Tridimensional Alginate Films with Cat’s Claw (Uncaria tomentosa) Extract or Aloe Vera (Aloe barbadensis) Gel for Potential Use as Wound Dressings †
Abstract
:1. Introduction
2. Experimental Part
2.1. Materials
2.2. Plant Extraction Process
2.3. Microparticles and Film Formation
2.4. Characterization of Films
2.4.1. Thickness and Morphological Analysis
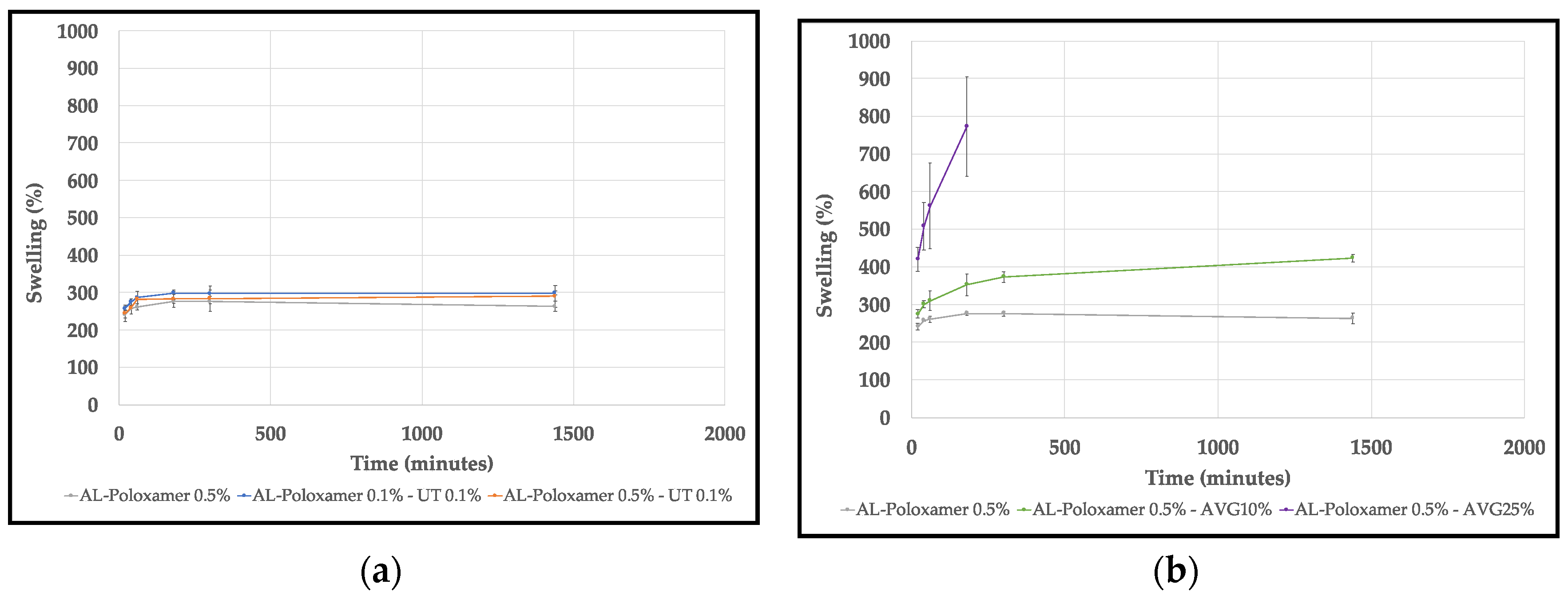
2.4.2. Swelling Behavior
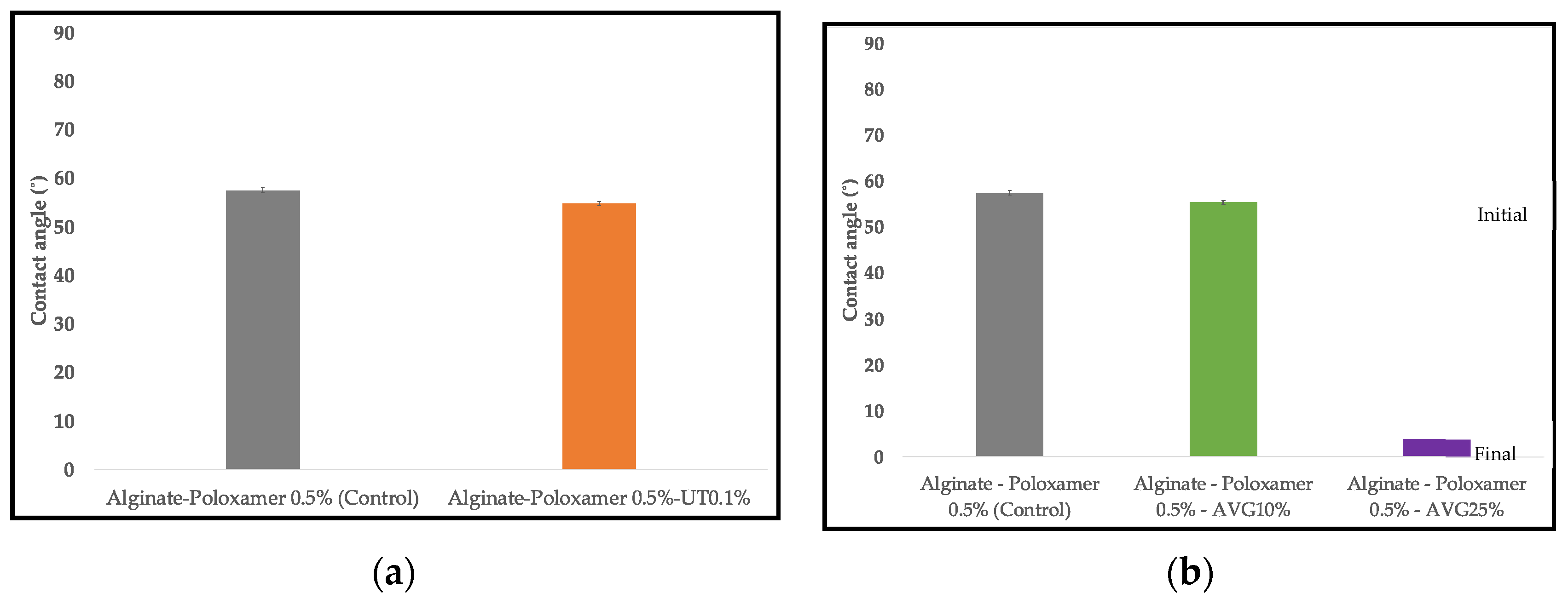
2.4.3. Wettability
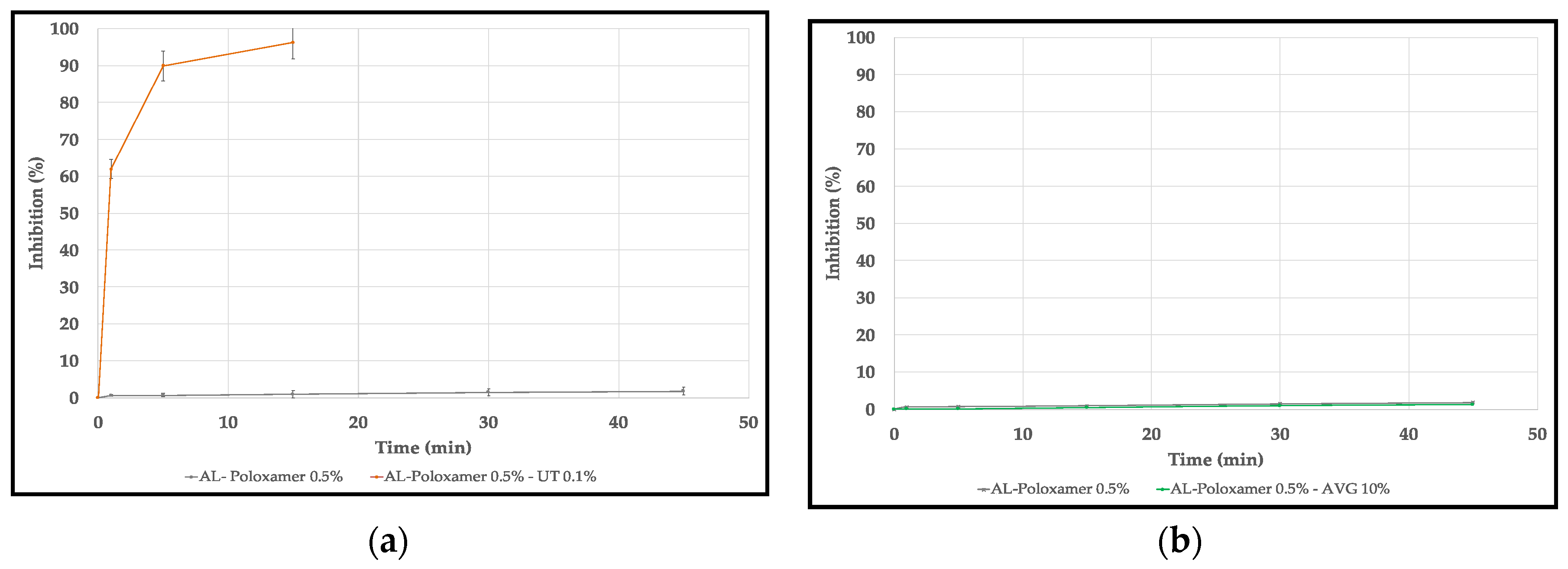
2.4.4. Antioxidant Activity
3. Results
3.1. Physical Appearance, Thickness and Morphological Analysis
3.2. Swelling Behavior
3.3. Wettability
3.4. Antioxidant Activity
4. Discussion
4.1. Physical Appearance, Thickness and Morphological Analysis
4.2. Swelling Behavior
4.3. Wettability
4.4. Antioxidant Activity
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Abbreviations
| UT: | Cat’s claw (Uncaria tomentosa) |
| AVG: | Aloe vera gel |
| AL: | Alginate |
| SEM: | Scanning electron microscopy |
| ABTS: | 2,2’-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid) |
References
- Kamoun, E.A.; Kenawy, E.-R.S.; Chen, X. A review on polymeric hydrogel membranes for wound dressing applications: PVA-based hydrogel dressings. J. Adv. Res. 2017, 8, 217–233. [Google Scholar] [CrossRef] [PubMed]
- Naseri-Nosar, M.; Ziora, Z.M. Wound dressings from naturally-occurring polymers: A review on homopolysaccharide-based composites. Carbohydr. Polym. 2018, 189, 379–398. [Google Scholar] [CrossRef] [PubMed]
