Evolution of Tuberculosis/Human Immunodeficiency Virus Services among Different Integrated Models in Myanmar: A Health Services Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Data Sources
2.2. Data Analysis
2.3. Ethics
3. Results
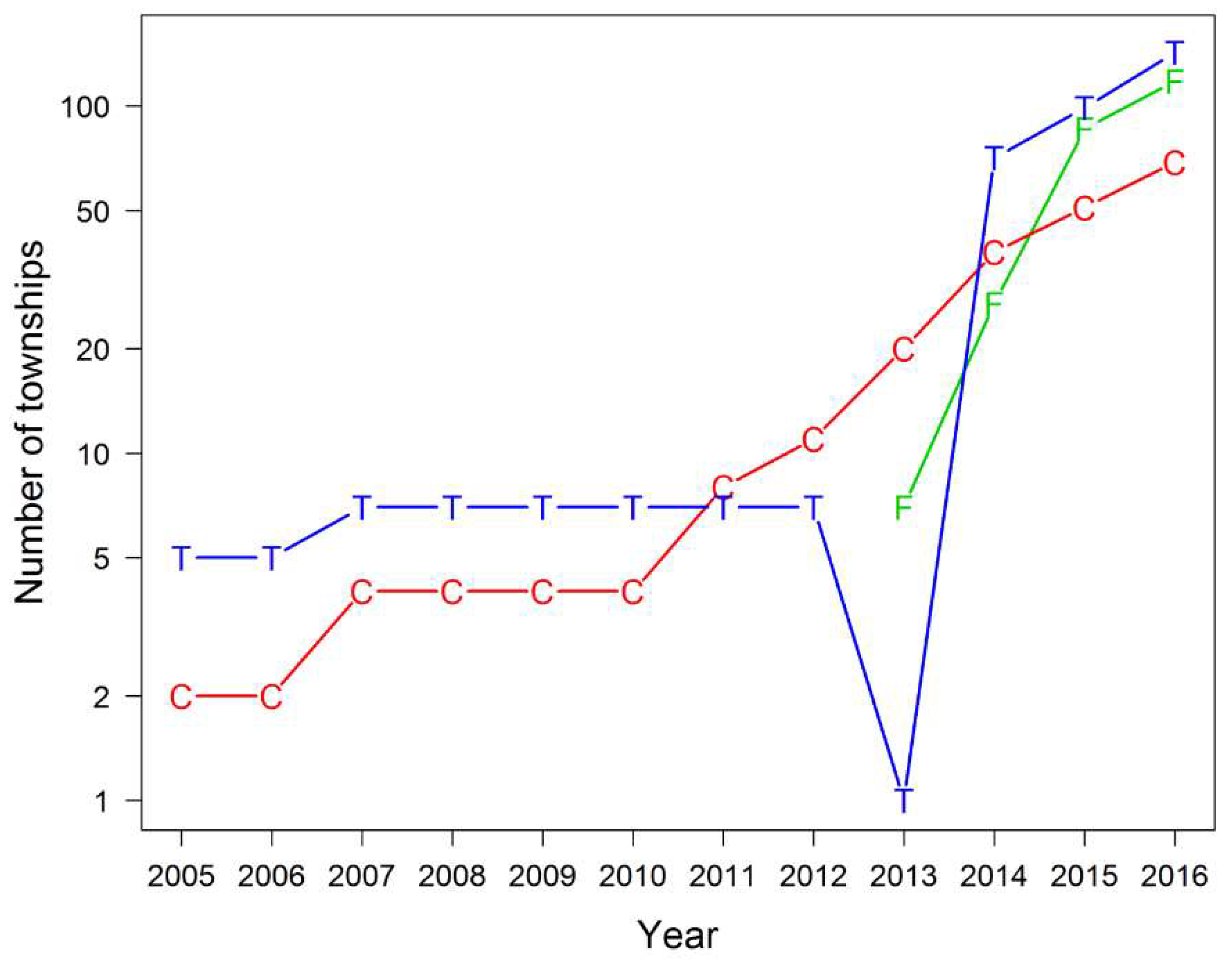
