Preparing for COVID-2x: Urban Planning Needs to Regard Urological Wastewater as an Invaluable Communal Public Health Asset and Not as a Burden
Abstract
1. Introduction
2. Sewage Testing as a Diagnostic Tool for Public Health
2.1. Testing Sewage for the Detection of Drugs and Pathogens
2.2. Sewage Testing as a Diagnostic Tool for the Presence of SARS-CoV-2
3. Contemporary Sewage and Wastewater Management
4. Planning
4.1. Concepts
4.2. Sampling Framework
4.3. Ensuring Future Testing Capacity
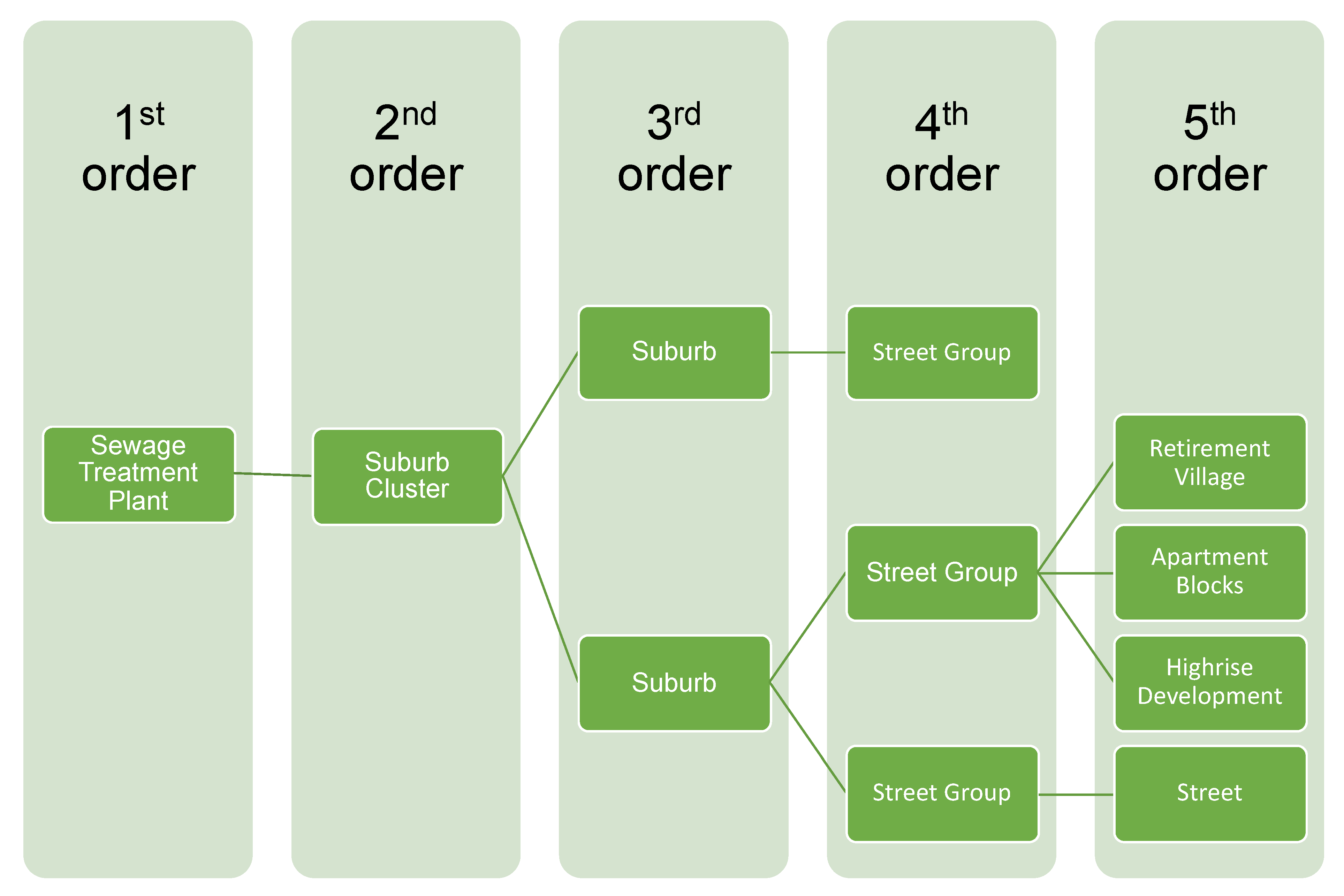
