Exploring Potential Antimalarial Candidate from Medicinal Plants of Kheaw Hom Remedy
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Collection and Management
2.2. Plant Extraction
2.3. Phytochemical Analysis
2.4. In Vitro Cultivation and Maintenance of Plasmodium falciparum
2.5. In Vitro Antimalarial Activity Assay
2.6. In Vitro Assessments of Cytotoxicity
2.7. Four-Day Suppressive Test (Peter’s Test)
2.8. Acute Toxicity Test
2.9. Selectivity Index
2.10. Ethical Statement
2.11. Statistical Analysis
3. Results
3.1. Phytochemical Screening
3.2. In Vitro Antimalarial Activity and Cytotoxicity of the Plant Extracts from Kheaw Hom Remedy
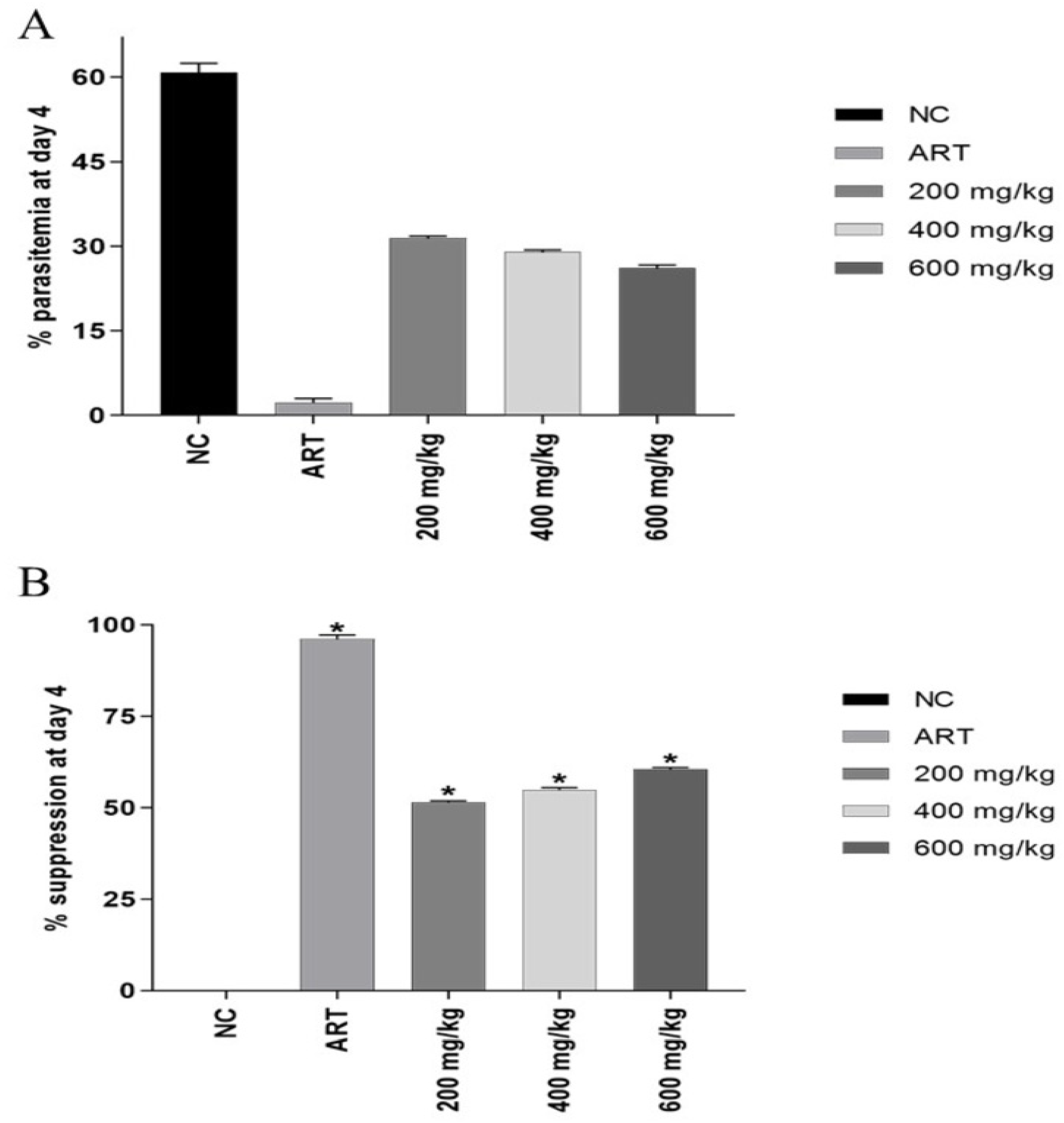
3.3. Four-Day Suppressive Test
3.4. Acute Toxicity Test
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- CDC. Malaria. Available online: https://www.cdc.gov/malaria/index.html (accessed on 20 May 2020).
