Feasibility of Oral Rabies Vaccination of Dogs in Mexico
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Bait Acceptance Study
2.3. Immunogenicity Study
2.4. Statistical Analysis
3. Results
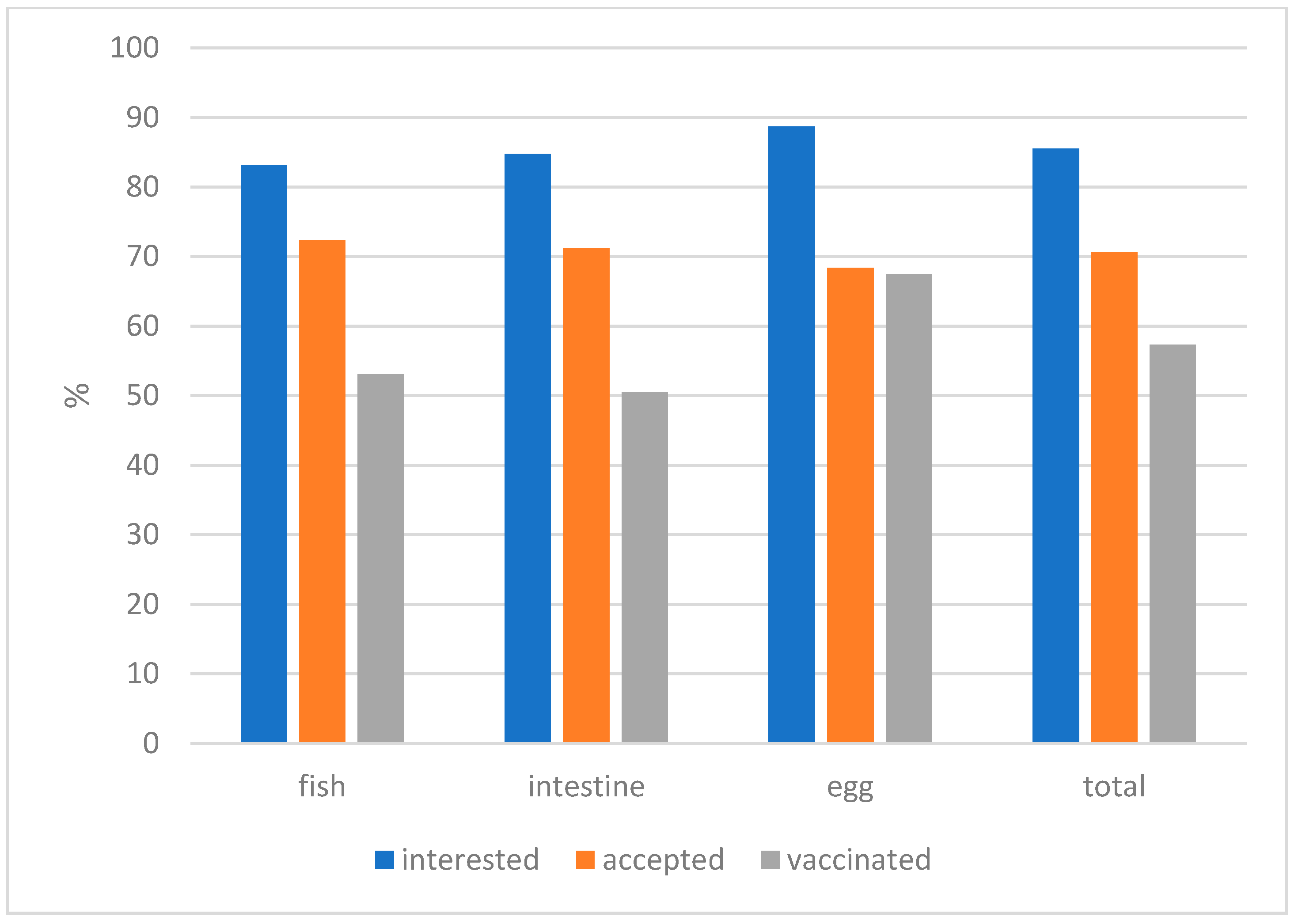
3.1. Bait Acceptance
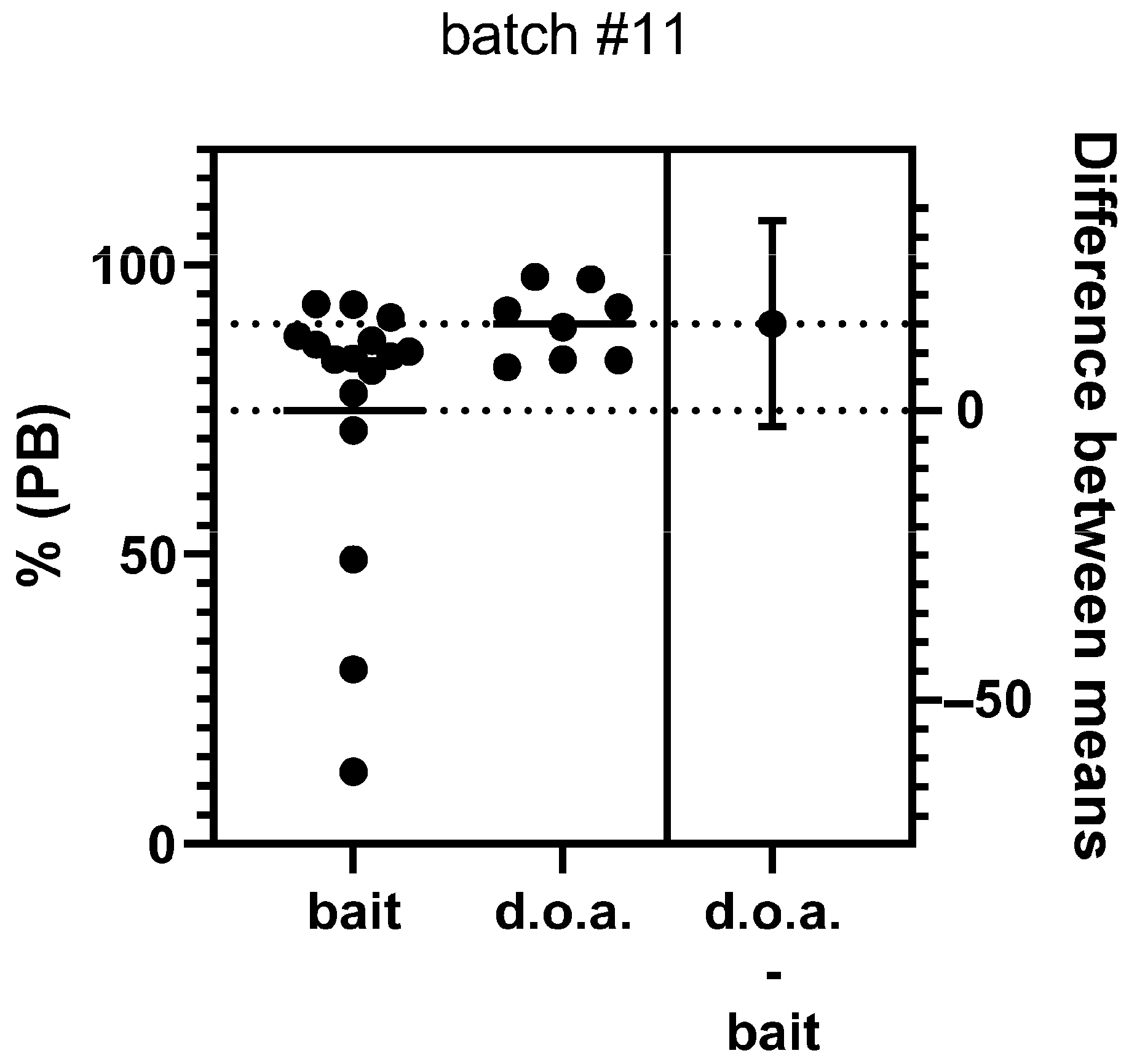
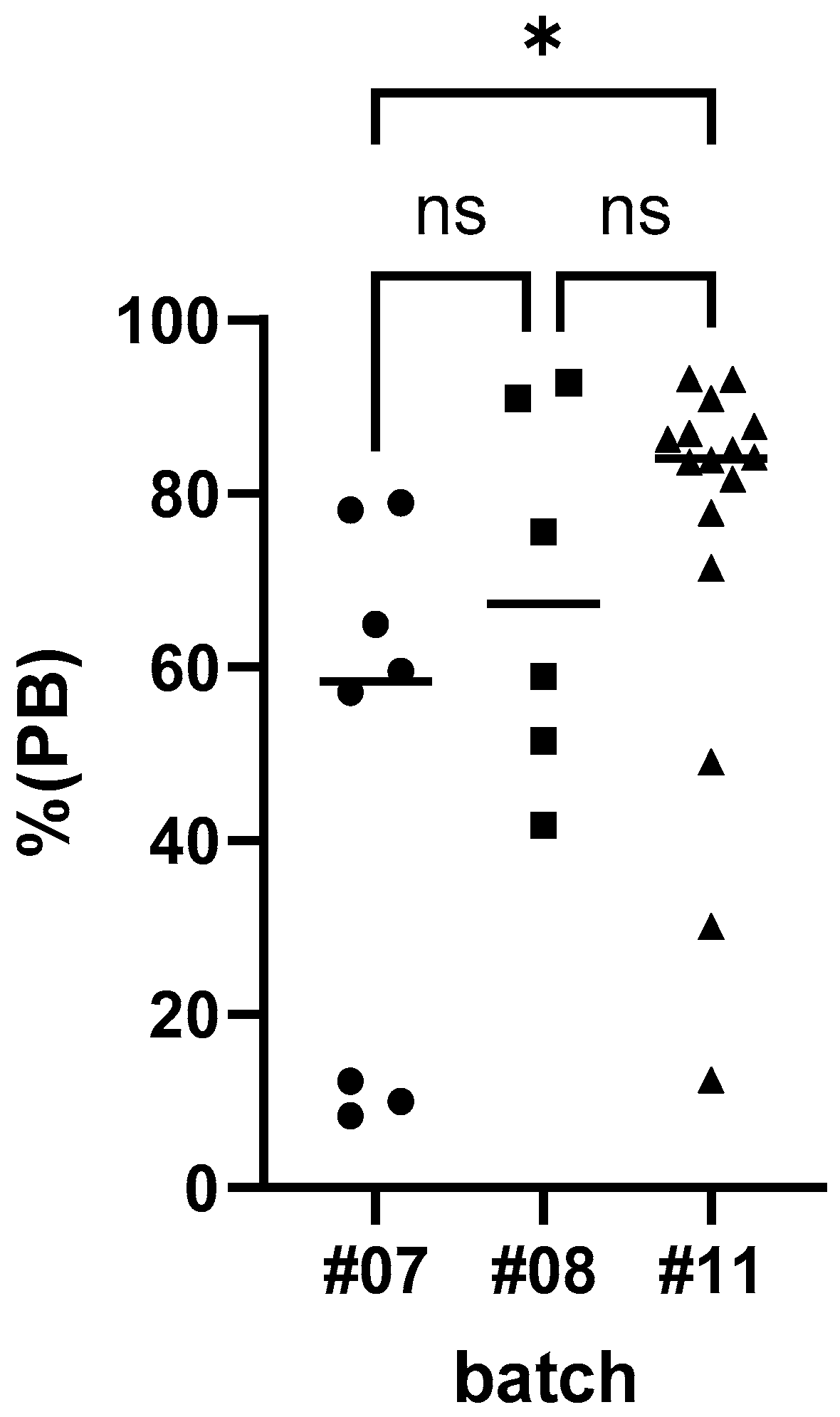
3.2. Immunogenicity
4. Discussion
5. Conclusions
- Rabies remains a public health threat in Mexico as the rabies virus continues to circulate among wild animals and can be transmitted to humans and domestic animals like dogs.
