Tuberculosis in Adolescents and Young Adults: Emerging Data on TB Transmission and Prevention among Vulnerable Young People
Abstract
1. Introduction
2. Methods
3. Epidemiology of Adolescent TB
3.1. Prevalence, Gender, and Risk Factors for AYA TB
3.2. TB Preventive Treatment Regimens for AYA
3.3. TB Disease Treatment for AYA
3.4. Outcomes for AYA
3.5. Vulnerable AYA Populations
3.5.1. Migrant AYA
3.5.2. AYA Living with HIV
3.5.3. AYA Experiencing Homelessness, Substance Use, or Incarceration
4. Settings of Adolescent TB Transmission
4.1. Household Exposures
4.2. School Exposures
4.3. Exposures on Public Transit
4.4. Other Community Exposures
4.5. Military Exposures
5. Identifying AYA with TB
5.1. Setting-Specific Opportunities in Case-Finding and Prevention
5.1.1. Households
5.1.2. Schools
5.1.3. Other Settings
5.2. Opportunities in Case-Finding and Prevention for AYA at Increased Risk
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Snow, K.J.; Sismanidis, C.; Denholm, J.; Sawyer, S.M.; Graham, S.M. The Incidence of Tuberculosis among Adolescents and Young Adults: A Global Estimate. Eur. Respir. J. 2018, 51, 1702352. [Google Scholar] [CrossRef]
- Seddon, J.A.; Chiang, S.S.; Esmail, H.; Coussens, A.K. The Wonder Years: What Can Primary School Children Teach Us About Immunity to Mycobacterium Tuberculosis? Front. Immunol. 2018, 9, 2946. [Google Scholar] [CrossRef]
- Snow, K.J.; Cruz, A.T.; Seddon, J.A.; Ferrand, R.A.; Chiang, S.S.; Hughes, J.A.; Kampmann, B.; Graham, S.M.; Dodd, P.J.; Houben, R.M.; et al. Adolescent Tuberculosis. Lancet Child. Adolesc Health 2020, 4, 68–79. [Google Scholar] [CrossRef]
- World Health Organization. Making Health Services Adolescent Friendly: Developing National Quality Standards for Adolescent-Friendly Health Services; World Health Organization: Geneva, Switzerland, 2012. [Google Scholar]
- World Health Organization. Global Tuberculosis Report 2020; World Health Organization: Geneva, Switzerland, 2020. [Google Scholar]
- Middelkoop, K.; Bekker, L.-G.; Liang, H.; Aquino, L.D.H.; Sebastian, E.; Myer, L.; Wood, R. Force of Tuberculosis Infection among Adolescents in a High HIV and TB Prevalence Community: A Cross-Sectional Observation Study. BMC Infect. Dis. 2011, 11, 156. [Google Scholar] [CrossRef]
- Marais, B.J.; Gie, R.P.; Schaaf, H.S.; Hesseling, A.C.; Obihara, C.C.; Nelson, L.J.; Enarson, D.A.; Donald, P.R.; Beyers, N. The Clinical Epidemiology of Childhood Pulmonary Tuberculosis: A Critical Review of Literature from the Pre-Chemotherapy Era. Int. J. Tuberc Lung Dis. 2004, 8, 278–285. [Google Scholar]
- Houben, R.M.G.J.; Dodd, P.J. The Global Burden of Latent Tuberculosis Infection: A Re-Estimation Using Mathematical Modelling. PLoS Med. 2016, 13, e1002152. [Google Scholar] [CrossRef] [PubMed]
- Cruz, A.T.; Hwang, K.M.; Birnbaum, G.D.; Starke, J.R. Adolescents with Tuberculosis: A Review of 145 Cases. Pediatr. Infect. Dis. J. 2013, 32, 937–941. [Google Scholar] [CrossRef]
- Chiang, S.S.; Dolynska, M.; Rybak, N.R.; Cruz, A.T.; Aibana, O.; Sheremeta, Y.; Petrenko, V.; Mamotenko, A.; Terleieva, I.; Horsburgh, C.R.; et al. Clinical Manifestations and Epidemiology of Adolescent Tuberculosis in Ukraine. ERJ Open Res. 2020, 6, 00308-2020. [Google Scholar] [CrossRef] [PubMed]
- Cogo, H.; Caseris, M.; Lachaume, N.; Cointe, A.; Faye, A.; Pommelet, V. Tuberculosis in Children Hospitalized in a Low-Burden Country: Description and Risk Factors of Severe Disease. Pediatr Infect. Dis. J. 2021, 40, 199–204. [Google Scholar] [CrossRef]
- Osman, M.; du Preez, K.; Seddon, J.A.; Claassens, M.M.; Dunbar, R.; Dlamini, S.S.; Welte, A.; Naidoo, P.; Hesseling, A.C. Mortality in South African Children and Adolescents Routinely Treated for Tuberculosis. Pediatrics 2021, 147, e2020032490. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Ren, Z.-H.; Qiang, H.; Wu, J.; Shen, M.; Zhang, L.; Lyu, J. Trends in the Incidence of Diabetes Mellitus: Results from the Global Burden of Disease Study 2017 and Implications for Diabetes Mellitus Prevention. BMC Public Health 2020, 20, 1415. [Google Scholar] [CrossRef]
- International Diabetes Federation. International Diabetes Federation Diabetes Atlas, 9th ed. Available online: https://diabetesatlas.org/en/ (accessed on 20 July 2021).