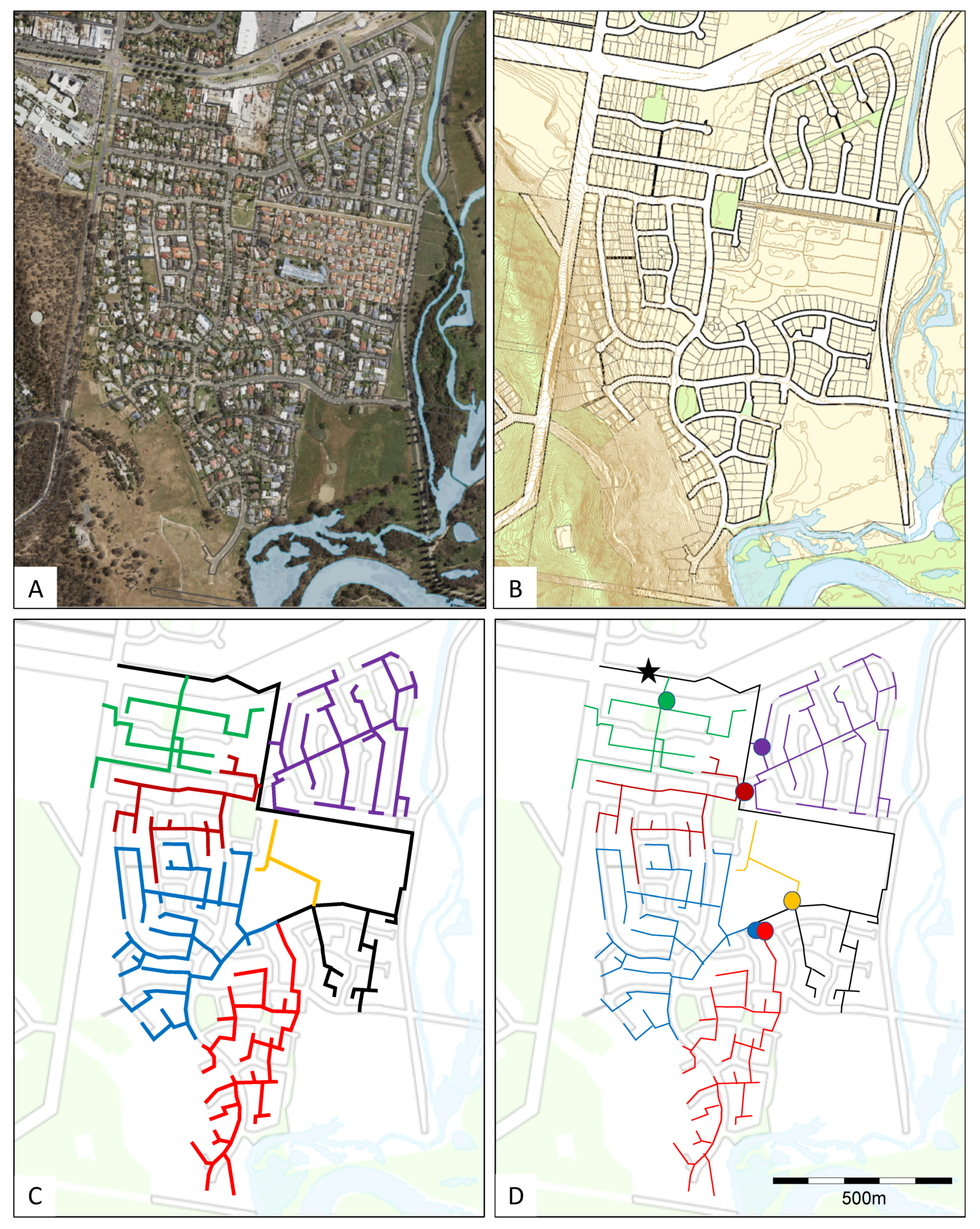
4.3.1. Design of Sewer Networks in New Housing Estates
4.3.2. Ensuring Ready Access to the Sewer Lines
4.3.3. Retrofitting of Existing Sewer Networks
5. Outlook
Funding
Conflicts of Interest
References
- WHO. Naming the Coronavirus Disease (COVID-19) and the Virus That Causes It. Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/technical-guidance/naming-the-coronavirus-disease-(covid-2019)-and-the-virus-that-causes-it (accessed on 10 August 2020).