- WHO. World Malaria Report 2019; WHO: Geneva, Switzerland, 2019. [Google Scholar]
- Bloland, P.B. Drug Resistance in Malaria; WHO: Geneva, Switzerland, 2001. [Google Scholar]
- Department for Development of Thai Traditional and Alternative Medicine. List of Herbal Medicine Products A.D 2011; The War Veterans Organization of Thailand under Royal Patronage of His Majesty the King: Bangkok, Thailand, 2011.
- Chusri, S.; Sinvaraphan, N.; Chaipak, P.; Luxsananuwong, A.; Voravuthikunchai, S.P. Evaluation of antibacterial activity, phytochemical constituents, and cytotoxicity effects of Thai household ancient remedies. J. Altern. Complement. Med. 2014, 20, 909–918. [Google Scholar] [CrossRef] [PubMed]
- Wutthithammawet, W. Textbook of Rattanakosin Pharmacy; Wutthithammawet Cooporation: Bangkok, Thailand, 2002. [Google Scholar]
- Sukkasem, K.; Panthong, S.; Itharat, A. Antimicrobial activities of Thai traditional remedy “Kheaw-Hom” and its plant ingredients for skin infection treatment in chickenpox. J. Med. Assoc. Thai. 2016, 99, 116. [Google Scholar] [PubMed]
- Ouncharoen, K.; Itharat, A.; Chaiyawatthanananthn, P. In vitro free radical scavenging and cell-based antioxidant activities of Kheaw-Hom remedy extracts and its plant ingredients. J. Med. Assoc. Thai. 2017, 100, 241. [Google Scholar]
- Sukkasem, K. Biological Activities of Thai Traditional Remedy Called Kheaw-Hom and Its Plant Ingredients; Thammasat University: Bangkok, Thailand, 2015. [Google Scholar]
- Anuthakoengkun, A.; Itharat, A. Inhibitory effect on nitric oxide production and free radical scavenging activity of Thai medicinal plants in osteoarthritic knee treatment. J. Med. Assoc. Thai. 2014, 97, 116. [Google Scholar]
- Makchuchit, S. Anti-Inflammatory and Anti-Allergic Activities of Thai Traditional Medicine Preparation Called Prasaprohyai; Thammasat University: Bangkok, Thailand, 2010. [Google Scholar]
- Chaniad, P.; Techarang, T.; Phuwajaroanpong, A.; Punsawad, C. Antimalarial activity and toxicological assessment of Betula alnoides extract against Plasmodium berghei infections in mice. Evid. Based Complementary Altern. Med. 2019, 2019, 2324679. [Google Scholar] [CrossRef] [PubMed]
- Senguttuvan, J.; Paulsamy, S.; Karthika, K. Phytochemical analysis and evaluation of leaf and root parts of the medicinal herb, Hypochaeris radicata L. for in vitro antioxidant activities. Asian. Pac. J. Trop. Biomed. 2014, 4, S359–S367. [Google Scholar] [CrossRef] [PubMed]
- Trager, W.; Jensen, J.B. Human malaria parasites in continuous culture. 1976. J. Parasitol. Res. 2005, 91, 484–486. [Google Scholar] [CrossRef]
- Makler, M.T.; Hinrichs, D.J. Measurement of the lactate dehydrogenase activity of Plasmodium falciparum as an assessment of parasitemia. Am. J. Trop. Med. Hyg. 1993, 48, 205–210. [Google Scholar] [CrossRef]
