Spatial Heterogeneity and Temporal Trends in Malaria on the Thai–Myanmar Border (2012–2017): A Retrospective Observational Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design and Study Areas
2.2. Malaria Data
- Thai cases, which referred to cases among Thai citizens.
- M1 cases, which referred to cases reported in migrants who had been in Thailand for >six months.
- M2 cases, which referred to cases reported in migrants who had been in Thailand for <six months.
2.3. Spatial Distribution
2.4. Spatial Clustering
2.5. Seasonality and Trends
3. Results
3.1. Summary Statistics
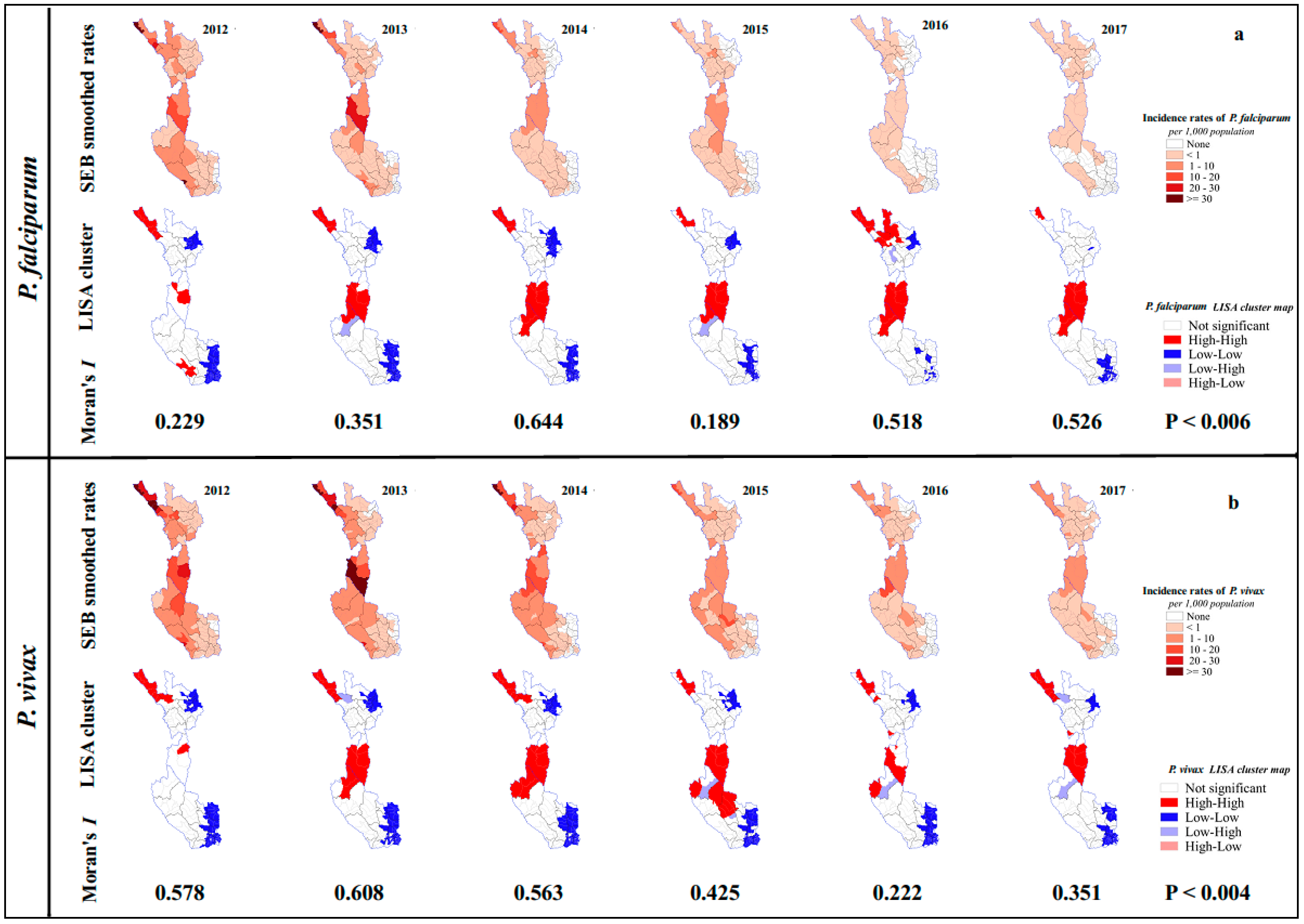
3.2. Spatial Distribution of P. falciparum and P. vivax Incidence
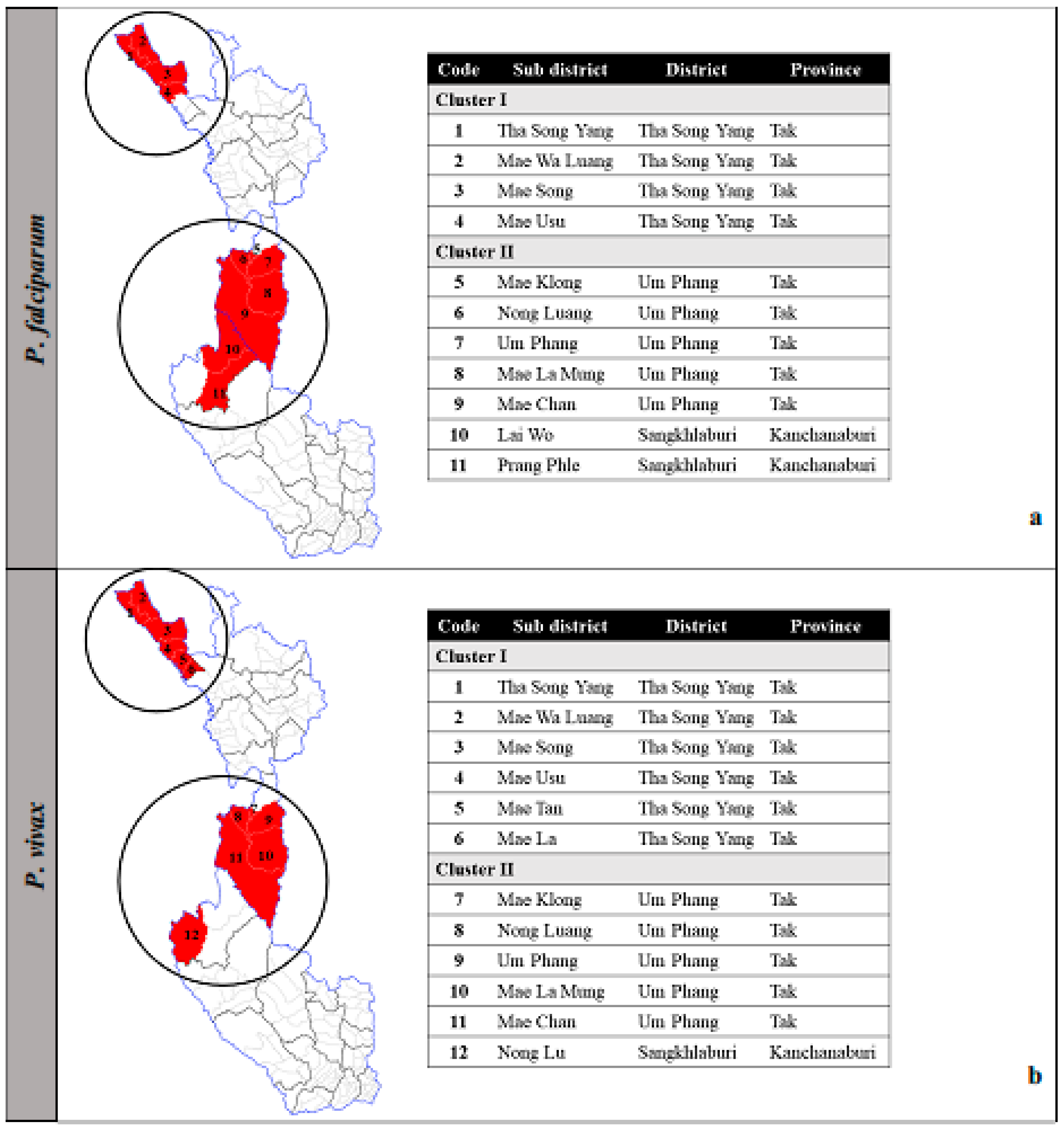
3.3. Clustered Areas
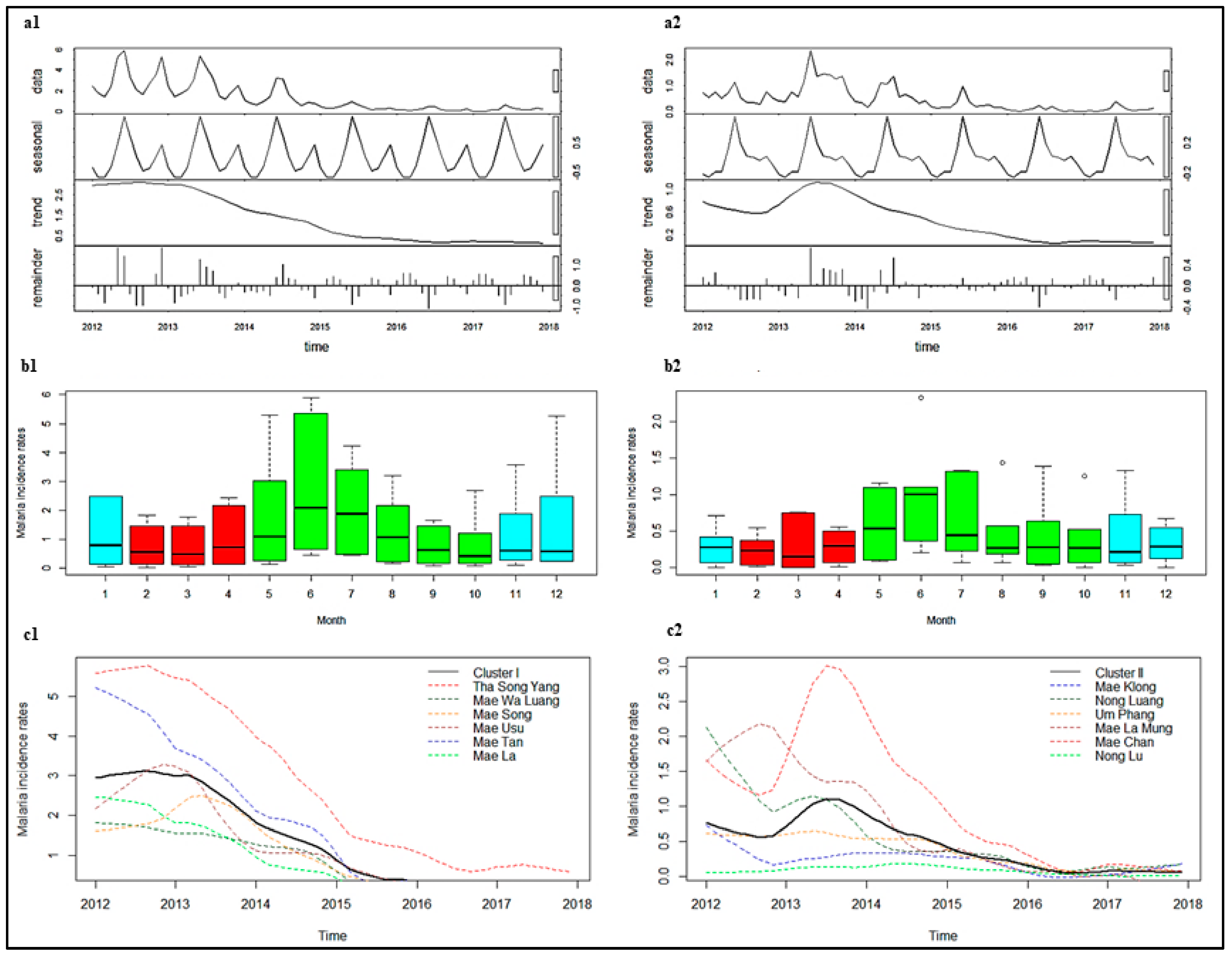
3.4. Temporal Distribution and Seasonality
3.5. Changes in Trends
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Ethical Issue
Abbreviations
| ACT | Artemisinin combination therapy |
| BOE | Bureau of Epidemiology |
| BVBD | Bureau of Vector-Borne Disease |
| CI | Confidence interval |
| GMS | Greater Mekong Sub-region |
| IRR | Incidence rate ratio |
| IRS | Indoor residual insecticide spraying |
| ITN | Insecticide-treated nets |
| LISA | Local indicators of spatial association |
| LLIN | long-lasting insecticide-treated bed nets |
| M1 | Long-term migrant |
| M2 | Short-term migrant |
| MOPH | Ministry of Public Health, Thailand |
| P | P-value |
| P. falciparum | Plasmodium falciparum |
| P. vivax | Plasmodium vivax |
| RDT | Rapid diagnostic test |
| SEB | Spatial Empirical Bayesian |
| STL | Seasonal decomposition of time series by Loess |
References
- World Health Organization. World Malaria Report 2017; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- World Health Organization. World Malaria Report 2016; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- World Health Organization. Strategy for Malaria Elimination in the Greater Mekong Subregion: 2015–2030; World Health Organization: Geneva, Switzerland, 2015. [Google Scholar]
