Melioidosis: An Australian Perspective
Abstract
1. History
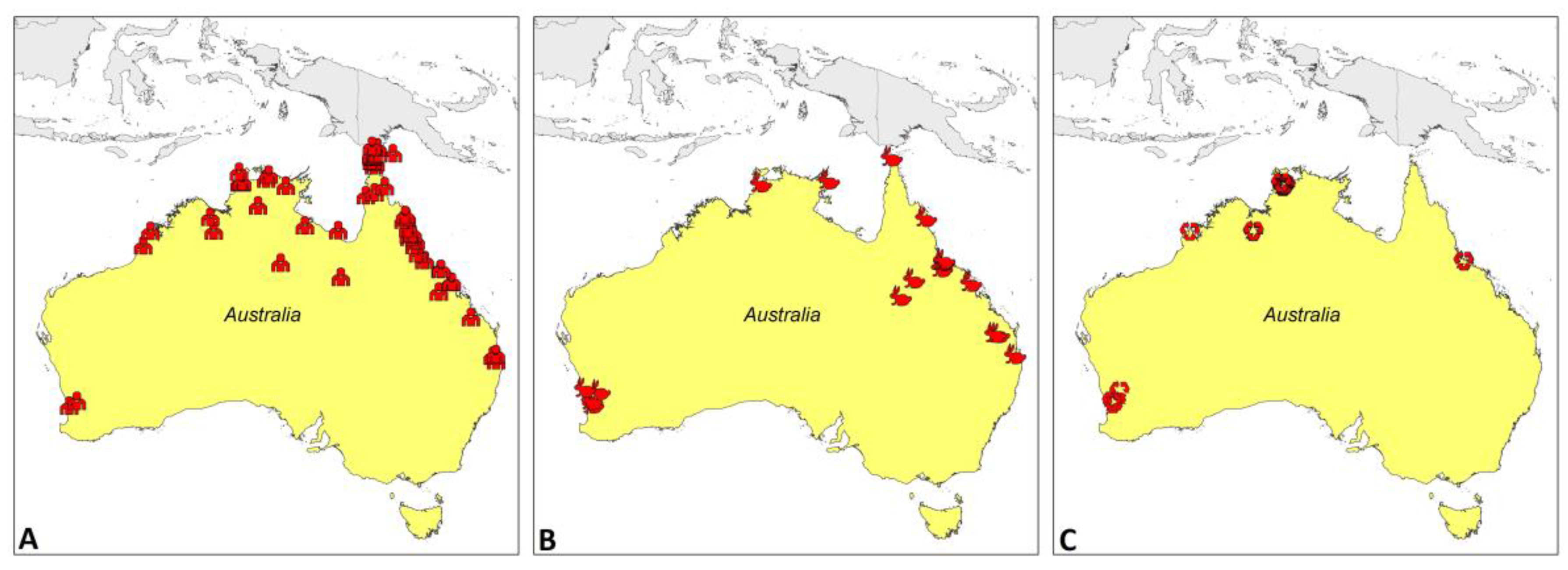
2. Melioidosis Cases and the Presence of B. pseudomallei
3. Surveillance Systems and Reporting
4. Diagnosis
5. Treatment
6. Awareness and Prevention of Melioidosis
7. Major Achievements
8. Current and Future Challenges
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cottew, G.S. Melioidosis in sheep in Queensland; a description of the causal organism. Aust. J. Exp. Biol. Med. Sci. 1950, 28, 677–683. [Google Scholar] [CrossRef] [PubMed]
- Rimington, R.A. Melioidosis in north Queensland. Med. J. Aust. 1962, 49, 50–53. [Google Scholar] [PubMed]
- Crotty, J.M.; Bromwich, A.F.; Quinn, J.V.; Brotherton, J. Meliodosis in the Northern Territory: A report of two cases. Med. J. Aust. 1963, 50, 274–275. [Google Scholar] [PubMed]
- Cheng, A.C.; Hanna, J.N.; Norton, R.; Hills, S.L.; Davis, J.; Krause, V.L.; Dowse, G.; Inglis, T.J.; Currie, B.J. Melioidosis in northern Australia, 2001–2002. Commun. Dis. Intell. Q. Rep. 2003, 27, 272–277. [Google Scholar] [PubMed]
- Faa, A.G.; Holt, P.J. Melioidosis in the Torres Strait Islands of Far North Queensland. Commun. Dis. Intell. Q. Rep. 2002, 26, 279–283. [Google Scholar] [PubMed]
- Currie, B.J.; Ward, L.; Cheng, A.C. The epidemiology and clinical spectrum of melioidosis: 540 cases from the 20 year Darwin prospective study. PLoS Negl. Trop. Dis. 2010, 4, e900. [Google Scholar] [CrossRef] [PubMed]
- Hanna, J.N.; Humphreys, J.L.; Brookes, D.L.; Messina, T.; Raulli, A. Melioidosis in north Queensland, 2000–2009. Commun. Dis. Intell. Q. Rep. 2010, 34, 444–447. [Google Scholar] [PubMed]
- Currie, B.; Smith-Vaughan, H.; Golledge, C.; Buller, N.; Sriprakash, K.S.; Kemp, D.J. Pseudomonas pseudomallei isolates collected over 25 years from a non-tropical endemic focus show clonality on the basis of ribotyping. Epidemiol. Infect. 1994, 113, 307–312. [Google Scholar] [CrossRef] [PubMed]
- Golledge, C.L.; Chin, W.S.; Tribe, A.E.; Condon, R.J.; Ashdown, L.R. A case of human melioidosis originating in south-west Western Australia. Med. J. Aust. 1992, 157, 332–334. [Google Scholar] [PubMed]
- Chapple, S.N.; Sarovich, D.S.; Holden, M.T.; Peacock, S.J.; Buller, N.; Golledge, C.; Mayo, M.; Currie, B.J.; Price, E.P. Whole-genome sequencing of a quarter-century melioidosis outbreak in temperate Australia uncovers a region of low-prevalence endemicity. Microb. Genom. 2016, 2, e000067. [Google Scholar] [CrossRef] [PubMed]
- Ketterer, P.J.; Webster, W.R.; Shield, J.; Arthur, R.J.; Blackall, P.J.; Thomas, A.D. Melioidosis in intensive piggeries in south eastern Queensland. Aust. Vet. J. 1986, 63, 146–149. [Google Scholar] [CrossRef] [PubMed]
- Scott, I.A.; Bell, A.M.; Staines, D.R. Fatal human melioidosis in south-eastern Queensland. Med. J. Aust. 1997, 166, 197–199. [Google Scholar] [PubMed]
- Munckhof, W.J.; Mayo, M.J.; Scott, I.; Currie, B.J. Fatal human melioidosis acquired in a subtropical Australian city. Am. J. Trop. Med. Hyg. 2001, 65, 325–328. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ketterer, P.J.; Donald, B.; Rogers, R.J. Bovine melioidosis in south-eastern Queensland. Aust. Vet. J. 1975, 51, 395–398. [Google Scholar] [CrossRef] [PubMed]