- Patel, S.; Gheewala, N.; Suthar, A.; Shah, A.M. In-vitro cytotoxicity activity of Solanum Nigrum extract against Hela cell line and Vero cell line. Int. J. Pharm. Pharm. Sci. 2009, 1, 38–46. [Google Scholar]
- Peters, W. The four-day suppressive in vivo antimalarial test. Ann. Trop. Med. Parasitol. 1975, 69, 155–171. [Google Scholar] [CrossRef]
- Muluye, A.B.; Desta, A.G.; Abate, S.K.; Dano, G.T. Anti-malarial activity of the root extract of Euphorbia abyssinica (Euphorbiaceae) against Plasmodium berghei infection in mice. Malar. J. 2019, 18, 261. [Google Scholar] [CrossRef] [PubMed]
- OECD. Test No. 425: Acute Oral Toxicity: Up-and-Down Procedure; OECD: Paris, France, 2008. [Google Scholar]
- Indrayanto, G.; Putra, G.; Suhud, F. Validation of in-vitro bioassay methods: Application in herbal drug research. Profiles Drug Subst. Excip. Relat. Methodol. 2021, 46, 273–307. [Google Scholar] [PubMed]
- Valdés, A.F.; Martínez, J.M.; Lizama, R.S.; Gaitén, Y.G.; Rodríguez, D.A.; Payrol, J.A. In vitro antimalarial activity and cytotoxicity of some selected cuban medicinal plants. Rev. Inst. Med. Trop. Sao Paulo 2010, 52, 197–201. [Google Scholar] [CrossRef]
- Amoa Onguéné, P.; Ntie-Kang, F.; Lifongo, L.L.; Ndom, J.C.; Sippl, W.; Mbaze, L.M. The potential of anti-malarial compounds derived from African medicinal plants, part I: A pharmacological evaluation of alkaloids and terpenoids. Malar. J. 2013, 12, 449. [Google Scholar] [CrossRef]
- Bidla, G.; Titanji, V.P.; Joko, B.; Ghazali, G.E.; Bolad, A. Antiplasmodial activity of seven plants used in African folk medicine. Indian. J. Pharmacol. 2004, 36, 245–246. [Google Scholar]
- Mbah, J.A.; Tane, P.; Ngadjui, B.T.; Connolly, J.D.; Okunji, C.C.; Iwu, M.M.; Schuster, B.M. Antiplasmodial agents from the leaves of Glossocalyx brevipes. Planta. Med. 2004, 70, 437–440. [Google Scholar] [CrossRef]
- Bringmann, G.; Dreyer, M.; Faber, J.H.; Dalsgaard, P.W.; Staerk, D.; Jaroszewski, J.W.; Ndangalasi, H.; Mbago, F.; Brun, R.; Christensen, S.B. Ancistrotanzanine C and related 5,1′- and 7,3′-coupled naphthylisoquinoline alkaloids from Ancistrocladus tanzaniensis. J. Nat. Prod. 2004, 67, 743–748. [Google Scholar] [CrossRef]
- Bringmann, G.; Messer, K.; Schwöbel, B.; Brun, R.; Aké Assi, L. Habropetaline A, an antimalarial naphthylisoquinoline alkaloid from Triphyophyllum peltatum. Phytochemistry 2003, 62, 345–349. [Google Scholar] [CrossRef]
- Bringmann, G.; Saeb, W.; Wohlfarth, M.; Messer, K.; Brun, R. Jozipeltine A, a Novel, Unnatural Dimer of the Highly Hydroxylated Naphthylisoquinoline Alkaloid Dioncopeltine A1. Tetrahedron 2000, 56, 5871–5875. [Google Scholar] [CrossRef]
- Sathaporn, O. Chemical Constituents and Their Inhibitions of Cyclic AMP Phosphodiesterase of the Rhizomes of Globba Malaccensis Ridl; Chulalongkorn University: Bangkok, Thailand, 2000. [Google Scholar]