- Imwong, M.; Suwannasin, K.; Kunasol, C.; Sutawong, K.; Mayxay, M.; Rekol, H.; Smithuis, F.M.; Hlaing, T.M.; Tun, K.M.; van der Pluijm, R.W.; et al. The spread of artemisinin-resistant Plasmodium falciparum in the Greater Mekong subregion: A molecular epidemiology observational study. Lancet Infect. Dis. 2017, 17, 491–497. [Google Scholar] [CrossRef]
- Imwong, M.; Jindakhad, T.; Kunasol, C.; Sutawong, K.; Vejakama, P.; Dondorp, A.M. An outbreak of artemisinin resistant falciparum malaria in Eastern Thailand. Sci. Rep. 2015, 5, 17412. [Google Scholar] [CrossRef] [PubMed]
- Bloland, P.B.; Williams, H.A. Malaria Control during Mass Population Movements and Natural Disasters; National Academies Press (US): Washington, DC, USA, 2002. [Google Scholar] [CrossRef]
- Smith, C.; Whittaker, M. Beyond mobile populations: A critical review of the literature on malaria and population mobility and suggestions for future directions. Malar. J. 2014, 13, 307. [Google Scholar] [CrossRef] [PubMed]
- Phyo, A.P.; Nkhoma, S.; Stepniewska, K.; Ashley, E.A.; Nair, S.; McGready, R.; ler Moo, C.; Al-Saai, S.; Dondorp, A.M.; Lwin, K.M.; et al. Emergence of artemisinin-resistant malaria on the western border of Thailand: A longitudinal study. Lancet 2012, 379, 1960–1966. [Google Scholar] [CrossRef]
- Payne, D. Did medicated salt hasten the spread of chloroquine resistance in Plasmodium falciparum? Parasitol. Today (Pers. Ed.) 1988, 4, 112–115. [Google Scholar] [CrossRef]
- Roper, C.; Pearce, R.; Nair, S.; Sharp, B.; Nosten, F.; Anderson, T. Intercontinental spread of pyrimethamine-resistant malaria. Science 2004, 305, 1124. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Public Health, Thailand. National Malaria Elimination Strategy, Thailand 2017–2026; Department of Disease Control: Nonthaburi, Thailand, 2017.
- World Health Organization. World Malaria Report 2018; World Health Organization: Geneva, Switzerland, 2018. [Google Scholar]
