The First Outbreak of Dengue Fever in Greater Darfur, Western Sudan
Abstract
1. Introduction
2. Materials and Methods
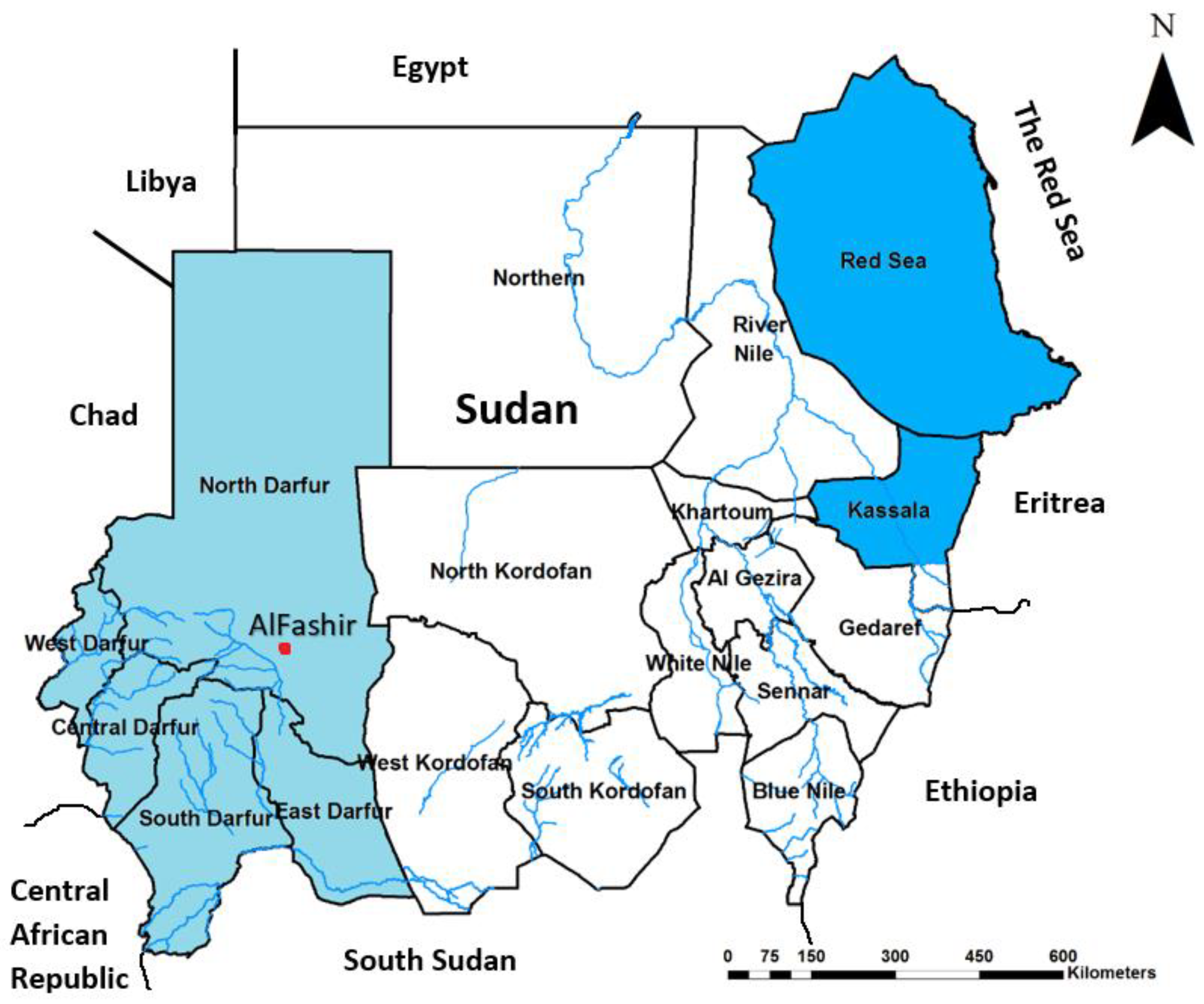
2.1. Study Area
2.2. The Outbreak Investigation
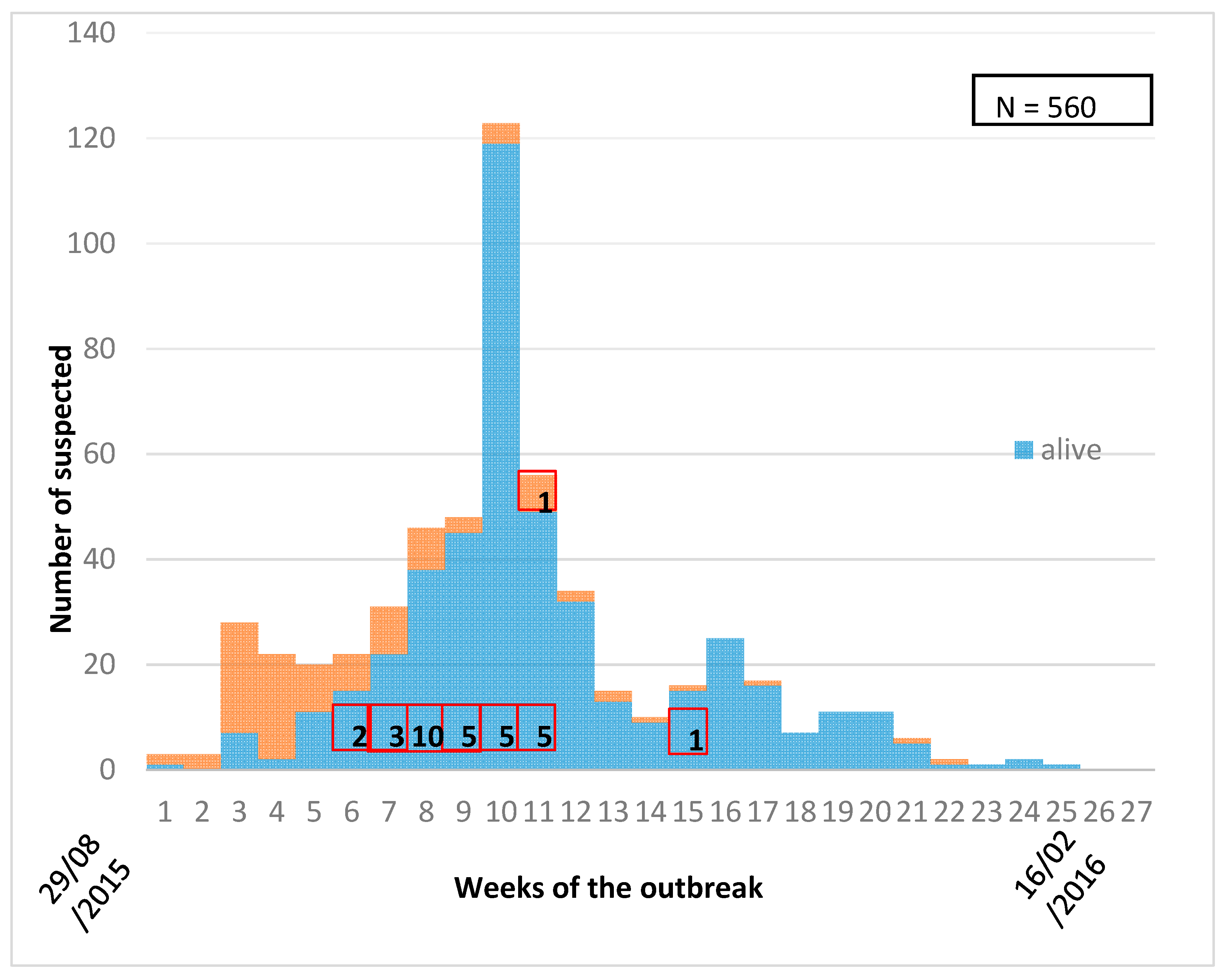
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Simmons, C.P.; Farrar, J.J.; van Vinh Chau, N.; Wills, B. Dengue. N. Engl. J. Med. 2012, 366, 1423–1432. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Dengue Guidelines for Diagnosis, Treatment, Prevention and Control: New Edition; World Health Organization: Geneva, Switzerland, 2009. [Google Scholar]
- Endy, T.P.; Anderson, K.B.; Nisalak, A.; Yoon, I.-K.; Green, S.; Rothman, A.L.; Thomas, S.J.; Jarman, R.G.; Libraty, D.H.; Gibbons, R.V. Determinants of Inapparent and Symptomatic Dengue Infection in a Prospective Study of Primary School Children in Kamphaeng Phet, Thailand. PLoS Negl. Trop. Dis. 2011, 5, e975. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, S.; Gething, P.W.; Brady, O.J.; Messina, J.P.; Farlow, A.W.; Moyes, C.L.; Drake, J.M.; Brownstein, J.S.; Hoen, A.G.; Sankoh, O.; et al. The global distribution and burden of dengue. Nature 2013, 496, 504–507. [Google Scholar] [CrossRef] [PubMed]
- Brady, O.J.; Gething, P.W.; Bhatt, S.; Messina, J.P.; Brownstein, J.S.; Hoen, A.G.; Moyes, C.L.; Farlow, A.W.; Scott, T.W.; Hay, S.I. Refining the global spatial limits of dengue virus transmission by evidence-based consensus. PLoS Negl. Trop. Dis. 2012, 6, e1760. [Google Scholar] [CrossRef] [PubMed]
- Guzman, M.G.; Halstead, S.B.; Artsob, H.; Buchy, P.; Farrar, J.; Gubler, D.J.; Hunsperger, E.; Kroeger, A.; Margolis, H.S.; Martínez, E.; et al. Dengue: A continuing global threat. Nat. Rev. Microbiol. 2010, 8, S7–S16. [Google Scholar] [CrossRef] [PubMed]