- Pereira, R.; Mendes, A.; Bártolo, P. Alginate/Aloe Vera Hydrogel Films for Biomedical Applications. Procedia CIRP 2013, 5, 210–215. [Google Scholar] [CrossRef]
- Eshun, K.; He, Q. Aloe Vera: A Valuable Ingredient for the Food, Pharmaceutical and Cosmetic Industries—A Review. Crit. Rev. Food Sci. Nutr. 2004, 44, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Hamman, J. Composition and Applications of Aloe vera Leaf Gel. Molecules 2008, 13, 1599–1616. [Google Scholar] [CrossRef] [PubMed]
- Saberian, H.; Abbasi, S.; Hamidi-Esfahani, Z. Effect of pasteurization and storage on bioactive components of Aloe vera gel. Nutr. Food Sci. 2013, 43, 175–183. [Google Scholar] [CrossRef]
- Silva, S.S.; Popa, E.G.; Gomes, M.E.; Cerqueira, M.T.; Marques, A.P.; Caridade, S.G.; Teixeira, P.; Sousa, C.; Mano, J.F.; Reis, R.L. An investigation of the potential application of chitosan/aloe-based membranes for regenerative medicine. Acta Biomater. 2013, 9, 6790–6797. [Google Scholar] [CrossRef] [PubMed]
- Heitzman, M.E.; Neto, C.C.; Winiarz, E.; Vaisberg, A.J.; Hammond, G.B. Ethnobotany, phytochemistry and pharmacology of Uncaria (Rubiaceae). Phytochemistry 2005, 66, 5–29. [Google Scholar] [CrossRef] [PubMed]
- Sheng, Y.; Pero, R.W.; Amiri, A.; Bryngelsson, C. Induction of apoptosis and inhibition of proliferation in human tumor cells treated with extracts of Uncaria tomentosa. Anticancer Res. 1998, 18, 3363–3368. [Google Scholar] [PubMed]