- Carrara, V.I.; Sirilak, S.; Thonglairuam, J.; Rojanawatsirivet, C.; Proux, S.; Gilbos, V.; Brockman, A.; Ashley, E.A.; McGready, R.; Krudsood, S.; et al. Deployment of Early Diagnosis and Mefloquine-Artesunate Treatment of Falciparum Malaria in Thailand: The Tak Malaria Initiative. PLoS Med. 2006, 3, e183. [Google Scholar] [CrossRef] [PubMed]
- Parker, D.M.; Carrara, V.I.; Pukrittayakamee, S.; McGready, R.; Nosten, F.H. Malaria ecology along the Thailand–Myanmar border. Malar. J. 2015, 14, 388. [Google Scholar] [CrossRef] [PubMed]
- Cui, L.; Cao, Y.; Kaewkungwal, J.; Khamsiriwatchara, A.; Lawpoolsri, S.; Soe, T.N.; Kyaw, M.P.; Sattabongkot, J. Malaria Elimination in the Greater Mekong Subregion: Challenges and Prospects. In Towards Malaria Elimination—A Leap Forward; IntechOpen: London, UK, 2018. [Google Scholar] [CrossRef]
- Lover, A.A.; Baird, J.K.; Gosling, R.; Price, R.N. Malaria Elimination: Time to Target All Species. Am. J. Trop. Med. Hyg. 2018, 99, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Parker, D.M.; Landier, J.; Thu, A.M.; Lwin, K.M.; Delmas, G.; Nosten, F.H. Scale up of a Plasmodium falciparum elimination program and surveillance system in Kayin State, Myanmar. Wellcome Open Res. 2017, 2, 98. [Google Scholar] [CrossRef] [PubMed]
- Taylor, A.R.; Schaffner, S.F.; Cerqueira, G.C.; Nkhoma, S.C.; Anderson, T.J.C.; Sriprawat, K.; Pyae Phyo, A.; Nosten, F.; Neafsey, D.E.; Buckee, C.O. Quantifying connectivity between local Plasmodium falciparum malaria parasite populations using identity by descent. PLoS Genet. 2017, 13, e1007065. [Google Scholar] [CrossRef] [PubMed]
- Maude, R.J.; Socheat, D.; Nguon, C.; Saroth, P.; Dara, P.; Li, G.; Song, J.; Yeung, S.; Dondorp, A.M.; Day, N.P.; et al. Optimising Strategies for Plasmodium falciparum Malaria Elimination in Cambodia: Primaquine, Mass Drug Administration and Artemisinin Resistance. PLoS ONE 2012, 7, e37166. [Google Scholar] [CrossRef] [PubMed]
