Hemoptysis in the Immunocompromised Patient: Do Not Forget Strongyloidiasis
Abstract
:1. Introduction
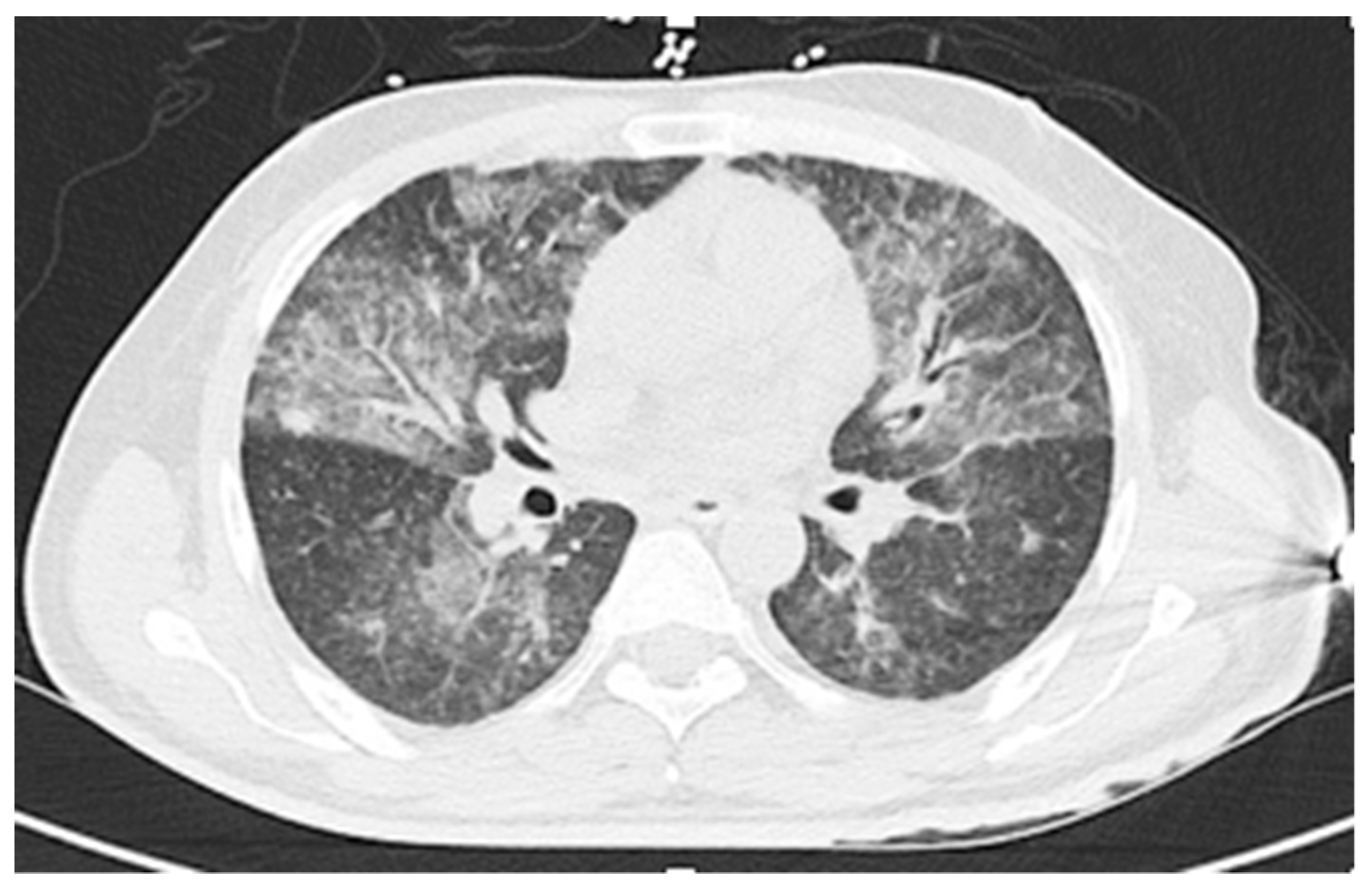
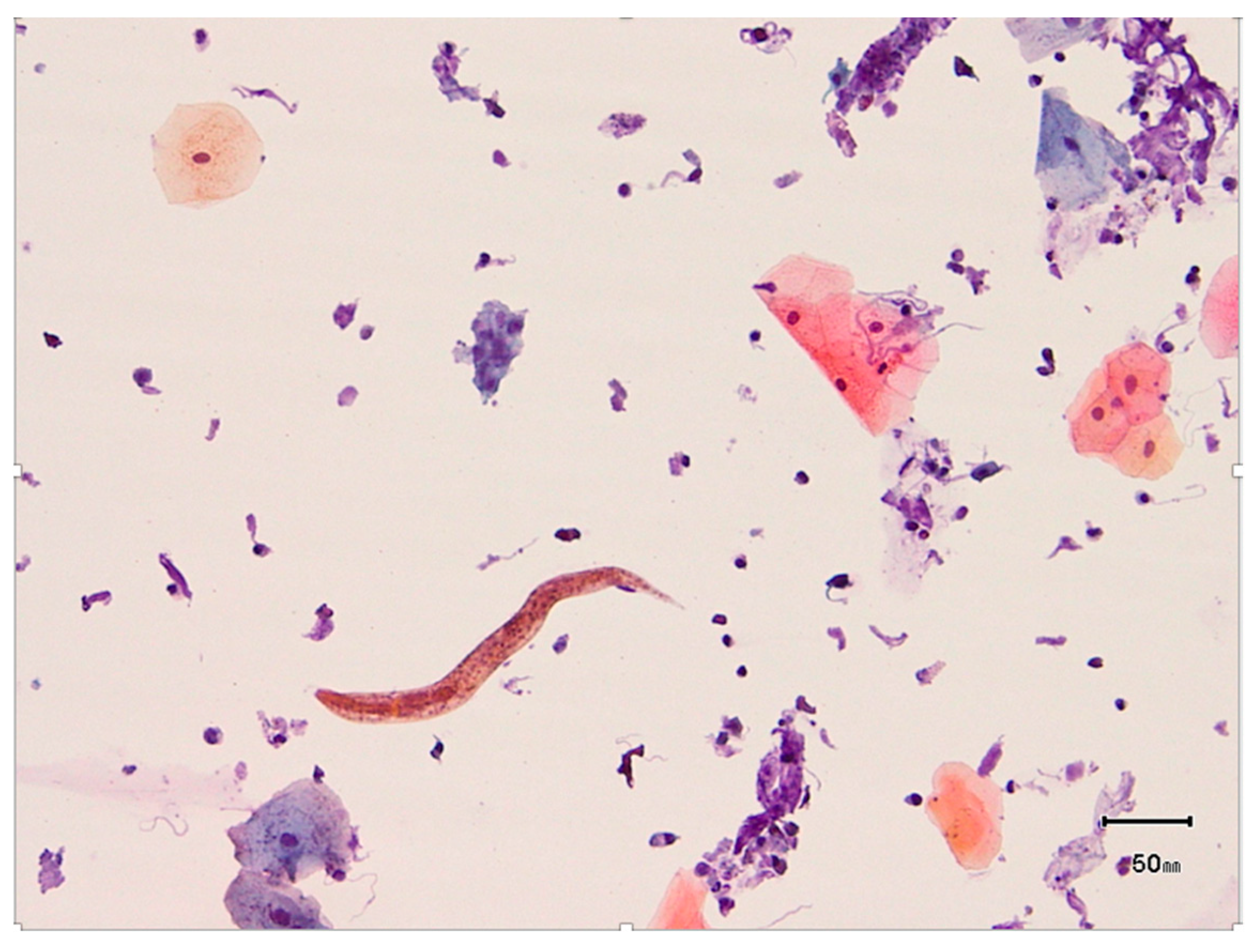
2. Case Presentations
2.1. Case 1
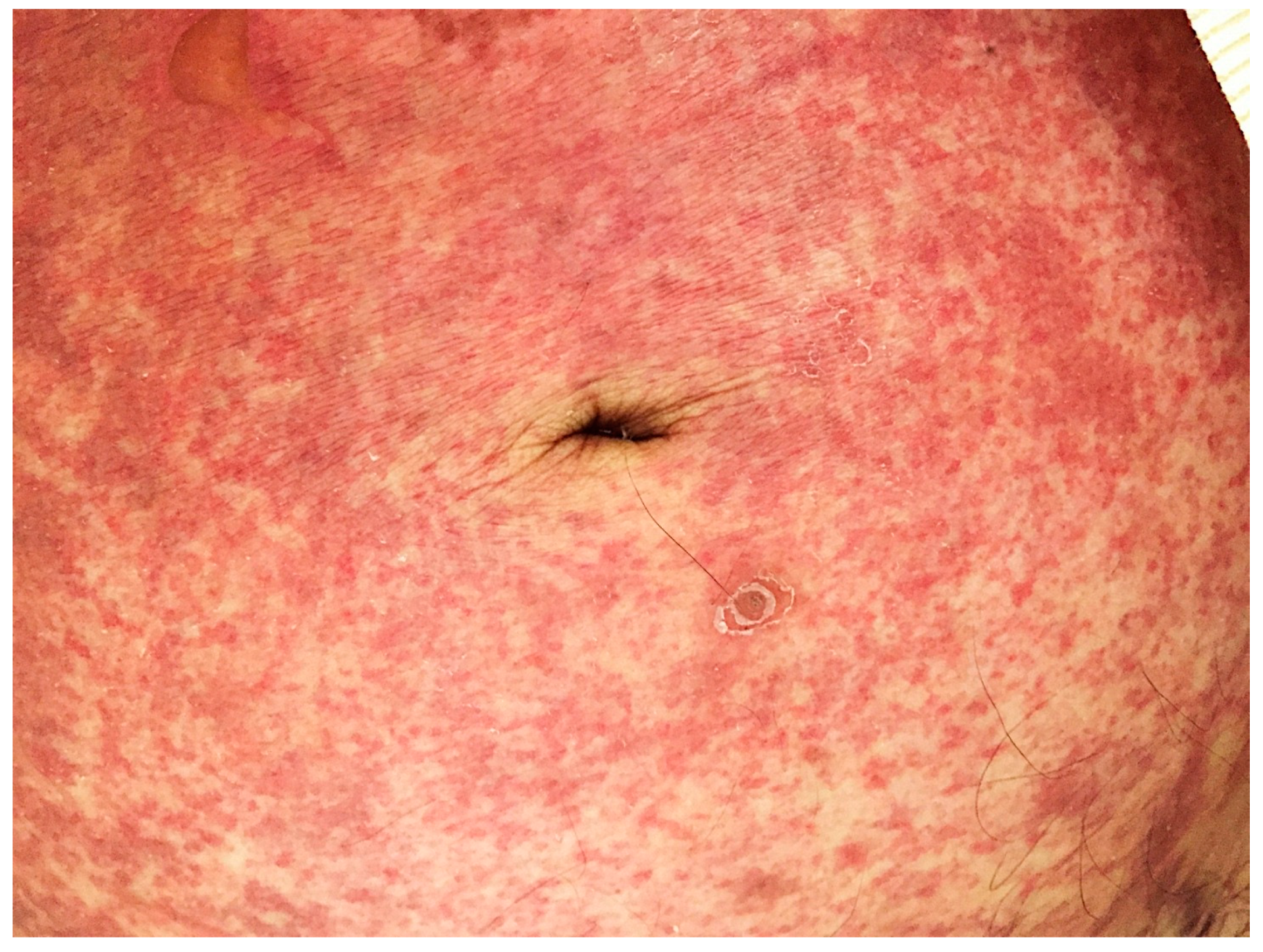
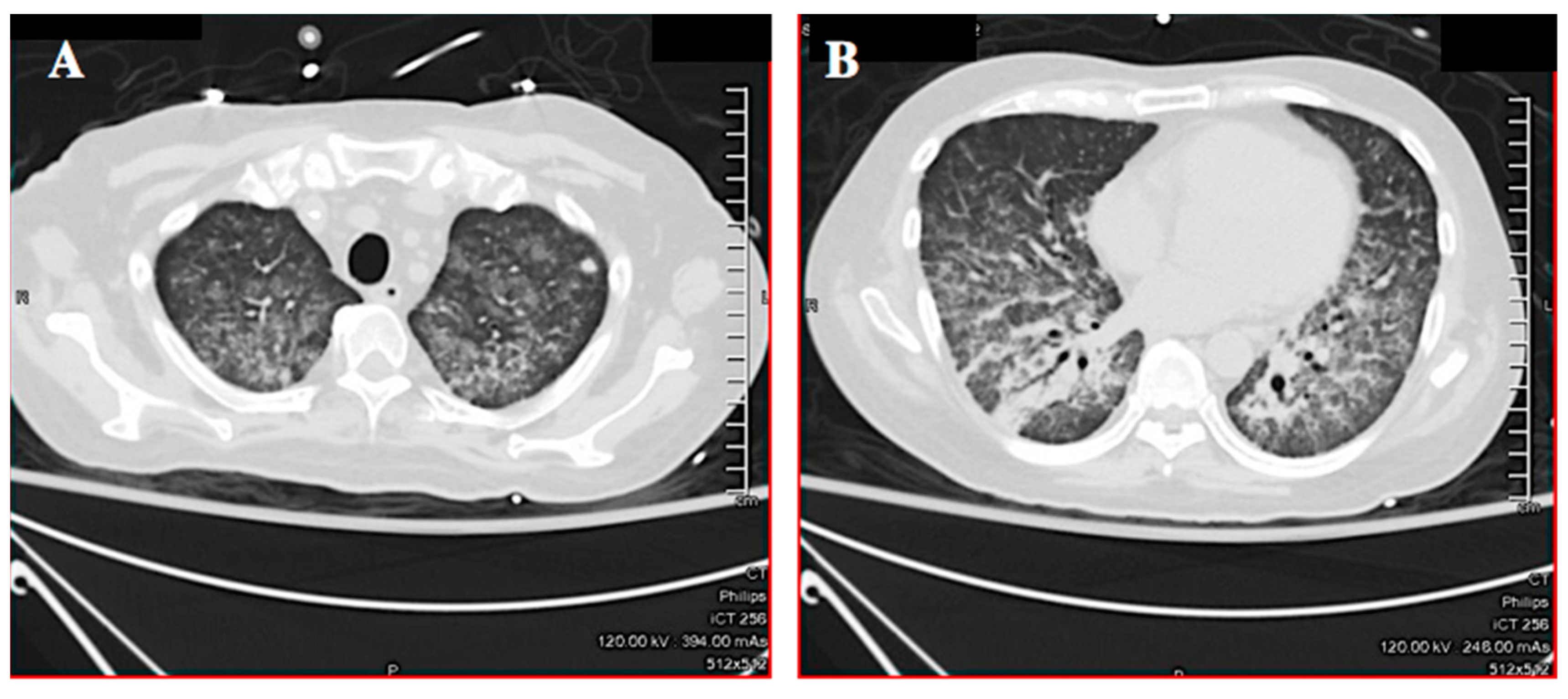
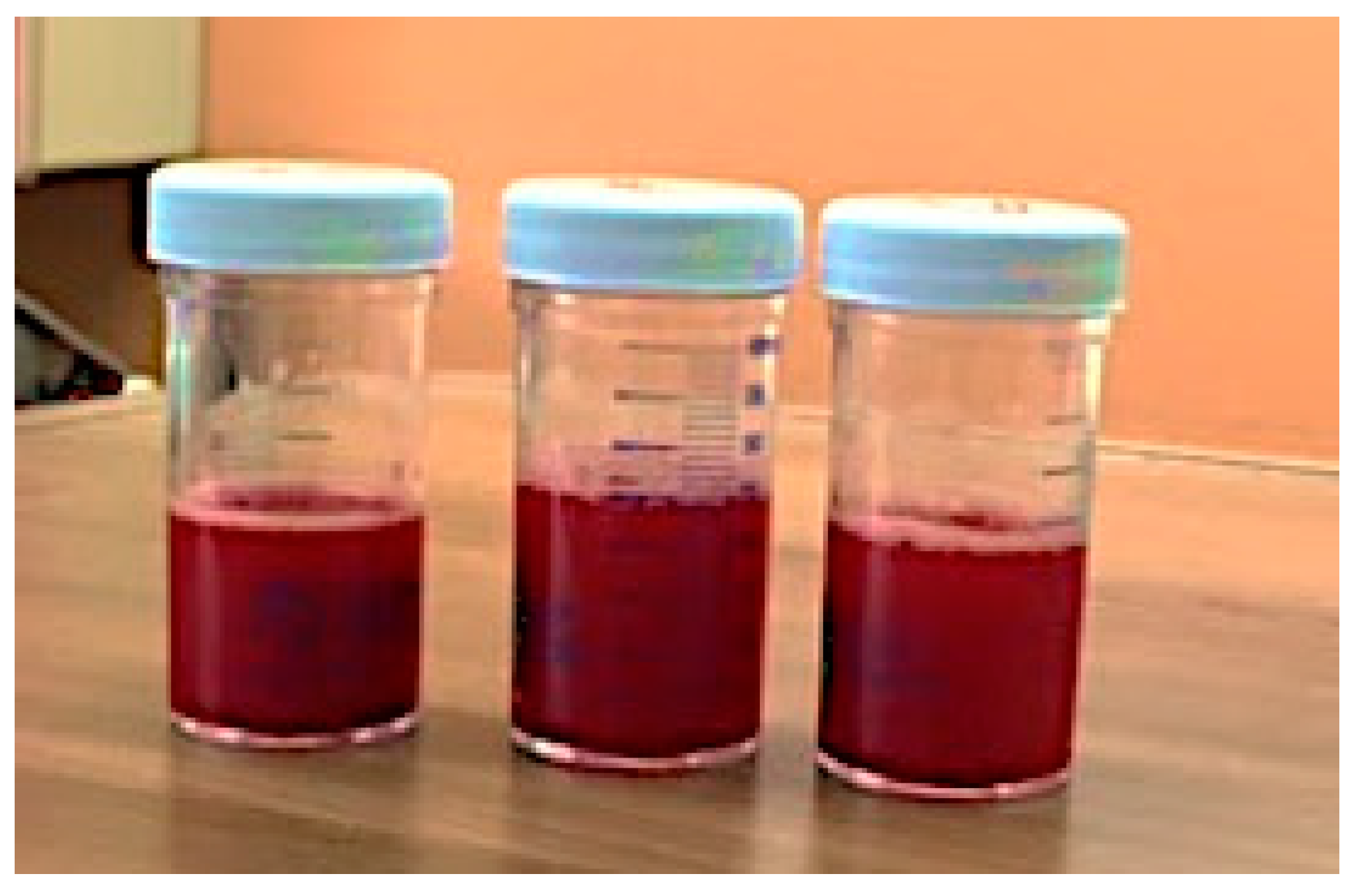
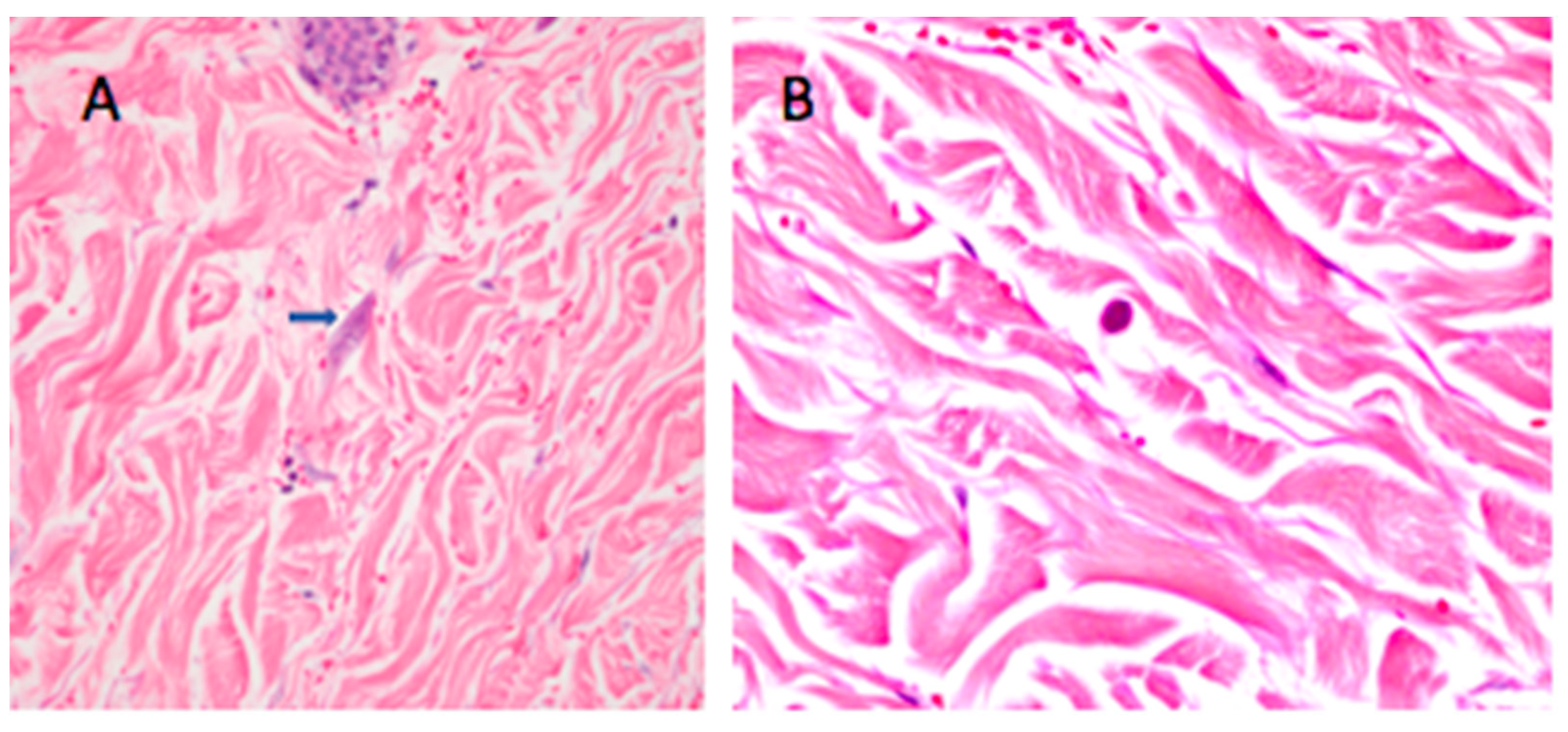
2.2. Case 2
3. Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bisoffi, Z.; Buonfrate, D.; Montresor, A.; Requena-Mendez, A.; Munoz, J.; Krolewiecki, A.J.; Gotuzzo, E.; Mena, M.A.; Chiodini, P.L.; Anselmi, M.; et al. Strongyloides stercoralis: A plea for action. PLoS Negl. Trop. Dis. 2013, 7, e2214. [Google Scholar] [CrossRef] [PubMed]
- Buonfrate, D.; Requena-Mendez, A.; Angheben, A.; Muñoz, J.; Gobbi, F.; Van Den Ende, J.; Bisoffi, Z. Severe strongyloidiasis: A systematic review of case reports. BMC Infect. Dis. 2013, 13, 78. [Google Scholar] [CrossRef] [PubMed]
- Genta, R.M. Global prevalence of strongyloidiasis: Critical review with epidemiologic insights into the prevention of disseminated disease. Rev. Infect. Dis. 1989, 11, 755–767. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, A.A.; Genta, R.M.; Maguilnik, I.; Berk, S.L. Strongyloidiasis. In Tropical Infectious Diseases: Principles, Pathogens, and Practice, 3rd ed.; Guerrant, R.L., Walker, D.H., Weller, P.F., Eds.; Churchill Livingstone: Philadelphia, PA, USA, 2010; pp. 805–812. [Google Scholar]