- Lynch, J.L.; Barrientos-Pérez, M.; Hafez, M.; Jalaludin, M.Y.; Kovarenko, M.; Rao, P.V.; Weghuber, D. Country-Specific Prevalence and Incidence of Youth-Onset Type 2 Diabetes: A Narrative Literature Review. Ann. Nutr. Metab. 2020, 76, 289–296. [Google Scholar] [CrossRef]
- Derges, J.; Kidger, J.; Fox, F.; Campbell, R.; Kaner, E.; Hickman, M. Alcohol Screening and Brief Interventions for Adults and Young People in Health and Community-Based Settings: A Qualitative Systematic Literature Review. BMC Public Health 2017, 17, 562. [Google Scholar] [CrossRef] [PubMed]
- Getahun, H.; Matteelli, A.; Chaisson, R.E.; Raviglione, M. Latent Mycobacterium Tuberculosis Infection. N Engl. J. Med. 2015, 372, 2127–2135. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO Consolidated Guidelines on Tuberculosis: Tuberculosis Preventive Treatment; World Health Organization: Geneva, Switzerland, 2020. [Google Scholar]
- Hatzenbuehler, L.A.; Starke, J.R.; Graviss, E.A.; Smith, E.O.; Cruz, A.T. School-Based Study to Identify and Treat Adolescent Students at Risk for Tuberculosis Infection. Pediatr. Infect. Dis. J. 2016, 35, 733–738. [Google Scholar] [CrossRef]
- Assefa, Y.; Assefa, Y.; Woldeyohannes, S.; Hamada, Y.; Getahun, H. 3-Month Daily Rifampicin and Isoniazid Compared to 6- or 9-Month Isoniazid for Treating Latent Tuberculosis Infection in Children and Adolescents Less than 15 Years of Age: An Updated Systematic Review. Eur. Respir. J. 2018, 52, 1800395. [Google Scholar] [CrossRef] [PubMed]
- Diallo, T.; Adjobimey, M.; Ruslami, R.; Trajman, A.; Sow, O.; Obeng Baah, J.; Marks, G.B.; Long, R.; Elwood, K.; Zielinski, D.; et al. Safety and Side Effects of Rifampin versus Isoniazid in Children. N. Engl. J. Med. 2018, 379, 454–463. [Google Scholar] [CrossRef]
- Enane, L.A.; Eby, J.; Arscott-Mills, T.; Argabright, S.; Caiphus, C.; Kgwaadira, B.; Steenhoff, A.P.; Lowenthal, E.D. TB and TB-HIV Care for Adolescents and Young Adults. Int. J. Tuberc. Lung Dis. 2020, 24, 240–249. [Google Scholar] [CrossRef] [PubMed]
- Laycock, K.M.; Eby, J.; Arscott-Mills, T.; Argabright, S.; Caiphus, C.; Kgwaadira, B.; Lowenthal, E.D.; Steenhoff, A.P.; Enane, L.A. Towards Quality Adolescent-Friendly Services in TB Care. Int. J. Tuberc. Lung Dis. 2021, 25, 579–583. [Google Scholar] [CrossRef] [PubMed]
- Sawyer, S.M.; Afifi, R.A.; Bearinger, L.H.; Blakemore, S.-J.; Dick, B.; Ezeh, A.C.; Patton, G.C. Adolescence: A Foundation for Future Health. Lancet 2012, 379, 1630–1640. [Google Scholar] [CrossRef]
- Van der Westhuizen, H.-M.; Dramowski, A. When Students Become Patients: TB Disease among Medical Undergraduates in Cape Town, South Africa. S. Afr. Med. J. 2017, 107, 475–479. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhou, L.; Liu, Z.-W.; Chai, C.-L.; Wang, X.-M.; Jiang, J.-M.; Chen, S.-H. Multidrug-Resistant Tuberculosis Transmission among Middle School Students in Zhejiang Province, China. Infect. Dis. Poverty 2020, 9, 57. [Google Scholar] [CrossRef] [PubMed]
- Enane, L.A.; Lowenthal, E.D.; Arscott-Mills, T.; Matlhare, M.; Smallcomb, L.S.; Kgwaadira, B.; Coffin, S.E.; Steenhoff, A.P. Loss to Follow-up among Adolescents with Tuberculosis in Gaborone, Botswana. Int. J. Tuberc. Lung Dis. 2016, 20, 1320–1325. [Google Scholar] [CrossRef]
- Berry, K.M.; Rodriguez, C.A.; Berhanu, R.H.; Ismail, N.; Mvusi, L.; Long, L.; Evans, D. Treatment Outcomes among Children, Adolescents, and Adults on Treatment for Tuberculosis in Two Metropolitan Municipalities in Gauteng Province, South Africa. BMC Public Health 2019, 19, 973. [Google Scholar] [CrossRef]
- Snow, K.; Hesseling, A.C.; Naidoo, P.; Graham, S.M.; Denholm, J.; du Preez, K. Tuberculosis in Adolescents and Young Adults: Epidemiology and Treatment Outcomes in the Western Cape. Int. J. Tuberc. Lung Dis. 2017, 21, 651–657. [Google Scholar] [CrossRef] [PubMed]
- Mulongeni, P.; Hermans, S.; Caldwell, J.; Bekker, L.-G.; Wood, R.; Kaplan, R. HIV Prevalence and Determinants of Loss-to-Follow-up in Adolescents and Young Adults with Tuberculosis in Cape Town. PLoS ONE 2019, 14, e0210937. [Google Scholar] [CrossRef]
- Enane, L.A.; Lowenthal, E.D.; Arscott-Mills, T.; Eby, J.; Caiphus, C.; Kgwaadira, B.; Coffin, S.E.; Steenhoff, A.P. Investigating Outcomes of Adolescents and Young Adults (10–24 Years of Age) Lost to Follow-up from Tuberculosis Treatment in Gaborone, Botswana. Pediatr. Infect. Dis. J. 2019, 38, e271–e274. [Google Scholar] [CrossRef]
- Lönnroth, K.; Mor, Z.; Erkens, C.; Bruchfeld, J.; Nathavitharana, R.R.; van der Werf, M.J.; Lange, C. Tuberculosis in Migrants in Low-Incidence Countries: Epidemiology and Intervention Entry Points. Int. J. Tuberc. Lung Dis. 2017, 21, 624–637. [Google Scholar] [CrossRef] [PubMed]