- Bhumiratana, A.; Intarapuk, A.; Sorosjinda-Nunthawarasilp, P.; Maneekan, P.; Koyadun, S. Border Malaria Associated with Multidrug Resistance on Thailand-Myanmar and Thailand-Cambodia Borders: Transmission Dynamic, Vulnerability, and Surveillance. BioMed Res. Int. 2013, 2013, 363417. [Google Scholar] [CrossRef] [PubMed]
- Soe, T.N.; Yeesoonsang, S.; Jiraphongsa, C.; Nguyen, H.A.P.; Thammapalo, S.; Tao, S.; Phanawadee, M.; Srethapranai, V.; Naung, S.Y. Malaria Surveillance at Thai-Myanmar Border, Mae Sot District, Tak Province, Thailand, July 2013. Outbreak Surveill. Investig. Rep. (OSIR) 2016, 9, 12–19. [Google Scholar]
- Ma, S.; Lawpoolsri, S.; Soonthornworasiri, N.; Khamsiriwatchara, A.; Jandee, K.; Taweeseneepitch, K.; Pawarana, R.; Jaiklaew, S.; Kijsanayotin, B.; Kaewkungwal, J. Effectiveness of Implementation of Electronic Malaria Information System as the National Malaria Surveillance System in Thailand. JMIR Public Health Surveill. 2016, 2, e20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khamsiriwatchara, A. Development and utilization of data in consolidated malaria reporting system in Thailand. In Proceedings of the Joint International Tropical Medicine Meeting 2014, Centara Grand & Bangkok Convention Centre at CentralWorld, Bangkok, Thailand, 2–4 December 2014; p. 23. [Google Scholar]
- Ministry of Public Health, Thailand. Thailand Malaria Elimination Program. Available online: http://203.157.41.215/malariaR10/index_newversion.php (accessed on 25 February 2018).
- Diallo, M.A.; Diongue, K.; Ndiaye, M.; Gaye, A.; Deme, A.; Badiane, A.S.; Ndiaye, D. Evaluation of CareStart™ Malaria HRP2/pLDH (Pf/pan) Combo Test in a malaria low transmission region of Senegal. Malar. J. 2017, 16, 328. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wong, D.W.S. The Modifiable Areal Unit Problem (MAUP). In WorldMinds: Geographical Perspectives on 100 Problems: Commemorating the 100th Anniversary of the Association of American Geographers 1904–2004; Janelle, D.G., Warf, B., Hansen, K., Eds.; Springer: Dordrecht, The Netherlands, 2004; pp. 571–575. [Google Scholar] [CrossRef]