- Nattaya, N.; Sathaporn, O.; Surachai, P.; Nongnuj, M.; Amorn, P. Cyclic AMP phosphodiesterase inhibitors from the rhizomes of Globba malaccensis. J. Trop. Med. Plants 2005, 6, 37–41. [Google Scholar]
- Kawamoto, F.; Alejo-Blanco, R.; Fleck, S.L.; Kawamoto, Y.; Sinden, R.E. Possible roles of Ca2+ and cGMP as mediators of the exflagellation of Plasmodium berghei and Plasmodium falciparum. Mol. Biochem. Parasitol. 1990, 42, 101–108. [Google Scholar] [CrossRef]
- Kawamoto, F.; Fujioka, H.; Murakami, R.; Syafruddin; Hagiwara, M.; Ishikawa, T.; Hidaka, H. The roles of Ca2+/calmodulin- and cGMP-dependent pathways in gametogenesis of a rodent malaria parasite, Plasmodium berghei. Eur. J. Cell Biol. 1993, 60, 101–107. [Google Scholar]
- Flueck, C.; Drought, L.G.; Jones, A.; Patel, A.; Perrin, A.J.; Walker, E.M.; Nofal, S.D.; Snijders, A.P.; Blackman, M.J.; Baker, D.A. Phosphodiesterase beta is the master regulator of cAMP signalling during malaria parasite invasion. PLoS Biol. 2019, 17, e3000154. [Google Scholar] [CrossRef] [PubMed]
- Yuasa, K.; Mi-Ichi, F.; Kobayashi, T.; Yamanouchi, M.; Kotera, J.; Kita, K.; Omori, K. PfPDE1, a novel cGMP-specific phosphodiesterase from the human malaria parasite Plasmodium falciparum. Biochem. J. 2005, 392, 221–229. [Google Scholar] [CrossRef] [PubMed]
- Lehane, A.M.; Saliba, K.J. Common dietary flavonoids inhibit the growth of the intraerythrocytic malaria parasite. BMC Res. Notes. 2008, 1, 26. [Google Scholar] [CrossRef] [PubMed]
- Rudrapal, M.; Chetia, D.D. Plant flavonoids as potential source of future antimalarial leads. Syst. Rev. Pharm. 2017, 8, 13–18. [Google Scholar] [CrossRef]
- Penna-Coutinho, J.; Aguiar, A.C.; Krettli, A.U. Commercial drugs containing flavonoids are active in mice with malaria and in vitro against chloroquine-resistant Plasmodium falciparum. Mem. Inst. Oswaldo. Cruz. 2018, 113, e180279. [Google Scholar] [CrossRef]
- Mahidol, C.; Kaweetripob, W.; Prawat, H.; Ruchirawat, S. Mammea coumarins from the flowers of Mammea siamensis. J. Nat. Prod. 2002, 65, 757–760. [Google Scholar] [CrossRef]
- Ninomiya, K.; Shibatani, K.; Sueyoshi, M.; Chaipech, S.; Pongpiriyadacha, Y.; Hayakawa, T.; Muraoka, O.; Morikawa, T. Aromatase inhibitory activity of geranylated coumarins, mammeasins C and D, isolated from the flowers of Mammea siamensis. Chem. Pharm. Bull. (Tokyo) 2016, 64, 880–885. [Google Scholar] [CrossRef]
- Moon, H.I.; Lee, J.H.; Lee, Y.C.; Kim, K.S. Antiplasmodial and cytotoxic activity of coumarin derivatives from dried roots of Angelica gigas Nakai in vitro. Immunopharmacol. Immunotoxicol. 2011, 33, 663–666. [Google Scholar] [CrossRef]
- Adib, A.; Yunos, N.; Chee, B.-J. Anti-cancer, antimicrobial, and antioxidative potentials of Mesua ferrea L. and its phytochemical constituents: A review. Asian. J. Pharm. 2019, 3, 5–19. [Google Scholar]