- Grove, D.I. Human strongyloidiasis. Adv. Parasitol. 1996, 38, 251–309. [Google Scholar] [PubMed]
- Croker, C.; Reporter, R.; Redelings, M.; Mascola, L. Strongyloidiasis-related deaths in the United States, 1991–2006. Am. J. Trop. Med. Hyg. 2010, 83, 422–426. [Google Scholar] [CrossRef] [PubMed]
- Posey, D.L.; Blackburn, B.G.; Weinberg, M.; Flagg, E.W.; Ortega, L.; Wilson, M.; Secor, W.E.; Sanders-Lewis, K.; Won, K.; Maguire, J.H. High prevalence and presumptive treatment of schistosomiasis and strongyloidiasis among African refugees. Clin. Infect. Dis. 2007, 45, 1310–1315. [Google Scholar] [CrossRef] [PubMed]
- Nuesch, R.; Zimmerli, L.; Stockli, R.; Gyr, N.; Christoph Hatz, F.R. Imported strongyloidosis: A longitudinal analysis of 31 cases. J. Travel Med. 2005, 12, 80–84. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Parasites—Strongyloides. Available online: http://www.cdc.gov/parasites/strongyloides/biology.html (accessed on 4 June 2018).
- Greaves, D.; Coggle, S.; Pollard, C.; Aliyu, S.H.; Moore, E.M. Strongyloides stercoralis infection. BMJ 2013, 347, f4610. [Google Scholar] [CrossRef] [PubMed]
- Prendki, V.; Fenaux, P.; Durand, R.; Thellier, M.; Bouchaud, O. Strongyloidiasis in man 75 years after initial exposure. Emerg. Infect. Dis. 2011, 17, 931–932. [Google Scholar] [CrossRef] [PubMed]
