Collagenase Biosensor Based on the Degradation of Peptide Cross-Linked Poly(Ethylene Glycol) Hydrogel Films †
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
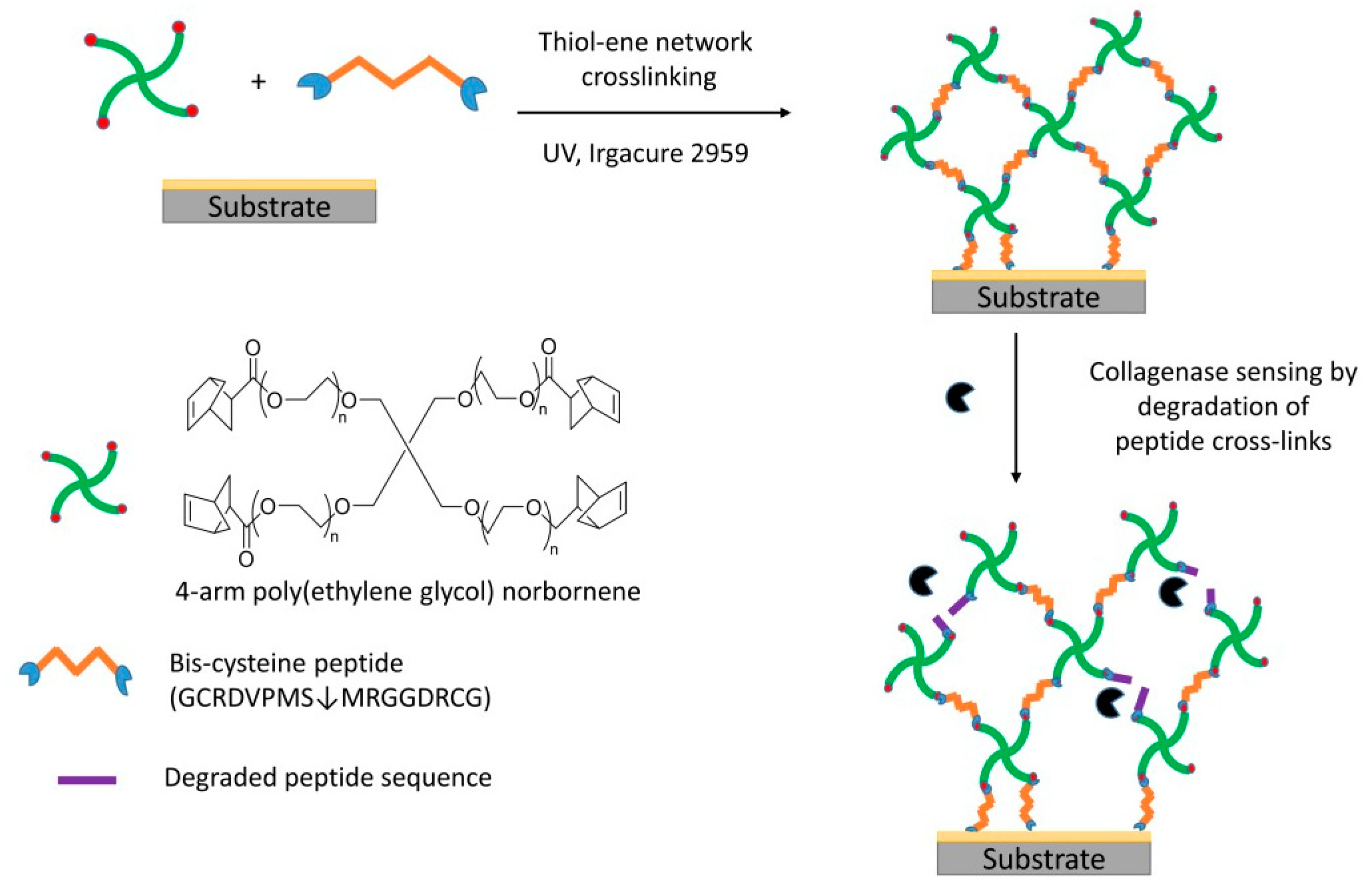
2.2. Sensor Fabrication
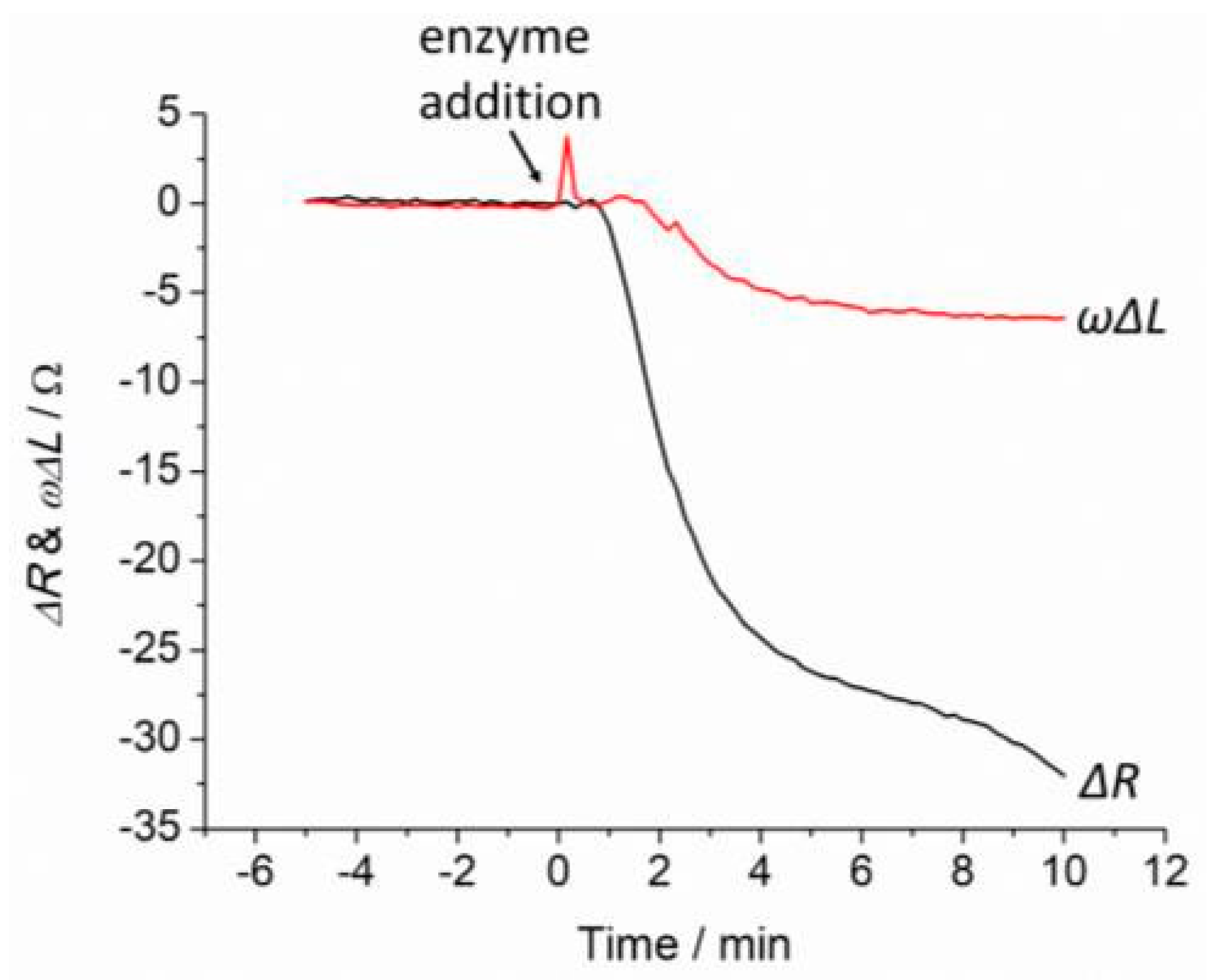
2.3. Monitoring Degradation of Hydrogel Using QCM Measurements
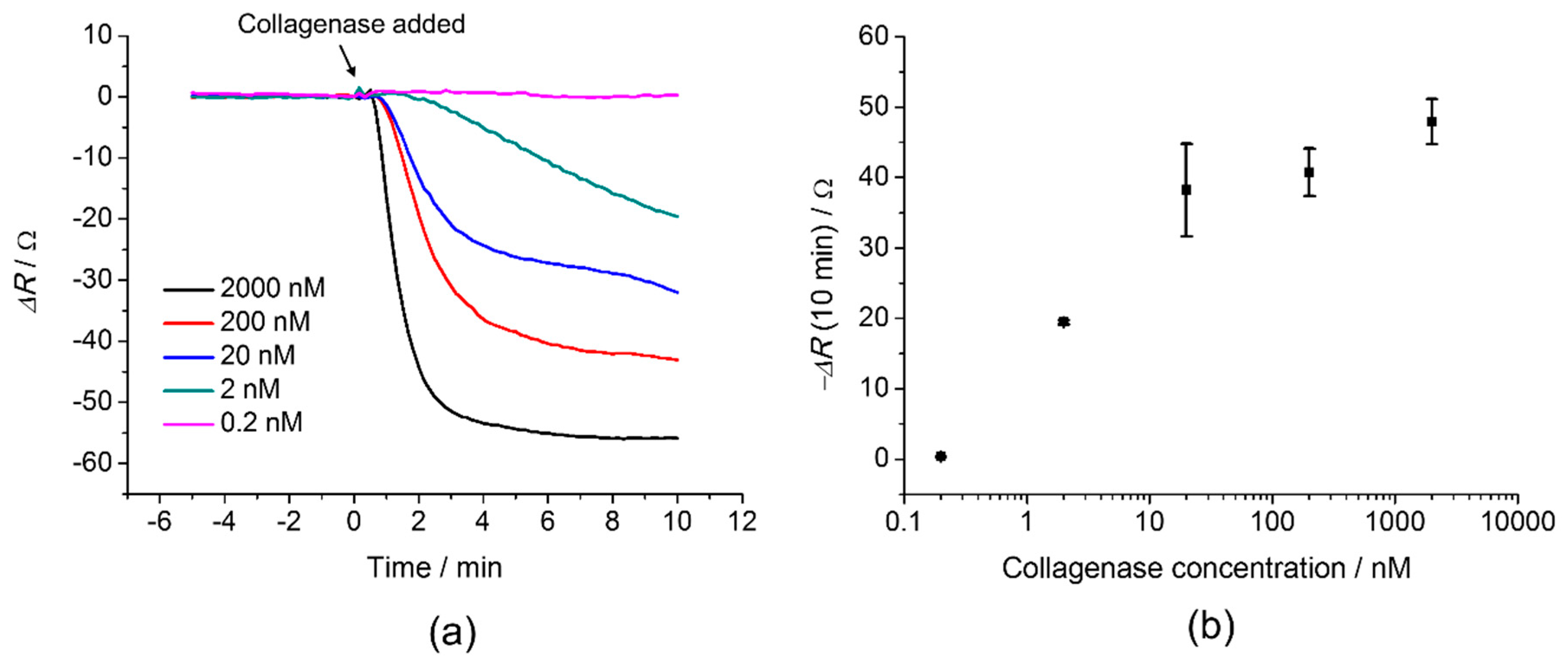
3. Results and Discussion
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Biela, A.; Watkinson, M.; Meier, U.C.; Baker, D.; Giovannoni, G.; Becer, C.R.; Krause, S. Disposable MMP-9 sensor based on the degradation of peptide cross-linked hydrogel films using electrochemical impedance spectroscopy. Biosens. Bioelectron. 2015, 68, 660–667. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Cook, J.P.; Watkinson, M.; Yang, S.; Douglas, I.; Rawlinson, A.; Krause, S. Generic protease detection technology for monitoring periodontal disease. Faraday Discuss. 