- Gabriel, H.; Sussmann, R.; Kimura, E.; Marin Rodriguez, A.; Bofill Verdaguer, I.; Leite, G.; Katzin, A. Terpenes as Potential Antimalarial Drugs; Intechopen: London, UK, 2018. [Google Scholar]
- Osman, C.; Ismail, N. Antiplasmodial anthraquinones from medicinal plants: The chemistry and possible mode of actions. Nat. Prod. Commun. 2018, 13, 1591–1597. [Google Scholar] [CrossRef]
- Puangpak, P. The accumulation of heavy metals (Pb and Cd) of the plant family Zingiberaceae at Srinakarind dam, Kanchanaburi province. Thai J. Toxicol. 2015, 30, 89–99. [Google Scholar]
- Mongkol, R.; Chavasiri, W.; Ishida, M.; Matsuda, K.; Morimoto, M. Phytotoxic and antiphytopathogenic compounds from Thai Alpinia galanga (L.) Willd. rhizomes. Weed Biol. Manag. 2015, 15, 87–93. [Google Scholar] [CrossRef]
| No | Plant Species | Family | Plant Part | Voucher Number |
|---|---|---|---|---|
| 1 | P. cablin (Blanco) Benth. | Lamiaceae | Leaves | SMD 142031002 |
| 2 | L. rugosa (Roth) Merr. | Scrophulariaceae | Leaves | SMD 211012017 |
| 3 | C. fruticosa (L.) A.Chev. | Asparagaceae | Red leaves | SMD 004002001 |
| 4 | C. fruticosa (L.) A.Chev. | Asparagaceae | Green leaves | SMD 004002002 |
| 5 | E. stoechadosmum Hance. | Asteraceae | Leaves | SMD 072036002 |
| 6 | V. zizanioides (L.) Nash ex Small | Gramineae | Roots | SMD 119094001 |
| 7 | K. galanga L. | Zingiberaceae | Rhizomes | SMD 288013007 |
| 8 | T. hoaensis Pit. | Rubiaceae | Stem | SMD 233076005 |
| 9 | D. loureiroi Gagnep. | Dracaenaceae | Stem | SMD 096001007 |
| 10 | A. evecta (G. Forst.) Hoffm. | Marattiaceae | Rhizomes | SMD 165001001 |
| 11 | G. malaccensis Ridl. | Zingiberaceae | Rhizomes | SMD 288010008 |
| 12 | T. chantrieri André. | Taccaceae | Rhizomes | SMD 262001001 |
| 13 | S. exigua Craib. | Leguminosae-Papilionoideae | Roots | SMD 148070001 |
| 14 | C. podophylla (Hook.) Copel. | Cyatheaceae | Stem | SMD 084001007 |
| 15 | M. elengi L. | Sapotaceae | Flowers | SMD 042004001 |
| 16 | M. ferrea L. | Guttiferae | Flowers | SMD 122007001 |
| 17 | M. siamensis Kosterm. | Guttiferae | Flowers | SMD 122006002 |
| 18 | N. nucifera Gaertn. | Nelumbonaceae | Stamens | SMD 181001001 |
| Groups (n = 5/group) | Extract/Drug | Dose (mg/kg) |
|---|---|---|
| Negative control | DMSO | - |
| Positive control | Artesunate | 6 |
| Experimental group 1 | G. malaccensis ethanolic extract | 200 |
| Experimental group 2 | G. malaccensis ethanolic extract | 400 |
| Experimental group 3 | G. malaccensis ethanolic extract | 600 |
| Plant | Extract | Phytochemical Constituents | |||||||
|---|---|---|---|---|---|---|---|---|---|
| FL | TN | AL | TA | AN | CG | SA | CM | ||
| P. cablin | Ethanolic | + | - | ++ | ++ | - | - | - | - |
| Aqueous | - | - | ++ | ++ | - | - | + | - | |