- Martuzzi, M.; Elliott, P. Empirical Bayes Estimation of Small Area Prevalence of Non-Rare Conditions. Stat. Med. 1996, 15, 1867–1873. [Google Scholar] [CrossRef]
- Getis, A. Spatial Autocorrelation. In Handbook of Applied Spatial Analysis: Software Tools, Methods and Applications; Fischer, M.M., Getis, A., Eds.; Springer: Berlin/Heidelberg, Germany, 2010; pp. 255–278. [Google Scholar] [CrossRef]
- Chuang, T.-W.; Wimberly, M.C. Remote Sensing of Climatic Anomalies and West Nile Virus Incidence in the Northern Great Plains of the United States. PLoS ONE 2012, 7, e46882. [Google Scholar] [CrossRef]
- Marshall, R.J. Mapping Disease and Mortality Rates Using Empirical Bayes Estimators. J. R. Stat. Soc. Ser. C Appl. Stat. 1991, 40, 283–294. [Google Scholar] [CrossRef]
- Wangdi, K.; Kaewkungwal, J.; Singhasivanon, P.; Silawan, T.; Lawpoolsri, S.; White, N.J. Spatio-temporal patterns of malaria infection in Bhutan: A country embarking on malaria elimination. Malar. J. 2011, 10, 89. [Google Scholar] [CrossRef]
- Gu, H.; Fan, W.; Liu, K.; Qin, S.; Li, X.; Jiang, J.; Chen, E.; Zhou, Y.; Jiang, Q. Spatio-temporal variations of typhoid and paratyphoid fevers in Zhejiang Province, China from 2005 to 2015. Sci. Rep. 2017, 7, 5780. [Google Scholar] [CrossRef] [Green Version]
- Saman, D.M.; Cole, H.P.; Odoi, A.; Myers, M.L.; Carey, D.I.; Westneat, S.C. A Spatial Cluster Analysis of Tractor Overturns in Kentucky from 1960 to 2002. PLoS ONE 2012, 7, e30532. [Google Scholar] [CrossRef]
- Anselin, L. Local Indicators of Spatial Association—LISA. Geogr. Anal. 1995, 27, 93–115. [Google Scholar] [CrossRef]
- Cleveland, R.B.; Cleveland, W.S.; McRae, J.E.; Terpenning, I. STL: A Seasonal-Trend Decomposition. J. Off. Stat. 1990, 6, 3–73. [Google Scholar]
- Hilbe, J.M. Poisson Regression. In Modeling Count Data; Cambridge University Press: Cambridge, UK, 2014; pp. 35–73. [Google Scholar] [CrossRef]
- Sriwichai, P.; Samung, Y.; Sumruayphol, S.; Kiattibutr, K.; Kumpitak, C.; Payakkapol, A.; Kaewkungwal, J.; Yan, G.; Cui, L.; Sattabongkot, J. Natural human Plasmodium infections in major Anopheles mosquitoes in western Thailand. Parasit. Vectors 2016, 9, 17. [Google Scholar] [CrossRef]