- Wild, V.; Jaff, D.; Shah, N.S.; Frick, M. Tuberculosis, Human Rights and Ethics Considerations along the Route of a Highly Vulnerable Migrant from Sub-Saharan Africa to Europe. Int. J. Tuberc. Lung Dis. 2017, 21, 1075–1085. [Google Scholar] [CrossRef]
- Thee, S.; Krüger, R.; von Bernuth, H.; Meisel, C.; Kölsch, U.; Kirchberger, V.; Feiterna-Sperling, C. Screening and Treatment for Tuberculosis in a Cohort of Unaccompanied Minor Refugees in Berlin, Germany. PLoS ONE 2019, 14, e0216234. [Google Scholar] [CrossRef]
- Ahmad, B.B.; Kristensen, K.L.; Glenthoej, J.P.; Poulsen, A.; Bryld, A.-G.; Huber, F.G.; Andersen, E.M.; Ravn, P. Latent Tuberculosis Infection among Minor Asylum Seekers in Denmark. Eur. Respir. J. 2020, 55, 1901688. [Google Scholar] [CrossRef]
- Walker, T.M.; Merker, M.; Knoblauch, A.M.; Helbling, P.; Schoch, O.D.; van der Werf, M.J.; Kranzer, K.; Fiebig, L.; Kröger, S.; Haas, W.; et al. MDR-TB Cluster Consortium. A Cluster of Multidrug-Resistant Mycobacterium Tuberculosis among Patients Arriving in Europe from the Horn of Africa: A Molecular Epidemiological Study. Lancet Infect. Dis. 2018, 18, 431–440. [Google Scholar] [CrossRef]
- Dhavan, P.; Dias, H.M.; Creswell, J.; Weil, D. An Overview of Tuberculosis and Migration. Int. J. Tuberc. Lung Dis. 2017, 21, 610–623. [Google Scholar] [CrossRef]
- Zenner, D. Crisis-Affected Populations and Tuberculosis. Microbiol. Spectr. 2017, 5. [Google Scholar] [CrossRef] [PubMed]
- Ncayiyana, J.R.; Bassett, J.; West, N.; Westreich, D.; Musenge, E.; Emch, M.; Pettifor, A.; Hanrahan, C.F.; Schwartz, S.R.; Sanne, I.; et al. Prevalence of Latent Tuberculosis Infection and Predictive Factors in an Urban Informal Settlement in Johannesburg, South Africa: A Cross-Sectional Study. BMC Infect. Dis. 2016, 16, 661. [Google Scholar] [CrossRef] [PubMed]
- Donnan, E.J.; Coulter, C.; Simpson, G.; Clark, J.; Nourse, C. Paediatric Tuberculosis in Queensland, Australia: Overrepresentation of Cross-Border and Indigenous Children. Int. J. Tuberc. Lung Dis. 2017, 21, 263–269. [Google Scholar] [CrossRef]
- Moya, E.M.; Chávez-Baray, S.M.; Wood, W.W.; Martinez, O. Nuestra Casa: An Advocacy Initiative to Reduce Inequalities and Tuberculosis along the US-Mexico Border. Int. Public Health J. 2016, 8, 107–119. [Google Scholar]
- Foster, J.; McBryde, E.; Taune, M.; Peniyamina, D. Cross-Border Tuberculosis: Opportunities, Challenges and Change. Int. J. Tuberc. Lung Dis. 2018, 22, 1107–1108. [Google Scholar] [CrossRef]
- Joint United Nations Programme on HIV/AIDS. Young People and HIV; UNAIDS: Geneva, Switzerland, 2021. [Google Scholar]
- Armstrong, A.; Nagata, J.M.; Vicari, M.; Irvine, C.; Cluver, L.; Sohn, A.H.; Ferguson, J.; Caswell, G.; Njenga, L.W.; Oliveras, C.; et al. A Global Research Agenda for Adolescents Living With HIV. J. Acquir. Immune Defic. Syndr. 2018, 78 (Suppl. 1), S16–S21. [Google Scholar] [CrossRef]
- Carlucci, J.G.; Blevins Peratikos, M.; Kipp, A.M.; Lindegren, M.L.; Du, Q.T.; Renner, L.; Reubenson, G.; Ssali, J.; Yotebieng, M.; Mandalakas, A.M.; et al. For the International Epidemiology Databases to Evaluate AIDS (IeDEA) Network. Tuberculosis Treatment Outcomes Among HIV/TB-Coinfected Children in the International Epidemiology Databases to Evaluate AIDS (IeDEA) Network. JAIDS J. Acquir. Immune Defic. Syndr. 2017, 75, 156–163. [Google Scholar] [CrossRef]
- Mandalakas, A.M.; Kay, A.W.; Bacha, J.M.; Devezin, T.; Golin, R.; Simon, K.R.; Dhillon, D.; Dlamini, S.; DiNardo, A.; Matshaba, M.; et al. Tuberculosis among Children and Adolescents at HIV Treatment Centers in Sub-Saharan Africa. Emerg. Infect. Dis. 2020, 26, 2933–2943. [Google Scholar] [CrossRef]
- Du Preez, K.; Osman, M.; Seddon, J.A.; Naidoo, P.; Schaaf, H.S.; Munch, Z.; Dunbar, R.; Mvusi, L.; Dlamini, S.S.; Hesseling, A.C. The Impact of the Evolving HIV Response on the Epidemiology of Tuberculosis in South African Children and Adolescents. Clin. Infect. Dis. 2021, 2021, ciab095. [Google Scholar] [CrossRef]
- Frigati, L.J.; Wilkinson, K.A.; le Roux, S.; Brown, K.; Ruzive, S.; Githinji, L.; Petersen, W.; Belard, S.; Cotton, M.F.; Myer, L.; et al. Tuberculosis Infection and Disease in South African Adolescents with Perinatally Acquired HIV on Antiretroviral Therapy: A Cohort Study. J. Int. AIDS Soc. 2021, 24, e25671. [Google Scholar] [CrossRef]
- Osman, M.; Lee, K.; Du Preez, K.; Dunbar, R.; Hesseling, A.C.; Seddon, J.A. Excellent Treatment Outcomes in Children Treated for Tuberculosis Under Routine Operational Conditions in Cape Town, South Africa. Clin. Infect. Dis 2017, 65, 1444–1452. [Google Scholar] [CrossRef]
- Reif, L.K.; Rivera, V.; Bertrand, R.; Rouzier, V.; Kutscher, E.; Walsh, K.; Charles, B.; Pape, J.W.; Fitzgerald, D.W.; Koenig, S.P.; et al. Outcomes across the Tuberculosis Care Continuum among Adolescents in Haiti. Public Health Action 2018, 8, 103–109. [Google Scholar] [CrossRef]