- Van Kleef, E.; Bambrick, H.; Hales, S. The geographic distribution of dengue fever and the potential influence of global climate change. Available online: http://eprints.qut.edu.au/103224/ (accessed on 11 February 2017).
- Tatem, A.J.; Hay, S.I.; Rogers, D.J. Global traffic and disease vector dispersal. Proc. Natl. Acad. Sci. USA 2006, 103, 6242–6247. [Google Scholar] [CrossRef] [PubMed]
- Cummings, D.A.T.; Irizarry, R.A.; Huang, N.E.; Endy, T.P.; Nisalak, A.; Ungchusak, K.; Burke, D.S. Travelling waves in the occurrence of dengue haemorrhagic fever in Thailand. Nature 2004, 427, 344–347. [Google Scholar] [CrossRef] [PubMed]
- Wilder-Smith, A.; Gubler, D.J. Geographic Expansion of Dengue: The Impact of International Travel. Med. Clin. 2008, 92, 1377–1390. [Google Scholar] [CrossRef] [PubMed]
- Wesolowski, A.; Qureshi, T.; Boni, M.F.; Sundsøy, P.R.; Johansson, M.A.; Rasheed, S.B.; Engø-Monsen, K.; Buckee, C.O. Impact of human mobility on the emergence of dengue epidemics in Pakistan. Proc. Natl. Acad. Sci. USA 2015, 112, 11887–11892. [Google Scholar] [CrossRef] [PubMed]
- Stoddard, S.T.; Morrison, A.C.; Vazquez-Prokopec, G.M.; Soldan, V.P.; Kochel, T.J.; Kitron, U.; Elder, J.P.; Scott, T.W. The Role of Human Movement in the Transmission of Vector-Borne Pathogens. PLoS Negl. Trop. Dis. 2009, 3, e481. [Google Scholar] [CrossRef] [PubMed]
- Gubler, D.J. Epidemic dengue/dengue hemorrhagic fever as a public health, social and economic problem in the 21st century. Trends Microbiol. 2002, 10, 100–103. [Google Scholar] [CrossRef]