- Salluh, J.I.; Bozza, F.A.; Pinto, T.S.; Toscano, L.; Weller, P.F.; Soares, M. Cutaneous periumbilical purpura in disseminated strongyloidiasis in cancer patients: A pathognomonic feature of potentially lethal disease? Braz. J. Infect. Dis. 2005, 9, 419–424. [Google Scholar] [CrossRef] [PubMed]
- Larici, A.R.; Franchi, P.; Occhipinti, M.; Contegiacomo, A.; del Ciello, A.; Calandriello, L.; Storto, M.L.; Marano, R.; Bonomo, L. Diagnosis and management of hemoptysis. Diagn. Intervent. Radiol. 2014, 20, 299–309. [Google Scholar] [CrossRef] [PubMed]
- von Ranke, F.M.; Zanetti, G.; Hochhegger, B.; Marchiori, E. Infectious diseases causing diffuse alveolar hemorrhage in immunocompetent patients: A state-of-the-art review. Lung 2013, 191, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Woodring, J.H.; Halfhill, H., 2nd; Berger, R.; Reed, J.C.; Moser, N. Clinical and imaging features of pulmonary strongyloidiasis. South Med. J. 1996, 89, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Setoyama, M.; Fukumaru, S.; Takasaki, T.; Yoshida, H.; Kanzaki, T. SLE with death from acute massive pulmonary hemorrhage caused by disseminated strongyloidiasis. Scand. J. Rheumatol. 1997, 26, 389–391. [Google Scholar] [CrossRef] [PubMed]
- El-Sameed, Y.A.; Beejay, N.; Al Maashari, R. Diffuse alveolar haemorrhage and severe hypoxemia from Strongyloides stercoralis hyperinfection syndrome. Clin. Respir. J. 2015, 9, 489–492. [Google Scholar] [CrossRef] [PubMed]