- De Oliveira, M.C.B.; Sant’Anna, C.C.; Raggio Luiz, R.; Kritski, A.L. Unfavorable Outcomes in Tuberculosis: Multidimensional Factors among Adolescents in Rio de Janeiro, Brazil. Am. J. Trop Med. Hyg. 2020, 103, 2492–2500. [Google Scholar] [CrossRef]
- Hino, P.; Yamamoto, T.T.; Bastos, S.H.; Beraldo, A.A.; de Figueiredo, T.M.R.M.; Bertolozzi, M.R. Tuberculosis in the Street Population: A Systematic Review. Rev. Esc. Enferm. USP 2021, 55, e03688. [Google Scholar] [CrossRef] [PubMed]
- Imtiaz, S.; Shield, K.D.; Roerecke, M.; Samokhvalov, A.V.; Lönnroth, K.; Rehm, J. Alcohol Consumption as a Risk Factor for Tuberculosis: Meta-Analyses and Burden of Disease. Eur. Respir. J. 2017, 50, 1700216. [Google Scholar] [CrossRef]
- Wang, E.Y.; Arrazola, R.A.; Mathema, B.; Ahluwalia, I.B.; Mase, S.R. The Impact of Smoking on Tuberculosis Treatment Outcomes: A Meta-Analysis. Int. J. Tuberc. Lung Dis. 2020, 24, 170–175. [Google Scholar] [CrossRef] [PubMed]
- Kinner, S.A.; Snow, K.; Wirtz, A.L.; Altice, F.L.; Beyrer, C.; Dolan, K. Age-Specific Global Prevalence of Hepatitis B, Hepatitis C, HIV, and Tuberculosis Among Incarcerated People: A Systematic Review. J. Adolesc. Health 2018, 62, S18–S26. [Google Scholar] [CrossRef] [PubMed]
- Szkwarko, D.; Mercer, T.; Kimani, S.; Braitstein, P.; Buziba, N.; Carter, E.J. Implementing Intensified Tuberculosis Case-Finding among Street-Connected Youth and Young Adults in Kenya. Public Health Action 2016, 6, 142–146. [Google Scholar] [CrossRef]
- Kerr, E.M.; Vonnahme, L.A.; Goswami, N.D. Impact of Targeted Local Interventions on Tuberculosis Awareness and Screening Among Persons Experiencing Homelessness During a Large Tuberculosis Outbreak in Atlanta, Georgia, 2015–2016. Public Health Rep. 2020, 135 (Suppl. 1), 90S–99S. [Google Scholar] [CrossRef]
- Stevens, H.; Ximenes, R.A.; Dantas, O.M.; Rodrigues, L.C. Risk Factors for Tuberculosis in Older Children and Adolescents: A Matched Case-Control Study in Recife, Brazil. Emerg. Themes Epidemiol. 2014, 11, 20. [Google Scholar] [CrossRef] [PubMed]
- Ganmaa, D.; Khudyakov, P.; Buyanjargal, U.; Baigal, D.; Baatar, M.; Enkhamgalan, N.; Erdenebaatar, S.; Ochirbat, B.; Burneebaatar, B.; Purevdorj, E.; et al. Risk Factors for Active Tuberculosis in 938 QuantiFERON-Positive Schoolchildren in Mongolia: A Community-Based Cross-Sectional Study. BMC Infect. Dis. 2019, 19, 532. [Google Scholar] [CrossRef] [PubMed]
- Patra, J.; Bhatia, M.; Suraweera, W.; Morris, S.K.; Patra, C.; Gupta, P.C.; Jha, P. Exposure to Second-Hand Smoke and the Risk of Tuberculosis in Children and Adults: A Systematic Review and Meta-Analysis of 18 Observational Studies. PLoS Med. 2015, 12, e1001835; discussion e1001835. [Google Scholar] [CrossRef]
- Soboka, M.; Tolessa, O.; Tesfaye, M.; Adorjan, K.; Krahl, W.; Tesfaye, E.; Yitayih, Y.; Strobl, R.; Grill, E. Magnitude and Predictors of Khat Use among Patients with Tuberculosis in Southwest Ethiopia: A Longitudinal Study. PLoS ONE 2020, 15, e0236154. [Google Scholar] [CrossRef] [PubMed]
- Wood, R.; Racow, K.; Bekker, L.-G.; Morrow, C.; Middelkoop, K.; Mark, D.; Lawn, S.D. Indoor Social Networks in a South African Township: Potential Contribution of Location to Tuberculosis Transmission. PLoS ONE 2012, 7, e39246. [Google Scholar] [CrossRef]
- Patterson, B.; Morrow, C.D.; Kohls, D.; Deignan, C.; Ginsburg, S.; Wood, R. Mapping Sites of High TB Transmission Risk: Integrating the Shared Air and Social Behaviour of TB Cases and Adolescents in a South African Township. Sci. Total Environ. 2017, 583, 97–103. [Google Scholar] [CrossRef]
- Yates, T.A.; Khan, P.Y.; Knight, G.M.; Taylor, J.G.; McHugh, T.D.; Lipman, M.; White, R.G.; Cohen, T.; Cobelens, F.G.; Wood, R.; et al. The Transmission of Mycobacterium Tuberculosis in High Burden Settings. Lancet Infect. Dis. 2016, 16, 227–238. [Google Scholar] [CrossRef]
- Mathema, B.; Andrews, J.R.; Cohen, T.; Borgdorff, M.W.; Behr, M.; Glynn, J.R.; Rustomjee, R.; Silk, B.J.; Wood, R. Drivers of Tuberculosis Transmission. J. Infect. Dis. 2017, 216 (Suppl. 6), S644–S653. [Google Scholar] [CrossRef]
- Martinez, L.; Shen, Y.; Mupere, E.; Kizza, A.; Hill, P.C.; Whalen, C.C. Transmission of Mycobacterium Tuberculosis in Households and the Community: A Systematic Review and Meta-Analysis. Am. J. Epidemiol. 2017, 185, 1327–1339. [Google Scholar] [CrossRef]
- Zelner, J.L.; Murray, M.B.; Becerra, M.C.; Galea, J.; Lecca, L.; Calderon, R.; Yataco, R.; Contreras, C.; Zhang, Z.; Grenfell, B.T.; et al. Age-Specific Risks of Tuberculosis Infection from Household and Community Exposures and Opportunities for Interventions in a High-Burden Setting. Am. J. Epidemiol. 2014, 180, 853–861. [Google Scholar] [CrossRef]