- Kendall, C.; Hudelson, P.; Leontsini, E.; Winch, P.; Lloyd, L.; Cruz, F. Urbanization, Dengue, and the Health Transition: Anthropological Contributions to International Health. Med. Anthropol. Q. 1991, 5, 257–268. [Google Scholar] [CrossRef]
- Gubler, D.J. Dengue, Urbanization and Globalization: The Unholy Trinity of the 21st Century. Trop. Med. Health 2011, 39, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Brathwaite Dick, O.; San Martín, J.L.; Montoya, R.H.; del Diego, J.; Zambrano, B.; Dayan, G.H. The History of Dengue Outbreaks in the Americas. Am. J. Trop. Med. Hyg. 2012, 87, 584–593. [Google Scholar] [CrossRef] [PubMed]
- Brady, O.J.; Smith, D.L.; Scott, T.W.; Hay, S.I. Dengue disease outbreak definitions are implicitly variable. Epidemics 2015, 11, 92–102. [Google Scholar] [CrossRef] [PubMed]
- Gayer, M.; Legros, D.; Formenty, P.; Connolly, M.A. Conflict and Emerging Infectious Diseases. Emerg. Infect. Dis. 2007, 13, 1625–1631. [Google Scholar] [CrossRef] [PubMed]
- Balfour, A.; Archibald, R.G. Review of Some of the Recent Advances in Tropical Medicine, Hygiene and Tropical Veterinary Science; Department of Education, Sudan Government, Khartoum, Baillière Tindall & Cox: London, UK, 1908; p. 262. Available online: http://archive.org/details/b21352161 (accessed on 11 April 2018).
- Soghaier, M.A.; Himatt, S.; Osman, K.E.; Okoued, S.I.; Seidahmed, O.E.; Beatty, M.E.; Elmusharaf, K.; Khogali, J.; Shingrai, N.H.; Elmangory, M.M. Cross-sectional community-based study of the socio-demographic factors associated with the prevalence of dengue in the eastern part of Sudan in 2011. BMC Public Health 2015, 15, 558. [Google Scholar] [CrossRef] [PubMed]