- The egg bait was not the most preferred bait by dogs in Mexico but achieved the highest vaccination rate of all three bait types offered.
- Although vaccination of dogs offered a vaccine bait that was slightly less effective (seroconversion rate) than parenteral vaccination under field conditions in Mexico, ORV can increase the overall vaccination coverage by reaching a significant part of the dog population that cannot readily be handled for vaccination.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO. Mexico Is Free from Human Rabies Transmitted by Dogs. 2019. Available online: https://www.who.int/news/item/21-12-2019-mexico-is-free-from-human-rabies-transmitted-by-dogs (accessed on 28 June 2024).
- Gutiérez, V. Mechanism and Benefits of Validation/Verification of Elimination of Human Rabies Transmitted by Dogs and Canine Rabies. In Proceedings of the 17th REDIPRA, Bogota, Columbia, 12–14 October 2023. [Google Scholar]
- Puebla-Rodríguez, P.; Almazán-Marín, C.; Garcés-Ayala, F.; Rendón-Franco, E.; Chávez-López, S.; Gómez-Sierra, M.; Sandoval-Borja, A.; Martínez-Solís, D.; Escamilla-Ríos, B.; Sauri-González, I.; et al. Rabies virus in white-nosed coatis (Nasua narica) in Mexico: What do we know so far? Front. Vet. Sci. 2023, 10, 1090222. [Google Scholar] [CrossRef]
- Arechiga Ceballos, N.; Puebla Rodrıguez, P.; Aguilar Setien, A. The New Face of Human Rabies in Mexico, What’s Next After Eradicating Rabies in Dogs. Vector-Borne Zoonotic Dis. 2022, 22, 69–75. [Google Scholar] [CrossRef]
- Alvarez Lucas, C.H.; Vargas Pino, F.; Baer, G.; Kuri Morales, P.; Gutiérrez Cedillo, V.; Llanas Blanco, M.A.; Hernandez Avila, M. Rabies control in Mexico. Dev. Biol. 2008, 131, 167–175. [Google Scholar]
- Gonzalez-Roldan, J.F.; Undurraga, E.A.; Meltzer, M.I.; Atkins, C.; Vargas-Pino, F.; Gutierrez Cedillo, V.; Ramon Hernandez-Perez, J. Cost-effectiveness of the national dog rabies prevention and control program in Mexico, 1990–2015. PLoS Negl. Trop. Dis. 2021, 15, e0009130. [Google Scholar] [CrossRef]
- Aboulfidaa, N.; Cliquet, F.; Robardet, E.; Darkaoui, S.; Wasniewski, M.; Kaiser, C.; Bobe, K.; Vos, A.; Fassi Fihri, O. Evaluation of bait acceptance and immune response in local dogs during an oral rabies vaccination field study in Morocco. Trop. Med. Infect. Dis. 2024, 9, 142. [Google Scholar] [CrossRef]
- Bender, S.; Bergman, D.; Vos, A.; Martin, A.; Chipman, R. Field studies evaluating bait acceptance and handling by dogs in Navajo Nation, USA. Trop. Med. Infect. Dis. 2017, 2, 17. [Google Scholar] [CrossRef]
- Husein, W.F.; Megawati-Saputra, I.L.; Suseno, P.P.; Arthawan, I.M.; Prayoga, I.M.A.; Daryono, J.; Vos, A.; Wicaksono, A.; Schoonman, L.; Weaver, J.; et al. Assessing Bait Acceptance of Local Dogs, Oral Vaccination Success and Human Contact Risk in Bali, Indonesia. One Health Implement. Res. 2023, 3, 16–29. [Google Scholar] [CrossRef]