- Ritchie, H.; Ortiz-Ospina, E.; Beltekian, D.; Mathieu, E.; Hasell, J.; Macdonald, B.; Giattino, C.; Roser, M. Coronavirus (COVID-19) Cases. Our World in Data. Available online: https://ourworldindata.org/covid-cases (accessed on 25 January 2021).
- Spreeuwenberg, P.; Kroneman, M.; Paget, J. Reassessing the global mortality burden of the 1918 influenza pandemic. Am. J. Epidemiol. 2018, 187, 2561–2567. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, P.; Clarke, S.; Kirk, A. Covid world map: Which countries have the most coronavirus cases and deaths? Guardian 2021. Available online: https://www.theguardian.com/world/2021/jun/11/covid-world-map-which-countries-have-the-most-coronavirus-vaccinations-cases-and-deaths (accessed on 11 June 2021).
- Inayatullah, S. Neither A Black Swan Nor A Zombie Apocalypse: The Futures Of A World With The COVID-19 Coronavirus. J. Futures Stud. 2020. Available online: https://jfsdigital.org/2020/03/18/neither-a-black-swan-nor-a-zombie-apocalypse-the-futures-of-a-world-with-the-covid-19-coronavirus/ (accessed on 25 January 2021).
- Goh, K.-T.; Cutter, J.; Heng, B.-H.; Ma, S.; Koh, B.K.; Kwok, C.; Toh, C.-M.; Chew, S.-K. Epidemiology and control of SARS in Singapore. Ann. Acad. Med. Singap. 2006, 35, 301. [Google Scholar] [PubMed]
- Cunha, C.B.; Opal, S.M. Middle East respiratory syndrome (MERS) A new zoonotic viral pneumonia. Virulence 2014, 5, 650–654. [Google Scholar] [CrossRef] [PubMed]
- Ye, Z.-W.; Yuan, S.; Yuen, K.-S.; Fung, S.-Y.; Chan, C.-P.; Jin, D.-Y. Zoonotic origins of human coronaviruses. Int. J. Biol. Sci. 2020, 16, 1686. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.-F.; Anderson, D.E.; Mackenzie, J.S.; Merson, M.H. From Hendra to Wuhan: What has been learned in responding to emerging zoonotic viruses. Lancet 2020, 395, e33–e34. [Google Scholar] [CrossRef]
- Peeri, N.C.; Shrestha, N.; Rahman, M.S.; Zaki, R.; Tan, Z.; Bibi, S.; Baghbanzadeh, M.; Aghamohammadi, N.; Zhang, W.; Haque, U. The SARS, MERS and novel coronavirus (COVID-19) epidemics, the newest and biggest global health threats: What lessons have we learned? Int. J. Epidemiol. 2020, 49, 717–726. [Google Scholar] [CrossRef]
- Neuwirth, L.S.; Jović, S.; Mukherji, B.R. Reimagining higher education during and post-COVID-19: Challenges and opportunities. J. Adult Contin. Educ. 2020. [Google Scholar] [CrossRef]
- Kramer, A.; Kramer, K.Z. The potential impact of the Covid-19 pandemic on occupational status, work from home, and occupational mobility. J. Vocat. Behav. 2020, 119, 103442. [Google Scholar] [CrossRef]
- Haywood, K.M. A post-COVID future: Tourism community re-imagined and enabled. Tour. Geogr. 2020, 22, 599–609. [Google Scholar] [CrossRef]
- Spennemann, D.H.R.; Whitsed, R. The impact of COVID-19 on the Australian outdoor recreation industry from the perspective of practitioners. J. Sust. Tour. 2021, 100445. [Google Scholar] [CrossRef]
- Musselwhite, C.; Avineri, E.; Susilo, Y. Editorial JTH 16–The Coronavirus Disease COVID-19 and implications for transport and health. J. Transp. Health 2020, 16, 100853. [Google Scholar] [CrossRef]