| L. rugosa | Ethanolic | - | + | - | + | - | - | - | + |
| Aqueous | - | - | - | + | - | - | - | - | |
| C. fruticosa | Ethanolic | + | - | ++ | + | - | - | - | - |
| (Red leaves) | Aqueous | - | - | - | + | - | - | ++ | - |
| C. fruticosa | Ethanolic | + | - | ++ | + | - | - | - | - |
| (Green leaves) | Aqueous | - | - | - | + | - | - | ++ | - |
| E. stoechadosmum | Ethanolic | - | + | - | + | - | - | - | - |
| Aqueous | - | - | + | + | - | - | - | - | |
| V. zizanioides | Ethanolic | + | + | - | - | - | - | - | + |
| Aqueous | + | - | ++ | + | - | - | + | - | |
| K. galanga | Ethanolic | - | + | - | - | - | - | - | - |
| Aqueous | + | - | +++ | - | - | - | + | - | |
| T. hoaensis | Ethanolic | - | + | - | + | + | - | - | - |
| Aqueous | + | - | ++ | + | - | - | + | - | |
| D. loureiroi | Ethanolic | ++ | + | - | - | - | - | - | - |
| Aqueous | ++ | + | - | + | - | - | - | + | |
| A. evecta | Ethanolic | + | - | - | + | + | - | + | - |
| Aqueous | + | - | + | + | + | - | - | - | |
| G. malaccensis | Ethanolic | ++ | + | - | - | - | - | + | - |
| Aqueous | + | + | + | - | - | - | ++ | + | |
| T. chantrieri | Ethanolic | + | + | - | - | - | - | - | - |
| Aqueous | + | - | + | + | - | - | ++ | - | |
| S. exigua | Ethanolic | +++ | + | - | - | - | - | ++ | + |
| Aqueous | + | - | +++ | - | - | - | - | - | |
| C. podophylla | Ethanolic | + | + | - | ++ | - | - | + | - |
| Aqueous | + | - | - | +++ | - | - | - | + | |
| M. elengi | Ethanolic | ++ | + | - | + | - | - | - | - |
| Aqueous | ++ | - | - | + | - | - | ++ | - | |
| M. ferrea | Ethanolic | + | + | - | ++ | + | - | ++ | + |
| Aqueous | + | - | - | ++ | - | - | + | - | |
| M. siamensis | Ethanolic | ++ | + | - | + | - | - | - | + |
| Aqueous | + | - | - | + | + | - | + | + | |
| N. nucifera | Ethanolic | + | + | - | - | - | - | - | - |
| Aqueous | + | - | - | + | + | - | + | + | |
| Plant | Ethanolic Extract | Aqueous Extract | ||||
|---|---|---|---|---|---|---|
| IC50 (µg/mL) | CC50 (µg/mL) | SI | IC50 (µg/mL) | CC50 (µg/mL) | SI | |
| P. cablin | 24.49 ± 0.01 | >80 | >3.27 | 549.30 ± 0.07 | >80 | >6.87 |
| L. rugosa | 54.02 ± 3.73 | >80 | >1.48 | 83.76 ± 1.58 | >80 | >1.05 |
| C. fruticosa (Red leaves) | 71.26 ± 2.40 | >80 | >1.12 | 245.00 ± 13.18 | >80 | >3.06 |
| C. fruticosa (Green leaves) | 68.31 ± 1.85 | >80 | >1.17 | 245.00 ± 13.18 | >80 | >3.06 |
| E. stoechadosmum | 28.15 ± 0.73 | >80 | >2.84 | 634.60 ± 32.60 | >80 | >7.93 |
| V. zizanioides | 158.70 ± 17.7 | >80 | >0.50 | 546.30 ± 13.20 | >80 | >6.83 |
| K. galanga | 25.80 ± 0.63 | >80 | >3.10 | 630.30 ± 14.05 | >80 | >7.88 |
| T. hoaensis | 72.02 ± 0.88 | >80 | >1.11 | 397.20 ± 13.20 | >80 | >4.97 |
| D. loureiroi | 10.47 ± 0.01 | 55.67 ± 3.80 | 5.32 | 104.00 ± 0.01 | >80 | >1.30 |
| A. evecta | 57.57 ± 2.89 | >80 | >1.39 | 105.10 ± 3.94 | >80 | >1.31 |