- Middelkoop, K.; Bekker, L.-G.; Morrow, C.; Lee, N.; Wood, R. Decreasing Household Contribution to TB Transmission with Age: A Retrospective Geographic Analysis of Young People in a South African Township. BMC Infect. Dis. 2014, 14, 221. [Google Scholar] [CrossRef] [PubMed]
- Marquez, C.; Atukunda, M.; Balzer, L.B.; Chamie, G.; Kironde, J.; Ssemmondo, E.; Ruel, T.D.; Mwangwa, F.; Tram, K.H.; Clark, T.D.; et al. The Age-Specific Burden and Household and School-Based Predictors of Child and Adolescent Tuberculosis Infection in Rural Uganda. PLoS ONE 2020, 15, e0228102. [Google Scholar] [CrossRef]
- Dolla, C.K.; Padmapriyadarsini, C.; Thiruvengadam, K.; Lokhande, R.; Kinikar, A.; Paradkar, M.; Bm, S.; Murali, L.; Gupte, A.; Gaikwad, S.; et al. Age-Specific Prevalence of TB Infection among Household Contacts of Pulmonary TB: Is It Time for TB Preventive Therapy? Trans. R Soc. Trop. Med. Hyg. 2019, 113, 632–640. [Google Scholar] [CrossRef]
- Richardson, E.T.; Morrow, C.D.; Kalil, D.B.; Ginsberg, S.; Bekker, L.-G.; Wood, R. Shared Air: A Renewed Focus on Ventilation for the Prevention of Tuberculosis Transmission. PLoS ONE 2014, 9, e96334. [Google Scholar] [CrossRef]
- Andrews, J.R.; Morrow, C.; Walensky, R.P.; Wood, R. Integrating Social Contact and Environmental Data in Evaluating Tuberculosis Transmission in a South African Township. J. Infect. Dis. 2014, 210, 597–603. [Google Scholar] [CrossRef]
- Moges, B.; Amare, B.; Yismaw, G.; Workineh, M.; Alemu, S.; Mekonnen, D.; Diro, E.; Tesema, B.; Kassu, A. Prevalence of Tuberculosis and Treatment Outcome among University Students in Northwest Ethiopia: A Retrospective Study. BMC Public Health 2015, 15, 15. [Google Scholar] [CrossRef]
- Mekonnen, A.; Petros, B. Burden of Tuberculosis Among Students in Two Ethiopian Universities. Ethiop Med. J. 2016, 54, 189–196. [Google Scholar]
- Steppacher, A.; Scheer, I.; Relly, C.; Začek, B.; Turk, A.; Altpeter, E.; Berger, C.; Nadal, D. Unrecognized Pediatric Adult-Type Tuberculosis Puts School Contacts at Risk. Pediatr. Infect. Dis. J. 2014, 33, 325–328. [Google Scholar] [CrossRef][Green Version]
- Piccini, P.; Venturini, E.; Bianchi, L.; Baretti, S.; Filidei, P.; Paliaga, L.; Mazzoli, F.; Chiappini, E.; de Martino, M.; Galli, L. The Risk of Mycobacterium Tuberculosis Transmission from Pediatric Index Cases to School Pupils. Pediatr. Infect. Dis. J. 2017, 36, 525–528. [Google Scholar] [CrossRef]
- Schepisi, M.S.; Motta, I.; Dore, S.; Costa, C.; Sotgiu, G.; Girardi, E. Tuberculosis Transmission among Children and Adolescents in Schools and Other Congregate Settings: A Systematic Review. New Microbiol. 2019, 41, 282–290. [Google Scholar]
- World Health Organization. Tuberculosis Outbreaks in Schools: Experiences from the Western Pacific Region; WHO Regional Office for the Western Pacific: Manila, Philippines, 2021. [Google Scholar]
- You, N.N.; Zhu, L.M.; Li, G.L.; Martinez, L.; Lu, W.; Liu, Q.; Yang, H.T. A Tuberculosis School Outbreak in China, 2018: Reaching an Often Overlooked Adolescent Population. Epidemiol. Infect. 2019, 147, e303. [Google Scholar] [CrossRef][Green Version]
- Hou, J.; Pang, Y.; Yang, X.; Chen, T.; Yang, H.; Yang, R.; Chen, L.; Xu, L. Outbreak of Mycobacterium Tuberculosis Beijing Strain in a High School in Yunnan, China. Am. J. Trop. Med. Hyg. 2020, 102, 728–730. [Google Scholar] [CrossRef]
- Anaraki, S.; Bell, A.J.; Perkins, S.; Murphy, S.; Dart, S.; Anderson, C. Expected Background Rates of Latent TB Infection in London Inner City Schools: Lessons from a TB Contact Investigation Exercise in a Secondary School. Epidemiol. Infect. 2018, 146, 2102–2106. [Google Scholar] [CrossRef]
- Stosic, M.B.; Plavsa, D.; Mavroeidi, N.; Jovanovic, D.; Vucinic, V.; Stevanovic, G.; Sagic, L.; Spahic, S.; Rakic, U.; Grgurevic, A. Tuberculosis Outbreak among High School Students in Novi Pazar, Serbia 2016: A Retrospective-Cohort Study. J. Infect. Dev. Ctries 2019, 13, 101–110. [Google Scholar] [CrossRef]
- Tasaka, M.; Shimamura, T.; Iwata, M.; Toyozawa, T.; Ota, M. A Tuberculosis Contact Investigation Involving a Large Number of Contacts Tested with Interferon-Gamma Release Assay at a Nursing School: Kanagawa, Japan, 2012. Western Pac. Surveill Response J. 2018, 9, 4–8. [Google Scholar] [CrossRef]
- Kim, Y.; Kim, B.K.; Choi, H.J.; Ryu, S.W.; Kim, E.S.; Chang, Y.S.; Kim, H.J.; Cha, J.H.; Kim, J.H.; Shin, C.; et al. Lessons Learned from Continued TB Outbreaks in a High School. PLoS ONE 2017, 12, e0188076. [Google Scholar] [CrossRef]
- Wu, X.; Pang, Y.; Song, Y.; Dong, W.; Zhang, T.; Wen, S.; Huang, H.; Gao, M. Implications of a School Outbreak of Multidrug-Resistant Tuberculosis in Northern China. Epidemiol. Infect. 2018, 146, 584–588. [Google Scholar] [CrossRef]
- Pan, D.; Lan, R.; Graviss, E.A.; Lin, D.; Liang, D.; McNeil, E.; Lin, M.; Chongsuvivatwong, V. Adolescent Tuberculosis Associated with Tuberculosis Exposure in Classrooms and Dorm Rooms in Guangxi, China. Int. J. Infect. Dis. 2019, 78, 8–14. [Google Scholar] [CrossRef]
