Protection Against CNS-Targeted Rabies Virus Infection is Dependent upon Type-1 Immune Mechanisms Induced by Live-Attenuated Rabies Vaccines
Abstract
:1. Introduction
2. Materials and Methods
2.1. Mice and Study Approval
2.2. Virus and Vaccine Strains
2.3. Virus Inactivation
2.4. Immunization and Challenge of Mice
2.5. Neutralizing Antibody (VNA)Titer and Serum Antibody Isotyping
2.6. StatisticalAnalysis
3. Results
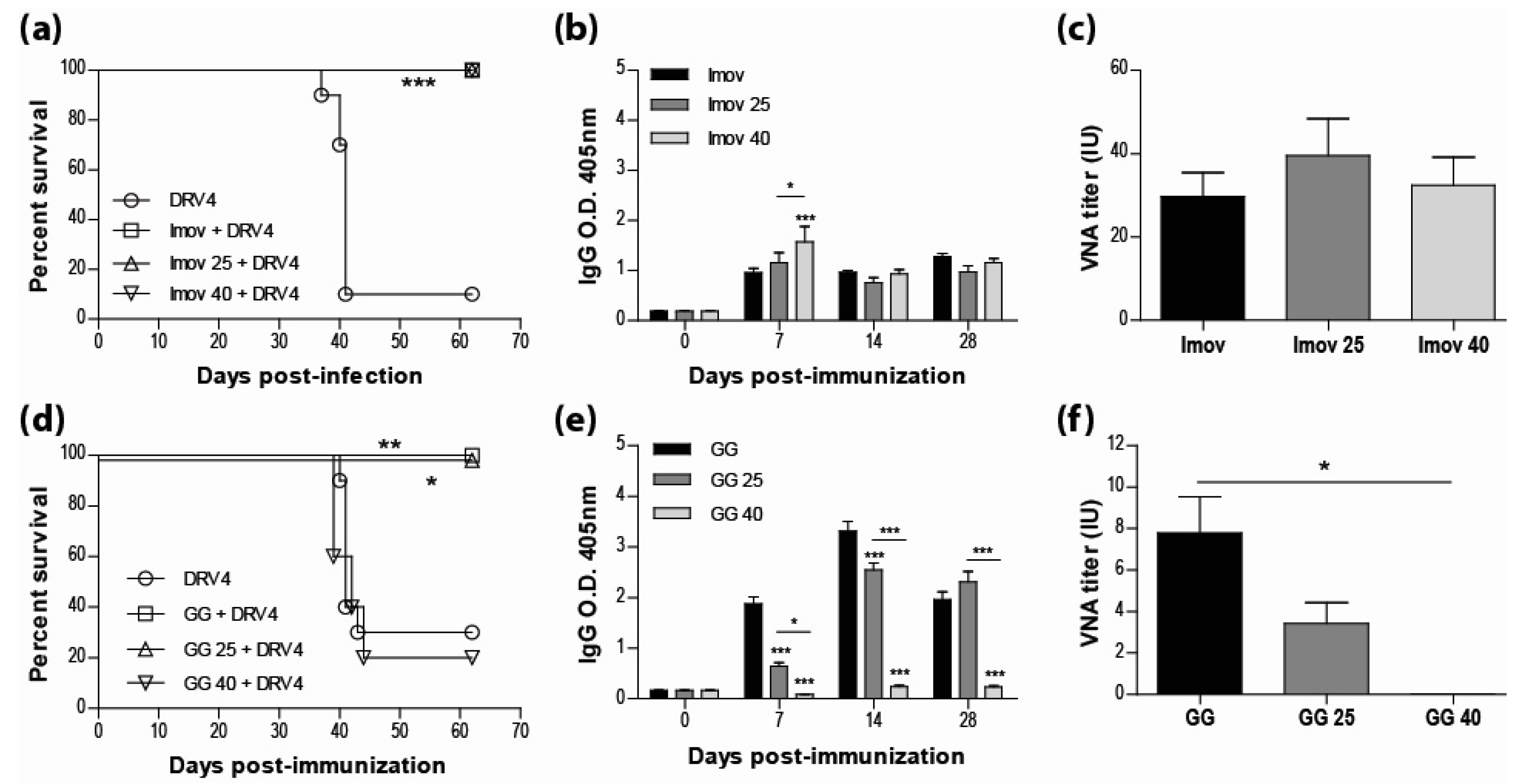
3.1. Live-Attenuated RABV are Protective and Stable at Room Temperature
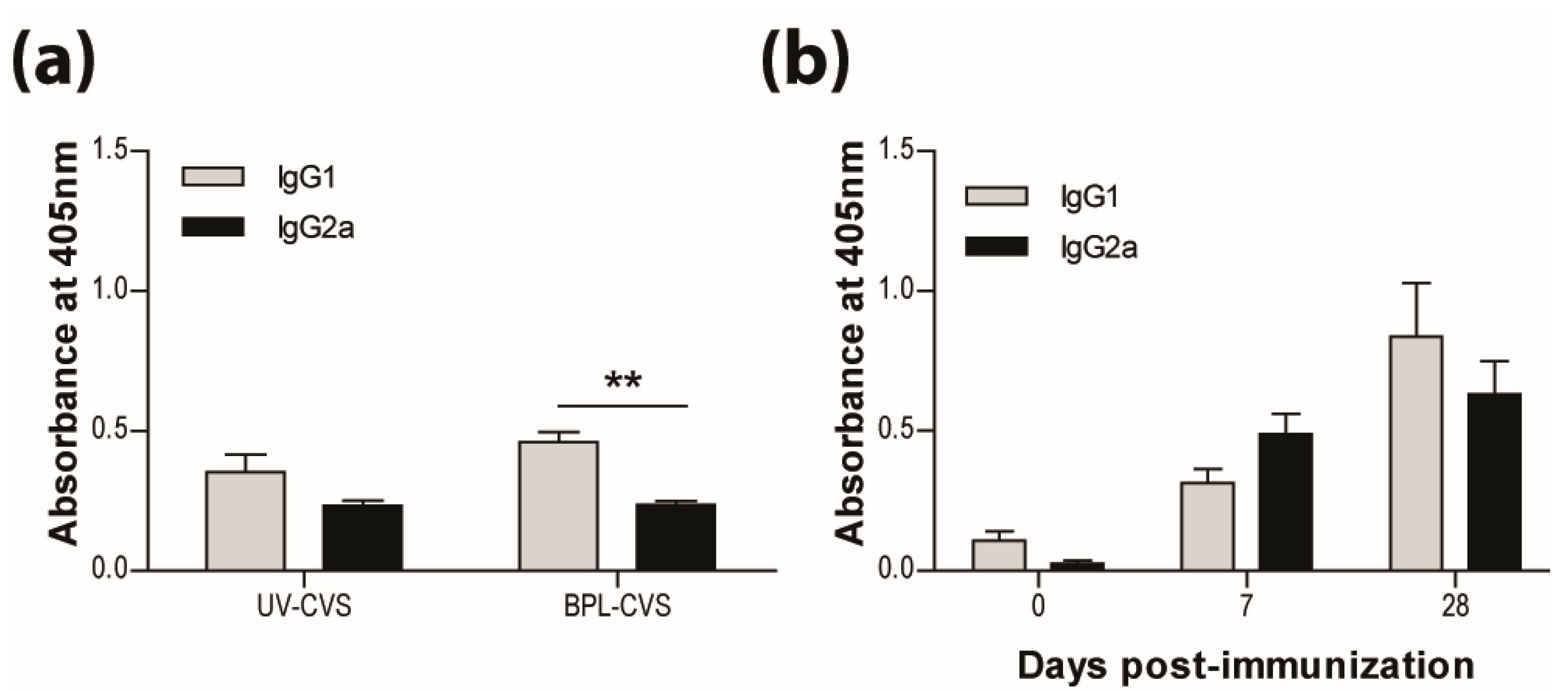
3.2. IMOVAX®Induces a Mixed Th1/Th2 Immune Response
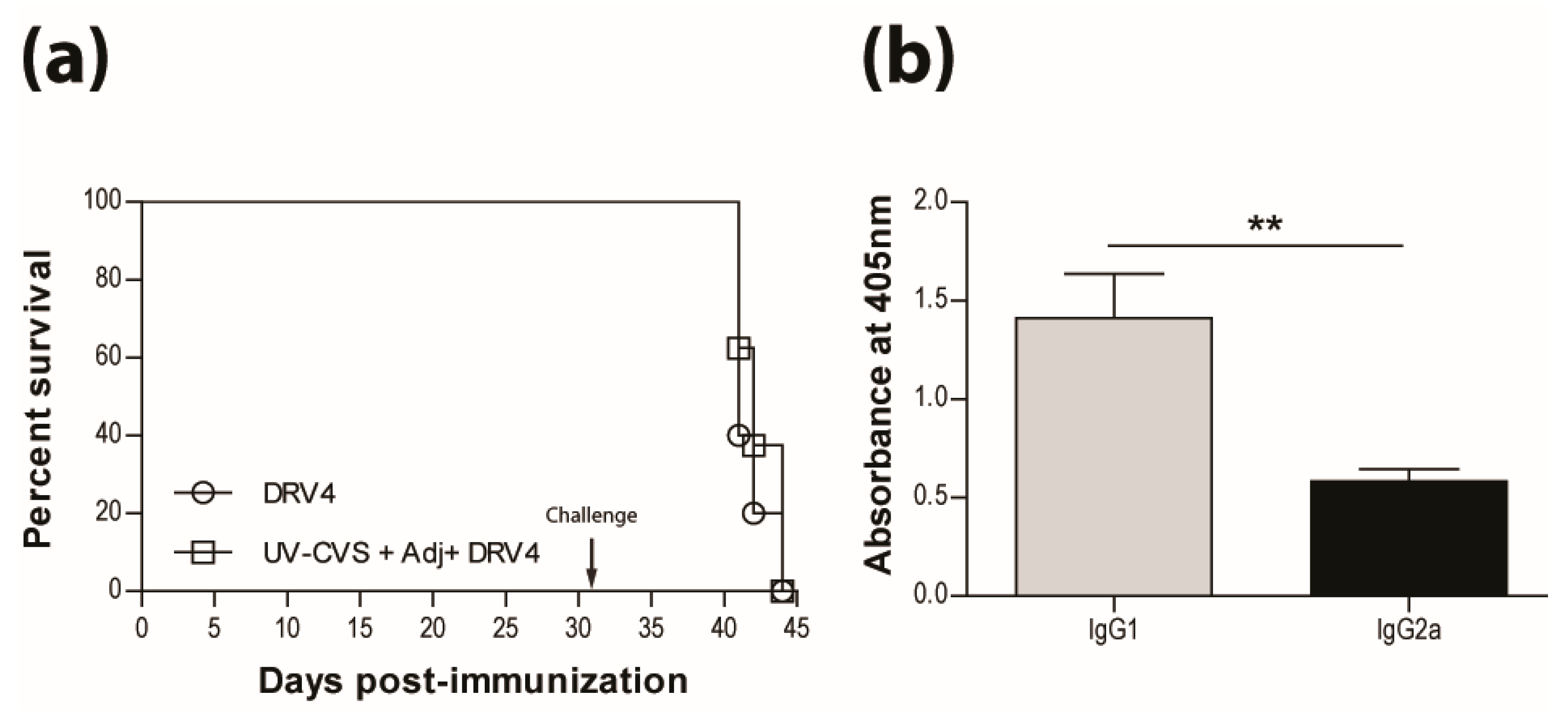
3.3. Addition of Adjuvant to Inactivated Vaccine does not Promote Protection
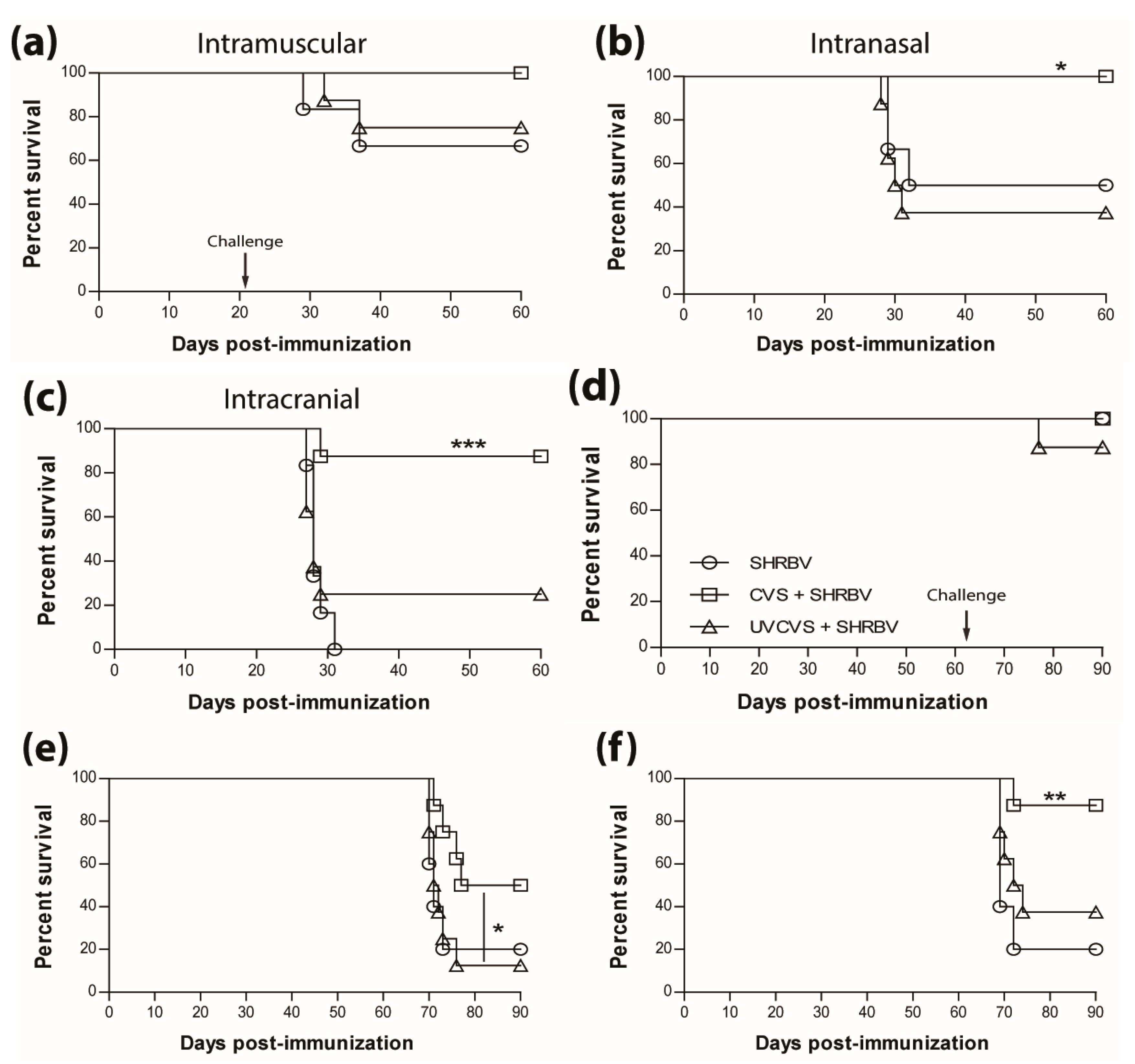
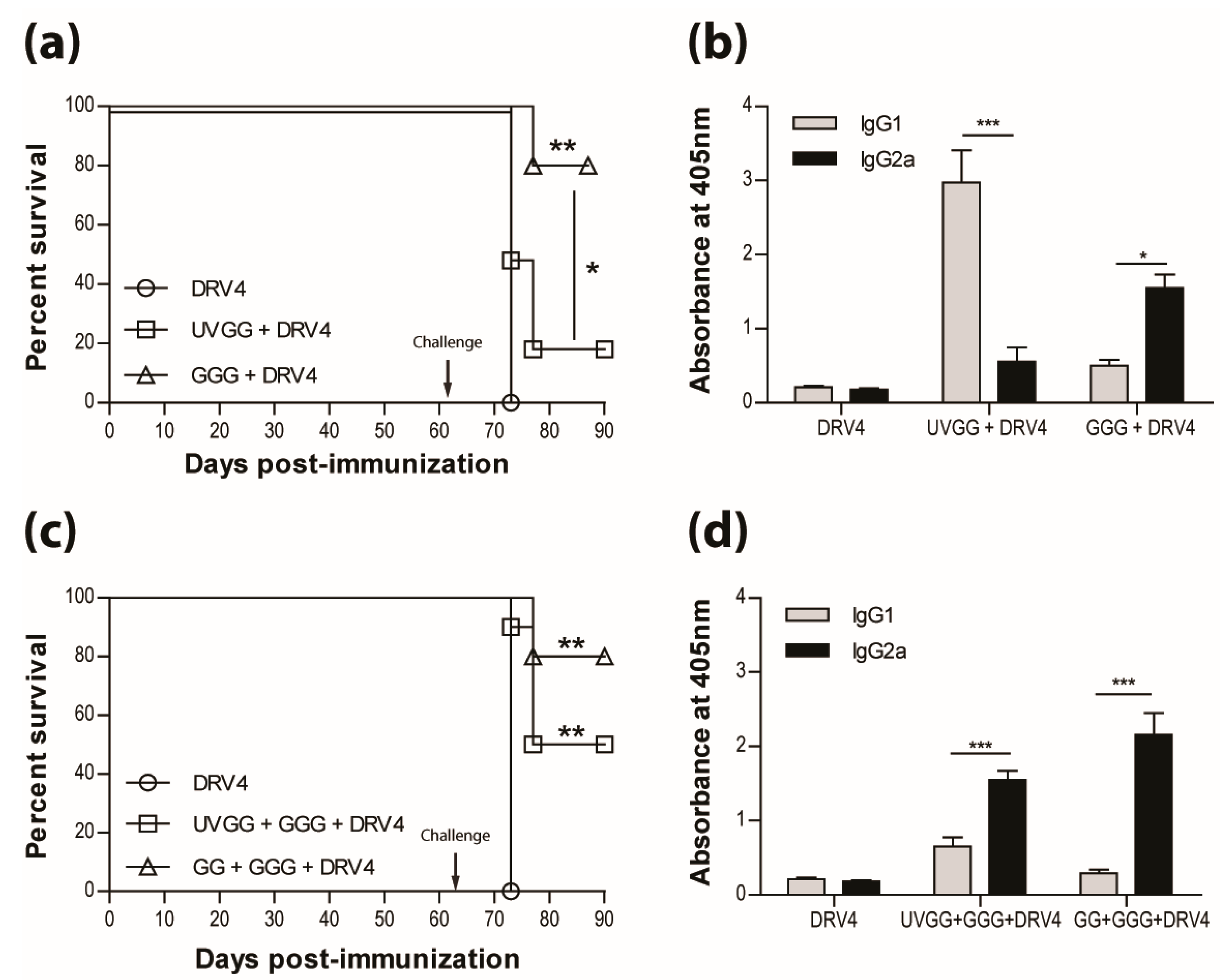
3.4. Vaccination Efficacy is Dictated by the Immunization Regimen and the Challenge Route
3.5. Type-1 Immunity is Critical for Protection AgainstWild-Type RABVInfection
