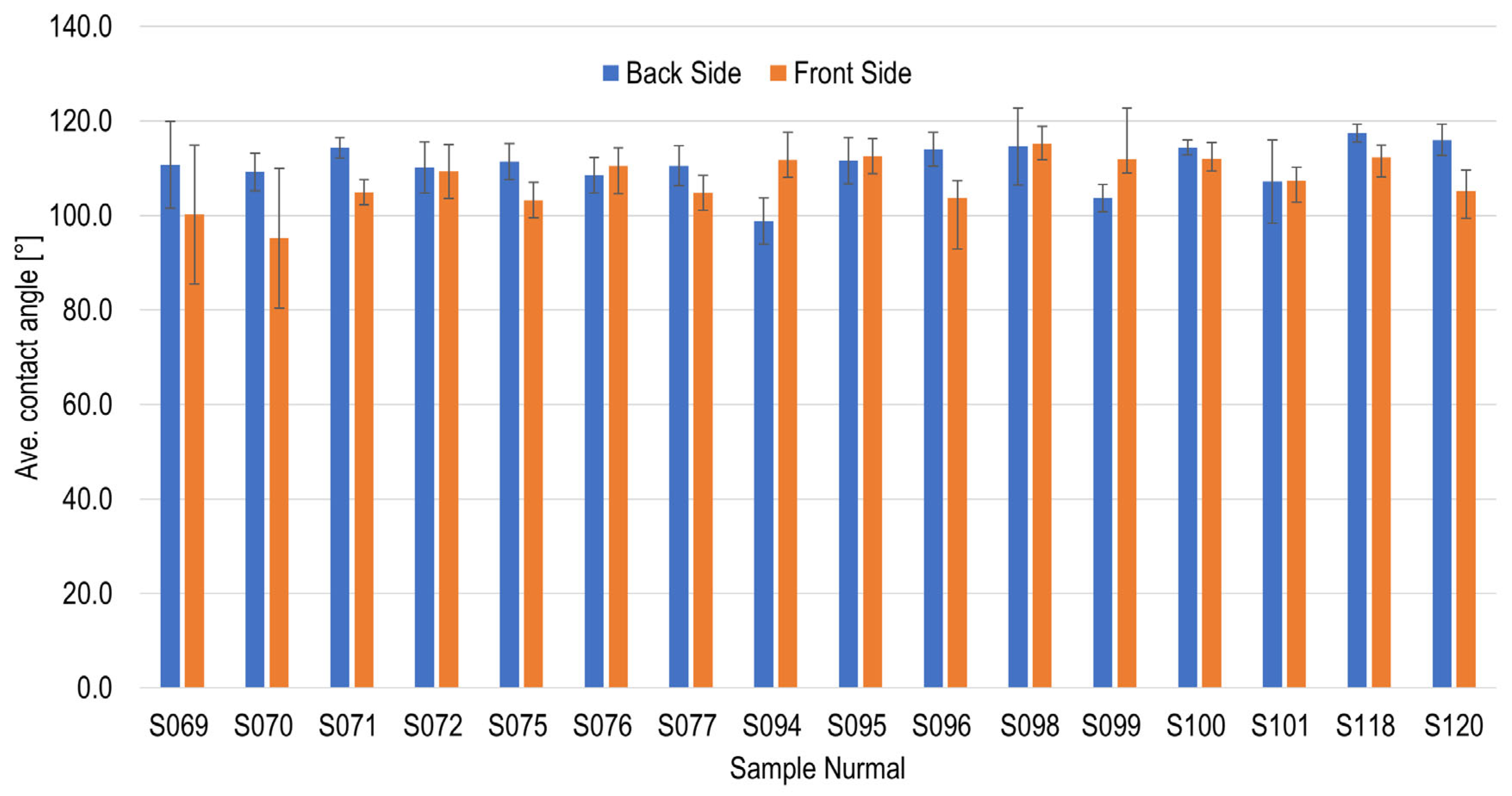
The contact angle results for the silicone samples after 500 cleaning, disinfection, and sterilization process is shown in
Figure 8. The mean contact angle was (109.1 ± 5.2)°, which corresponds to a slight increase (up to 5%) in the mean contact angle and a significant decrease in the standard deviation compared to the previous series of measurements (samples with 200 cycles). This slight increase in contact angle may be attributed to continued surface erosion, which expanded the affected area and exposed a smoother underlying layer by removing the previously weakened surface.