- Patel, T.; Singh, R.; Reddy, V.; Hodowanec, A.; Singh, K. Photo quiz: Sepsis, confusion, rash, and pulmonary hemorrhage in a 36-year-old man with lymphoma. J. Clin. Microbiol. 2015, 53, 758. [Google Scholar] [CrossRef]
- Keiser, P.B.; Nutman, T.B. Strongyloides stercoralis in the immunocompromised population. Clin. Microbiol. Rev. 2004, 17, 208–217. [Google Scholar] [CrossRef] [PubMed]
- Fardet, L.; Généreau, T.; Cabane, J.; Kettaneh, A. Severe strongyloidiasis in corticosteroid-treated patients. Clin. Microbiol. Infect. 2006, 12, 945–947. [Google Scholar] [CrossRef] [PubMed]
- Weiser, J.A.; Scully, B.E.; Bulman, W.A.; Husain, S.; Grossman, M.E. Periumbilical parasitic thumbprint purpura: Strongyloides hyperinfection syndrome acquired from a cadaveric renal transplant. Transplant Infect. Dis. 2011, 13, 58–62. [Google Scholar] [CrossRef] [PubMed]
- Zaha, O.; Hirata, T.; Kinjo, F.; Saito, A. Strongyloidiasis-progress in diagnosis and treatment. Intern. Med. 2000, 39, 695–700. [Google Scholar] [CrossRef] [PubMed]
- Geri, G.; Rabbat, A.; Mayaux, J.; Zafrani, L.; Chalumeau-Lemoine, L.; Guidet, B.; Azoulay, E.; Pène, F. Strongyloides stercoralis hyperinfection syndrome: A case series and a review of the literature. Infection 2015, 43, 691–698. [Google Scholar] [CrossRef] [PubMed]
- Mejia, R.; Nutman, T.B. Screening, prevention, and treatment for hyperinfection syndrome and disseminated infections caused by Strongyloides stercoralis. Curr. Opin. Infect. Dis. 2012, 25, 458–463. [Google Scholar] [CrossRef] [PubMed]