- Kasemsuwan, S.; Chanachai, K.; Pinyopummintr, T.; Leelalapongsathon, K.; Sujit, K.; Vos, A. Field studies evaluating bait acceptance and—Handling by free-roaming dogs in Thailand. Vet. Sci. 2018, 5, 47. [Google Scholar] [CrossRef]
- Smith, T.G.; Vos, A.; Acky Crowdis, K.; Chirodea, C.; Medley, A.; Chipman, R.; Millien, M.; Qin, Y.; Blanton, J.; Wallace, R. Evaluation of Immune Responses in Dogs to Oral Rabies Vaccine under Field Conditions. Vaccine 2019, 37, 4743–4749. [Google Scholar] [CrossRef] [PubMed]
- Saputra, I.L.M.; Suwarno, S.; Husein, W.F.; Suseno, P.P.; Prayoga, I.M.A.; Vos, A.; Arthawan, I.M.; Schoonman, L.; Weaver, J.; Zainuddin, N. Immunogenicity of Oral Rabies Vaccine Strain SPBN GASGAS in Local Dogs in Bali, Indonesia. Viruses 2023, 15, 1405. [Google Scholar] [CrossRef] [PubMed]
- Molini, U.; Hassel, R.; Ortmann, S.; Vos, A.; Loschke, M.; Shilongo, A.; Freuling, C.M.; Müller, T. Immunogenicity of the oral rabies vaccine strain SPBN GASGAS in dogs under field settings in Namibia. Front. Vet. Sci. 2021, 8, 737250. [Google Scholar] [CrossRef]
- Bonwitt, J.; Bonaparte, S.; Blanton, J.; Gibson, A.D.; Hoque, M.; Kennedy, E.; Islam, K.; Siddiqi, U.R.; Wallace, R.M.; Azam, S. Oral bait preferences and feasibility of oral rabies vaccination in Bangladeshi dogs. Vaccine 2020, 38, 5021–5026. [Google Scholar] [CrossRef]
- Gibson, A.D.; Mazeri, S.; Yale, G.; Desai, S.; Naik, V.; Corfmat, J.; Ortmann, S.; King, A.; Müller, T.; Handel, I.; et al. Development of a non-meat based, mass producible and effective bait for oral vaccination of dogs against rabies in Goa State, India. Trop. Med. Infect. Dis. 2019, 4, 118. [Google Scholar] [CrossRef]
- Undurraga, E.A.; Millien, M.F.; Allel, K.; Etheart, M.D.; Cleaton, J.; Ross, Y.; Vaccine Evaluation Team; Wallace, R.M. Costs and effectiveness of alternative dog vaccination strategies to improve dog population coverage in rural and urban settings during a rabies outbreak. Vaccine 2020, 38, 6162–6173. [Google Scholar] [CrossRef]
- Chanachai, K.; Wongphruksasoong, V.; Vos, A.; Leelahapongsathon, K.; Tangwangvivat, R.; Sagarasaeranee, O.; Lekcharoen, P.; Trinuson, P.; Kasemsuwan, S. Feasibility and Effectiveness Studies with Oral Vaccination of Free-Roaming Dogs against Rabies in Thailand. Viruses 2021, 13, 571. [Google Scholar] [CrossRef]
- Freuling, C.M.; Busch, F.; Vos, A.; Ortmann, S.; Lohr, F.; Hedimbi, N.; Josephat, P.; Nelson, H.A.; Shoombe, K.; Shilongo, A.; et al. Oral vaccination of dogs—Experiences from a field trial in Namibia. PLoS Negl. Trop. Dis. 2022, 6, e0010422. [Google Scholar] [CrossRef] [PubMed]