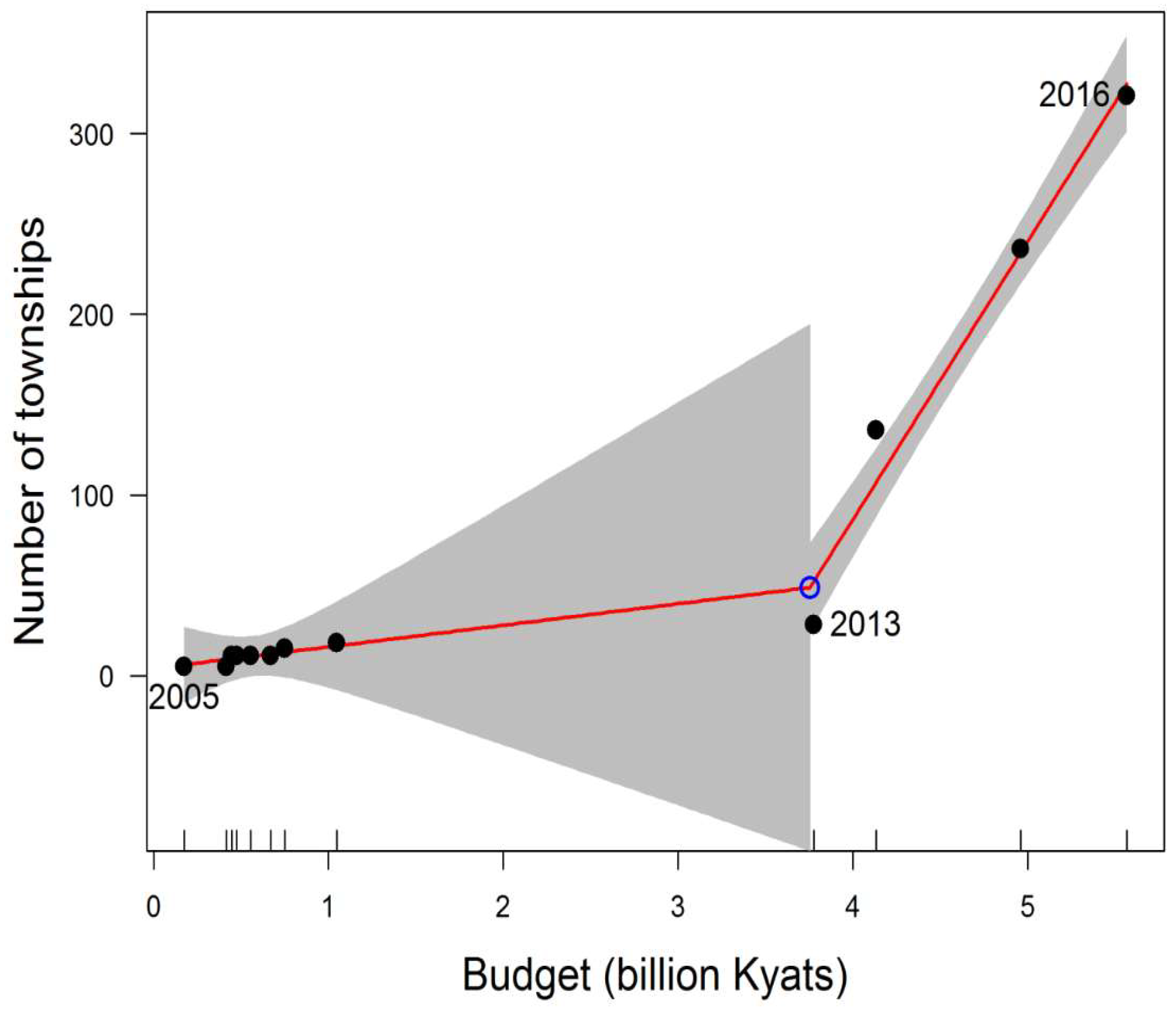
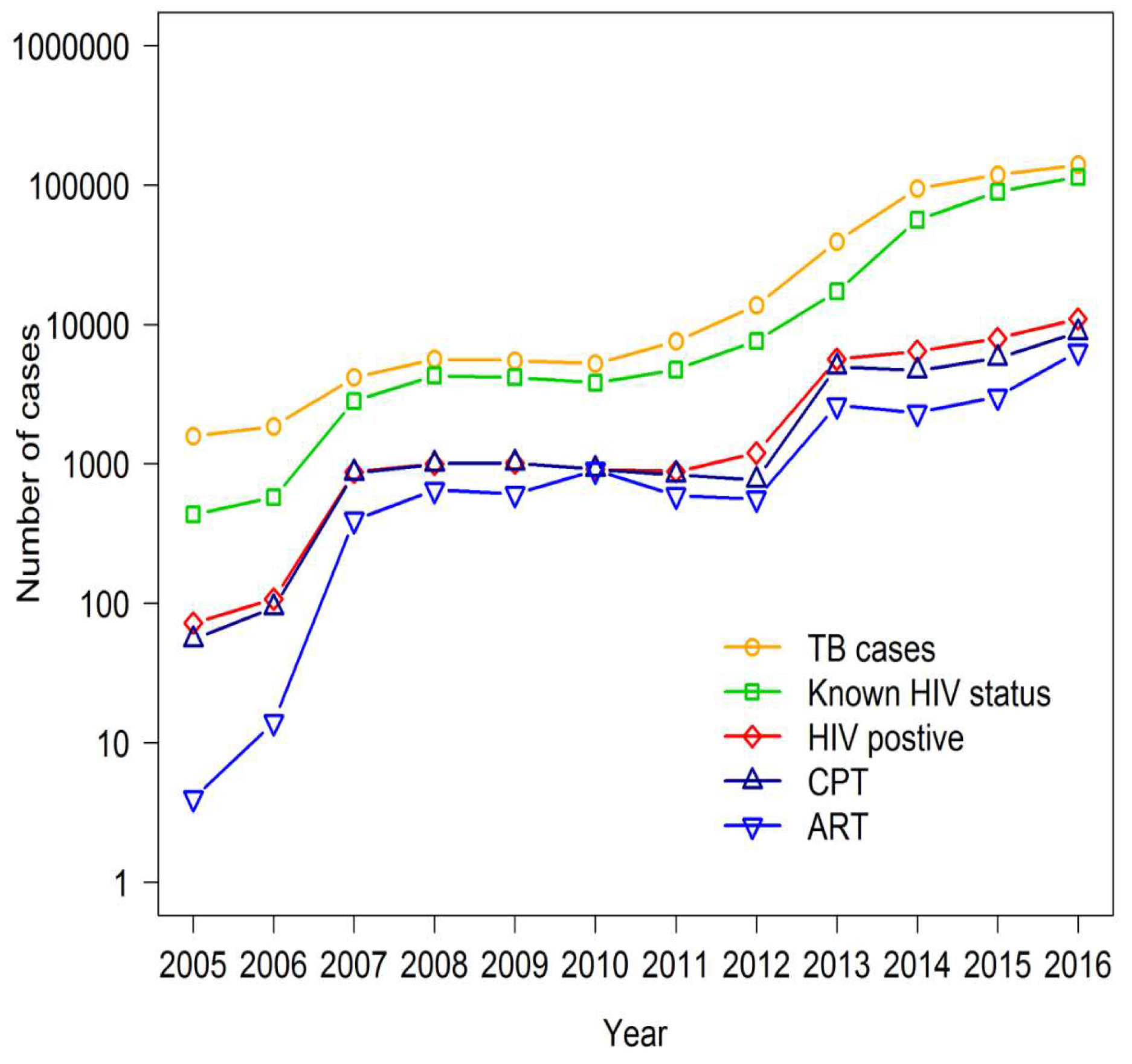
3.1. Evolution of TB/HIV Services
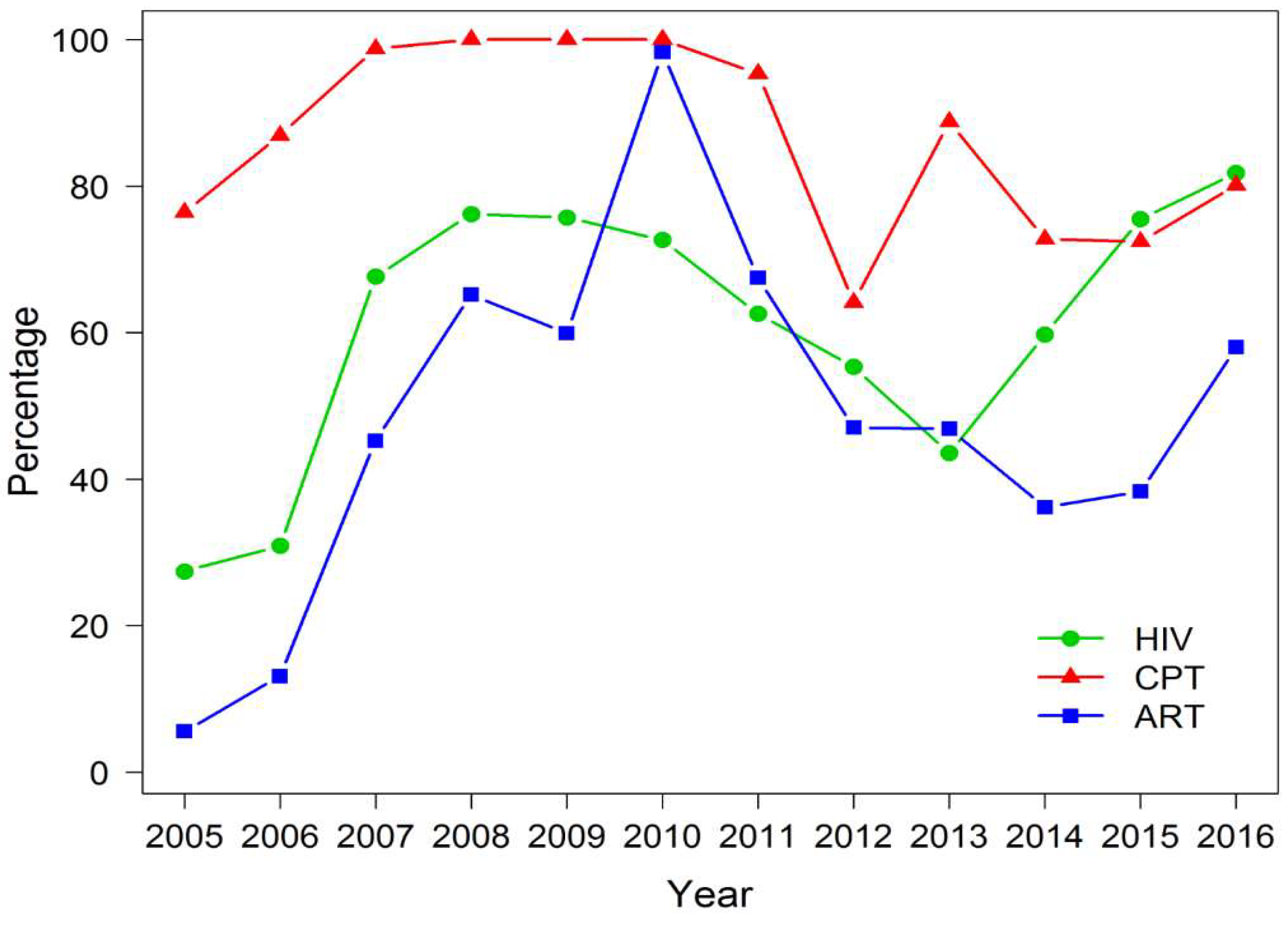
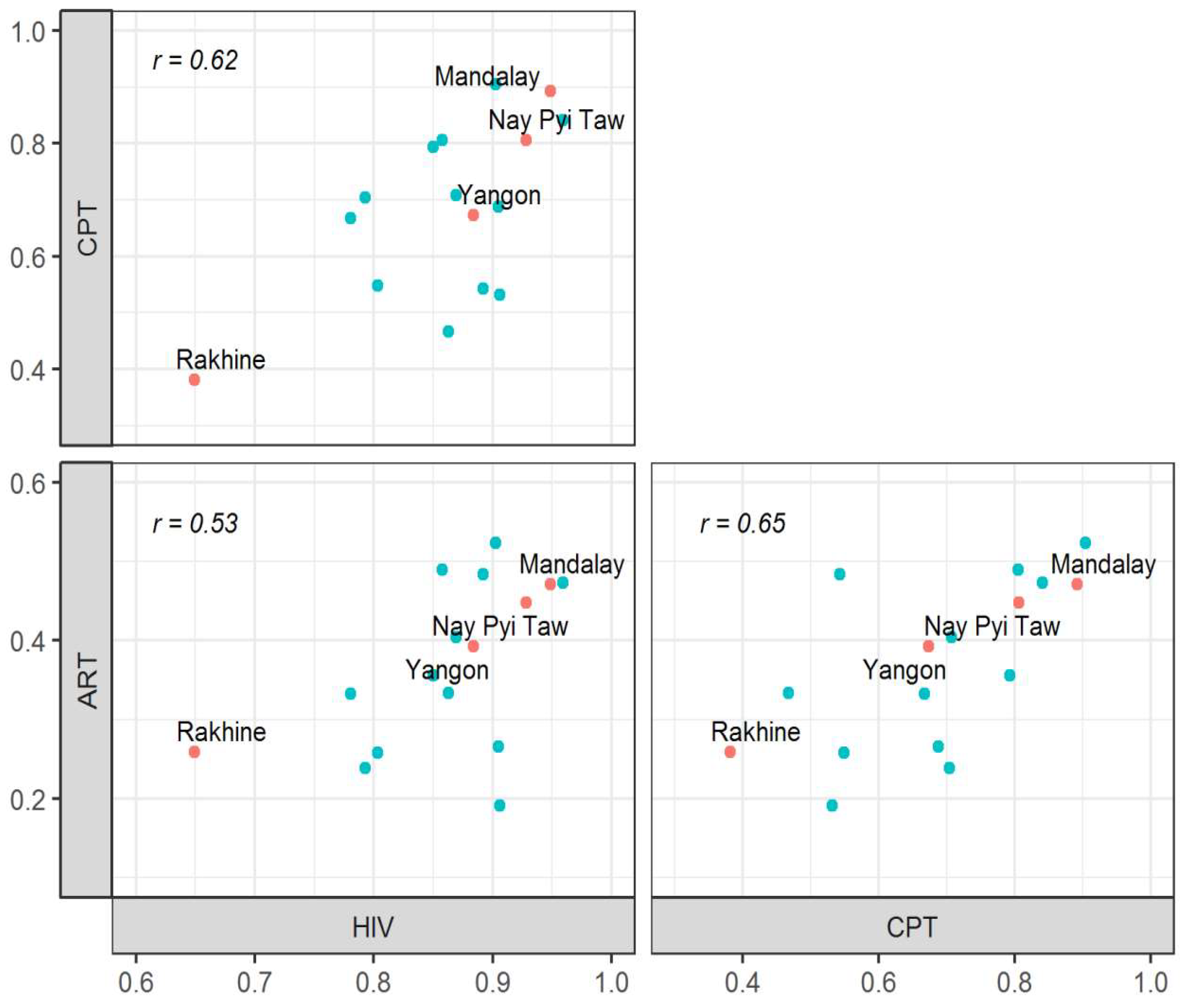
3.2. Performance of TB/HIV Services
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- National TB Programme, Myanmar. Report on National Prevalent Survey 2009–2010, Myanmar. 2010. Available online: http://www.searo.who.int/myanmar/documents/TB_Prevelence_Survey_report.pdf (accessed on 7 December 2011).
- National AIDS Programme, Myanmar. National Strategic Plan on HIV and AIDS, Myanmar (2016–2020). Available online: https://www.aidsdatahub.org/sites/default/files/highlight-reference/document/Myanmar_National_Strategic_Plan_on_HIV_and_AIDS_2016-2020.pdf (accessed on 12 December 2016).
- Annual Report 2016, National Tuberculosis Programme Myanmar. National TB Programme, Myanmar. 2018. Available online: https://myanmarhscc.org/wp-content/uploads/2016/12/2008.pdf (accessed on 12 December 2016).