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Afonso, C.L.; Amarasinghe, G.K.; Banyai, K.; Bao, Y.; Basler, C.F.; Bavari, S.; Bejerman, N.; Blasdell, K.R.; Briand, F.X.; Briese, T.; et al. Taxonomy of the order mononegavirales: Update 2016. Arch. Virol. 2016, 161, 2351–2360. [Google Scholar] [CrossRef] [PubMed]
- Hampson, K.; Coudeville, L.; Lembo, T.; Sambo, M.; Kieffer, A.; Attlan, M.; Barrat, J.; Blanton, J.D.; Briggs, D.J.; Cleaveland, S.; et al. Estimating the global burden of endemic canine rabies. PLoS Neglect. Trop. Dis. 2015, 9, e0003709. [Google Scholar]
- Crowcroft, N.S.; Thampi, N. The prevention and management of rabies. BMJ 2015, 350, g7827. [Google Scholar] [CrossRef] [PubMed]
- Wallace, R.M.; Undurraga, E.A.; Blanton, J.D.; Cleaton, J.; Franka, R. Elimination of dog-mediated human rabies deaths by 2030: Needs assessment and alternatives for progress based on dog vaccination. Front. Vet. Sci. 2017, 4, 9. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Who Expert Committee on Biological Standardization; World Health Organization: Geneva, Switzerland, 2007; pp. 1–340. [Google Scholar]