- Kar, N.P.; Kumar, A.; Singh, O.P.; Carlton, J.M.; Nanda, N. A review of malaria transmission dynamics in forest ecosystems. Parasit. Vectors 2014, 7, 265. [Google Scholar] [CrossRef]
- Sriwichai, P.; Karl, S.; Samung, Y.; Kiattibutr, K.; Sirichaisinthop, J.; Mueller, I.; Cui, L.; Sattabongkot, J. Imported Plasmodium falciparum and locally transmitted Plasmodium vivax: Cross-border malaria transmission scenario in northwestern Thailand. Malar. J. 2017, 16, 258. [Google Scholar] [CrossRef]
- Smith, P.; Bourgeois Lüthi, N.; Huachun, L.; Naing Oo, K.; Phonvisay, A.; Premashthira, S.; Abila, R.; Widders, P.; Karan, K.; Miller, C. Movement Pathways and Market Chains of Large Ruminants in the Greater Mekong Sub-Region; Sub-regional representation for South-East Asia: Bangkok, Thailand, 2015. [Google Scholar]
- Pim, M.; Lisbeth, H. Malaria on the move: Human population movement and malaria transmission. Emerg. Infect. Dis. 2000, 6, 103. [Google Scholar] [CrossRef]
- Jitthai, N. Migration and malaria. Southeast Asian J. Trop. Med. Public Health 2013, 44 (Suppl. 1), 166–200. [Google Scholar]
- Singhanetra-Renard, A. Malaria and mobility in Thailand. Soc. Sci. Med. (1982) 1993, 37, 1147–1154. [Google Scholar] [CrossRef]
- Landier, J.; Parker, D.M.; Thu, A.M.; Lwin, K.M.; Delmas, G.; Nosten, F.H.; Andolina, C.; Aguas, R.; Ang, S.M.; Aung, E.P.; et al. Effect of generalised access to early diagnosis and treatment and targeted mass drug administration on Plasmodium falciparum malaria in Eastern Myanmar: An observational study of a regional elimination programme. Lancet 2018, 391, 1916–1926. [Google Scholar] [CrossRef]
- President’s Malaria Initiative Thailand, Lao PDR and Regional. Malaria Operational Plan FY 2018 USAID: 2018. Available online: https://www.pmi.gov/docs/default-source/default-document-library/malaria-operational-plans/fy-2018/fy-2018-thailand-regional-malaria-operational-plan.pdf?sfvrsn=8 (accessed on 28 June 2018).