- Yip, T.W.; Hewagama, S.; Mayo, M.; Price, E.P.; Sarovich, D.S.; Bastian, I.; Baird, R.W.; Spratt, B.G.; Currie, B.J. Endemic melioidosis in residents of desert region after atypically intense rainfall in central Australia, 2011. Emerg. Infect. Dis. 2015, 21, 1038–1040. [Google Scholar] [CrossRef] [PubMed]
- Parameswaran, U.; Baird, R.W.; Ward, L.M.; Currie, B.J. Melioidosis at Royal Darwin Hospital in the big 2009-2010 wet season: Comparison with the preceding 20 years. Med. J. Aust. 2012, 196, 345–348. [Google Scholar] [CrossRef] [PubMed]
- Stewart, J.D.; Smith, S.; Binotto, E.; McBride, W.J.; Currie, B.J.; Hanson, J. The epidemiology and clinical features of melioidosis in Far North Queensland: Implications for patient management. PLoS Negl. Trop. Dis. 2017, 11, e0005411. [Google Scholar] [CrossRef] [PubMed]
- Ashdown, L.R.; Guard, R.W. The prevalence of human melioidosis in northern Queensland. Am. J. Trop. Med. Hyg. 1984, 33, 474–478. [Google Scholar] [CrossRef] [PubMed]
- Currie, B.J.; Fisher, D.A.; Howard, D.M.; Burrow, J.N.; Selvanayagam, S.; Snelling, P.L.; Anstey, N.M.; Mayo, M.J. The epidemiology of melioidosis in Australia and Papua New Guinea. Acta Trop. 2000, 74, 121–127. [Google Scholar] [CrossRef]
- Pearson, T.; Giffard, P.; Beckstrom-Sternberg, S.; Auerbach, R.; Hornstra, H.; Tuanyok, A.; Price, E.P.; Glass, M.B.; Leadem, B.; Beckstrom-Sternberg, J.S.; et al. Phylogeographic reconstruction of a bacterial species with high levels of lateral gene transfer. BMC Biol. 2009, 7, 78. [Google Scholar] [CrossRef] [PubMed]
- Cheng, A.C.; Ward, L.; Godoy, D.; Norton, R.; Mayo, M.; Gal, D.; Spratt, B.G.; Currie, B.J. Genetic diversity of Burkholderia pseudomallei isolates in Australia. J. Clin. Microbial. 2008, 46, 249–254. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Price, E.P.; Sarovich, D.S.; Smith, E.J.; MacHunter, B.; Harrington, G.; Theobald, V.; Hall, C.M.; Hornstra, H.M.; McRobb, E.; Podin, Y.; et al. Unprecedented melioidosis cases in northern Australia caused by an Asian Burkholderia pseudomallei strain identified by using large-scale comparative genomics. Appl. Environ. Microbial. 2016, 82, 954–963. [Google Scholar] [CrossRef] [PubMed]
- Thomas, A.D.; Spinks, G.A.; D’Arcy, T.L.; Norton, J.H.; Trueman, K.F. Evaluation of four serological tests for the diagnosis of caprine melioidosis. Aust. Vet. J. 1988, 65, 261–264. [Google Scholar] [CrossRef] [PubMed]
- Forbes-Faulkner, J.C.; Townsend, W.L.; Thomas, A.D. Pseudomonas pseudomallei infection in camels. Aust. Vet. J. 1992, 69, 148. [Google Scholar] [CrossRef] [PubMed]
- Janmaat, A.; Choy, J.L.; Currie, B.J. Melioidosis in an alpaca (Lama pacos). Aust. Vet. J. 2004, 82, 622–623. [Google Scholar] [CrossRef] [PubMed]
- Choy, J.L.; Mayo, M.; Janmaat, A.; Currie, B.J. Animal melioidosis in Australia. Acta Trop. 2000, 74, 153–158. [Google Scholar] [CrossRef]
- Ladds, P.W.; Thomas, A.D.; Pott, B. Melioidosis with acute meningoencephalomyelitis in a horse. Aust. Vet. J. 1981, 57, 36–38. [Google Scholar] [CrossRef] [PubMed]