- Bereitschaft, B.; Scheller, D. How Might the COVID-19 Pandemic Affect 21st Century Urban Design, Planning, and Development? Urban Sci. 2020, 4, 56. [Google Scholar] [CrossRef]
- Honey-Rosés, J.; Anguelovski, I.; Chireh, V.K.; Daher, C.; Konijnendijk van den Bosch, C.; Litt, J.S.; Mawani, V.; McCall, M.K.; Orellana, A.; Oscilowicz, E. The impact of COVID-19 on public space: An early review of the emerging questions–design, perceptions and inequities. Cities Health 2020, 1–17. [Google Scholar] [CrossRef]
- Spennemann, D.H.R. Residential Architecture in a post-pandemic world: Implications of COVID-19 for new construction and for adapting heritage buildings. J. Green Build. 2021, 16, 199–215. [Google Scholar] [CrossRef]
- Thevis, M.; Geyer, H.; Sigmund, G.; Schänzer, W. Sports drug testing: Analytical aspects of selected cases of suspected, purported, and proven urine manipulation. J. Pharm. Biomed. Anal. 2012, 57, 26–32. [Google Scholar] [CrossRef]
- Macdonald, S.; Hall, W.; Roman, P.; Stockwell, T.; Coghlan, M.; Nesvaag, S. Testing for cannabis in the work-place: A review of the evidence. Addiction 2010, 105, 408–416. [Google Scholar] [CrossRef] [PubMed]
- Thomas, K.V.; Bijlsma, L.; Castiglioni, S.; Covaci, A.; Emke, E.; Grabic, R.; Hernández, F.; Karolak, S.; Kasprzyk-Hordern, B.; Lindberg, R.H. Comparing illicit drug use in 19 European cities through sewage analysis. Sci. Total Environ. 2012, 432, 432–439. [Google Scholar] [CrossRef]
- Van Nuijs, A.L.; Mougel, J.-F.; Tarcomnicu, I.; Bervoets, L.; Blust, R.; Jorens, P.G.; Neels, H.; Covaci, A. Sewage epidemiology—a real-time approach to estimate the consumption of illicit drugs in Brussels, Belgium. Environ. Int. 2011, 37, 612–621. [Google Scholar] [CrossRef] [PubMed]
- Khan, U.; van Nuijs, A.L.N.; Li, J.; Maho, W.; Du, P.; Li, K.; Hou, L.; Zhang, J.; Meng, X.; Li, X.; et al. Application of a sewage-based approach to assess the use of ten illicit drugs in four Chinese megacities. Sci. Total Environ. 2014, 487, 710–721. [Google Scholar] [CrossRef]
- Daughton, C.G. Illicit Drugs: Contaminants in the Environment and Utility in Forensic Epidemiology. In Reviews of Environmental Contamination and Toxicology; Whitacre, D.M., Ed.; Springer: New York, NY, USA, 2011; Volume 210, pp. 59–110. [Google Scholar] [CrossRef]
- Irvine, R.J.; Kostakis, C.; Felgate, P.D.; Jaehne, E.J.; Chen, C.; White, J.M. Population drug use in Australia: A wastewater analysis. Forensic Sci. Int. 2011, 210, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Prichard, J.; Lai, F.Y.; Kirkbride, P.; Bruno, R.; Ort, C.; Carter, S.; Hall, W.; Gartner, C.; Thai, P.; Mueller, J. Measuring drug use patterns in Queensland through wastewater analysis. Trends Issues Crime Crim. Justice 2012, 442, 1–8. [Google Scholar]
- Zuccato, E.; Chiabrando, C.; Castiglioni, S.; Bagnati, R.; Fanelli, R. Estimating community drug abuse by wastewater analysis. Environ. Health Perspect. 2008, 116, 1027–1032. [Google Scholar] [CrossRef]