- Freuling, C.M.; Busch, F.; Shikongo, M.B.; Silume, N.; van der Westhuizen, J.; Khaiseb, S.; Shilongo, A.; Müller, T. Emergency response using oral rabies vaccination of dogs -field data from Namibia demonstrate high efficiency. One Health 2023, 16, 100562. [Google Scholar] [CrossRef] [PubMed]
- Leelahapongsathon, K.; Kasemsuwan, S.; Pinyopummintr, T.; Boodde, O.; Phawaphutayanchai, P.; Aiyara, N.; Bobe, K.; Vos, A.; Friedrichs, V.; Müller, T.; et al. Humoral immune response of Thai dogs after oral vaccination against rabies with the SPBN GASGAS vaccine strain. Vaccines 2020, 8, 573. [Google Scholar] [CrossRef]
- Langguth, A.; Leelahapongsathon, K.; Wannapong, N.; Kasemsuwan, S.; Ortmann, S.; Vos, A.; Böer, M. Comparative Study of Optical Markers to assess Bait System Efficiency concerning Vaccine Release in the Oral Cavity of Dogs. Viruses 2021, 13, 1382. [Google Scholar] [CrossRef]
- Bobe, K.; Ortmann, S.; Kaiser, C.; Perez-Bravo, D.; Gethmann, J.; Kliemt, J.; Körner, S.; Theuss, T.; Lindner, T.; Freuling, C.; et al. Efficacy of oral rabies vaccine baits containing SPBN GASGAS in domestic dogs according to international standards. Vaccines 2023, 11, 307. [Google Scholar] [CrossRef] [PubMed]
- Faber, M.; Dietzschold, B.; Li, J. Immunogenicity and safety of recombinant rabies viruses used for oral vaccination of stray dogs and wildlife. Zoonoses Public Health 2009, 56, 262–269. [Google Scholar] [CrossRef] [PubMed]
- Faber, M.; Faber, M.L.; Papaneri, A.; Bette, M.; Weihe, E.; Dietzschold, B.; Schnell, M.J. A single amino acid change in rabies virus glycoprotein increases virus spread and enhances virus pathogenicity. J. Virol. 2005, 79, 14141–14148. [Google Scholar] [CrossRef] [PubMed]
- Faber, M.; Pulmanausahakul, R.; Hodawadekar, S.S.; Spitsin, S.; McGettigan, J.P.; Schnell, M.J.; Dietzschold, B. Overexpression of the rabies virus glycoprotein results in enhancement of apoptosis and antiviral immune response. J. Virol. 2002, 76, 3374–3381. [Google Scholar] [CrossRef]
- Zöfel, P. Statistik in der Praxis; Fischer: Stuttgart, Germany, 1988. [Google Scholar]
- Velasco-Villa, A.; Escobar, L.E.; Sanchez, A.; Shi, M.; Streicker, D.G.; Gallardo-Romero, N.F.; Vargas-Pino, F.; Gutierrez-Cedillo, V.; Damon, I.; Emerson, G. Successful strategies implemented towards the elimination of canine rabies in the Western Hemi-sphere. Antivir. Res. 2017, 143, 1–12. [Google Scholar] [CrossRef]
- Vargas Pino, F.; Gutiérrez Cedillo, V.; Ëlvarez Lucas, C. Acceptance of Baits and Development of Immune Response Against the Rabies Virus in Canine Population of Mexico (Preliminary Report). In Rabies in the Americas 2009; Abstract Book; 2009; p. 62. Available online: https://rabiesintheamericas.com/wp-content/uploads/2009_RITA_XX.pdf (accessed on 4 July 2025).