Figure 9 shows the surface of the examined sample after 500 cycles of cleaning, disinfection, and sterilization, revealing a noticeably larger area affected by erosion.
Figure 10 indicates the contact angle results after 1000 cycles of cleaning, disinfection, and sterilization. The mean contact angle was (112.8 ± 3.2)°, which represents a slight increase (up to 4%) in the mean contact angle and a significant decrease in the standard deviation compared to the previous series of measurements. The samples remained clearly hydrophobic.
Figure 11 shows the overall condition of the examined surface before and after 1000 cycles, indicating that erosion had progressed over a larger area, eventually leading to a more uniform surface appearance across the worn regions.
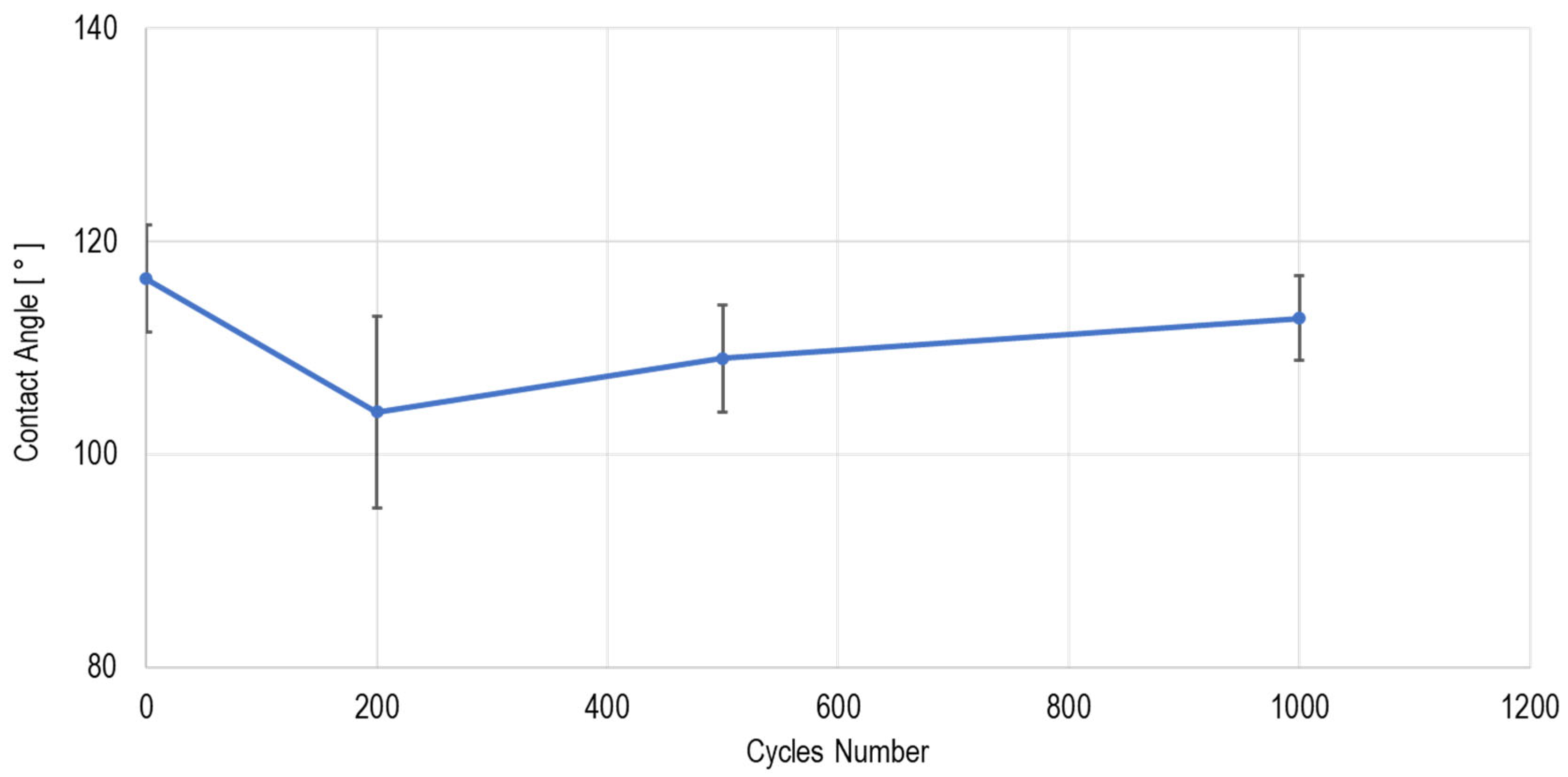
Figure 12 presents a graph comparing the average contact angle values across all processing steps, including standard deviation. It shows that as the number of cleaning, disinfection, and sterilization cycles increases, the average contact angle gradually returns closer to the initial value.