- Seidahmed, O.M.; Hassan, S.A.; Soghaier, M.A.; Siam, H.A.; Ahmed, F.T.; Elkarsany, M.M.; Sulaiman, S.M. Spatial and temporal patterns of dengue transmission along a Red Sea coastline: A longitudinal entomological and serological survey in Port Sudan city. PLoS Negl. Trop. Dis. 2012, 6, e1821. [Google Scholar] [CrossRef] [PubMed]
- Abdallah, T.M.; Ali, A.A.A.; Karsany, M.S.; Adam, I. Epidemiology of dengue infections in Kassala, Eastern Sudan. J. Med. Virol. 2012, 84, 500–503. [Google Scholar] [CrossRef] [PubMed]
- Malik, A.; Earhart, K.; Mohareb, E.; Saad, M.; Saeed, M.; Ageep, A.; Soliman, A. Dengue hemorrhagic fever outbreak in children in Port Sudan. J. Infect. Public Health 2011, 4, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Adam, I.; Jumaa, A.M.; Elbashir, H.M.; Karsany, M.S. Maternal and perinatal outcomes of dengue in PortSudan, Eastern Sudan. Virol. J. 2010, 7, 153. [Google Scholar] [CrossRef] [PubMed]
- Markoff, L. Yellow Fever Outbreak in Sudan. N. Engl. J. Med. 2013, 368, 689–691. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, A.M.; Adam, I.A.; Osman, B.T.; Aradaib, I.E. Epidemiological survey of Crimean Congo hemorrhagic fever virus in cattle in East Darfur State, Sudan. Ticks Tick-Borne Dis. 2015, 6, 439–444. [Google Scholar] [CrossRef] [PubMed]
- Seidahmed, O.M.E.; Siam, H.A.M.; Soghaier, M.A.; Abubakr, M.; Osman, H.A.; Abd Elrhman, L.S.; Elmagbol, B.; Velayudhan, R. Dengue Vector Control and Surveillance during a Major Outbreak in a Coastal Red Sea Area in Sudan. 2012. Available online: http://www.who.int/iris/handle/10665/118472 (accessed on 10 February 2018).