- Checkley, A.M.; Chiodini, P.L.; Dockrell, D.H.; Bates, I.; Thwaites, G.E.; Booth, H.L.; Brown, M.; Wright, S.G.; Grant, A.D.; Mabey, D.C.; et al. British Infection Society and Hospital for Tropical Diseases. Eosinophilia in returning travellers and migrants from the tropics: UK recommendations for investigation and initial management. J. Infect. 2010, 60, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Santiago, M.; Leitão, B. Prevention of strongyloides hyperinfection syndrome: A rheumatological point of view. Eur. J. Intern. Med. 2009, 20, 744–748. [Google Scholar] [CrossRef] [PubMed]
- Genta, R.M. Dysregulation of strongyloidiasis: A new hypothesis. Clin. Microbiol. Rev. 1992, 5, 345–355. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, P.B.; Mojon, M. Improved diagnosis of Strongyloides stercoralis by seven consecutive stool specimens. Zentralbl. Bakteriol. Mikrobiol. Hyg. A 1987, 263, 616–618. [Google Scholar] [CrossRef]
- Loutfy, M.R.; Wilson, M.; Keystone, J.S.; Kain, K.C. Serology and eosinophil count in the diagnosis and management of strongyloidiasis in a non-endemic area. Am. J. Trop. Med. Hyg. 2002, 66, 749–752. [Google Scholar] [CrossRef] [PubMed]
- Albonico, M.; Becker, S.L.; Odermatt, P.; Angheben, A.; Anselmi, M.; Amor, A.; Barda, B.; Buonfrate, D.; Cooper, P.; Gétaz, L.; et al. StrongNet: An international network to improve diagnostics and access to treatment for strongyloidiasis control. PLoS Negl. Trop. Dis. 2016, 10, e0004898. [Google Scholar] [CrossRef] [PubMed]