- Dorjee, K.; Topgyal, S.; Dorjee, C.; Tsundue, T.; Namdol, T.; Tsewang, T.; Nangsel, T.; Lhadon, D.; Choetso, T.; Dawa, T.; et al. High Prevalence of Active and Latent Tuberculosis in Children and Adolescents in Tibetan Schools in India: The Zero TB Kids Initiative in Tibetan Refugee Children. Clin. Infect. Dis. 2019, 69, 760–768. [Google Scholar] [CrossRef]
- Popovici, O.; Monk, P.; Chemtob, D.; Chiotan, D.; Freidlin, P.J.; Groenheit, R.; Haanperä, M.; Homorodean, D.; Mansjö, M.; Robinson, E.; et al. Cross-Border Outbreak of Extensively Drug-Resistant Tuberculosis Linked to a University in Romania. Epidemiol. Infect. 2018, 146, 824–831. [Google Scholar] [CrossRef]
- Chou, K.H.; Kam, K.M.; Ieong, S.K.; Yip, C.W.; Ip, P.K.; Yew, W.W.; Leung, C.C.; Wong, N.S.; Lau, S.M.; Lee, S.S. Concurrent Outbreaks of Tuberculosis in a School and the Wider Community in Macau. J. Pediatric Infect. Dis. Soc. 2015, 4, 359–362. [Google Scholar] [CrossRef][Green Version]
- Bunyasi, E.W.; Geldenhuys, H.; Mulenga, H.; Shenje, J.; Luabeya, A.K.K.; Tameris, M.; Nemes, E.; Mahomed, H.; Rozot, V.; Wood, R.; et al. Temporal Trends in the Prevalence of Mycobacterium Tuberculosis Infection in South African Adolescents. Int. J. Tuberc. Lung Dis. 2019, 23, 571–578. [Google Scholar] [CrossRef]
- Pan, D.; Lin, M.; Lan, R.; Graviss, E.A.; Lin, D.; Liang, D.; Long, X.; Qin, H.; Huang, L.; Huang, M.; et al. Tuberculosis Transmission in Households and Classrooms of Adolescent Cases Compared to the Community in China. Int. J. Environ. Res. Public Health 2018, 15, 2803. [Google Scholar] [CrossRef]
- Waako, J.; Verver, S.; Wajja, A.; Ssengooba, W.; Joloba, M.L.; Colebunders, R.; Musoke, P.; Mayanja-Kizza, H. Burden of Tuberculosis Disease among Adolescents in a Rural Cohort in Eastern Uganda. BMC Infect. Dis. 2013, 13, 349. [Google Scholar] [CrossRef]
- Mumpe-Mwanja, D.; Verver, S.; Yeka, A.; Etwom, A.; Waako, J.; Ssengooba, W.; Matovu, J.K.; Wanyenze, R.K.; Musoke, P.; Mayanja-Kizza, H. Prevalence and Risk Factors of Latent Tuberculosis among Adolescents in Rural Eastern Uganda. Afr. Health Sci. 2015, 15, 851–860. [Google Scholar] [CrossRef]
- Nduba, V.; Van’t Hoog, A.H.; de Bruijn, A.; Mitchell, E.M.H.; Laserson, K.; Borgdorff, M. Estimating the Annual Risk of Infection with Mycobacterium Tuberculosis among Adolescents in Western Kenya in Preparation for TB Vaccine Trials. BMC Infect. Dis. 2019, 19, 682. [Google Scholar] [CrossRef]
- Zamudio, C.; Krapp, F.; Choi, H.W.; Shah, L.; Ciampi, A.; Gotuzzo, E.; Heymann, J.; Seas, C.; Brewer, T.F. Public Transportation and Tuberculosis Transmission in a High Incidence Setting. PLoS ONE 2015, 10, e0115230. [Google Scholar] [CrossRef]
- Edelson, P.J.; Phypers, M. TB Transmission on Public Transportation: A Review of Published Studies and Recommendations for Contact Tracing. Travel. Med. Infect. Dis. 2011, 9, 27–31. [Google Scholar] [CrossRef]
- Hella, J.; Morrow, C.; Mhimbira, F.; Ginsberg, S.; Chitnis, N.; Gagneux, S.; Mutayoba, B.; Wood, R.; Fenner, L. Tuberculosis Transmission in Public Locations in Tanzania: A Novel Approach to Studying Airborne Disease Transmission. J. Infect. 2017, 75, 191–197. [Google Scholar] [CrossRef]
- Chee, C.B.E.; Gan, S.-H.; Ong, R.T.; Sng, L.-H.; Wong, C.W.; Cutter, J.; Gong, M.; Seah, H.-M.; Hsu, L.Y.; Solhan, S.; et al. Multidrug-Resistant Tuberculosis Outbreak in Gaming Centers, Singapore, 2012. Emerg. Infect. Dis. 2015, 21, 179–180. [Google Scholar] [CrossRef]
- Black, A.T.; Hamblion, E.L.; Buttivant, H.; Anderson, S.R.; Stone, M.; Casali, N.; Drobniewski, F.; Nwoguh, F.; Marshall, B.G.; Booth, L. Tracking and Responding to an Outbreak of Tuberculosis Using MIRU-VNTR Genotyping and Whole Genome Sequencing as Epidemiological Tools. J. Public Health 2018, 40, e66–e73. [Google Scholar] [CrossRef]
- Mancuso, J.D.; Aaron, C.L. Tuberculosis Trends in the U.S. Armed Forces, Active Component, 1998–2012. MSMR 2013, 20, 4–8. [Google Scholar]
- Kumar, H.K.V.S.; Patnaik, S.K. Incidence of Mycobacterial Disorders in Indian Adult Male Service Population Followed for over Two Decades. J. Infect. Public Health 2018, 11, 581–583. [Google Scholar] [CrossRef]
- Mancuso, J.D. Tuberculosis Screening and Control in the US Military in War and Peace. Am. J. Public Health 2017, 107, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, J.L.; Sanchez, J.L.; Cooper, M.J.; Hiser, M.J.; Mancuso, J.D. Tuberculosis as a Force Health Protection Threat to the United States Military. Mil. Med. 2015, 180, 276–284. [Google Scholar] [CrossRef] [PubMed]
- German, V.; Giannakos, G.; Kopterides, P.; Falagas, M.E. Prevalence and Predictors of Tuberculin Skin Positivity in Hellenic Army Recruits. BMC Infect. Dis. 2006, 6, 102. [Google Scholar] [CrossRef]