The changes in contact angle reflect surface energy alterations driven by both physical and chemical surface changes during cycling. The initial 12% decrease after 200 cycles is likely due to increased surface roughness caused by micro-erosion, which enhances liquid spreading. However, as the cycles progress, weakened or oxidized surface layers may be gradually removed, leading to smoother surface reformation and partial recovery of the original contact angle. This behavior aligns with the Wenzel and Cassie–Baxter models, where wetting properties are sensitive to both surface texture and chemical composition. These findings suggest that silicone surfaces experience dynamic topographical and chemical evolution under sterilization, which may impact wettability and, by extension, device performance in contact with biological fluids.
3.2. Samples Hardness
Hardness measurements of the processed samples provide valuable insight into their mechanical properties, as the polymeric bonds in the material can be affected even by low-temperature cleaning processes.
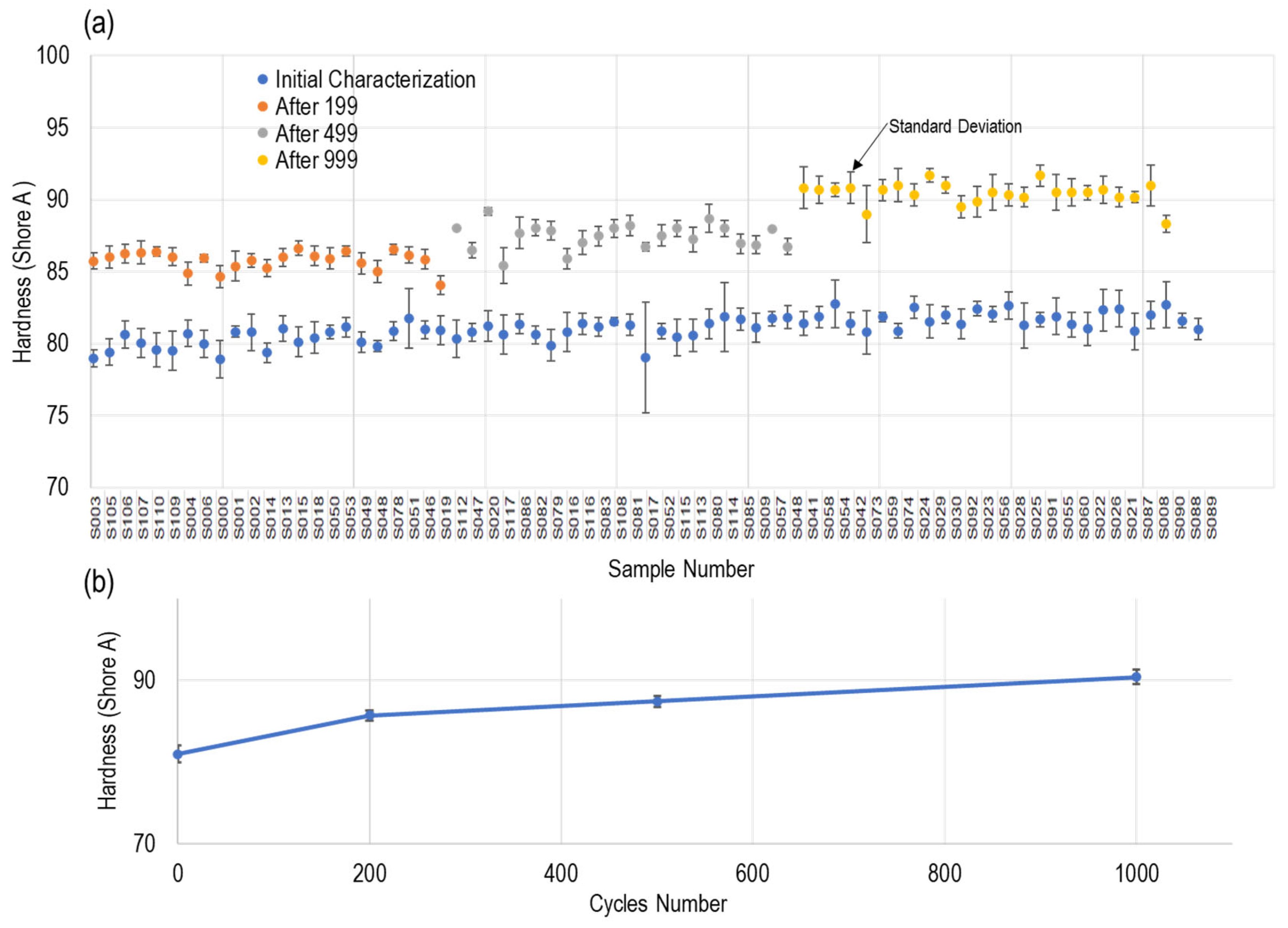
Figure 13 presents the hardness values of the samples subjected to 200, 500, and 1000 cycles.