- Valerio, L.; Roure, S.; Fernández-Rivas, G.; Basile, L.; Martínez-Cuevas, O.; Ballesteros, Á.L.; Ramos, X.; Sabrià, M. North Metropolitan Working Group on Imported Diseases. Strongyloides stercoralis, the hidden worm. Epidemiological and clinical characteristics of 70 cases diagnosed in the North Metropolitan Area of Barcelona, Spain, 2003–2012. Trans. R. Soc. Trop. Med. Hyg. 2013, 107, 465–470. [Google Scholar] [PubMed]
- Newberry, A.M.; Williams, D.N.; Stauffer, W.M.; Boulware, D.R.; Hendel-Paterson, B.R.; Walker, P.F. Strongyloides hyperinfection presenting as acute respiratory failure and gram-negative sepsis. Chest 2005, 128, 3681–3684. [Google Scholar] [CrossRef] [PubMed]
- Luvira, V.; Trakulhun, K.; Mungthin, M.; Naaglor, T.; Chantawat, N.; Pakdee, W.; Phiboonbanakit, D.; Dekumyoy, P. Comparative Diagnosis of Strongyloidiasis in Immunocompromised Patients. Am. J. Trop. Med. Hyg. 2016, 95, 401–414. [Google Scholar] [CrossRef] [PubMed]
- Repetto, S.A.; Ruybal, P.; Batalla, E.; López, C.; Fridman, V.; Sierra, M.; Radisic, M.; Bravo, P.M.; Risso, M.G.; González Cappa, S.M.; et al. Strongyloidiasis outside endemic areas: Long-term parasitological and clinical follow-up after ivermectin treatment. Clin. Infect. Dis. 2018, 66, 1558–1565. [Google Scholar] [CrossRef] [PubMed]