- World Health Organization. WHO Policy on Collaborative TB/HIV Activities: Guidelines for National Programmes and Other Stakeholders; WHO: Geneva, Switzerland, 2012. [Google Scholar]
- Pfeiffer, J.; Montoya, P.; Baptista, A.J.; Karagianis, M.; Pugas Mde, M.; Micek, M.; Johnson, W.; Sherr, K.; Gimbel, S.; Baird, S.; et al. Integration of HIV/AIDS services into African primary health care: Lessons learned for health system strengthening in Mozambique—A case study. J. Int. AIDS Soc. 2010, 13, 3. [Google Scholar] [CrossRef] [PubMed]
- Owiti, P.; Zachariah, R.; Bissell, K.; Kumar, A.M.V.; Diero, L.; Carter, E.J.; Gardner, A. Integrating tuberculosis and HIV services in rural Kenya: Uptake and outcomes. Public Health Action 2015, 5, 36–44. [Google Scholar] [CrossRef] [PubMed]
- Hermans, S.M.; Castelnuovo, B.; Katabira, C.; Mbidde, P.; Lange, J.M.A.; Hoepelman, A.I.M.; Coutinho, A.; Manabe, Y.C. Integration of HIV and TB Services Results in Improved TB Treatment Outcomes and Earlier Prioritized ART Initiation in a Large Urban HIV Clinic in Uganda. J. Acquir. Immune Defic. Syndr. 2012, 60, e29–e35. [Google Scholar] [CrossRef] [PubMed]
- Kerschberger, B.; Hilderbrand, K.; Boulle, A.M.; Coetzee, D.; Goemaere, E.; De Azevedo, V.; Cutsem, G.V. The Effect of Complete Integration of HIV and TB Services on Time to Initiation of Antiretroviral Therapy: A Before-After Study. PLoS ONE 2012, 7, e46988. [Google Scholar] [CrossRef] [PubMed]
- Pathmanathan, I.; Pasipamire, M.; Pals, S.; Dokubo, E.K.; Preko, P.; Ao, T.; Mazibuko, S.; Ongole, J.; Dhlamini, T.; Haumba, S. High uptake of antiretroviral therapy among HIV-positive TB patients receiving co-located services in Swaziland. PLoS ONE 2018, 13, e0196831. [Google Scholar] [CrossRef] [PubMed]
- Nash, D.; Tymejczyk, O.; Gadisa, T.; Kulkarni, S.G.; Hoffman, S.; Yigzaw, M.; Elul, B.; Remien, R.H.; Lahuerta, M.; Daba, S.; et al. Factors associated with initiation of antiretroviral therapy in the advanced stages of HIV infection in six Ethiopian HIV clinics, 2012 to 2013. J. Int. AIDS Soc. 2016, 19, 20637. [Google Scholar] [CrossRef] [PubMed]
- Phiri, S.; Khan, P.Y.; Grant, A.D.; Gareta, D.; Tweya, H.; Kalulu, M.; Chaweza, T.; Mbetewa, L.; Kanyerere, H.; Weigel, R.; et al. Integrated tuberculosis and HIV care in a resource-limited setting: Experience from the Martin Preuss centre, Malawi: Integrated TB and HIV care in Malawi. Trop. Med. Int. Health 2011, 16, 1397–1403. [Google Scholar] [CrossRef]
- Kapata, N.; Chanda-Kapata, P.; Grobusch, M.P.; O’Grady, J.; Schwank, S.; Bates, M.; Jansenn, S.; Mwinga, A.; Cobelens, F.; Mwaba, P.; et al. Scale-up of TB and HIV programme collaborative activities in Zambia—A 10-year review: Scale-up of TB and HIV programme collaborative activities. Trop. Med. Int. Health 2012, 17, 760–766. [Google Scholar] [CrossRef]
- Pevzner, E.S.; Vandebriel, G.; Lowrance, D.W.; Gasana, M.; Finlay, A. Evaluation of the Rapid Scale-up of Collaborative TB/HIV Activities in TB Facilities in Rwanda, 2005–2009. BMC Public Health 2011, 11, 550. [Google Scholar] [CrossRef]
- Ansa, G.A.; Walley, J.D.; Siddiqi, K.; Wei, X. Assessing the impact of TB/HIV services integration on TB treatment outcomes and their relevance in TB/HIV monitoring in Ghana. Infect. Dis. Poverty 2012, 1, 13. [Google Scholar] [CrossRef]
- Schulz, S.A.; Draper, H.R.; Naidoo, P. A comparative study of tuberculosis patients initiated on ART and receiving different models of TB-HIV care. Int. J. Tuberc. Lung Dis. 2013, 17, 1558–1563. [Google Scholar] [CrossRef] [PubMed]