Figure 13a shows the hardness values for each sample before and after cleaning, disinfection, and sterilization, clearly demonstrating the significant impact of these processes. In each case, hardness increased compared to the initial state.
Figure 13b displays the average hardness values along with standard deviation, illustrating a gradual increase with a rate of almost 5% in hardness with each step of processing cycles.
The primary reason for this phenomenon is the gradual curing of the polymeric bonds, promoted by the elevated temperatures involved in the repeated cleaning, disinfection, and sterilization processes. Although these processes are conducted at relatively low temperatures compared to industrial thermal treatments, repeated exposure over hundreds of cycles can lead to continued crosslinking or rearrangement within the polymer matrix. This gradual post-curing effect enhances the rigidity and structural integrity of the material, resulting in increased hardness values.
In polymeric materials, incomplete curing during initial fabrication is not uncommon, particularly in systems that rely on time- or temperature-dependent polymerization. As a result, subsequent thermal exposure, even at moderate temperatures, can activate residual reactive groups, leading to further network formation. This hardening trend, observed consistently after 200, 500, and 1000 cycles, suggests that the material becomes more structurally stable and resistant to deformation over time. However, while the increase in hardness may enhance certain mechanical properties, it could also introduce brittleness or reduce flexibility factors that must be carefully considered when evaluating the long-term performance and biocompatibility of medical devices.
The observed increase in hardness across all sterilization stages is indicative of progressive polymer crosslinking within the silicone matrix. Thermoset silicones, though initially cured during manufacturing, can continue to undergo post-curing under repeated thermal exposure, especially at elevated temperatures such as the 134 °C used in sterilization. This additional crosslinking results in a denser, more rigid polymer network, which explains the consistent ~5% hardness increase observed after each cycle group. The gradual stiffening of the material may also be linked to the loss of low-molecular-weight species or plasticizers, further reducing molecular mobility. This mechanism provides insight into the long-term mechanical behavior of silicone under repetitive thermal stress, which is particularly relevant to reusable medical devices.
3.3. Notch Growth Test
To evaluate the impact of the cleaning, disinfection, and sterilization processes on the mechanical properties of the silicone samples, a notch growth test under loading was performed. In this method, identical loading and load-holding conditions were applied to the samples both before and after the cycling process to assess the extent of notch propagation. It was hypothesized that repeated processing would degrade the mechanical integrity of the material, thereby facilitating further notch growth under the applied loading. This approach allows for a comparative assessment of the material’s resistance to crack propagation and mechanical deterioration as a result of the sterilization cycles.