- Suputtamongkol, Y.; Premasathian, N.; Bhumimuang, K.; Waywa, D.; Nilganuwong, S.; Karuphong, E.; Anekthananon, T.; Wanachiwanawin, D.; Silpasakorn, S. Efficacy and safety of single and double doses of ivermectin versus 7-day high dose albendazole for chronic strongyloidiasis. PLoS Negl. Trop. Dis. 2011, 5, e1044. [Google Scholar] [CrossRef] [PubMed]
- Henriquez-Camacho, C.; Gotuzzo, E.; Echevarria, J.; White, A.C., Jr.; Terashima, A.; Samalvides, F.; Pérez-Molina, J.A.; Plana, M.N. Ivermectin versus albendazole or thiabendazole for Strongyloides stercoralis infection. Cochrane Database Syst. Rev. 2016, 1, CD007745. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Perez, A.; Díez, S.R.; Belhasen-Garcia, M.; Torrús-Tendero, D.; Perez-Arellano, J.L.; Cabezas, T.; Soler, C.; Díaz-Menéndez, M.; Navarro, M.; Treviño, B.; et al. Management of severe strongyloidiasis attended at reference centers in Spain. PLoS Negl. Trop. Dis. 2018, 12, e0006272. [Google Scholar] [CrossRef] [PubMed]
- Pornsuriyasak, P.; Niticharoenpong, K.; Sakapibunnan, A. Disseminated strongyloidiasis successfully treated with extended duration ivermectin combined with albendazole: A case report of intractable strongyloidiasis. Southeast Asian J. Trop. Med. Public Health 2004, 35, 531–534. [Google Scholar] [PubMed]
- Tarr, P.E.; Miele, P.S.; Peregoy, K.S.; Smith, M.A.; Neva, F.A.; Lucey, D.R. Case report: Rectal administration of ivermectin to a patient with Strongyloides hyperinfection syndrome. Am. J. Trop. Med. Hyg. 2003, 68, 453–455. [Google Scholar] [CrossRef] [PubMed]