- Olsson, O. After Janjaweed? Socioeconomic Impacts of the Conflict in Darfur. World Bank Econ. Rev. 2010, 24, 386–411. [Google Scholar] [CrossRef]
- Ahmed, S.S.; Soghaier, M.A.; Mohammed, S.; Khogali, H.S.; Osman, M.M.; Abdalla, A.M. Concomitant outbreaks of yellow fever and hepatitis E virus in Darfur States, Sudan, 2012. J. Infect. Dev. Ctries 2016, 10, 24–29. [Google Scholar] [CrossRef] [PubMed]
- Hamid, Z.; Hamid, T.; Alsedig, K.; Abdallah, T.; Elaagip, A.; Ahmed, A.; Khalid, F.; Abdel Hamid, M. Molecular Investigation of Dengue virus serotype 2 Circulation in Kassala State, Sudan. Jpn. J. Infect. Dis. 2019, 72, 58–61. [Google Scholar] [CrossRef] [PubMed]
- Soghaier, M.A.; Hagar, A.; Abbas, M.A.; Elmangory, M.M.; Eltahir, K.M.; Sall, A.A. Yellow Fever outbreak in Darfur, Sudan in October 2012; the initial outbreak investigation report. J. Infect. Public Health 2013, 6, 370–376. [Google Scholar] [CrossRef] [PubMed]

| State | Suspected Cases | Samples Collected (Percentage) | DENV | WNV | CCHFV | Negative |
|---|---|---|---|---|---|---|
| Central Darfur | 74 | 55 (74%) | 5 | 0 | 0 | 50 |
| East Darfur | 26 | 22 (85%) | 0 | 0 | 3 | 19 |
| North Darfur | 128 | 27 (21%) | 1 | 1 | 0 | 25 |
| South Darfur | 20 | 15 (75%) | 2 | 0 | 0 | 13 |
| West Darfur | 312 | 85 (27%) | 24 | 5 | 0 | 56 |
| Total | 560 | 204 (36%) | 32 | 6 | 3 | 163 |
| Age Group | DF | WNV | CCHF | F:M |
|---|---|---|---|---|
| 0–10 years | 12 | 0 | 0 | 1:2 |
| 11–25 years | 11 | 5 | 0 | 1.3:1 |
| 26–45 years | 8 | 1 | 3 | 1:1 |
| Elder than 46 years | 1 | 0 | 0 | 1:0 |
| Total | 32 | 6 | 3 | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahmed, A.; Elduma, A.; Magboul, B.; Higazi, T.; Ali, Y. The First Outbreak of Dengue Fever in Greater Darfur, Western Sudan. Trop. Med. Infect. Dis. 2019, 4, 43. https://doi.org/10.3390/tropicalmed4010043
Ahmed A, Elduma A, Magboul B, Higazi T, Ali Y. The First Outbreak of Dengue Fever in Greater Darfur, Western Sudan. Tropical Medicine and Infectious Disease. 2019; 4(1):43. https://doi.org/10.3390/tropicalmed4010043
Chicago/Turabian StyleAhmed, Ayman, Adel Elduma, Babiker Magboul, Tarig Higazi, and Yousif Ali. 2019. "The First Outbreak of Dengue Fever in Greater Darfur, Western Sudan" Tropical Medicine and Infectious Disease 4, no. 1: 43. https://doi.org/10.3390/tropicalmed4010043
APA StyleAhmed, A., Elduma, A., Magboul, B., Higazi, T., & Ali, Y. (2019). The First Outbreak of Dengue Fever in Greater Darfur, Western Sudan. Tropical Medicine and Infectious Disease, 4(1), 43. https://doi.org/10.3390/tropicalmed4010043