- Miti, S.; Mfungwe, V.; Reijer, P.; Maher, D. Integration of tuberculosis treatment in a community-based home care programme for persons living with HIV/AIDS in Ndola, Zambia. Int. J. Tuberc. Lung Dis. 2003, 7, S92–S98. [Google Scholar] [PubMed]
- Burnett, S.M.; Zawedde-Muyanja, S.; Hermans, S.M.; Weaver, M.R.; Colebunders, R.; Manabe, Y.C. Effect of TB/HIV Integration on TB and HIV Indicators in Rural Ugandan Health Facilities. J. Acquir. Immune Defic. Syndr. 2018, 79, 7. [Google Scholar] [CrossRef] [PubMed]
- Friedland, G.; Harries, A.; Coetzee, D. Implementation Issues in Tuberculosis/HIV Program Collaboration and Integration: 3 Case Studies. J. Infect. Dis. 2007, 196, S114–S123. [Google Scholar] [CrossRef] [PubMed]
- Theingi, P.; Harries, A.D.; Wai, K.T.; Shewade, H.D.; Saw, S.; Win, T.; Thein, S.; Kyi, M.S.; Nyunt Oo, H.; Aung, S.T. National scale-up of tuberculosis–human immunodeficiency virus collaborative activities in Myanmar from 2005 to 2016 and tuberculosis treatment outcomes for patients with human immunodeficiency virus-positive tuberculosis in the Mandalay Region in 2015. Trans. R. Soc. Trop. Med. Hyg. 2017, 111, 402–409. [Google Scholar] [CrossRef] [PubMed]
- National TB Programme, Myanmar, National Strategic Plan for Tuberculosis, Myanmar (2016–2020). Available online: https://www.aidsdatahub.org/sites/default/files/publication/Myanmar_National_Strategic_Plan_for_Tuberculosis_2016-2020.pdf (accessed on 22 December 2016).
- Kunii, O.; Yassin, M.A.; Wandwalo, E. Investing to end epidemics: The role of the Global Fund to control TB by 2030. Trans. R. Soc. Trop. Med. Hyg. 2016, 110, 153–154. [Google Scholar] [CrossRef] [PubMed]
- National TB Programme, Myanmar, National AIDS programme, Myanmar. Guidelines for the Programmatic management of TB/HIV in Myanmar. Available online: https://www.aidsdatahub.org/sites/default/files/toolandguide/document/Myanmar_Guidelines_for_the_Management_of_Multidrug-Resistant_Tuberculosis_2017.pdf (accessed on 15 February 2017).
- Kanchar, A.; Getahun, H.; Joint United Nations Programme on HIV/AIDS; United States, President’s Emergency Plan for AIDS Relief; WHO Global TB Programme Ed. A Guide to Monitoring and Evaluation for Collaborative TB/HIV Activities; WHO: Geneva, Switzerland, 2015. [Google Scholar]
- Kim, H.J.; Fay, M.P.; Feuer, E.J.; Midthune, D.N. Permutation tests for joinpoint regression with applications to cancer rates. Stat. Med. 2000, 19, 335–351. [Google Scholar] [CrossRef]
- Pagan, E.; Rea, F.; Compagnoni, M.M.; Cantarutti, A.; Pugni, P.; Bagnardi, V.; Corrao, G. Joinpoint Regression Analysis with Time-on-Study as Time-Scale. Application to Three ITALIAN Population-Based Cohort Studies; Epidemiol Biostat and Public Health: Milano, Italy, 2017. [Google Scholar]
- Organisation Mondiale de la Santé. Global Tuberculosis Report 2017; Organisation Mondiale de la Santé: Geneva, Switzerland, 2017. [Google Scholar]
- Legido-Quigley, H.; Montgomery, C.M.; Khan, P.; Atun, R.; Fakoya, A.; Getahun, H.; Grant, A.D. Integrating tuberculosis and HIV services in low- and middle-income countries: A systematic review. Trop. Med. Int. Health 2013, 18, 199–211. [Google Scholar] [CrossRef] [PubMed]
- Wandwalo, E.; Kapalata, N.; Tarimo, E.; Corrigan, C.B.; Morkve, O. Collaboration between the national tuberculosis programme and a non governmental organisation in TB/HIV care at a district level: Experience from Tanzania. Afr. Health Sci. 2004, 4, 6. [Google Scholar]
