Apoptosis Signalling Pathway in Cervical Cancer Cells Treated with Gama Radiation †
Abstract
:1. Introduction
2. Materials and Methods
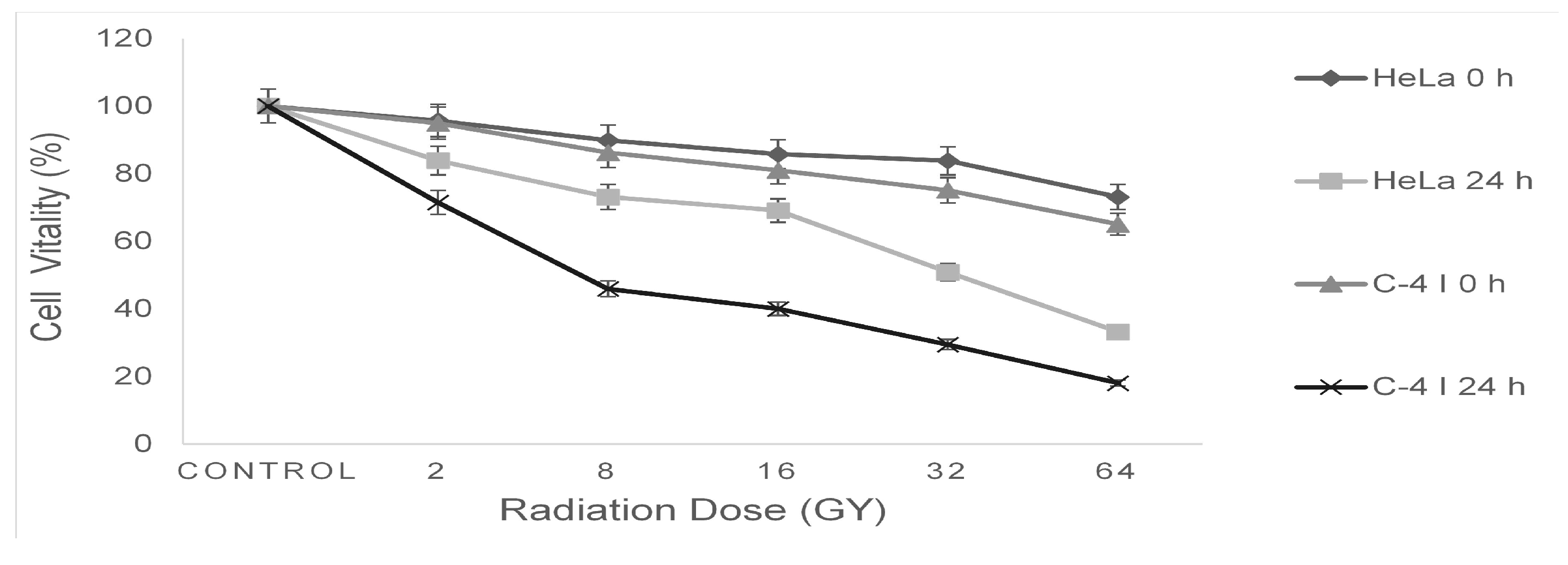
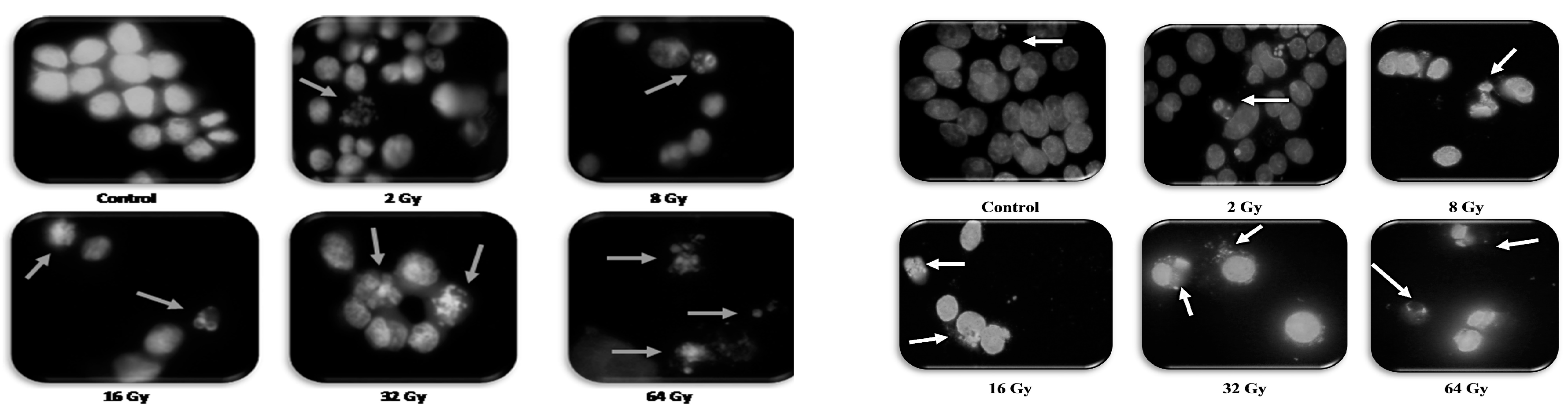
3. Results
4. Discussion
References
- Nair, S.; Nair, R.R.K.; Srinivas, P.; Srinivas, G.; Pillai, M.R. Radiosensitizing effects of plumbagin in cervical cancer cells is through modulation of apoptotic pathway. Mol. Carcinog. 2008, 47, 22–33. [Google Scholar] [CrossRef] [PubMed]
- Wyllie, A.H. A Brief Review of Apoptosis Biology. Mol. Neurobiol. 2010, 42, 4–9. [Google Scholar] [CrossRef] [PubMed]
- Bian, J.; Wang, X.; Yun, J.; Cao, R.; Cao, Y.; Liang, J.; Ma, B.F. Single-fraction γ−60Co radiation induces apoptosis in cultured rat C6 cells. Ann. Saudi Med. 2012, 32, 269–275. [Google Scholar] [CrossRef] [PubMed]
- Özcan, G.; Dağdeviren, Ö.; Akman, G.; Khalilia, W.; Tezel Yetiz, B.; Karagöz, A.; Melikoglu, G.; Anil, S.; Kultur, S.; Sutlupinar, N. Screening for antitumour activity of various plant extracts on HeLa and C 4-I cell lines. J. Buon 2016, 21, 1552–1560. [Google Scholar] [PubMed]
- Khalilia, W.M.; Özcan, G.; Karaçam, S. Gene Expression and Pathway Analysis of Radiation-Induced Apoptosis in C-4 I Cervical Cancer Cells. J. Autoimmun. Res. 2017, 4, 1014. [Google Scholar]
- Mi, H.; Muruganujan, A.; Casagrande, J.T.; Thomas, P.D. Large-scale gene function analysis with the PANTHER classification system. Nat. Protocols 2013, 8, 1551–1566. [Google Scholar] [CrossRef] [PubMed]