- Dodet, B.; Durrheim, D.N.; Rees, H. Rabies: Underused vaccines, unnecessary deaths. Vaccine 2014, 32, 2017–2019. [Google Scholar] [CrossRef] [PubMed]
- Pichon, S.; Guinet-Morlot, F.; Minutello, M.; Donazzolo, Y.; Rouzier, R.; Chassard, D.; Fitoussi, S.; Hou, V. A serum-free, purified vero cell rabies vaccine is safe and as immunogenic as the reference vaccine verorab for pre-exposure use in healthy adults: Results from a randomized controlled phase-ii trial. Vaccine 2013, 31, 2295–2301. [Google Scholar] [CrossRef] [PubMed]
- Keates, L. Rabies vaccines: WHO position paper-recommendations. Vaccine 2010, 28, 7140–7142. [Google Scholar]
- Wilde, H. Failures of post-exposure rabies prophylaxis. Vaccine 2007, 25, 7605–7609. [Google Scholar] [CrossRef] [PubMed]
- Shantavasinkul, P.; Wilde, H. Postexposure prophylaxis for rabies in resource-limited/poor countries. Adv. Virus Res. 2011, 79, 291–307. [Google Scholar] [PubMed]
- Wilde, H.; Lumlertdacha, B.; Meslin, F.X.; Ghai, S.; Hemachudha, T. Worldwide rabies deaths prevention—A focus on the current inadequacies in postexposure prophylaxis of animal bite victims. Vaccine 2016, 34, 187–189. [Google Scholar] [CrossRef] [PubMed]
- Barkhouse, D.A.; Faber, M.; Hooper, D.C. Pre- and post-exposure safety and efficacy of attenuated rabies virus vaccines are enhanced by their expression of IFN-gamma. Virology 2015, 474, 174–180. [Google Scholar] [CrossRef] [PubMed]
- Barkhouse, D.A.; Garcia, S.A.; Bongiorno, E.K.; Lebrun, A.; Faber, M.; Hooper, D.C. Expression of interferon gamma by a recombinant rabies virus strongly attenuates the pathogenicity of the virus via induction of type 1 interferon. J. Virol. 2015, 89, 312–322. [Google Scholar] [CrossRef] [PubMed]