- Vos, A. Oral Vaccination of Dogs against Rabies. Int. Anim. Health J. 2019, 6, 25–29. [Google Scholar]
- Frontini, M.G.; Fishbein, D.B.; Garza Ramos, J.; Flores Collins, E.; Balderas Torres, J.M.; Quiroz Huerta, G.; Gamez Rodriguez, J.J.; Belotto, A.J.; Dobbins, J.G.; Linhart, S.B.; et al. A field evaluation in Mexico of four baits for oral rabies vaccination of dogs. Am. J. Trop. Med. Hyg. 1992, 47, 310–316. [Google Scholar] [CrossRef]
- Bergman, D.; Bender, S.; Wenning, K.; Slate, D.; Rupprecht, C.; Heuser, C.; Delibertoi, T. Bait Acceptability for Delivery of Oral Rabies Vaccine to Free-Ranging Dogs on the Navajo and Hopi Nations. Dev. Biol. 2008, 131, 145–150. [Google Scholar]
- Freuling, C.M.; Eggerbauer, E.; Finke, S.; Kaiser, C.; Kaiser, C.; Kretzschmar, A.; Lange, E.; Nolden, T.; Ortmann, S.; Schröder, C.; et al. Efficacy of the oral rabies virus vaccine strain SPBN GASGAS in foxes and raccoon dogs. Vaccine 2019, 37, 4750–4757. [Google Scholar] [CrossRef] [PubMed]
- Moore, S.; Gilbert, A.; Vos, A.; Freuling, C.; Ellis, C.; Kliemt, J.; Müller, T. Review of rabies virus antibodies from oral vaccination as a correlate of protection against lethal infection in animals. Trop. Med. Infect. Dis. 2017, 2, 31. [Google Scholar] [CrossRef]
- Freuling, C.M.; Te Kamp, V.; Klein, A.; Günther, M.; Zaeck, L.; Potratz, M.; Eggerbauer, E.; Bobe, K.; Kaiser, C.; Kretzschmar, A.; et al. Long-term immunogenicity and efficacy of the oral rabies virus vaccine strain SPBN GASGAS in foxes. Viruses 2019, 11, 790. [Google Scholar] [CrossRef]
- Vos, A.; Kasemsuwan, S.; Leelahapongsathon, K.; Bobe, K.; Perez-Bravo, D.; Kliemt, J.; Phawaphutayanchi, P.; Aiyara, N.; Freuling, C.M.; Müller, T. Long-term immunity in orally vaccinated dogs against rabies induced using SPBN GASGAS. Vaccines X 2023, 15, 100410. [Google Scholar] [CrossRef] [PubMed]
- Te Kamp, V.; Freuling, C.M.; Vos, A.; Schuster, P.; Kaiser, C.; Ortmann, S.; Kretzschmar, A.; Nemitz, S.; Eggerbauer, E.; Ulrich, R.; et al. Oral rabies vaccination of wildlife: What drives vaccine uptake efficiency in different reservoir species? Sci. Rep. 2020, 10, 2919. [Google Scholar]
- Gutiérrez Cedillo, V.; Montoya Mondragón, L.A.; Ramón, J.; Colon, F.; Bobe, K.; Vos, A.; Lecuona Olivares, L.A.; López Ridaura, R. Feasibility of oral rabies vaccination of dogs in Mexico: Bait acceptance and immunogenicity studies. Rev. Asoc. Colomb. Infectologica 2023, 27, 17–18. [Google Scholar]



| Variable | Settings |
|---|---|
| date | 26 April; 27 April; 28 April |
| time (hh:mm) | 09:00–10:59; 11:00–12:59; 13:00–14:59; 15:00–16:59; 17:00–19:00 |
| level of restriction | restricted, free-roaming |
| social status | solitary; multiple |
| ownership | owned, ownerless, community |
| size | small; medium; large |
| sex | female; male |
| age | <12 months; ≥12 months |
| bait type | egg, intestine, fish meal |
| Variable | Settings | ||||
