The Development of a Mobile Application to Support Peripheral Health Workers to Diagnose and Treat People with Skin Diseases in Resource-Poor Settings
Abstract
:1. Introduction
1.1. Short Communication
1.2. Epidemiology of Skin Diseases in Low- and Middle-Income Countries
1.3. Psycological and Socioeconomic Burden
1.4. Capacity Gap
2. Mobile Health (mHealth)
2.1. Technologies to Support Dermatology Services
2.2. mHealth to Improve Peripheral Services forSkin Diseases
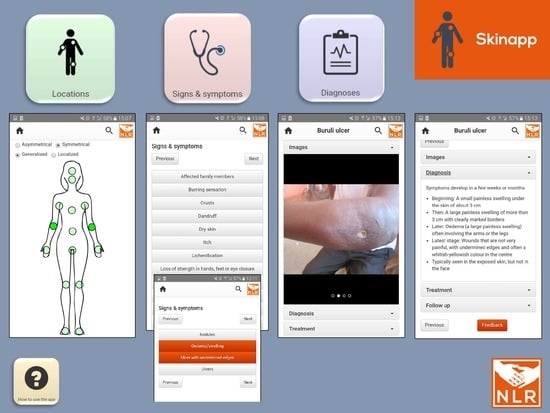
2.3. Development of NLR SkinApp
3. Discussion and Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hay, R.; Fuller, L. The assessment of dermatological needs in resource-poor regions. Int. J. Dermatol. 2011, 50, 552–557. [Google Scholar] [CrossRef] [PubMed]
- Vos, T.; Allen, C.; Arora, M.; Barber, R.; Bhutta, Z.; Brown, A.; Carter, A.; Casey, D.; Charlson, F.; Murray, C.; et al. Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990–2015: A systematic analysis for the Global Burden of Disease Study 2015. Lancet 2016, 388, 1545–1602. [Google Scholar] [CrossRef]
- Hay, R.; Johns, N.; Williams, H.; Bolliger, I.; Dellavalle, R.; Margolis, D.; Marks, R.; Naldi, L.; Weinstock, M.; Wulf, S.; et al. The global burden of skin disease in 2010: An analysis of the prevalence and impact of skin conditions. J. Investig. Dermatol. 2014, 134, 1527–1534. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; McKoy, K.; Papier, A.; Klaus, S.; Ryan, T.; Grossman, H.; Masenga, E.J.; Sethi, A.; Craft, N. Dermatology and HIV/AIDS in Africa. J. Glob. Infect. Dis. 2011, 3, 275–280. [Google Scholar] [PubMed]
- Sanclemente, G.; Burgos, C.; Nova, J.; Hernández, F.; González, C.; Reyes, M.; Córdoba, N.; Arévalo, Á.; Meléndez, E.; Colmenares, J.; et al. The impact of skin diseases on quality of life: A multicenter study. Actas Dermosifiliogr. 2017, 108, 244–252. [Google Scholar] [CrossRef] [PubMed]
- Barankin, B.; DeKoven, J. Psychosocial effect of common skin diseases. Can. Fam. Physician 2002, 48, 712–716. [Google Scholar] [PubMed]
- Figueroa, J.; Fuller, L.; Abraha, A.; Hay, R. Dermatology in southwestern Ethiopia: Rationale for a community approach. Int. J. Dermatol. 1998, 37, 752–758. [Google Scholar] [CrossRef] [PubMed]
- Mitjà, O.; Marks, M.; Bertran, L.; Kollie, K.; Argaw, D.; Fahal, A.; Fitzpatrick, C.; Fuller, L.; Garcia Izquierdo, B.; Hay, R.; et al. Integrated control and management of neglected tropical skin diseases. PLoS Negl. Trop. Dis. 2017, 11, e0005136. [Google Scholar] [CrossRef] [PubMed]
- Mahe, A.; N’Diaye, H.; Bobin, P. The proportion of medical consultations motivated by skin diseases in the health centers of Bamako (Republic of Mali). Int. J. Dermatol. 1997, 36, 185–186. [Google Scholar] [CrossRef] [PubMed]
- Odueko, O.M.; Onayemi, O.; Oyedeji, G.A. A prevalence survey of skin diseases in Nigerian children. Niger. J. Med. 2001, 10, 64–67. [Google Scholar] [PubMed]
- Seth, D.; Cheldize, K.; Brown, D.; Freeman, E. Global burden of skin disease: Inequities and innovations. Curr. Dermatol. Rep. 2017, 6, 204–210. [Google Scholar] [CrossRef] [PubMed]
- Surovi, N.A.; Kiber, A.; Kashem, A.; Babi, K.N. Study and Development of Algorithm of Different Skin Diseases Analysis Using Image Processing Method. Available online: http://www.alliedacademies.org/articles/study-and-development-of-algorithm-of-different-skin-diseases-analysis-using-image-processing-method.html (accessed on 26 June 2018).
- Desai, B.; McKoy, K.; Kovarik, C. Overview of international teledermatology. Pan Afr. Med. J. 2010, 6, 3. [Google Scholar] [CrossRef] [PubMed]
- Yadav, N.; Narang, V.; Shrivastava, U. Skin diseases detection models using image processing: A survey Utpal Shrivastava. Int. J. Comput. Appl. 2016, 137, 34–39. [Google Scholar]
- WHO. Classification of Digital Health Interventions v1.0. World Health Organization. Available online: http://www.who.int/reproductivehealth/publications/mhealth/classification-digital-health-interventions/en/ (accessed on 26 June 2018).