- Bogoch, I.I.; Khan, K.; Abrams, H.; Nott, C.; Leung, E.; Fleckenstein, L.; Keystone, J.S. Failure of ivermectin per rectum to achieve clinically meaningful serum levels in two cases of Strongyloides hyperinfection. Am. J. Trop. Med. Hyg. 2015, 93, 94–96. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shrestha, P.; O’Neil, S.E.; Taylor, B.S.; Bode-Omoleye, O.; Anstead, G.M. Hemoptysis in the Immunocompromised Patient: Do Not Forget Strongyloidiasis. Trop. Med. Infect. Dis. 2019, 4, 35. https://doi.org/10.3390/tropicalmed4010035
Shrestha P, O’Neil SE, Taylor BS, Bode-Omoleye O, Anstead GM. Hemoptysis in the Immunocompromised Patient: Do Not Forget Strongyloidiasis. Tropical Medicine and Infectious Disease. 2019; 4(1):35. https://doi.org/10.3390/tropicalmed4010035
Chicago/Turabian StyleShrestha, Prakash, Sean E. O’Neil, Barbara S. Taylor, Olaoluwa Bode-Omoleye, and Gregory M. Anstead. 2019. "Hemoptysis in the Immunocompromised Patient: Do Not Forget Strongyloidiasis" Tropical Medicine and Infectious Disease 4, no. 1: 35. https://doi.org/10.3390/tropicalmed4010035
APA StyleShrestha, P., O’Neil, S. E., Taylor, B. S., Bode-Omoleye, O., & Anstead, G. M. (2019). Hemoptysis in the Immunocompromised Patient: Do Not Forget Strongyloidiasis. Tropical Medicine and Infectious Disease, 4(1), 35. https://doi.org/10.3390/tropicalmed4010035