- Chaumeau, V.; Cerqueira, D.; Zadrozny, J.; Kittiphanakun, P.; Andolina, C.; Chareonviriyaphap, T.; Nosten, F.; Corbel, V. Insecticide resistance in malaria vectors along the Thailand-Myanmar border. Parasit. Vectors 2017, 10, 165. [Google Scholar] [CrossRef]
- Luxemburger, C.; Perea, W.A.; Delmas, G.; Pruja, C.; Pecoul, B.; Moren, A. Permethrin-impregnated bed nets for the prevention of malaria in schoolchildren on the Thai-Burmese border. Trans. R. Soc. Trop. Med. Hyg. 1994, 88, 155–159. [Google Scholar] [CrossRef]

| Characteristics | Tak Province | Kanchanaburi Province | |||
|---|---|---|---|---|---|
| No. of public hospitals | 9 | 15 | |||
| No. of malaria clinics | 26 | 15 | |||
| No. of malaria posts | 76 | 41 | |||
| 2012 | 2017 | 2012 | 2017 | ||
| P. vivax : P. falciparum † | 1.62 | 11.45 | 1.06 | 13.06 | |
| P. falciparum cases | 4735 | 90 | 1719 | 16 | |
| Percent by age | <5 years | 9.27 | 10.00 | 4.42 | 6.23 |
| 5 to 14 years | 31.83 | 27.78 | 18.62 | 0.00 | |
| 15 to 24 years | 22.52 | 18.89 | 23.79 | 25.00 | |
| 25 to 44 years | 24.19 | 26.67 | 35.78 | 37.50 | |
| ≥45 years | 12.20 | 16.67 | 17.39 | 31.25 | |
| By occupation (%) | Agriculture | 13.72 | 14.44 | 44.56 | 62.50 |
| Student | 39.78 | 30.00 | 21.29 | 12.50 | |
| Laborer | 45.23 | 54.44 | 31.41 | 18.75 | |
| Others * | 1.27 | 1.11 | 2.73 | 6.25 | |
| By type (%) | Thais | 33.04 | 42.22 | 30.31 | 37.50 |
| M1 | 33.69 | 23.33 | 50.79 | 56.25 | |
| M2 | 33.27 | 34.44 | 18.90 | 6.25 | |
| Male : Female | 1.95 | 1.37 | 2.39 | 1.29 | |
| P. vivax cases | 7660 | 1041 | 1820 | 209 | |
| Percent by age | < 5 years | 11.87 | 7.77 | 7.15 | 6.22 |
| 5 to 14 years | 33.79 | 31.77 | 23.53 | 22.97 | |
| 15 to 24 years | 22.13 | 23.03 | 22.32 | 23.92 | |
| 25 to 44 years | 22.00 | 23.99 | 28.48 | 26.79 | |
| ≥45 years | 10.21 | 13.44 | 18.53 | 20.10 | |
| By occupation (%) | Agriculture | 11.52 | 16.31 | 31.45 | 42.58 |
| Student | 44.41 | 36.66 | 30.07 | 28.71 | |
| Laborer | 42.88 | 45.20 | 35.02 | 24.40 | |
| Others * | 1.19 | 1.82 | 3.46 | 4.31 | |
| By type (%) | Thais | 43.49 | 43.76 | 43.32 | 44.50 |
| M1 | 27.85 | 32.73 | 41.95 | 43.54 | |
| M2 | 28.66 | 23.51 | 14.73 | 11.96 | |
| Male : Female | 1.55 | 1.82 | 2.06 | 1.90 | |
| Incidence Rates | 2012 | 2013 | 2014 | 2015 | 2016 | 2017 |
|---|---|---|---|---|---|---|
| P. falciparum | ||||||
| 0 | 25 | 45 | 74 | 80 | 103 | 105 |
| <1 | 93 | 93 | 72 | 74 | 58 | 56 |
| 1–10 | 38 | 19 | 14 | 7 | 0 | 0 |
| 10–20 | 2 | 2 | 1 | 0 | 0 | 0 |
| 20–30 | 1 | 1 | 0 | 0 | 0 | 0 |
| ≥30 | 2 | 1 | 0 | 0 | 0 | 0 |
| P. vivax | ||||||
| 0 | 7 | 24 | 45 | 47 | 70 | 66 |
| <1 | 101 | 88 | 74 | 74 | 71 | 77 |
| 1–10 | 39 | 38 | 33 | 33 | 19 | 18 |