2011, 149, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Clark, I.M.; Powell, L.K.; Ramsey, S.; Hazleman, B.L.; Cawston, T.E. The measurement of collagenase, tissue inhibitor of metalloproteinases (timp), and collagenase—Timp complex in synovial fluids from patients with osteoarthritis and rheumatoid arthritis. Arthritis Rheum. 1993, 36, 372–379. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, I.; Pangule, R.C.; Kane, R.S. Antifouling coatings: Recent developments in the design of surfaces that prevent fouling by proteins, bacteria, and marine organisms. Adv. Mater. 2011, 23, 690–718. [Google Scholar] [CrossRef] [PubMed]
- Colak, B.; Da Silva, J.C.S.; Soares, T.A.; Gautrot, J.E. Impact of the Molecular Environment on Thiol- Ene Coupling for Biofunctionalization and Conjugation. Bioconjug. Chem. 2016, 27, 2111–2123. [Google Scholar] [CrossRef] [PubMed]
- Sabot, A.; Krause, S. Simultaneous quartz crystal microbalance impedance and electrochemical impedance measurements. Investigation into the degradation of thin polymer films. Anal. Chem. 2002, 74, 3304–3311. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahmad, N.; Colak, B.; Gibbs, M.J.; Zhang, D.-W.; Becer, C.R.; Watkinson, M.; Gautrot, J.E.; Krause, S. Collagenase Biosensor Based on the Degradation of Peptide Cross-Linked Poly(Ethylene Glycol) Hydrogel Films. Proceedings 2018, 2, 961. https://doi.org/10.3390/proceedings2130961
Ahmad N, Colak B, Gibbs MJ, Zhang D-W, Becer CR, Watkinson M, Gautrot JE, Krause S. Collagenase Biosensor Based on the Degradation of Peptide Cross-Linked Poly(Ethylene Glycol) Hydrogel Films. Proceedings. 2018; 2(13):961. https://doi.org/10.3390/proceedings2130961
Chicago/Turabian StyleAhmad, Norlaily, Burcu Colak, Martin John Gibbs, De-Wen Zhang, C. Remzi Becer, Michael Watkinson, Julien E. Gautrot, and Steffi Krause. 2018. "Collagenase Biosensor Based on the Degradation of Peptide Cross-Linked Poly(Ethylene Glycol) Hydrogel Films" Proceedings 2, no. 13: 961. https://doi.org/10.3390/proceedings2130961
APA StyleAhmad, N., Colak, B., Gibbs, M. J., Zhang, D.-W., Becer, C. R., Watkinson, M., Gautrot, J. E., & Krause, S. (2018). Collagenase Biosensor Based on the Degradation of Peptide Cross-Linked Poly(Ethylene Glycol) Hydrogel Films. Proceedings, 2(13), 961. https://doi.org/10.3390/proceedings2130961