- Hooper, D.C.; Phares, T.W.; Fabis, M.J.; Roy, A. The production of antibody by invading B cells is required for the clearance of rabies virus from the central nervous system. PLoS Neglect. Trop. Dis. 2009, 3, e535. [Google Scholar] [CrossRef] [PubMed]
- Lebrun, A.; Portocarrero, C.; Kean, R.B.; Barkhouse, D.A.; Faber, M.; Hooper, D.C. T-bet is required for the rapid clearance of attenuated rabies virus from central nervous system tissue. J. Immunol. 2015, 195, 4358–4368. [Google Scholar] [CrossRef] [PubMed]
- Faber, M.; Li, J.; Kean, R.B.; Hooper, D.C.; Alugupalli, K.R.; Dietzschold, B. Effective preexposure and postexposure prophylaxis of rabies with a highly attenuated recombinant rabies virus. Pro. Natl. Acad. Sci. USA 2009, 106, 11300–11305. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; McGettigan, J.P.; Faber, M.; Schnell, M.J.; Dietzschold, B. Infection of monocytes or immature dendritic cells (DCS) with an attenuated rabies virus results in DC maturation and a strong activation of the nfkappab signaling pathway. Vaccine 2008, 26, 419–426. [Google Scholar] [CrossRef] [PubMed]