| 10–20 | 6 | 5 | 7 | 7 | 1 | 0 |
| 20–30 | 5 | 3 | 1 | 1 | 0 | 0 |
| ≥30 | 3 | 3 | 1 | 1 | 0 | 0 |
| Clustered Areas and Individual Sub-Districts | P. falciparum | P. vivax | ||
|---|---|---|---|---|
| IRR | (95% CI) | IRR | (95% CI) | |
| Cluster I | 0.949 | (0.947–0.952) | 0.955 | (0.953–0.956) |
| Tha Song Yang | 0.950 | (0.945–0.953) | 0.965 | (0.963–0.967) |
| Mae Wa Luang | 0.974 | (0.966–0.981) | 0.962 | (0.957–0.968) |
| Mae Song | 0.965 | (0.960–0.969) | 0.960 | (0.956–0.963) |
| Mae Usu | 0.945 | (0.939–0.951) | 0.951 | (0.947–0.954) |
| Mae Tan | - | - | 0.948 | (0.945–0.952) |
| Mae La | - | - | 0.952 | (0.947–0.956) |
| Cluster II | 0.962 | (0.958–0.965) | 0.968 | (0.965–0.970) |
| Mae Klong | 0.985 | (0.975–0.995) | 0.988 | (0.979–0.997) |
| Nong Luang | 0.977 | (0.968–0.987) | 0.973 | (0.965–0.981) |
| Um Phang | 0.980 | (0.971–0.989) | 0.981 | (0.974–0.989) |
| Mae La Mung | 0.984 | (0.975–0.992) | 0.971 | (0.963–0.978) |
| Mae Chan | 0.961 | (0.957–0.965) | 0.965 | (0.962–0.968) |
| Lai Wo | 0.991 | (0.982–1.000) | - | - |
| Prang Phle | 0.996 | (0.986–1.007) | - | - |
| Nong Lu | - | - | 0.986 | (0.980–0.992) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saita, S.; Silawan, T.; Parker, D.M.; Sriwichai, P.; Phuanukoonnon, S.; Sudathip, P.; Maude, R.J.; White, L.J.; Pan-ngum, W. Spatial Heterogeneity and Temporal Trends in Malaria on the Thai–Myanmar Border (2012–2017): A Retrospective Observational Study. Trop. Med. Infect. Dis. 2019, 4, 62. https://doi.org/10.3390/tropicalmed4020062
Saita S, Silawan T, Parker DM, Sriwichai P, Phuanukoonnon S, Sudathip P, Maude RJ, White LJ, Pan-ngum W. Spatial Heterogeneity and Temporal Trends in Malaria on the Thai–Myanmar Border (2012–2017): A Retrospective Observational Study. Tropical Medicine and Infectious Disease. 2019; 4(2):62. https://doi.org/10.3390/tropicalmed4020062
Chicago/Turabian StyleSaita, Sayambhu, Tassanee Silawan, Daniel M. Parker, Patchara Sriwichai, Suparat Phuanukoonnon, Prayuth Sudathip, Richard J. Maude, Lisa J. White, and Wirichada Pan-ngum. 2019. "Spatial Heterogeneity and Temporal Trends in Malaria on the Thai–Myanmar Border (2012–2017): A Retrospective Observational Study" Tropical Medicine and Infectious Disease 4, no. 2: 62. https://doi.org/10.3390/tropicalmed4020062
APA StyleSaita, S., Silawan, T., Parker, D. M., Sriwichai, P., Phuanukoonnon, S., Sudathip, P., Maude, R. J., White, L. J., & Pan-ngum, W. (2019). Spatial Heterogeneity and Temporal Trends in Malaria on the Thai–Myanmar Border (2012–2017): A Retrospective Observational Study. Tropical Medicine and Infectious Disease, 4(2), 62. https://doi.org/10.3390/tropicalmed4020062