- Labrique, A.; Vasudevan, L.; Kochi, E.; Fabricant, R.; Mehl, G. mHealth innovations as health system strengthening tools: 12 common applications and a visual framework. Glob. Health Sci. Pract. 2013, 1, 160–171. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agarwal, S.; Perry, H.; Long, L.; Labrique, A. Evidence on feasibility and effective use of mHealth strategies by frontline health workers in developing countries: Systematic review. Trop. Med. Int. Health 2015, 20, 1003–1014. [Google Scholar] [CrossRef] [PubMed]
- Flaten, H.K.; Claire, C.S.; Schlager, E.; Dunnick, C.A.; Dellavalle, R.P. Growth of mobile applications in dermatology 2017 update. Dermatol. Online J. 2018, 24, 13. [Google Scholar]
- Brewer, A.C.; Endly, D.C.; Henley, J.; Amir, M.; Sampson, B.P.; Moreau, J.F.; Dellavalle, R.P. Mobile applications in dermatology. JAMA Dermatol. 2013, 149, 1300–1304. [Google Scholar] [CrossRef] [PubMed]
- Lipoff, J.B.; Cobos, G.; Kaddu, S.; Kovarik, C.L. The Africa Teledermatology Project: A retrospective case review of 1229 consultations from sub-Saharan Africa. J. Am. Acad. Dermatol. 2015, 72, 1084–1085. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, A.; Tran, D.; Uemura, M.; Bardin, R.L.; Shitabata, P.K. Practical and sustainable teledermatology and teledermatopathology: Specialty care in Cameroon Africa. J. Clin. Aesthet. Dermatol. 2017, 10, 47–56. [Google Scholar] [PubMed]
- Mahe, A.; Faye, O.; N’Diaye, H.T.; Ly, F.; Konare, H.; Keita, S.; Traore, A.K.; Hay, R. Definition of an algorithm for the management of common skin diseases at primary health care level in sub-Saharan Africa. Trans. Soc. Trop. Med. Hyg. 2005, 99, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Taal, A.; Hussaini, T.; Gayus, B.; Dahiru, T.; Post, E. First-line health care provider performance in the management of common skin diseases using an algorithmic approach as a diagnostic tool in Kano State, Nigeria. Res. Rep. Trop. Med. 2015, 6, 85–94. [Google Scholar] [CrossRef]
- Van Hees, C.; Naafs, B. Common Skin Diseases in Africa, an Illustrated Guide, 3rd ed.; Stichting Troderma: Voorburg, The Netherlands, 2014. [Google Scholar]
- Kousa, M.; Sanders, C. HIV Related Skin Diseases and Sexually Transmitted Infections. Available online: https://www.ntvg.nl/artikelen/hiv-related-skin-diseases-and-sexually-transmitted-infections-africa/volledig (accessed on 29 June 2018).
- WHO. Neglected Tropical Diseases. Available online: http://www.who.int/neglected_diseases/en/ (accessed on 16 July 2018).
| Common Skin Diseases | NTDs Manifesting in Skin Lesions | HIV-Related Skin Diseases | Others |
|---|---|---|---|
| Acne 1 | Buruli ulcer 1 | Angular cheilitis 1 | Albinism |
| Atopic eczema 1 | Cutaneous leishmaniasis 1 | Herpes simplex 1 | Blistering diseases |
| Contact eczema 1 | Leprosy 1 | Herpes zoster 1 | Creeping eruption |
| Impetigo 1 | Lymphatic filiariasis 1 | Kaposi sarcoma 1 | Vitiligo |
| Pityriasis versicolor 1 | Mycetoma | Molluscum contagiosum 1 | |
| Psoriasis | Onchocerciasis 1 | Oral candidiasis 1 | |
| Seborrhoeic eczema 1 (*) | Podoconiosis | Pruritic papular eruption 1 | |
| Tinea capitis 1 | Scabies 1 | ||
| Tinea corporis 1 (*) | Yaws |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mieras, L.F.; Taal, A.T.; Post, E.B.; Ndeve, A.G.Z.; Van Hees, C.L.M. The Development of a Mobile Application to Support Peripheral Health Workers to Diagnose and Treat People with Skin Diseases in Resource-Poor Settings. Trop. Med. Infect. Dis. 2018, 3, 102. https://doi.org/10.3390/tropicalmed3030102
Mieras LF, Taal AT, Post EB, Ndeve AGZ, Van Hees CLM. The Development of a Mobile Application to Support Peripheral Health Workers to Diagnose and Treat People with Skin Diseases in Resource-Poor Settings. Tropical Medicine and Infectious Disease. 2018; 3(3):102. https://doi.org/10.3390/tropicalmed3030102
Chicago/Turabian StyleMieras, Liesbeth F., Anna T. Taal, Erik B. Post, Alcino G. Z. Ndeve, and Colette L. M. Van Hees. 2018. "The Development of a Mobile Application to Support Peripheral Health Workers to Diagnose and Treat People with Skin Diseases in Resource-Poor Settings" Tropical Medicine and Infectious Disease 3, no. 3: 102. https://doi.org/10.3390/tropicalmed3030102