- Dietzschold, B.; Morimoto, K.; Hooper, D.C.; Smith, J.S.; Rupprecht, C.E.; Koprowski, H. Genotypic and phenotypic diversity of rabies virus variants involved in human rabies: Implications for postexposure prophylaxis. J. Hum. Virol. 2000, 3, 50–57. [Google Scholar] [PubMed]
- Dietzschold, B.; Wunner, W.H.; Wiktor, T.J.; Lopes, A.D.; Lafon, M.; Smith, C.L.; Koprowski, H. Characterization of an antigenic determinant of the glycoprotein that correlates with pathogenicity of rabies virus. Proc. Natl. Acad. Sci. USA 1983, 80, 70–74. [Google Scholar] [CrossRef]
- Manning, S.E.; Rupprecht, C.E.; Fishbein, D.; Hanlon, C.A.; Lumlertdacha, B.; Guerra, M.; Meltzer, M.I.; Dhankhar, P.; Vaidya, S.A.; Jenkins, S.R.; et al. Human Rabies Prevention--United States, 2008: Recommendations of the Advisory Committee On Immunization Practices; MMWR. Morbidity and Mortality Weekly Report; Recommendations and Reports/Centers for Disease Control: Atlanta, GA, USA, 2008; 57, pp. 1–28.