| G. malaccensis | 5.33 ± 0.08 | >80 | >15.01 | 103.10 ± 11.23 | >80 | >1.29 |
| T. chantrieri | 65.45 ± 3.54 | >80 | >1.22 | 80.63 ± 2.51 | >80 | >1.01 |
| S. exigua | 224.40 ± 3.82 | 18.56 ± 0.56 | 0.08 | 173.20 ± 25.81 | >80 | >2.17 |
| C. podophylla | 312.50 ± 5.05 | >80 | >0.26 | 329.40 ± 5.05 | >80 | >4.12 |
| M. elengi | 49.80 ± 2.44 | >80 | >1.61 | 106.20 ± 7.63 | >80 | >1.33 |
| M. ferrea | 7.39 ± 0.89 | 11.44 ± 1.14 | 1.55 | 112.20 ± 8.05 | >80 | >1.40 |
| M. siamensis | 1.50 ± 0.03 | <5 | <3.33 | 82.29 ± 1.54 | >80 | >1.03 |
| N. nucifera | 157.90 ± 8.61 | >80 | >0.51 | 53.08 ± 1.43 | >80 | >0.66 |
| Kheaw Hom remedy | 14.16 ± 0.65 | >80 | >5.65 | 292.90 ± 15.15 | >80 | >3.66 |
| Artesunate | 1.28 ± 0.71 * | ND | ND | ND | ||
| Doxorubicin | ND | ND | 2.07 ± 0.13 | ND | ||
| Group | Dose (mg/kg) | % Parasitemia | % Suppression |
|---|---|---|---|
| Negative control | - | 64.22 ± 0.27 | - |
| Artesunate | 6 | 2.56 ± 0.83 | 96.02 ± 1.35 a,c,d,e |
| G. malaccensis extract (Treated groups) | 200 | 31.10 ± 0.21 | 51.57 ± 0.36 a,b,e |
| 400 | 28.94 ± 12.94 | 54.94 ± 0.58 a,b,e | |
| 600 | 25.34 ± 11.33 | 60.53 ± 0.47 a,b,c,d |
| Group | Dose (mg/kg) | Day 0 (g) | Day 4 (g) | Change (%) |
|---|---|---|---|---|
| Negative control | - | 36.56 ± 0.56 | 35.47 ± 0.49 | −3.82 ± 0.33 b,c,d,e |
| Artesunate | 6 | 35.95 ± 0.90 | 37.04 ± 0.97 | 6.42 ± 0.72 a,c,d,e |
| G. malaccensis extract (Treated groups) | 200 | 35.19 ± 0.29 | 34.56 ± 0.34 | −1.78 ± 0.68 a,b,d,e |
| 400 | 35.08 ± 0.23 | 32.58 ± 0.36 | −7.11 ± 0.67 a,b,c,e | |
| 600 | 34.60 ± 1.15 | 30.37 ± 1.04 | −12.21 ± 0.72 a,b,c,d |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chaniad, P.; Techarang, T.; Phuwajaroanpong, A.; Horata, N.; Septama, A.W.; Punsawad, C. Exploring Potential Antimalarial Candidate from Medicinal Plants of Kheaw Hom Remedy. Trop. Med. Infect. Dis. 2022, 7, 368. https://doi.org/10.3390/tropicalmed7110368
Chaniad P, Techarang T, Phuwajaroanpong A, Horata N, Septama AW, Punsawad C. Exploring Potential Antimalarial Candidate from Medicinal Plants of Kheaw Hom Remedy. Tropical Medicine and Infectious Disease. 2022; 7(11):368. https://doi.org/10.3390/tropicalmed7110368
Chicago/Turabian StyleChaniad, Prapaporn, Tachpon Techarang, Arisara Phuwajaroanpong, Natharinee Horata, Abdi Wira Septama, and Chuchard Punsawad. 2022. "Exploring Potential Antimalarial Candidate from Medicinal Plants of Kheaw Hom Remedy" Tropical Medicine and Infectious Disease 7, no. 11: 368. https://doi.org/10.3390/tropicalmed7110368
APA StyleChaniad, P., Techarang, T., Phuwajaroanpong, A., Horata, N., Septama, A. W., & Punsawad, C. (2022). Exploring Potential Antimalarial Candidate from Medicinal Plants of Kheaw Hom Remedy. Tropical Medicine and Infectious Disease, 7(11), 368. https://doi.org/10.3390/tropicalmed7110368