- Ort, C.; Van Nuijs, A.L.; Berset, J.D.; Bijlsma, L.; Castiglioni, S.; Covaci, A.; de Voogt, P.; Emke, E.; Fatta-Kassinos, D.; Griffiths, P. Spatial differences and temporal changes in illicit drug use in Europe quantified by wastewater analysis. Addiction 2014, 109, 1338–1352. [Google Scholar] [CrossRef]
- Brewer, A.J.; Banta-Green, C.J.; Ort, C.; Robel, A.E.; Field, J. Wastewater testing compared with random urinalyses for the surveillance of illicit drug use in prisons. Drug Alcohol Rev. 2016, 35, 133–137. [Google Scholar] [CrossRef]
- Foppe, K.S.; Hammond-Weinberger, D.R.; Subedi, B. Estimation of the consumption of illicit drugs during special events in two communities in Western Kentucky, USA using sewage epidemiology. Sci. Total Environ. 2018, 633, 249–256. [Google Scholar] [CrossRef]
- Benaglia, L.; Udrisard, R.; Bannwarth, A.; Gibson, A.; Béen, F.; Lai, F.Y.; Esseiva, P.; Delémont, O. Testing wastewater from a music festival in Switzerland to assess illicit drug use. Forensic Sci. Int. 2020, 309, 110148. [Google Scholar] [CrossRef]
- Lai, F.Y.; Thai, P.K.; O’Brien, J.; Gartner, C.; Bruno, R.; Kele, B.; Ort, C.; Prichard, J.; Kirkbride, P.; Hall, W.; et al. Using quantitative wastewater analysis to measure daily usage of conventional and emerging illicit drugs at an annual music festival. Drug Alcohol Rev. 2013, 32, 594–602. [Google Scholar] [CrossRef]
- Mackuľak, T.; Birošová, L.; Gál, M.; Bodík, I.; Grabic, R.; Ryba, J.; Škubák, J. Wastewater analysis: The mean of the monitoring of frequently prescribed pharmaceuticals in Slovakia. Environ. Monit. Assess. 2016, 188, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Q.-D.; Lin, J.-G.; Pei, W.; Guo, M.-X.; Wang, Z.; Wang, D.-G. Estimating nicotine consumption in eight cities using sewage epidemiology based on ammonia nitrogen equivalent population. Sci. Total Environ. 2017, 590–591, 226–232. [Google Scholar] [CrossRef]
- Banks, A.P.W.; Lai, F.Y.; Mueller, J.F.; Jiang, G.; Carter, S.; Thai, P.K. Potential impact of the sewer system on the applicability of alcohol and tobacco biomarkers in wastewater-based epidemiology. Drug Test. Anal. 2018, 10, 530–538. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, F.; Tscharke, B.; O’Brien, J.; Thompson, J.; Samanipour, S.; Choi, P.; Li, J.; Mueller, J.F.; Thomas, K. Wastewater-based estimation of the prevalence of gout in Australia. Sci. Total Environ. 2020, 715, 136925. [Google Scholar] [CrossRef]
- Moll, D.M.; Frick, E.A.; Henderson, A.K.; Furlong, E.T.; Meyer, M.T. Presence of pharmaceuticals in treated wastewater effluent and surface water supply systems, metropolitan Atlanta, Georgia, July–September 1999. In Proceedings of the 2nd International Conference on Pharmaceuticals and Endocrine Disrupting Chemicals in Water, Minneapolis, MN, USA, 9–11 October 2001. [Google Scholar]
- Singer, A.C.; Järhult, J.D.; Grabic, R.; Khan, G.A.; Lindberg, R.H.; Fedorova, G.; Fick, J.; Bowes, M.J.; Olsen, B.; Söderström, H. Intra-and inter-pandemic variations of antiviral, antibiotics and decongestants in wastewater treatment plants and receiving rivers. PLoS ONE 2014, 9, e108621. [Google Scholar] [CrossRef]