| date | 26 April | 27 April | 28 April | ||
| acceptance | 107/170 (64.85%) | 174/240 (73.11%) | 151/208 (73.30%) | ||
| vaccination | 90/170 (56.25%) | 132/240 (58.41%) | 127/208 (62.25%) | ||
| time | 9:00–10:59 | 11:00–12:59 | 13:00–14:59 | 15:00–16:59 | 17:00–18:59 |
| acceptance | 83/126 (66.40%) | 199/277 (73.70%) | 71/104 (68.93%) | 58/73 (79.45%) | 19/35 (54.29%) |
| vaccination | 67/126 (54.92%) | 166/277 (61.94%) | 55/104 (56.70%) | 45/73 (67.16%) | 15/35 (45.45%) |
| restriction | restricted | free-roaming | |||
| acceptance | 229/313 (73.40%) | 187/283 (67.51%) | |||
| vaccination | 179/313 (59.67%) | 159/283 (58.89) | |||
| social status | solitary | multiple | |||
| acceptance | 246/343 (69.82%) | 183/256 (72.33) | |||
| vaccination | 194/343 (59.33%) | 159/283 (59.35%) | |||
| ownership | owned | ownerless | community | ||
| acceptance | 252/349 (72.62%) | 104/150 (71.23%) | 16/27 (61.54%) | ||
| vaccination | 201/349 (60.18%) | 82/150 (56.94%) | 16/27 (61.54%) | ||
| size | small | medium | large | ||
| acceptance | 67/118 (57.26%) | 146/202 (73.00%) | 206/277 (75.74%) | ||
| vaccination | 56/118 (50.45%) | 116/202 (59.18%) | 170/277 (64.39) | ||
| sex | female | male | |||
| acceptance | 179/246 (73.36%) | 229/332 (70.03%) | |||
| vaccination | 143/246 (60.59%) | 186/332 (58.68%) | |||
| age | <12 months | ≥12 months | |||
| acceptance | 45/74 (60.81) | 320/435 (74.42%) | |||
| vaccination | 43/71 (58.11%) | 247/435 (59.66%) | |||
| bait type | egg | intestine | fish meal | ||
| acceptance | 145/216 (69.58%) | 131/185 (70.81%) | 154/217 (70.97%) | ||
| vaccination | 143/216 (66.20%) | 93/185 (50.27%) | 113/217 (52.07%) | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gutiérrez Cedillo, V.; Montoya Mondragón, L.A.; Fernández Colín, J.R.; Bobe, K.; Vos, A.; Lecuona Olivares, L.A.; López Ridaura, R. Feasibility of Oral Rabies Vaccination of Dogs in Mexico. Trop. Med. Infect. Dis. 2025, 10, 244. https://doi.org/10.3390/tropicalmed10090244
Gutiérrez Cedillo V, Montoya Mondragón LA, Fernández Colín JR, Bobe K, Vos A, Lecuona Olivares LA, López Ridaura R. Feasibility of Oral Rabies Vaccination of Dogs in Mexico. Tropical Medicine and Infectious Disease. 2025; 10(9):244. https://doi.org/10.3390/tropicalmed10090244
Chicago/Turabian StyleGutiérrez Cedillo, Verónica, Luis Antonio Montoya Mondragón, Jose Ramón Fernández Colín, Katharina Bobe, Ad Vos, Luis Armando Lecuona Olivares, and Ruy López Ridaura. 2025. "Feasibility of Oral Rabies Vaccination of Dogs in Mexico" Tropical Medicine and Infectious Disease 10, no. 9: 244. https://doi.org/10.3390/tropicalmed10090244
APA StyleGutiérrez Cedillo, V., Montoya Mondragón, L. A., Fernández Colín, J. R., Bobe, K., Vos, A., Lecuona Olivares, L. A., & López Ridaura, R. (2025). Feasibility of Oral Rabies Vaccination of Dogs in Mexico. Tropical Medicine and Infectious Disease, 10(9), 244. https://doi.org/10.3390/tropicalmed10090244







