Quantifying Topographical Changes in Muscle Activation: A Statistical Parametric Mapping Approach †
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants and Experimental Protocol
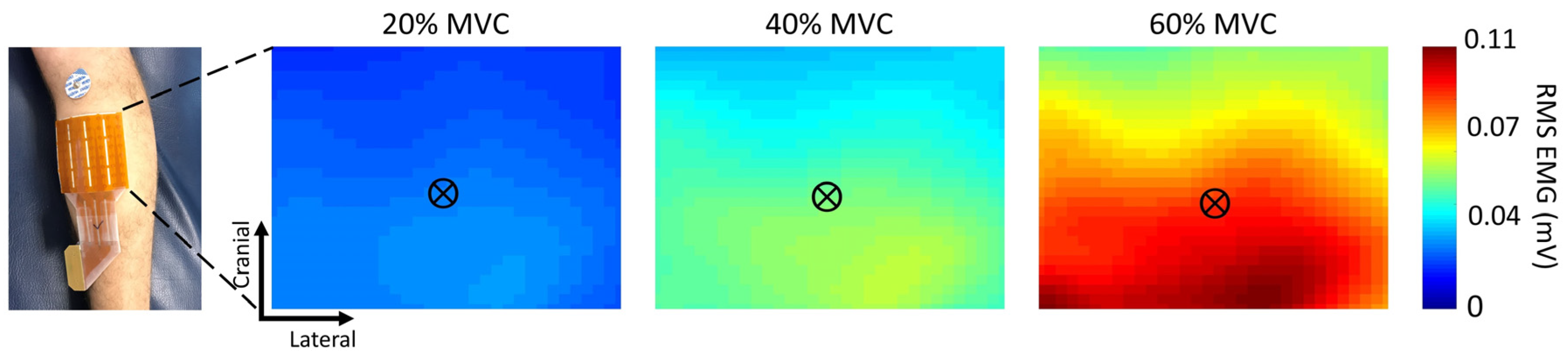
2.2. HDEMG Recordings
2.3. Data Processing
2.4. HDEMG Topographical Map Processing
2.5. SPM and Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Gallina, A.; Garland, S.J.; Wakeling, J.M. Identification of regional activation by factorization of high-density surface EMG signals: A comparison of Principal Component Analysis and Non-negative Matrix factorization. J. Electromyogr. Kinesiol. 2018, 41, 116–123. [Google Scholar] [CrossRef] [PubMed]
- Friston, K.J.; Ashburner, J.T.; Kiebel, S.J.; Nichols, T.E.; Penny, W.D. Statistical Parametric Mapping: The Analysis of Functional Brain Images; Elsevier: Amsterdam, The Netherlands, 2011; ISBN 978-0-08-046650-7. [Google Scholar]
- Kinugasa, R.; Kawakami, Y.; Sinha, S.; Fukunaga, T. Unique spatial distribution of in vivo human muscle activation. Exp. Physiol. 2011, 96, 938–948. [Google Scholar] [CrossRef] [PubMed]
- NIfTI-1 Data Format—Neuroimaging Informatics Technology Initiative. Available online: https://nifti.nimh.nih.gov/nifti-1/ (accessed on 18 October 2019).
- Litvak, V.; Mattout, J.; Kiebel, S.; Phillips, C.; Henson, R.; Kilner, J.; Barnes, G.; Oostenveld, R.; Daunizeau, J.; Flandin, G.; et al. EEG and MEG Data Analysis in SPM8. Comput. Intell. Neurosci. 2011, 2011, 852961. [Google Scholar] [CrossRef] [PubMed]
- Vinti, M.; Gracies, J.-M.; Gazzoni, M.; Vieira, T. Localised sampling of myoelectric activity may provide biased estimates of cocontraction for gastrocnemius though not for soleus and tibialis anterior muscles. J. Electromyogr. Kinesiol. 2018, 38, 34–43. [Google Scholar] [CrossRef] [PubMed]

| Height Threshold 1 | KE 2 | PFWEcorr 3 | Test Statistic 4 | Peak Coordinates 5 | ||
|---|---|---|---|---|---|---|
| RM ANOVA | F = 3.7 | 896 | <0.001 | F = 117 | 68 | 39 |
| 40% vs. 20% | T = 2.8 | 896 | <0.001 | T = 9.48 | 68 | 56 |
| 60% vs. 20% | T = 2.8 | 896 | <0.001 | T = 7.06 | 68 | 54 |
| 60% vs. 40% | T = 2.8 | 896 | <0.001 | T = 4.77 | 2 | 9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pincheira, P.A.; Martinez-Valdes, E.; De la Fuente, C.; Palma, F.; Valencia, O.; Redenz, G.; Guzman-Venegas, R. Quantifying Topographical Changes in Muscle Activation: A Statistical Parametric Mapping Approach. Proceedings 2020, 49, 71. https://doi.org/10.3390/proceedings2020049071
Pincheira PA, Martinez-Valdes E, De la Fuente C, Palma F, Valencia O, Redenz G, Guzman-Venegas R. Quantifying Topographical Changes in Muscle Activation: A Statistical Parametric Mapping Approach. Proceedings. 2020; 49(1):71. https://doi.org/10.3390/proceedings2020049071
Chicago/Turabian StylePincheira, Patricio A., Eduardo Martinez-Valdes, Carlos De la Fuente, Felipe Palma, Oscar Valencia, Gunther Redenz, and Rodrigo Guzman-Venegas. 2020. "Quantifying Topographical Changes in Muscle Activation: A Statistical Parametric Mapping Approach" Proceedings 49, no. 1: 71. https://doi.org/10.3390/proceedings2020049071
APA StylePincheira, P. A., Martinez-Valdes, E., De la Fuente, C., Palma, F., Valencia, O., Redenz, G., & Guzman-Venegas, R. (2020). Quantifying Topographical Changes in Muscle Activation: A Statistical Parametric Mapping Approach. Proceedings, 49(1), 71. https://doi.org/10.3390/proceedings2020049071