- Burgard, D.A.; Fuller, R.; Becker, B.; Ferrell, R.; Dinglasan-Panlilio, M. Potential trends in Attention Deficit Hyperactivity Disorder (ADHD) drug use on a college campus: Wastewater analysis of amphetamine and ritalinic acid. Sci. Total Environ. 2013, 450, 242–249. [Google Scholar] [CrossRef] [PubMed]
- Leknes, H.; Sturtzel, I.E.; Dye, C. Environmental release of oseltamivir from a Norwegian sewage treatment plant during the 2009 influenza A (H1N1) pandemic. Sci. Total Environ. 2012, 414, 632–638. [Google Scholar] [CrossRef] [PubMed]
- Shinohara, N.; Tanaka, H.; Saito, T.; Deguchi, J.; Soda, K.; Sonoda, S.; Sugiyama, T.; Ishimaru, Y. Surveillance for typhoid fever in Matsuyama city during 1974-1981 and detection of Salmonella typhi in sewage and river waters. Jpn. J. Med Sci. Biol. 1983, 36, 191–197. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yanagimoto, K.; Yamagami, T.; Uematsu, K.; Haramoto, E. Characterization of Salmonella isolates from wastewater treatment plant influents to estimate unreported cases and infection sources of salmonellosis. Pathogens 2020, 9, 52. [Google Scholar] [CrossRef]
- Cai, L.; Zhang, T. Detecting human bacterial pathogens in wastewater treatment plants by a high-throughput shotgun sequencing technique. Environ. Sci. Technol. 2013, 47, 5433–5441. [Google Scholar] [CrossRef]
- Huijbers, P.M.; Flach, C.-F.; Larsson, D.J. A conceptual framework for the environmental surveillance of antibiotics and antibiotic resistance. Environ. Int. 2019, 130, 104880. [Google Scholar] [CrossRef]
- Nakamura, T.; Hamasaki, M.; Yoshitomi, H.; Ishibashi, T.; Yoshiyama, C.; Maeda, E.; Sera, N.; Yoshida, H. Environmental surveillance of poliovirus in sewage water around the introduction period for inactivated polio vaccine in Japan. Appl. Environ. Microbiol. 2015, 81, 1859–1864. [Google Scholar] [CrossRef]
- Smith, D.B.; Paddy, J.O.; Simmonds, P. The use of human sewage screening for community surveillance of hepatitis E virus in the UK. J. Med. Virol. 2016, 88, 915–918. [Google Scholar] [CrossRef]
- Filipidou, A.; Parasidis, T.; Alexandropoulou, I.; Stavrou, E.; Karlou, K.; Vantarakis, A. Detection of Infectious Pathogenic Viruses in Untreated and Treated Wastewater Samples from An Urbanised Area. Int. J. Infect. Dis. 2008, 12, e104. [Google Scholar] [CrossRef][Green Version]
- Heijnen, L.; Medema, G. Surveillance of influenza A and the pandemic influenza A (H1N1) 2009 in sewage and surface water in the Netherlands. J. Water Health 2011, 9, 434–442. [Google Scholar] [CrossRef] [PubMed]
- Amdiouni, H.; Faouzi, A.; Fariat, N.; Hassar, M.; Soukri, A.; Nourlil, J. Detection and molecular identification of human adenoviruses and enteroviruses in wastewater from Morocco. Lett. Appl. Microbiol. 2012, 54, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Battistone, A.; Buttinelli, G.; Bonomo, P.; Fiore, S.; Amato, C.; Mercurio, P.; Cicala, A.; Simeoni, J.; Foppa, A.; Triassi, M. Detection of enteroviruses in influent and effluent flow samples from wastewater treatment plants in Italy. Food Environ. Virol. 2014, 6, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Pennino, F.; Nardone, A.; Montuori, P.; Aurino, S.; Torre, I.; Battistone, A.; Delogu, R.; Buttinelli, G.; Fiore, S.; Amato, C. Large-scale survey of human enteroviruses in wastewater treatment plants of a metropolitan area of southern Italy. Food Environ. Virol. 2018, 10, 187–192. [Google Scholar] [CrossRef]