- Thomas, A.D. Prevalence of melioidosis in animals in northern Queensland. Aust. Vet. J. 1981, 57, 146–148. [Google Scholar] [CrossRef] [PubMed]
- Parkes, H.M.; Shilton, C.M.; Jerrett, I.V.; Benedict, S.; Spratt, B.G.; Godoy, D.; O’Brien, C.R.; Krockenberger, M.B.; Mayo, M.; Currie, B.J.; et al. Primary ocular melioidosis due to a single genotype of Burkholderia pseudomallei in two cats from Arnhem Land in the Northern Territory of Australia. J. Feline Med. Surg. 2009, 11, 856–863. [Google Scholar] [CrossRef] [PubMed]
- Ladds, P.W.; Thomas, A.D.; Speare, R.; Brown, A.S. Melioidosis in a koala. Aust. Vet. J. 1990, 67, 304–305. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mekisic, A.P.; Wardill, J.R. Crocodile attacks in the Northern Territory of Australia. Med. J. Aust. 1992, 157, 751–754. [Google Scholar] [PubMed]
- Thomas, A.D.; Norton, J.H.; Pott, B.W. Melioidosis in a galah (Cacatua roseicapilla). Aust. Vet. J. 1980, 56, 192–193. [Google Scholar] [CrossRef] [PubMed]
- Thomas, A.D.; Wilson, A.J.; Aubrey, J.N. Melioidosis in a sulphur-crested cockatoo (Cacatua galerita). Aust. Vet. J. 1978, 54, 306–307. [Google Scholar] [CrossRef] [PubMed]
- Hampton, V.; Kaestli, M.; Mayo, M.; Choy, J.L.; Harrington, G.; Richardson, L.; Benedict, S.; Noske, R.; Garnett, S.T.; Godoy, D.; et al. Melioidosis in birds and Burkholderia pseudomallei dispersal, Australia. Emerg. Infect. Dis. 2011, 17, 1310–1312. [Google Scholar] [CrossRef] [PubMed]
- Hoger, A.C.; Mayo, M.; Price, E.P.; Theobald, V.; Harrington, G.; Machunter, B.; Choy, J.L.; Currie, B.J.; Kaestli, M. The melioidosis agent Burkholderia pseudomallei and related opportunistic pathogens detected in faecal matter of wildlife and livestock in northern Australia. Epidemiol. Infect. 2016, 144, 1924–1932. [Google Scholar] [CrossRef] [PubMed]
- Currie, B.J.; Mayo, M.; Anstey, N.M.; Donohoe, P.; Haase, A.; Kemp, D.J. A cluster of melioidosis cases from an endemic region is clonal and is linked to the water supply using molecular typing of Burkholderia pseudomallei isolates. Am. J. Trop. Med. Hyg. 2001, 65, 177–179. [Google Scholar] [CrossRef] [PubMed]
- Inglis, T.J.; Garrow, S.C.; Henderson, M.; Clair, A.; Sampson, J.; O’Reilly, L.; Cameron, B. Burkholderia pseudomallei traced to water treatment plant in Australia. Emerg. Infect. Dis. 2000, 6, 56–59. [Google Scholar] [PubMed]
- Currie, B.J.; Jacups, S.P. Intensity of rainfall and severity of melioidosis, Australia. Emerg. Infect. Dis. 2003, 9, 1538–1542. [Google Scholar] [CrossRef] [PubMed]
- Cheng, A.C.; Jacups, S.P.; Ward, L.; Currie, B.J. Melioidosis and aboriginal seasons in northern Australia. Trans. R. Soc. Trop. Med. Hyg. 2008, 102 (Suppl. 1), S26–S29. [Google Scholar] [CrossRef]
- Kaestli, M.; Grist, E.P.M.; Ward, L.; Hill, A.; Mayo, M.; Currie, B.J. The association of melioidosis with climatic factors in Darwin, Australia: A 23-year time-series analysis. J. Infect. 2016, 72, 687–697. [Google Scholar] [CrossRef] [PubMed]