- Elkoreh, G.; Blais, V.; Béliveau, E.; Guillemette, G.; Denault, J.B. Type1inositol-1,4,5 trisphosphate receptor isalate substrate of caspases during apoptosis. J. Cell. Biochem. 2012, 113, 2775–2784. [Google Scholar] [CrossRef] [PubMed]
- Davies, L.; Gray, D.; Spiller, D.; White, M.R.H.; Damato, B.; Grierson, I.; Paraoan, L. P53 apoptosis mediator PERP: Localization, function and caspase activation in uveal melanoma. J. Cell. Mol. Med. 2009, 13, 1995–2007. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khalilia, W.M.; Özcan, G.; Karaçam, S. Apoptosis Signalling Pathway in Cervical Cancer Cells Treated with Gama Radiation. Proceedings 2018, 2, 1531. https://doi.org/10.3390/proceedings2251531
Khalilia WM, Özcan G, Karaçam S. Apoptosis Signalling Pathway in Cervical Cancer Cells Treated with Gama Radiation. Proceedings. 2018; 2(25):1531. https://doi.org/10.3390/proceedings2251531
Chicago/Turabian StyleKhalilia, Walid Mahmoud, Gül Özcan, and Songül Karaçam. 2018. "Apoptosis Signalling Pathway in Cervical Cancer Cells Treated with Gama Radiation" Proceedings 2, no. 25: 1531. https://doi.org/10.3390/proceedings2251531
APA StyleKhalilia, W. M., Özcan, G., & Karaçam, S. (2018). Apoptosis Signalling Pathway in Cervical Cancer Cells Treated with Gama Radiation. Proceedings, 2(25), 1531. https://doi.org/10.3390/proceedings2251531