- McCall, C.; Wu, H.; Miyani, B.; Xagoraraki, I. Identification of multiple potential viral diseases in a large urban center using wastewater surveillance. Water Res. 2020, 184, 116160. [Google Scholar] [CrossRef]
- Santiso-Bellón, C.; Randazzo, W.; Pérez-Cataluña, A.; Vila-Vicent, S.; Gozalbo-Rovira, R.; Muñoz, C.; Buesa, J.; Sanchez, G.; Rodríguez Díaz, J. Epidemiological surveillance of norovirus and rotavirus in sewage (2016–2017) in Valencia (Spain). Microorganisms 2020, 8, 458. [Google Scholar] [CrossRef]
- Gourinat, A.-C.; O’Connor, O.; Calvez, E.; Goarant, C.; Dupont-Rouzeyrol, M. Detection of Zika virus in urine. Emerg. Infect. Dis. 2015, 21, 84. [Google Scholar] [CrossRef]
- Muirhead, A.; Zhu, K.; Brown, J.; Basu, M.; Brinton, M.A.; Costa, F.; Hayat, M.J.; Stauber, C.E. Zika Virus RNA persistence in sewage. Environ. Sci. Technol. Lett. 2020, 7, 659–664. [Google Scholar] [CrossRef]
- Chandra, F.; Lee, W.L.; Armas, F.; Leifels, M.; Gu, X.; Chen, H.; Wuertz, S.; Alm, E.J.; Thompson, J. Persistence of Dengue (Serotypes 2 and 3), Zika, Yellow Fever, and Murine Hepatitis Virus RNA in Untreated Wastewater. Environ. Sci. Technol. Lett. 2021, 8, 785–791. [Google Scholar] [CrossRef]
- Barzon, L.; Pacenti, M.; Franchin, E.; Pagni, S.; Martello, T.; Cattai, M.; Cusinato, R.; Palù, G. Excretion of West Nile virus in urine during acute infection. J. Infect. Dis. 2013, 208, 1086–1092. [Google Scholar] [CrossRef]
- Hirayama, T.; Mizuno, Y.; Takeshita, N.; Kotaki, A.; Tajima, S.; Omatsu, T.; Sano, K.; Kurane, I.; Takasaki, T. Detection of dengue virus genome in urine by real-time reverse transcriptase PCR: A laboratory diagnostic method useful after disappearance of the genome in serum. J. Clin. Microbiol. 2012, 50, 2047–2052. [Google Scholar] [CrossRef]
- Mizuno, Y.; Kotaki, A.; Harada, F.; Tajima, S.; Kurane, I.; Takasaki, T. Confirmation of dengue virus infection by detection of dengue virus type 1 genome in urine and saliva but not in plasma. Trans. R. Soc. Trop. Med. Hyg. 2007, 101, 738–739. [Google Scholar] [CrossRef] [PubMed]
- Thai, P.K.; O’Brien, J.; Jiang, G.; Gernjak, W.; Yuan, Z.; Eaglesham, G.; Mueller, J.F. Degradability of creatinine under sewer conditions affects its potential to be used as biomarker in sewage epidemiology. Water Res. 2014, 55, 272–279. [Google Scholar] [CrossRef]
- Li, J.; Gao, J.; Thai, P.K.; Mueller, J.F.; Yuan, Z.; Jiang, G. Transformation of Illicit Drugs and Pharmaceuticals in Sewer Sediments. Environ. Sci. Technol. 2020, 54, 13056–13065. [Google Scholar] [CrossRef] [PubMed]
- Gundy, P.M.; Gerba, C.P.; Pepper, I.L. Survival of coronaviruses in water and wastewater. Food Environ. Virol. 2009, 1, 10–14. [Google Scholar] [CrossRef]