- Cheng, A.C.; Jacups, S.P.; Gal, D.; Mayo, M.; Currie, B.J. Extreme weather events and environmental contamination are associated with case-clusters of melioidosis in the Northern Territory of Australia. Int. J. Epidemiol. 2006, 35, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Inglis, T.J.; O’Reilly, L.; Merritt, A.J.; Levy, A.; Heath, C.H. The aftermath of the Western Australian melioidosis outbreak. Am. J. Trop. Med. Hyg. 2011, 84, 851–857. [Google Scholar] [CrossRef] [PubMed]
- Stewart, J.D.; Smith, S.; Hanson, J. Melioidosis in Far North Queensland is not correlated with severe weather events. Med. J. Aust. 2017, 207, 394. [Google Scholar] [CrossRef] [PubMed]
- Merritt, A.J.; Inglis, T.J.J. The role of climate in the epidemiology of melioidosis. Curr. Trop. Med. Rep. 2017, 4, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Meumann, E.M.; Cheng, A.C.; Ward, L.; Currie, B.J. Clinical features and epidemiology of melioidosis pneumonia: Results from a 21-year study and review of the literature. Clin. Infect. Dis. 2012, 54, 362–369. [Google Scholar] [CrossRef] [PubMed]
- Morse, L.P.; Moller, C.C.; Harvey, E.; Ward, L.; Cheng, A.C.; Carson, P.J.; Currie, B.J. Prostatic abscess due to Burkholderia pseudomallei: 81 cases from a 19-year prospective melioidosis study. J. Urol. 2009, 182, 542–547. [Google Scholar] [CrossRef] [PubMed]
- Kozlowska, J.; Smith, S.; Roberts, J.; Pridgeon, S.; Hanson, J. Prostatic abscess due to Burkholderia pseudomallei: Facilitating diagnosis to optimize management. Am. J. Trop. Med. Hyg. 2017, 98, 227–230. [Google Scholar] [CrossRef] [PubMed]
- Shetty, R.P.; Mathew, M.; Smith, J.; Morse, L.P.; Mehta, J.A.; Currie, B.J. Management of melioidosis osteomyelitis and septic arthritis. Bone Jt. J. 2015, 97-B, 277–282. [Google Scholar] [CrossRef] [PubMed]
- McLeod, C.; Morris, P.S.; Bauert, P.A.; Kilburn, C.J.; Ward, L.M.; Baird, R.W.; Currie, B.J. Clinical presentation and medical management of melioidosis in children: A 24-year prospective study in the Northern Territory of Australia and review of the literature. Clin. Infect. Dis. 2015, 60, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Sanderson, C.; Currie, B.J. Melioidosis: A pediatric disease. Pediatr. Infect. Dis. J. 2014, 33, 770–771. [Google Scholar] [CrossRef] [PubMed]
- Young, A.; Tacon, C.; Smith, S.; Reeves, B.; Wiseman, G.; Hanson, J. Case report: Fatal pediatric melioidosis despite optimal intensive care. Am. J. Trop. Med. Hyg. 2017, 97, 1691–1694. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.; Stewart, J.D.; Tacon, C.; Archer, N.; Hanson, J. Children with melioidosis in Far North Queensland are commonly bacteraemic and have a high case fatality rate. Commun. Dis. Intell. Q. Rep. 2018, in press. [Google Scholar]
- Sarovich, D.S.; Ward, L.; Price, E.P.; Mayo, M.; Pitman, M.C.; Baird, R.W.; Currie, B.J. Recurrent melioidosis in the Darwin prospective melioidosis study: Improving therapies mean that relapse cases are now rare. J. Clin. Microbial. 2014, 52, 650–653. [Google Scholar] [CrossRef] [PubMed]
- Currie, B.J.; Fisher, D.A.; Howard, D.M.; Burrow, J.N. Neurological melioidosis. Acta Trop. 2000, 74, 145–151. [Google Scholar] [CrossRef]