- Food and Drug Administration: Imovax. Available online: https://www.fda.gov/BiologicsBloodVaccines/Vaccines/ApprovedProducts/ucm180097.htm (accessed on 4 April 2017).
- Perrin, P.; Morgeaux, S. Inactivation of DNA by beta-propiolactone. Biologicals 1995, 23, 207–211. [Google Scholar] [CrossRef] [PubMed]
- Wiktor, T.J.; Macfarlan, R.I.; Foggin, C.M.; Koprowski, H. Antigenic analysis of rabies and mokola virus from Zimbabwe using monoclonal antibodies. Dev. Biol. Stand. 1984, 57, 199–211. [Google Scholar]
- Phares, T.W.; Kean, R.B.; Mikheeva, T.; Hooper, D.C. Regional differences in blood-brain barrier permeability changes and inflammation in the apathogenic clearance of virus from the central nervous system. J. Immunol. 2006, 176, 7666–7675. [Google Scholar] [CrossRef] [PubMed]
- Hooper, D.C.; Morimoto, K.; Bette, M.; Weihe, E.; Koprowski, H.; Dietzschold, B. Collaboration of antibody and inflammation in clearance of rabies virus from the central nervous system. J. Virol. 1998, 72, 3711–3719. [Google Scholar] [PubMed]
- Roy, A.; Phares, T.W.; Koprowski, H.; Hooper, D.C. Failure to open the blood-brain barrier and deliver immune effectors to central nervous system tissues leads to the lethal outcome of silver-haired bat rabies virus infection. J. Virol. 2007, 81, 1110–1118. [Google Scholar] [CrossRef] [PubMed]
- Phares, T.W.; Fabis, M.J.; Brimer, C.M.; Kean, R.B.; Hooper, D.C. A peroxynitrite-dependent pathway is responsible for blood-brain barrier permeability changes during a central nervous system inflammatory response: TNF-alpha is neither necessary nor sufficient. J. Immunol. 2007, 178, 7334–7343. [Google Scholar] [CrossRef] [PubMed]
- Champion, J.M.; Kean, R.B.; Rupprecht, C.E.; Notkins, A.L.; Koprowski, H.; Dietzschold, B.; Hooper, D.C. The development of monoclonal human rabies virus-neutralizing antibodies as a substitute for pooled human immune globulin in the prophylactic treatment of rabies virus exposure. J. Immunol. Methods 2000, 235, 81–90. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, S.; Li, L.; Hu, R.; Lin, H.; Liu, H.; Liu, F.; Shao, H.; Liu, Y. Ineffectiveness of rabies vaccination alone for post-exposure protection against rabies infection in animal models. Antivir. Res. 2016, 135, 56–61. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, S.; Li, W.; Hu, Y.; Zhao, J.; Liu, F.; Lin, H.; Liu, Y.; Wang, L.; Xu, S.; et al. A novel rabies vaccine based-on toll-like receptor 3 (TLR3) agonist pika adjuvant exhibiting excellent safety and efficacy in animal studies. Virology 2016, 489, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Franka, R.; Henderson, H.; Rupprecht, C.E. Live attenuated rabies virus co-infected with street rabies virus protects animals against rabies. Vaccine 2011, 29, 4195–4201. [Google Scholar] [CrossRef] [PubMed]
- Smith, T.G.; Siirin, M.; Wu, X.; Hanlon, C.A.; Bronshtein, V. Rabies vaccine preserved by vaporization is thermostable and immunogenic. Vaccine 2015, 33, 2203–2206. [Google Scholar] [CrossRef] [PubMed]