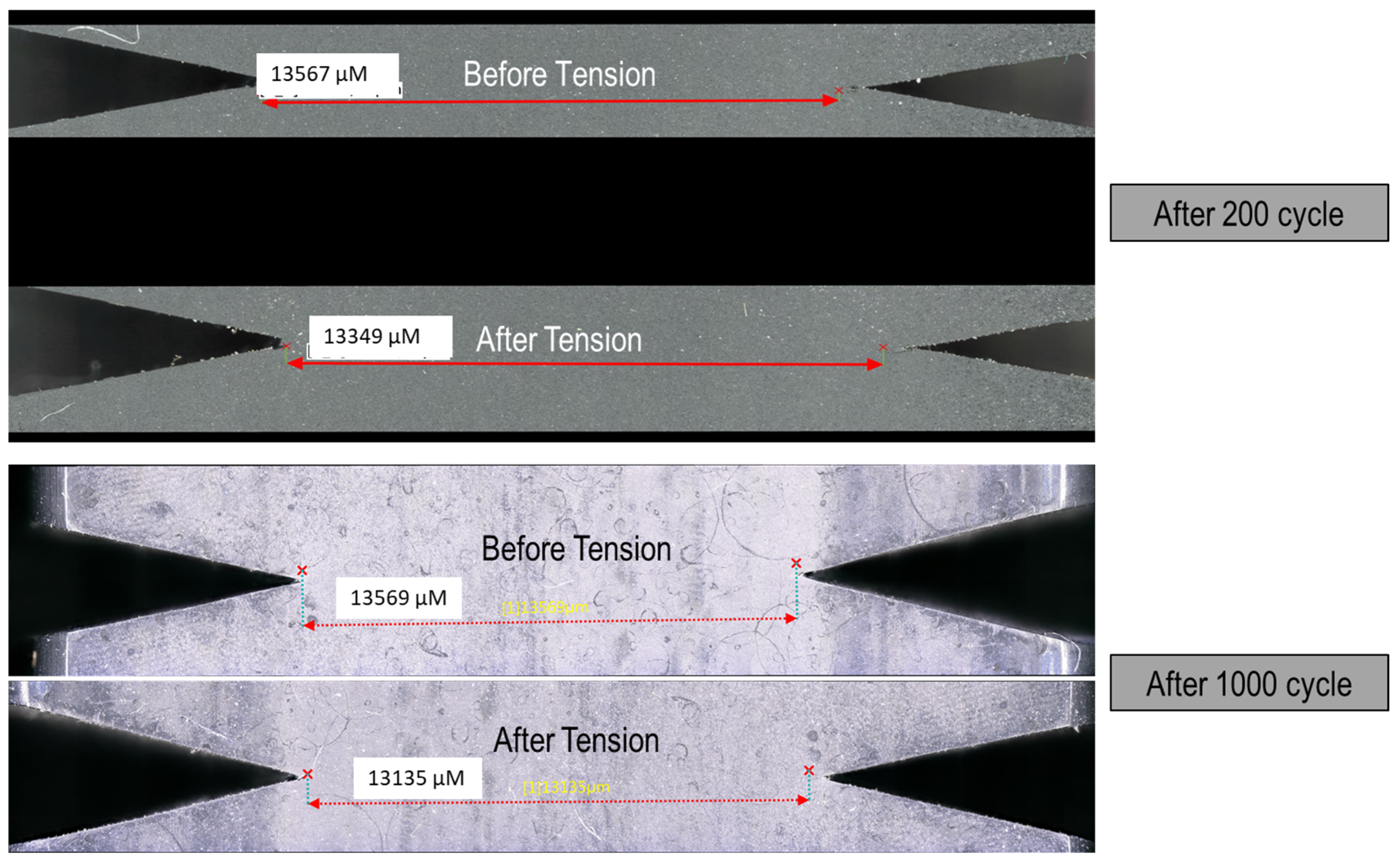
Figure 14 presents the measured notch distances after 200 and 1000 cycles, highlighting a significant difference in notch growth between the two sets of experiments. This indicates that prolonged processing notably influences the material’s susceptibility to crack propagation.