- Sarovich, D.S.; Price, E.P.; Webb, J.R.; Ward, L.M.; Voutsinos, M.Y.; Tuanyok, A.; Mayo, M.; Kaestli, M.; Currie, B.J. Variable virulence factors in Burkholderia pseudomallei (melioidosis) associated with human disease. PLoS ONE 2014, 9, e91682. [Google Scholar] [CrossRef] [PubMed]
- Morris, J.L.; Fane, A.; Sarovich, D.S.; Price, E.P.; Rush, C.M.; Govan, B.L.; Parker, E.; Mayo, M.; Currie, B.J.; Ketheesan, N. Increased neurotropic threat from Burkholderia pseudomallei strains with a B. Mallei-like variation in the bima motility gene, Australia. Emerg. Infect. Dis. 2017, 23. [Google Scholar] [CrossRef] [PubMed]
- Baker, A.; Mayo, M.; Owens, L.; Burgess, G.; Norton, R.; McBride, W.J.; Currie, B.J.; Warner, J. Biogeography of Burkholderia pseudomallei in the Torres Strait Islands of northern Australia. J. Clin. Microbial. 2013, 51, 2520–2525. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hill, A.A.; Mayo, M.; Kaestli, M.; Price, E.P.; Richardson, L.J.; Godoy, D.; Spratt, B.G.; Currie, B.J. Melioidosis as a consequence of sporting activity. Am. J. Trop. Med. Hyg. 2013, 89, 365–366. [Google Scholar] [CrossRef] [PubMed]
- Baker, A.L.; Ezzahir, J.; Gardiner, C.; Shipton, W.; Warner, J.M. Environmental attributes influencing the distribution of Burkholderia pseudomallei in northern Australia. PLoS ONE 2015, 10, e0138953. [Google Scholar] [CrossRef] [PubMed]
- McRobb, E.; Kaestli, M.; Mayo, M.; Price, E.P.; Sarovich, D.S.; Godoy, D.; Spratt, B.G.; Currie, B.J. Melioidosis from contaminated bore water and successful UV sterilization. Am. J. Trop. Med. Hyg. 2013, 89, 367–368. [Google Scholar] [CrossRef] [PubMed]
- Kaestli, M.; Mayo, M.; Harrington, G.; Ward, L.; Watt, F.; Hill, J.V.; Cheng, A.C.; Currie, B.J. Landscape changes influence the occurrence of the melioidosis bacterium Burkholderiapseudomallei in soil in northern Australia. PLoS Negl. Trop. Dis. 2009, 3, e364. [Google Scholar] [CrossRef] [PubMed]
- Kaestli, M.; Schmid, M.; Mayo, M.; Rothballer, M.; Harrington, G.; Richardson, L.; Hill, A.; Hill, J.; Tuanyok, A.; Keim, P.; et al. Out of the ground: Aerial and exotic habitats of the melioidosis bacterium Burkholderia pseudomallei in grasses in Australia. Environ. Microbiol. 2012, 14, 2058–2070. [Google Scholar] [CrossRef] [PubMed]
- Currie, B.J.; Price, E.P.; Mayo, M.; Kaestli, M.; Theobald, V.; Harrington, I.; Harrington, G.; Sarovich, D.S. Use of whole-genome sequencing to link Burkholderia pseudomallei from air sampling to mediastinal melioidosis, Australia. Emerg. Infect. Dis. 2015, 21, 2052–2054. [Google Scholar] [CrossRef] [PubMed]
- Merritt, A.J.; Peck, M.; Gayle, D.; Levy, A.; Ler, Y.H.; Raby, E.; Gibbs, T.M.; Inglis, T.J. Cutaneous melioidosis cluster caused by contaminated wound irrigation fluid. Emerg. Infect. Dis. 2016, 22, 1420. [Google Scholar] [CrossRef] [PubMed]
- Government of Western Australia. Department of Primary Industries and Regional Development. Melioidosis in Animals. Available online: https://www.Agric.Wa.Gov.Au/livestock-biosecurity/melioidosis-animals (accessed on 21 January 2018).