- Keplinger, K.; Laus, G.; Wurm, M.; Dierich, M.P.; Teppner, H. Uncaria tomentosa (Willd.) DC.—Ethnomedicinal use and new pharmacological, toxicological and botanical results. J. Ethnopharmacol. 1998, 64, 23–34. [Google Scholar] [CrossRef]
- Navarro-Hoyos, M.; Alvarado-Corella, D.; Moreira-Gonzalez, I.; Arnaez-Serrano, E.; Monagas-Juan, M. Polyphenolic Composition and Antioxidant Activity of Aqueous and Ethanolic Extracts from Uncaria tomentosa Bark and Leaves. Antioxidants 2018, 7, 65. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Aguilera, A.; Rull, A.; Rodríguez-Gallego, E.; Riera-Borrull, M.; Luciano-Mateo, F.; Camps, J.; Menéndez, J.A.; Joven, J. Mitochondrial Dysfunction: A Basic Mechanism in Inflammation-Related Non-Communicable Diseases and Therapeutic Opportunities. Mediat. Inflamm. 2013, 2013, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Navarro-Hoyos, M.; Lebrón-Aguilar, R.; Quintanilla-López, J.E.; Cueva, C.; Hevia, D.; Quesada, S.; Azofeifa, G.; Moreno-Arribas, M.; Monagas, M.; Bartolomé, B. Proanthocyanidin Characterization and Bioactivity of Extracts from Different Parts of Uncaria tomentosa L. (Cat’s Claw). Antioxidants 2017, 6, 12. [Google Scholar] [CrossRef] [PubMed]
- Navarro, M.; Zamora, W.; Quesada, S.; Azofeifa, G.; Alvarado, D.; Monagas, M. Fractioning of Proanthocyanidins of Uncaria tomentosa. Composition and Structure-Bioactivity Relationship. Antioxidants 2017, 6, 60. [Google Scholar] [CrossRef] [PubMed]