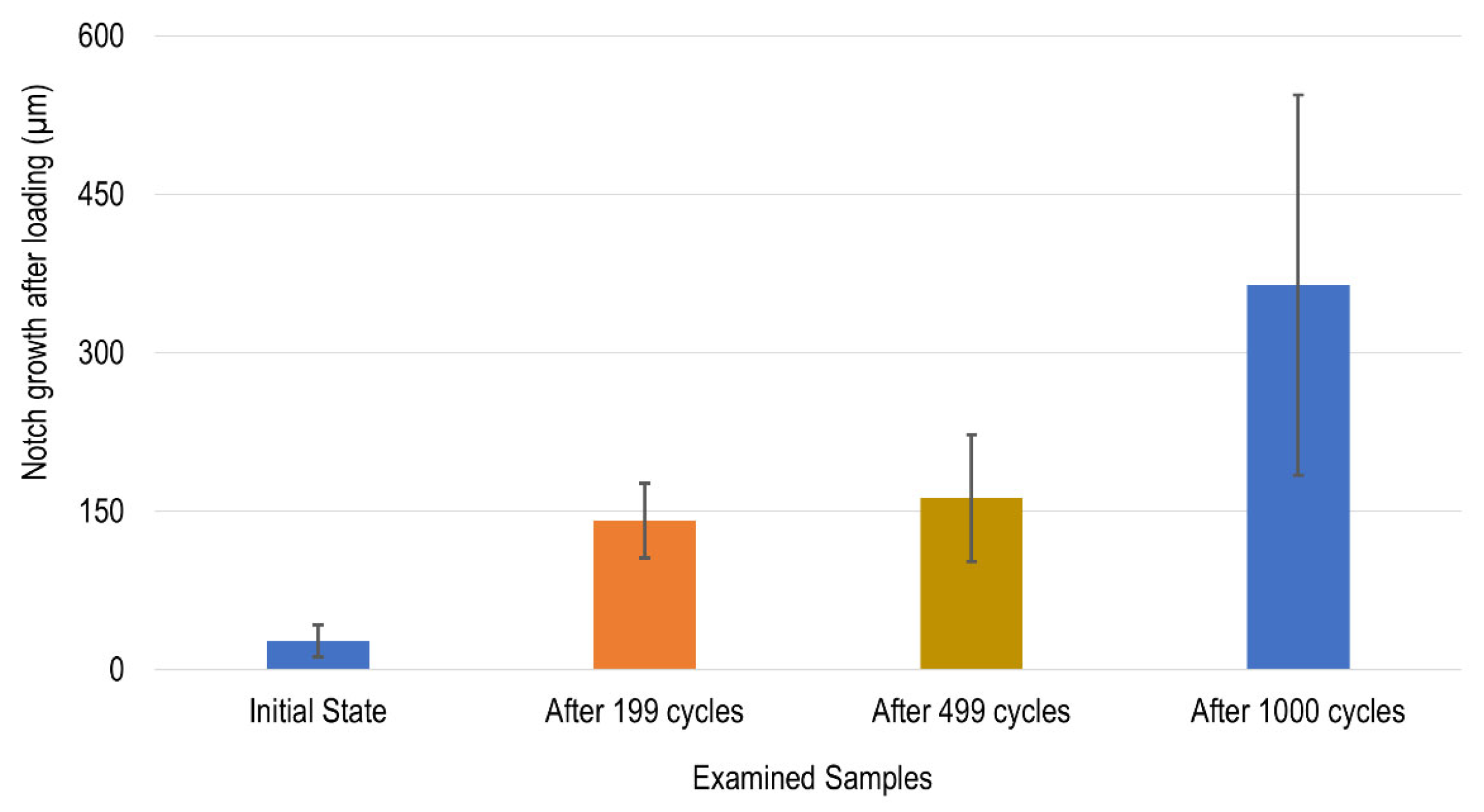
Figure 15 illustrates the effect of the cycling process on the notch growth in the silicone samples before and after different cycling numbers, including 200, 500, and 1000. The graphs demonstrate that increasing the number of cleaning, disinfection, and sterilization cycles led to a notable increase in notch growth. After 200 cycles, the average notch growth was approximately six times higher than that of the unprocessed samples. Extending the process to 500 cycles resulted in a moderate increase of around 14% compared to the 200-cycle samples. However, further processing up to 1000 cycles caused a substantial rise in notch growth, reaching more than twice the value observed after 500 cycles. These findings suggest a progressive deterioration in the material’s resistance to crack propagation with prolonged cyclic processing. It is also evident that a considerable drop in mechanical properties occurred after the initial 200 cycles of cleaning, disinfection, and sterilization. However, extending the process to 500 cycles did not result in a significant additional change, which could be attributed to the material’s temporary resistance to further deterioration. In contrast, a substantial decline in mechanical performance was observed again after 1000 cycles, suggesting the onset of a second phase of pronounced degradation. This indicates that between 500 and 1000 cycles, the material underwent another critical stage of mechanical deterioration.
The significant increase in notch growth, especially after 200 and 1000 cycles, points to a reduction in fracture resistance and increased brittleness of the silicone material. This behavior is attributed to thermal aging and over-curing effects, where repeated thermal cycles enhance crosslink density beyond optimal levels, decreasing the material’s ability to undergo plastic deformation. Additionally, accumulated microstructural damage such as chain scission or void formation may facilitate crack initiation and propagation. These results demonstrate a shift in the fracture mechanics of the material, underlining the importance of evaluating crack resistance for components subjected to cyclic thermal and mechanical loading, especially in load-bearing or stress-prone regions of medical devices.