- Podin, Y.; Kaestli, M.; McMahon, N.; Hennessy, J.; Ngian, H.U.; Wong, J.S.; Mohana, A.; Wong, S.C.; William, T.; Mayo, M.; et al. Reliability of automated biochemical identification of Burkholderia pseudomallei is regionally dependent. J. Clin. Microbial. 2013, 51, 3076–3078. [Google Scholar] [CrossRef] [PubMed]
- Inglis, T.J.; Healy, P.E.; Fremlin, L.J.; Golledge, C.L. Use of matrix-assisted laser desorption/ionization time-of-flight mass spectrometry analysis for rapid confirmation of Burkholderia pseudomallei in septicemic melioidosis. Am. J. Trop. Med. Hyg. 2012, 86, 1039–1042. [Google Scholar] [CrossRef] [PubMed]
- Robertson, G.; Sorenson, A.; Govan, B.; Ketheesan, N.; Houghton, R.; Chen, H.; AuCoin, D.; Dillon, M.; Norton, R. Rapid diagnostics for melioidosis: A comparative study of a novel lateral flow antigen detection assay. J. Med. Microbial. 2015, 64, 845–848. [Google Scholar] [CrossRef] [PubMed]
- Inglis, T.J.; Merritt, A.; Chidlow, G.; Aravena-Roman, M.; Harnett, G. Comparison of diagnostic laboratory methods for identification of Burkholderia pseudomallei. J. Med. Microbial. 2005, 43, 2201–2206. [Google Scholar] [CrossRef] [PubMed]
- Haase, A.; Brennan, M.; Barrett, S.; Wood, Y.; Huffam, S.; O’Brien, D.; Currie, B. Evaluation of PCR for diagnosis of melioidosis. J. Med. Microbial. 1998, 36, 1039–1041. [Google Scholar]
- Kaestli, M.; Richardson, L.J.; Colman, R.E.; Tuanyok, A.; Price, E.P.; Bowers, J.R.; Mayo, M.; Kelley, E.; Seymour, M.L.; Sarovich, D.S.; et al. Comparison of Taqman PCR assays for detection of the melioidosis agent Burkholderia pseudomallei in clinical specimens. J. Med. Microbial. 2012, 50, 2059–2062. [Google Scholar] [CrossRef] [PubMed]
- Cheng, A.C.; O’Brien, M.; Freeman, K.; Lum, G.; Currie, B.J. Indirect hemagglutination assay in patients with melioidosis in northern Australia. Am. J. Trop. Med. Hyg. 2006, 74, 330–334. [Google Scholar] [PubMed]
- Davis, J.S.; Currie, B.J.; Fisher, D.A.; Huffam, S.E.; Anstey, N.M.; Price, R.N.; Krause, V.L.; Zweck, N.; Lawton, P.D.; Snelling, P.L.; et al. Prevention of opportunistic infections in immunosuppressed patients in the tropical top end of the Northern Territory. Commun. Dis. Intell. Q. Rep. 2003, 27, 526–532. [Google Scholar]
- Maloney, S.; Engler, C.; Norton, R. Epidemiological cut-off value of clinical isolates of Burkholderia pseudomallei from northern Queensland to meropenem, ceftazidime, trimethoprim/sulfamethoxazole and doxycycline by the microbroth dilution method. J. Glob. Antimicrob. Resist. 2017, 10, 291–294. [Google Scholar] [CrossRef] [PubMed]
- Currie, B.J. Melioidosis: Evolving concepts in epidemiology, pathogenesis, and treatment. Semin. Respir. Crit. Care Med. 2015, 36, 111–125. [Google Scholar] [CrossRef] [PubMed]
- Jenney, A.W.; Lum, G.; Fisher, D.A.; Currie, B.J. Antibiotic susceptibility of Burkholderia pseudomallei from tropical northern Australia and implications for therapy of melioidosis. Int. J. Antimicrob. Agents 2001, 17, 109–113. [Google Scholar] [CrossRef]