- Morimoto, K.; McGettigan, J.P.; Foley, H.D.; Hooper, D.C.; Dietzschold, B.; Schnell, M.J. Genetic engineering of live rabies vaccines. Vaccine 2001, 19, 3543–3551. [Google Scholar] [CrossRef]
- Morimoto, K.; Hooper, D.C.; Spitsin, S.; Koprowski, H.; Dietzschold, B. Pathogenicity of different rabies virus variants inversely correlates with apoptosis and rabies virus glycoprotein expression in infected primary neuron cultures. J. Virol. 1999, 73, 510–518. [Google Scholar] [PubMed]
- Schutsky, K.; Curtis, D.; Bongiorno, E.K.; Barkhouse, D.A.; Kean, R.B.; Dietzschold, B.; Hooper, D.C.; Faber, M. Intramuscular inoculation of mice with the live-attenuated recombinant rabies virus trigas results in a transient infection of the draining lymph nodes and a robust, long-lasting protective immune response against rabies. J. Virol. 2013, 87, 1834–1841. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Ertel, A.; Portocarrero, C.; Barkhouse, D.A.; Dietzschold, B.; Hooper, D.C.; Faber, M. Postexposure treatment with the live-attenuated rabies virus (RV) vaccine trigas triggers the clearance of wild-type RV from the central nervous system (CNS) through the rapid induction of genes relevant to adaptive immunity in CNS tissues. J. Virol. 2012, 86, 3200–3210. [Google Scholar] [CrossRef] [PubMed]
- Vos, A.; Kretzschmar, A.; Ortmann, S.; Lojkic, I.; Habla, C.; Muller, T.; Kaiser, C.; Hundt, B.; Schuster, P. Oral vaccination of captive small Indian mongoose (Herpestes auropunctatus) against rabies. J. Wildl. Dis. 2013, 49, 1033–1036. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lebrun, A.; Garcia, S.; Li, J.; Kean, R.B.; Hooper, D.C. Protection Against CNS-Targeted Rabies Virus Infection is Dependent upon Type-1 Immune Mechanisms Induced by Live-Attenuated Rabies Vaccines. Trop. Med. Infect. Dis. 2017, 2, 22. https://doi.org/10.3390/tropicalmed2030022
Lebrun A, Garcia S, Li J, Kean RB, Hooper DC. Protection Against CNS-Targeted Rabies Virus Infection is Dependent upon Type-1 Immune Mechanisms Induced by Live-Attenuated Rabies Vaccines. Tropical Medicine and Infectious Disease. 2017; 2(3):22. https://doi.org/10.3390/tropicalmed2030022
Chicago/Turabian StyleLebrun, Aurore, Samantha Garcia, Jianwei Li, Rhonda B. Kean, and D. Craig Hooper. 2017. "Protection Against CNS-Targeted Rabies Virus Infection is Dependent upon Type-1 Immune Mechanisms Induced by Live-Attenuated Rabies Vaccines" Tropical Medicine and Infectious Disease 2, no. 3: 22. https://doi.org/10.3390/tropicalmed2030022
APA StyleLebrun, A., Garcia, S., Li, J., Kean, R. B., & Hooper, D. C. (2017). Protection Against CNS-Targeted Rabies Virus Infection is Dependent upon Type-1 Immune Mechanisms Induced by Live-Attenuated Rabies Vaccines. Tropical Medicine and Infectious Disease, 2(3), 22. https://doi.org/10.3390/tropicalmed2030022