3.4. Steel–Silicone Samples
Many applications of silicone materials in medical devices involve not only pure silicone components but also parts that interact with other materials, such as stainless steel. To evaluate the impact of repeated cleaning, disinfection, and sterilization cycles on the interaction between silicone and steel, the same cyclic processing was applied to samples featuring silicone–steel contact. This approach aimed to assess any changes in adhesion or mechanical integrity at the material interface resulting from prolonged exposure to processing conditions.
Figure 16 presents optical microscopy images of multiple samples at the silicone–steel interface after 200, 500, and 1000 cycles. Significant changes at the interface were observed during the inspection process. After 200 cycles, the formation of cracks, holes, and signs of erosion became evident. These surface defects progressively worsened with an increasing number of cycles. In the samples subjected to 500 and 1000 cycles, a substantial enlargement of cracks and pronounced surface deterioration were clearly visible, indicating a progressive breakdown of the silicone–steel interface under prolonged processing. Intense erosion along the silicone–steel interface was observed in all samples, with the most severe deterioration occurring after 1000 cycles. This progressive degradation is primarily attributed to the inherent vulnerability of the interface, which acts as a stress concentration zone during thermal and mechanical loading. The repeated cleaning, disinfection, and sterilization cycles expose the interface to fluctuating temperatures, moisture, and potential chemical interactions, all of which can compromise the structural integrity of the contact region. At the microscopic level, differences in thermal expansion coefficients between silicone and steel can generate interfacial stresses during each cycle, promoting the initiation of microcracks. Over time, these microcracks can propagate and coalesce, leading to the formation of larger cracks and surface erosion. Additionally, the interface may be subject to mechanical fatigue due to the cyclic expansion and contraction of the materials, further weakening the bond and accelerating damage. The accumulation of defects such as cracks, voids, and delamination reduces the mechanical stability and adhesion between the two materials. This is particularly critical in medical devices, where the durability and reliability of bonded interfaces are essential for long-term performance and patient safety.
4. Conclusions
This study investigated the effects of repeated cleaning, disinfection, and sterilization cycles on the durability of silicone-based medical devices. Since these processes are critical for infection control, their impact on the mechanical and surface properties of silicone components must be understood. The results show that repeated processing can cause surface damage, such as cracks and erosion, which compromise material integrity and may promote contamination. To evaluate these effects, a series of experiments was conducted on both pure silicone and silicone–steel samples subjected to up to 1000 cycles. Mechanical and surface properties were assessed at four stages: the initial state and after 200, 500, and 1000 cycles. The key findings are summarized below:
- (1)
The application of multiple cleaning, disinfection, and sterilization cycles influenced the contact angle of the silicone samples, initially showing a decreasing trend up to 200 cycles (12% reduction), followed by a gradual increase toward the initial value with continued processing. This behavior was primarily attributed to surface erosion caused by the cycling process, which initially increased surface roughness and reduced the contact angle, followed by the removal of weakened surface layers over time, gradually restoring the contact angle toward its original value.
- (2)
The hardness of the samples exhibited an incremental trend at each processing stage, with an approximately constant growth rate of 5%. This increase was attributed to the continued curing of the silicone material, promoted by the mechanical and thermal effects experienced during the repeated cleaning, disinfection, and sterilization cycles.
- (3)
The notch growth of the silicone samples under loading showed significant changes, particularly after 200 and 1000 cycles, reaching approximately more than sixfold and twofold increases, respectively, compared to the previous stages. This behavior can be attributed to increased brittleness in the polymer structure, likely caused by over-curing and excessive crosslinking within the polymer chains during the repeated thermal and mechanical processing.
- (4)
The cycling process had a significant impact on the steel–silicone interfacial area, leading to pronounced crack initiation and propagation over time. Additionally, intensive surface erosion and partial debonding at the steel–silicone interface were observed, indicating that repeated cleaning, disinfection, and sterilization cycles can compromise the integrity of the bonded interface and potentially affect the performance and durability of multi-material medical components.
For future work, additional analyses such as long-term fatigue testing, chemical characterization, and microbiological assessments could provide deeper insight into the cumulative effects of repeated sterilization on silicone-based medical devices. Expanding the study to include different silicone formulations and sterilization methods (e.g., ethylene oxide, gamma irradiation) would also help generalize the findings and support broader material selection and device design recommendations.