- Mancuso, J.D.; Geurts, M. Challenges in Obtaining Estimates of the Risk of Tuberculosis Infection During Overseas Deployment. Am. J. Trop. Med. Hyg. 2015, 93, 1172–1178. [Google Scholar] [CrossRef] [PubMed]
- O’Shea, M.K.; Wilson, D. Tuberculosis and the Military. J. R Army Med. Corps 2013, 159, 190–199. [Google Scholar] [CrossRef]
- Aksenova, V.A.; Vasilyeva, I.A.; Kasaeva, T.C.; Samoilova, A.G.; Pshenichnaya, N.Y.; Tyulkova, T.E. Latent Tuberculosis Infection in Children and Adolescents in Russia. Int. J. Infect. Dis. 2020, 92S, S26–S30. [Google Scholar] [CrossRef]
- Gupta, R.K.; Calderwood, C.J.; Yavlinsky, A.; Krutikov, M.; Quartagno, M.; Aichelburg, M.C.; Altet, N.; Diel, R.; Dobler, C.C.; Dominguez, J.; et al. Discovery and Validation of a Personalized Risk Predictor for Incident Tuberculosis in Low Transmission Settings. Nat. Med. 2020, 26, 1941–1949. [Google Scholar] [CrossRef] [PubMed]
- Hamada, Y.; Glaziou, P.; Sismanidis, C.; Getahun, H. Prevention of Tuberculosis in Household Members: Estimates of Children Eligible for Treatment. Bull. World Health Organ. 2019, 97, 534D–547D. [Google Scholar] [CrossRef] [PubMed]
- Dodd, P.J.; Yuen, C.M.; Becerra, M.C.; Revill, P.; Jenkins, H.E.; Seddon, J.A. Potential Effect of Household Contact Management on Childhood Tuberculosis: A Mathematical Modelling Study. Lancet Glob. Health 2018, 6, e1329–e1338. [Google Scholar] [CrossRef]
- Usdin, M.; Dedicoat, M.; Gajraj, R.; Harrison, P.; Kaur, H.; Duffield, K.; Walker, C.-L.; Akram, Y.; Aiyedun, V.; Mohamed, H.; et al. Latent Tuberculous Screening of Recent Migrants Attending Language Classes: A Cohort Study and Cost Analysis. Int. J. Tuberc. Lung Dis. 2017, 21, 175–180. [Google Scholar] [CrossRef]
- Walker, C.-L.; Duffield, K.; Kaur, H.; Dedicoat, M.; Gajraj, R. Acceptability of Latent Tuberculosis Testing of Migrants in a College Environment in England. Public Health 2018, 158, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Jiang, H.; Wang, X.; Liu, H.; Zhou, L.; Lu, X. ESMPE: A Combined Strategy for School Tuberculosis Prevention and Control Proposed by Dalian, China. PLoS ONE 2017, 12, e0185646. [Google Scholar] [CrossRef]
- Lewinsohn, D.M.; Leonard, M.K.; LoBue, P.A.; Cohn, D.L.; Daley, C.L.; Desmond, E.; Keane, J.; Lewinsohn, D.A.; Loeffler, A.M.; Mazurek, G.H.; et al. Official American Thoracic Society/Infectious Diseases Society of America/Centers for Disease Control and Prevention Clinical Practice Guidelines: Diagnosis of Tuberculosis in Adults and Children. Clin. Infect. Dis. 2017, 64, 111–115. [Google Scholar] [CrossRef]
- Faust, L.; McCarthy, A.; Schreiber, Y. Recommendations for the Screening of Paediatric Latent Tuberculosis Infection in Indigenous Communities: A Systematic Review of Screening Strategies among High-Risk Groups in Low-Incidence Countries. BMC Public Health 2018, 18, 979. [Google Scholar] [CrossRef]
- Dorjee, K.; Topgyal, S.; Tsewang, T.; Tsundue, T.; Namdon, T.; Bonomo, E.; Kensler, C.; Lhadon, D.; Choetso, T.; Nangsel, T.; et al. Risk of Developing Active Tuberculosis Following Tuberculosis Screening and Preventive Therapy for Tibetan Refugee Children and Adolescents in India: An Impact Assessment. PLoS Med. 2021, 18, e1003502. [Google Scholar] [CrossRef]
- Ustero, P.A.; Kay, A.W.; Ngo, K.; Golin, R.; Tsabedze, B.; Mzileni, B.; Glickman, J.; Wisile Xaba, M.; Mavimbela, G.; Mandalakas, A.M. School and Household Tuberculosis Contact Investigations in Swaziland: Active TB Case Finding in a High HIV/TB Burden Setting. PLoS ONE 2017, 12, e0178873. [Google Scholar] [CrossRef]
- Nduba, V.; Hoog, A.H.V.; Mitchell, E.; Onyango, P.; Laserson, K.; Borgdorff, M. Prevalence of Tuberculosis in Adolescents, Western Kenya: Implications for Control Programs. Int. J. Infect. Dis. 2015, 35, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Hatzenbuehler, L.A.; Starke, J.R.; Smith, E.O.; Turner, T.L.; Balmer, D.F.; Arif, Z.; Guzman, F.; Cruz, A.T. Increased Adolescent Knowledge and Behavior Following a One-Time Educational Intervention about Tuberculosis. Patient Educ. Couns. 2017, 100, 950–956. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zheng, Y.H.; Lu, L.P.; Yang, M.X.; Zhou, C.M.; Yuan, Z.A.; Hu, Y.; Xu, B. Acceptance of Chemo-Prophylaxis for Latent Tuberculosis Infection among High School/College Student Contacts of Tuberculosis Patients in Shanghai, China. Biomed. Environ. Sci. 2018, 31, 317–321. [Google Scholar] [CrossRef]
- Freier, G.; Wright, A.; Nelson, G.; Brenner, E.; Mase, S.; Tasker, S.; Matthews, K.L.; Bohnker, B.K. Multidrug-Resistant Tuberculosis in Military Recruits. Emerg. Infect. Dis. 2006, 12, 760–762. [Google Scholar] [CrossRef] [PubMed]