- Crowe, A.; McMahon, N.; Currie, B.J.; Baird, R.W. Current antimicrobial susceptibility of first-episode melioidosis Burkholderia pseudomallei isolates from the Northern Territory, Australia. Int. J. Antimicrob. Agents 2014, 44, 160–162. [Google Scholar] [CrossRef] [PubMed]
- Price, E.P.; Smith, M.L.; Paxinos, E.E.; Tallon, L.J.; Sadzewicz, L.; Sengamalay, N.; Baird, R.W.; Currie, B.J.; Sarovich, D.S. Whole-genome sequences of Burkholderia pseudomallei isolates exhibiting decreased meropenem susceptibility. Genome Announc. 2017, 5, e00053-17. [Google Scholar] [CrossRef] [PubMed]
- Viberg, L.T.; Sarovich, D.S.; Kidd, T.J.; Geake, J.B.; Bell, S.C.; Currie, B.J.; Price, E.P. Within-host evolution of Burkholderia pseudomallei during chronic infection of seven Australasian cystic fibrosis patients. mBio 2017, 8, e00356-17. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.C.; Singh, D.; Kwan, G.; Deuble, M.; Aquilina, C.; Korah, I.; Norton, R. Neuromelioidosis: Craniospinal MRI findings in Burkholderia pseudomallei infection. J. Neuroimag. 2016, 26, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Deuble, M.; Aquilina, C.; Norton, R. Neurologic melioidosis. Am. J. Trop. Med. Hyg. 2013, 89, 535–539. [Google Scholar] [CrossRef] [PubMed]
- Huffam, S.; Jacups, S.P.; Kittler, P.; Currie, B.J. Out of hospital treatment of patients with melioidosis using ceftazidime in 24 h elastomeric infusors, via peripherally inserted central catheters. Trop. Med. Int. Health 2004, 9, 715–717. [Google Scholar] [CrossRef] [PubMed]
- Cheng, A.C.; Chierakul, W.; Chaowagul, W.; Chetchotisakd, P.; Limmathurotsakul, D.; Dance, D.A.; Peacock, S.J.; Currie, B.J. Consensus guidelines for dosing of amoxicillin-clavulanate in melioidosis. Am. J. Trop. Med. Hyg. 2008, 78, 208–209. [Google Scholar] [PubMed]
- Stephens, D.P.; Thomas, J.H.; Ward, L.M.; Currie, B.J. Melioidosis causing critical illness: A review of 24 years of experience from the Royal Darwin Hospital ICU. Crit. Care Med. 2016, 44, 1500–1505. [Google Scholar] [CrossRef] [PubMed]
- Cheng, A.C.; Stephens, D.P.; Anstey, N.M.; Currie, B.J. Adjunctive granulocyte colony-stimulating factor for treatment of septic shock due to melioidosis. Clin. Infect. Dis. 2004, 38, 32–37. [Google Scholar] [CrossRef] [PubMed]
- Cheng, A.C.; Limmathurotsakul, D.; Chierakul, W.; Getchalarat, N.; Wuthiekanun, V.; Stephens, D.P.; Day, N.P.; White, N.J.; Chaowagul, W.; Currie, B.J.; et al. A randomized controlled trial of granulocyte colony-stimulating factor for the treatment of severe sepsis due to melioidosis in Thailand. Clin. Infect. Dis. 2007, 45, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Northern Territory Government. Melioidosis. Available online: https://nt.Gov.Au/wellbeing/health-conditions-treatments/bacterial/melioidosis (accessed on 21 January 2018).
- Boyd, R.; Mcguinness, S.; Draper, A.; Neilson, M.; Krause, V. Melioidosis Awareness Campaign; The Northern Territory Disease Control Bulletin, Centre for Disease Control: Darwin, Australia, 2016; Volume 23, pp. 1–4.