- Dong, K.; Thabethe, Z.; Hurtado, R.; Sibaya, T.; Dlwati, H.; Walker, B.; Wilson, D. Challenges to the Success of HIV and Tuberculosis Care and Treatment in the Public Health Sector in South Africa. J. Infect. Dis. 2007, 196, S491–S496. [Google Scholar] [CrossRef]
- Gandhi, N.R.; Moll, A.P.; Lalloo, U.; Pawinski, R.; Zeller, K.; Moodley, P.; Meyer, E.; Friedland, G. Tugela Ferry Care and Research (TFCaRes) Collaboration. Successful Integration of Tuberculosis and HIV Treatment in Rural South Africa: The Sizonq’oba Study. J. Acquir. Immune Defic. Syndr. 2008, 50, 37–43. [Google Scholar] [CrossRef] [PubMed]
| Core Indicator | Numerator | Denominator |
|---|---|---|
| Proportion of TB patients with known HIV status | Number of TB patients registered during the reporting period that had a HIV test result and recorded in the TB register. | Total number of TB patients registered during the reporting period. |
| Proportion of HIV-positive TB patients who received CPT | Number of HIV-positive TB patients, registered over the reporting period, starting or continuing CPT treatment during their TB treatment. | Total number of HIV positive TB patients registered during the reporting period. |
| Proportion of HIV-positive TB patients who received ART | Number of HIV-positive TB patients registered over the reporting period starting or continuing ART treatment during their TB treatment. | Total number of HIV positive TB patients registered during the reporting period. |
| Services | Fully Integrated Model (118 †) | Partially Integrated Model (212 †) | |
|---|---|---|---|
| TB Clinics | HIV Clinics | ||
| HIV services | |||
| HIV counseling and testing | + | + | + |
| ART provision | + | Refer to HIV clinic | + |
| CPT provision | + | Refer to HIV clinic | + |
| TB services | |||
| TB symptom screening | + | + | + |
| Diagnosis and treatment of TB (including Xpert MTB/RIF * assay) | + | + | Refer to TB clinic |
| IPT provision | + | + | + |
| Integrated Model | ||||
|---|---|---|---|---|
| Partial | ||||
| Full | TB and HIV co-Existed | TB Only | p Value † | |
| TB patients with known HIV status | 93 (77.9–98.1) | 93 (82.5–98.6) | 90 (76.3–98.5) | 0.63 |
| HIV-positive TB patients who received CPT | 81 (50.0–100) | 82 (62.2–91.1) | 74 (33.3–100) | 0.43 |
| HIV-positive TB patients who received ART | 32 (19.4–50.0) | 45 (28.4–59.0) | 33 (8.3–57.1) | 0.02 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kyi, M.S.; Aung, S.T.; McNeil, E.; Chongsuvivatwong, V. Evolution of Tuberculosis/Human Immunodeficiency Virus Services among Different Integrated Models in Myanmar: A Health Services Review. Trop. Med. Infect. Dis. 2019, 4, 2. https://doi.org/10.3390/tropicalmed4010002
Kyi MS, Aung ST, McNeil E, Chongsuvivatwong V. Evolution of Tuberculosis/Human Immunodeficiency Virus Services among Different Integrated Models in Myanmar: A Health Services Review. Tropical Medicine and Infectious Disease. 2019; 4(1):2. https://doi.org/10.3390/tropicalmed4010002
Chicago/Turabian StyleKyi, Myo Su, Si Thu Aung, Edward McNeil, and Virasakdi Chongsuvivatwong. 2019. "Evolution of Tuberculosis/Human Immunodeficiency Virus Services among Different Integrated Models in Myanmar: A Health Services Review" Tropical Medicine and Infectious Disease 4, no. 1: 2. https://doi.org/10.3390/tropicalmed4010002
APA StyleKyi, M. S., Aung, S. T., McNeil, E., & Chongsuvivatwong, V. (2019). Evolution of Tuberculosis/Human Immunodeficiency Virus Services among Different Integrated Models in Myanmar: A Health Services Review. Tropical Medicine and Infectious Disease, 4(1), 2. https://doi.org/10.3390/tropicalmed4010002