- Thomas, T.A.; Heysell, S.K.; Houpt, E.R.; Moore, J.L.; Keller, S.J. Outbreak of Pyrazinamide-Monoresistant Tuberculosis Identified Using Genotype Cluster and Social Media Analysis. Int. J. Tuberc. Lung Dis. 2014, 18, 552–558. [Google Scholar] [CrossRef][Green Version]
- Pareek, M.; Greenaway, C.; Noori, T.; Munoz, J.; Zenner, D. The Impact of Migration on Tuberculosis Epidemiology and Control in High-Income Countries: A Review. BMC Med. 2016, 14, 48. [Google Scholar] [CrossRef]
- Charan, J.; Goyal, J.P.; Reljic, T.; Emmanuel, P.; Patel, A.; Kumar, A. Isoniazid for the Prevention of Tuberculosis in HIV-Infected Children: A Systematic Review and Meta-Analysis. Pediatr. Infect. Dis. J. 2018, 37, 773–780. [Google Scholar] [CrossRef] [PubMed]
- Jerene, D.; Abebe, W.; Taye, K.; Suarez, P.G.; Feleke, Y.; Hallström, I.; Ruff, A.J. Tuberculosis along the Continuum of HIV Care in a Cohort of Adolescents Living with HIV in Ethiopia. Int. J. Tuberc. Lung Dis. 2017, 21, 32–37. [Google Scholar] [CrossRef] [PubMed]
- Mudzviti, T.; Shamu, T.; Chimbetete, C.; Munengerwa, T.; Bote, S.; Pascoe, M. Tolerability of Isoniazid Preventive Therapy in an HIV-Infected Cohort of Paediatric and Adolescent Patients on Antiretroviral Therapy from a Resource-Limited Setting: A Retrospective Cohort Study. Drugs Real. World Outcomes 2019, 6, 37–42. [Google Scholar] [CrossRef]
- Hunter, O.F.; Kyesi, F.; Ahluwalia, A.K.; Daffé, Z.N.; Munseri, P.; von Reyn, C.F.; Adams, L.V. Successful Implementation of Isoniazid Preventive Therapy at a Pediatric HIV Clinic in Tanzania. BMC Infect. Dis. 2020, 20, 738. [Google Scholar] [CrossRef]
- Kay, A.W.; Thivalapill, N.; Skinner, D.; Dube, G.S.; Dlamini, N.; Mzileni, B.; Fuentes, P.; Ustero, P.; Adams, L.V.; Mandalakas, A.M. Predictors of Suboptimal Adherence to Isoniazid Preventive Therapy among Adolescents and Children Living with HIV. PLoS ONE 2020, 15, e0243713. [Google Scholar] [CrossRef] [PubMed]
- Glatstein, M.; Carbell, G.; Scolnik, D.; Rimon, A.; Banerji, S.; Hoyte, C. Pyridoxine for the Treatment of Isoniazid-Induced Seizures in Intentional Ingestions: The Experience of a National Poison Center. Am. J. Emerg. Med. 2018, 36, 1775–1778. [Google Scholar] [CrossRef] [PubMed]
- Bennet, R.; Eriksson, M. Tuberculosis Infection and Disease in the 2015 Cohort of Unaccompanied Minors Seeking Asylum in Northern Stockholm, Sweden. Infect. Dis. 2017, 49, 501–506. [Google Scholar] [CrossRef]
- Mekonnen, A.; Collins, J.M.; Klinkenberg, E.; Assefa, D.; Aseffa, A.; Ameni, G.; Petros, B. Tuberculosis Knowledge and Attitude among Non-Health Science University Students Needs Attention: A Cross-Sectional Study in Three Ethiopian Universities. BMC Public Health 2020, 20, 631. [Google Scholar] [CrossRef] [PubMed]
- Idris, N.A.; Zakaria, R.; Muhamad, R.; Nik Husain, N.R.; Ishak, A.; Wan Mohammad, W.M.Z. The Effectiveness of Tuberculosis Education Programme in Kelantan, Malaysia on Knowledge, Attitude, Practice and Stigma Towards Tuberculosis among Adolescents. Malays. J. Med. Sci. 2020, 27, 102–114. [Google Scholar] [CrossRef]
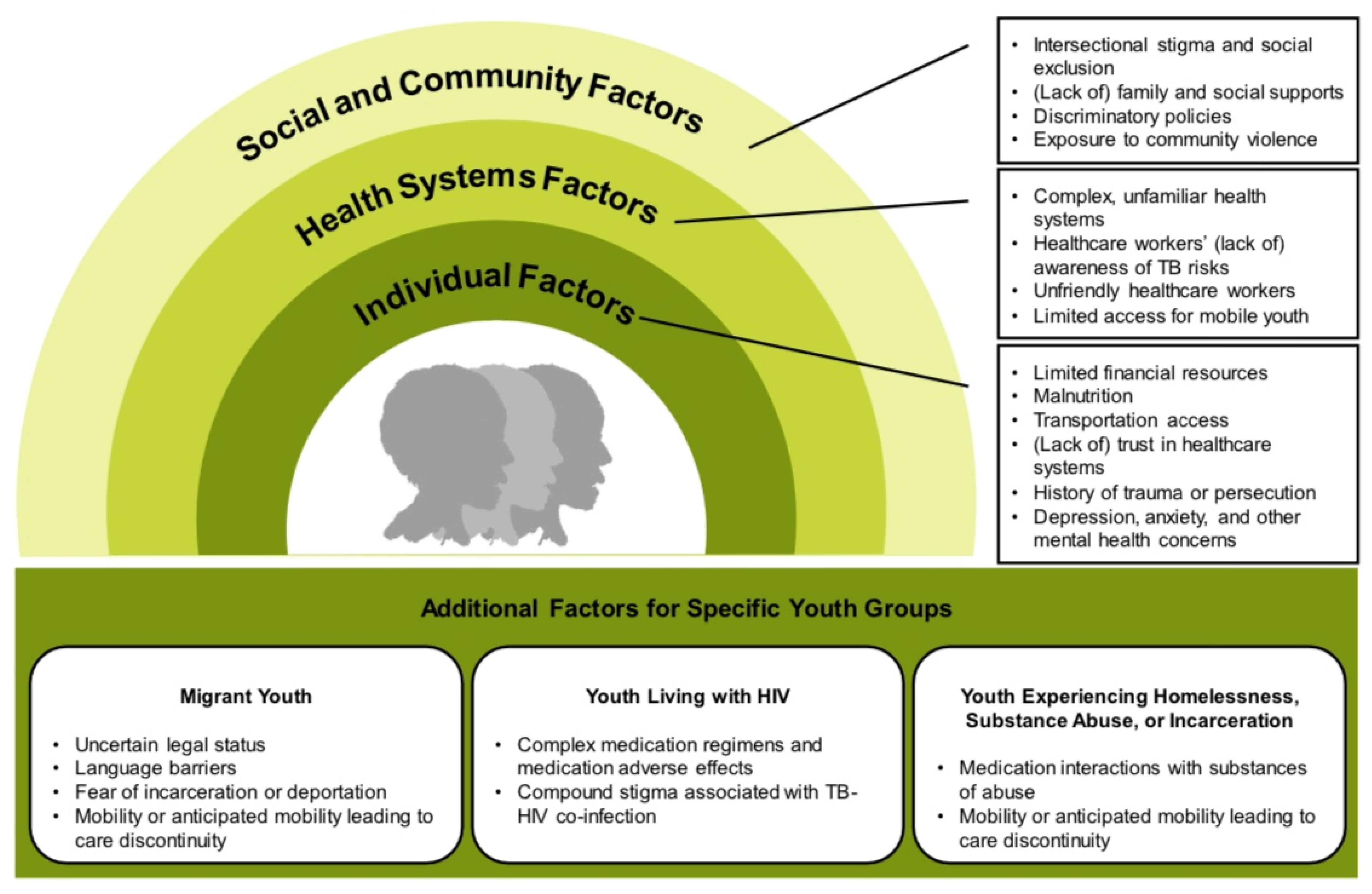
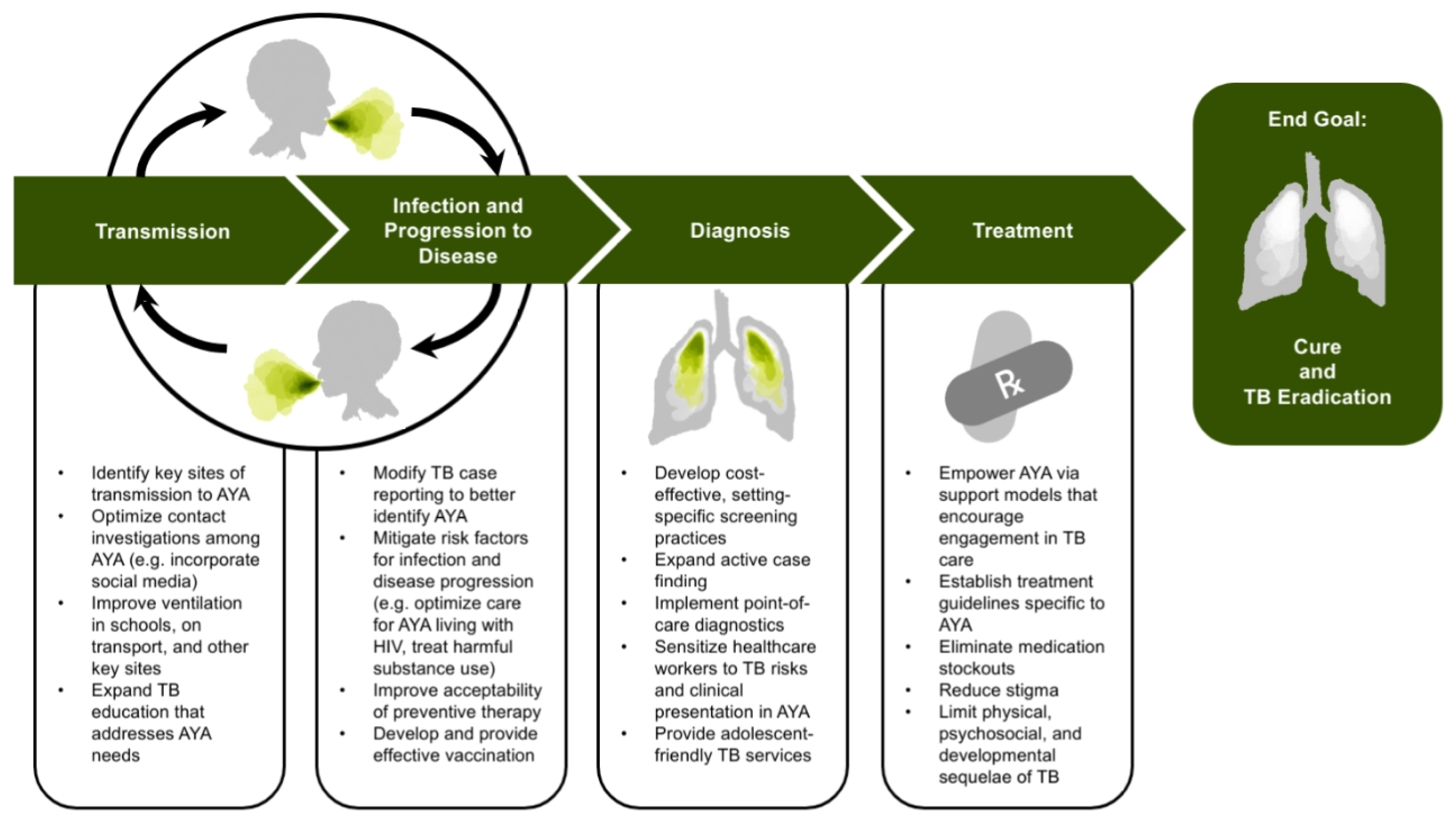
| Individual Factors | Community Factors |
|---|---|
|
|
| Medication (s) | Dosing Interval | Duration |
|---|---|---|
| For drug-susceptible TB: | ||
| Isoniazid monotherapy | Daily | 6 months (6H) or 9 months (9H) |
| Rifampicin monotherapy (4R) | Daily | 4 months |
| Rifampicin plus Isoniazid (3HR) | Daily | 3 months |
| Rifapentine plus Isoniazid | Weekly (3HP) | 3 months (12 doses) |
| Rifapentine plus Isoniazid | Daily (1HP) * | 1 month (28 doses) |
| For multidrug-resistant (MDR) TB: | ||
| Levofloxacin | Daily | 6 months |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Laycock, K.M.; Enane, L.A.; Steenhoff, A.P. Tuberculosis in Adolescents and Young Adults: Emerging Data on TB Transmission and Prevention among Vulnerable Young People. Trop. Med. Infect. Dis. 2021, 6, 148. https://doi.org/10.3390/tropicalmed6030148
Laycock KM, Enane LA, Steenhoff AP. Tuberculosis in Adolescents and Young Adults: Emerging Data on TB Transmission and Prevention among Vulnerable Young People. Tropical Medicine and Infectious Disease. 2021; 6(3):148. https://doi.org/10.3390/tropicalmed6030148
Chicago/Turabian StyleLaycock, Katherine M., Leslie A. Enane, and Andrew P. Steenhoff. 2021. "Tuberculosis in Adolescents and Young Adults: Emerging Data on TB Transmission and Prevention among Vulnerable Young People" Tropical Medicine and Infectious Disease 6, no. 3: 148. https://doi.org/10.3390/tropicalmed6030148
APA StyleLaycock, K. M., Enane, L. A., & Steenhoff, A. P. (2021). Tuberculosis in Adolescents and Young Adults: Emerging Data on TB Transmission and Prevention among Vulnerable Young People. Tropical Medicine and Infectious Disease, 6(3), 148. https://doi.org/10.3390/tropicalmed6030148