- Chalmers, R.M.; Majoni, S.W.; Ward, L.; Perry, G.J.; Jabbar, Z.; Currie, B.J. Melioidosis and end-stage renal disease in tropical northern Australia. Kidney Int. 2014, 86, 867–870. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Majoni, S.W.; Hughes, J.T.; Heron, B.; Currie, B.J. Trimethoprim + sulfamethoxazole reduces rates of melioidosis in high-risk hemodialysis patients. Kidney Int. Rep. 2018, 3, 160–167. [Google Scholar] [CrossRef] [PubMed]
- Fitzpatrick, S.; Kearney, S. Melioidosis. Department of Regional Development, Primary Industry, Fisheries and Resources, Northern Territory Government. Available online: https://dpir.nt.gov.au/__data/assets/pdf_file/0011/232976/840.pdf (accessed on 21 January 2018).
- McRobb, E.; Kaestli, M.; Price, E.P.; Sarovich, D.S.; Mayo, M.; Warner, J.; Spratt, B.G.; Currie, B.J. Distribution of Burkholderia pseudomallei in northern Australia, a land of diversity. Appl. Environ. Microbial. 2014, 80, 3463–3468. [Google Scholar] [CrossRef] [PubMed]
- Aziz, A.; Sarovich, D.S.; Harris, T.M.; Kaestli, M.; McRobb, E.; Mayo, M.; Currie, B.J.; Price, E.P. Suspected cases of intracontinental Burkholderia pseudomallei sequence type homoplasy resolved using whole-genome sequencing. Microb. Genom. 2017, 3. [Google Scholar] [CrossRef] [PubMed]
- Chewapreecha, C.; Holden, M.T.; Vehkala, M.; Valimaki, N.; Yang, Z.; Harris, S.R.; Mather, A.E.; Tuanyok, A.; De Smet, B.; Le Hello, S.; et al. Global and regional dissemination and evolution of Burkholderia pseudomallei. Nat. Microbial. 2017, 2, 16263. [Google Scholar] [CrossRef] [PubMed]
| Phase | Antibiotic | Adult Dose | Child Dose |
|---|---|---|---|
| Intensive | Meropenem | 1 g intravenously 8-hourly 2 g intravenously 8-hourly 1 | 25 mg/kg up to 1 g intravenously 8-hourly 50 mg/kg up to 2 g intravenously 8-hourly 1 |
| Intensive | Ceftazidime | 2 g intravenously 6-hourly | 50 mg/kg up to 2 g intravenously 6-hourly |
| Intensive and eradication | Trimethoprim-sulfamethoxazole | ≥60 kg: 320 + 1600 mg orally 12-hourly 40–60 kg: 240 + 1200 mg orally 12-hourly | 6 + 30 mg/kg up to 240 + 1200 mg orally 12-hourly |
| Eradication | Amoxicillin-clavulanate | 20/5 mg/kg orally 8-hourly | 20/5 mg/kg orally 8-hourly |
| Eradication | Doxycycline | 100 mg orally 12-hourly | Not recommended |
| Site of Infection | Minimum Intensive Phase Duration (Weeks) | Eradication Phase Duration (Months) |
|---|---|---|
| Cutaneous infection only | 2 | 3 |
| Bacteraemia without focus | 2 | 3 |
| Pneumonia without lymphadenopathy or ICU admission | 2 | 3 |
| Pneumonia with lymphadenopathy 1 or ICU admission | 4 | 3 |
| Deep seated collection 2 | 4 3 | 3 |
| Septic arthritis | 4 3 | 3 |
| Osteomyelitis | 6 | 6 |
| Central nervous system infection | 8 | 6 |
| Mycotic aneurysm or other arterial infection | 8 | 6 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Smith, S.; Hanson, J.; Currie, B.J. Melioidosis: An Australian Perspective. Trop. Med. Infect. Dis. 2018, 3, 27. https://doi.org/10.3390/tropicalmed3010027
Smith S, Hanson J, Currie BJ. Melioidosis: An Australian Perspective. Tropical Medicine and Infectious Disease. 2018; 3(1):27. https://doi.org/10.3390/tropicalmed3010027
Chicago/Turabian StyleSmith, Simon, Josh Hanson, and Bart J. Currie. 2018. "Melioidosis: An Australian Perspective" Tropical Medicine and Infectious Disease 3, no. 1: 27. https://doi.org/10.3390/tropicalmed3010027
APA StyleSmith, S., Hanson, J., & Currie, B. J. (2018). Melioidosis: An Australian Perspective. Tropical Medicine and Infectious Disease, 3(1), 27. https://doi.org/10.3390/tropicalmed3010027