- Shankar, S.; Kasapis, S.; Rhim, J.-W. Alginate-based nanocomposite films reinforced with halloysite nanotubes functionalized by alkali treatment and zinc oxide nanoparticles. Int. J. Biol. Macromol. 2018, 118, 1824–1832. [Google Scholar] [CrossRef] [PubMed]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elgegren, M.; Donayre, A.; Kim, S.; Galarreta, B.C.; Nakamatsu, J. Tridimensional Alginate Films with Cat’s Claw (Uncaria tomentosa) Extract or Aloe Vera (Aloe barbadensis) Gel for Potential Use as Wound Dressings. Proceedings 2021, 69, 24. https://doi.org/10.3390/CGPM2020-07225
Elgegren M, Donayre A, Kim S, Galarreta BC, Nakamatsu J. Tridimensional Alginate Films with Cat’s Claw (Uncaria tomentosa) Extract or Aloe Vera (Aloe barbadensis) Gel for Potential Use as Wound Dressings. Proceedings. 2021; 69(1):24. https://doi.org/10.3390/CGPM2020-07225
Chicago/Turabian StyleElgegren, Mariela, Alonso Donayre, Suyeon Kim, Betty C. Galarreta, and Javier Nakamatsu. 2021. "Tridimensional Alginate Films with Cat’s Claw (Uncaria tomentosa) Extract or Aloe Vera (Aloe barbadensis) Gel for Potential Use as Wound Dressings" Proceedings 69, no. 1: 24. https://doi.org/10.3390/CGPM2020-07225
APA StyleElgegren, M., Donayre, A., Kim, S., Galarreta, B. C., & Nakamatsu, J. (2021). Tridimensional Alginate Films with Cat’s Claw (Uncaria tomentosa) Extract or Aloe Vera (Aloe barbadensis) Gel for Potential Use as Wound Dressings. Proceedings, 69(1), 24. https://doi.org/10.3390/CGPM2020-07225