- Wang, X.-W.; Li, J.-S.; Guo, T.-K.; Zhen, B.; Kong, Q.-X.; Yi, B.; Li, Z.; Song, N.; Jin, M.; Xiao, W.-J. Concentration and detection of SARS coronavirus in sewage from Xiao Tang Shan Hospital and the 309th Hospital. J. Virol. Methods 2005, 128, 156–161. [Google Scholar] [CrossRef] [PubMed]
- Izquierdo-Lara, R.; Elsinga, G.; Heijnen, L.; Munnink, B.B.O.; Schapendonk, C.M.; Nieuwenhuijse, D.; Kon, M.; Lu, L.; Aarestrup, F.M.; Lycett, S. Monitoring SARS-CoV-2 circulation and diversity through community wastewater sequencing, the netherlands and belgium. Emerg. Infect. Dis. 2021, 27, 1405. [Google Scholar] [CrossRef] [PubMed]
- Wilton, T.; Bujaki, E.; Klapsa, D.; Majumdar, M.; Zambon, M.; Fritzsche, M.; Mate, R.; Martin, J. Rapid increase of SARS-CoV-2 variant B. 1.1. 7 detected in sewage samples from England between October 2020 and January 2021. Msystems 2021, 6, e00353-21. [Google Scholar] [CrossRef] [PubMed]
- Lesté-Lasserre, C. Coronavirus found in Paris sewage points to early warning system. Science 2020. [Google Scholar] [CrossRef]
- Westhaus, S.; Weber, F.-A.; Schiwy, S.; Linnemann, V.; Brinkmann, M.; Widera, M.; Greve, C.; Janke, A.; Hollert, H.; Wintgens, T. Detection of SARS-CoV-2 in raw and treated wastewater in Germany–suitability for COVID-19 surveillance and potential transmission risks. Sci. Total Environ. 2021, 751, 141750. [Google Scholar] [CrossRef] [PubMed]
- Róka, E.; Khayer, B.; Kis, Z.; Kovács, L.B.; Schuler, E.; Magyar, N.; Málnási, T.; Oravecz, O.; Pályi, B.; Pándics, T. Ahead of the second wave: Early warning for COVID-19 by wastewater surveillance in Hungary. Sci. Total Environ. 2021, 786, 147398. [Google Scholar] [CrossRef] [PubMed]
- La Rosa, G.; Iaconelli, M.; Mancini, P.; Ferraro, G.B.; Veneri, C.; Bonadonna, L.; Lucentini, L.; Suffredini, E. First detection of SARS-CoV-2 in untreated wastewaters in Italy. Sci. Total Environ. 2020, 736, 139652. [Google Scholar] [CrossRef] [PubMed]
- Medema, G.; Heijnen, L.; Elsinga, G.; Italiaander, R.; Brouwer, A. Presence of SARS-Coronavirus-2 RNA in sewage and correlation with reported COVID-19 prevalence in the early stage of the epidemic in the Netherlands. Environ. Sci. Technol. Lett. 2020, 7, 511–516. [Google Scholar] [CrossRef]
- Kuryntseva, P.; Karamova, K.; Fomin, V.; Selivanovskaya, S.; Galitskaya, P. A simplified approach to monitoring the COVID-19 epidemiologic situation using waste water analysis and its application in Russia. medRxiv 2020. [Google Scholar] [CrossRef]
- Chavarria-Miró, G.; Anfruns-Estrada, E.; Martínez-Velázquez, A.; Vázquez-Portero, M.; Guix, S.; Paraira, M.; Galofré, B.; Sánchez, G.; Pintó, R.M.; Bosch, A. Time evolution of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in wastewater during the first pandemic wave of COVID-19 in the metropolitan area of Barcelona, Spain. Appl. Environ. Microbiol. 2021, 87, e02750-20. [Google Scholar] [CrossRef]
- Randazzo, W.; Cuevas-Ferrando, E.; Sanjuán, R.; Domingo-Calap, P.; Sánchez, G. Metropolitan wastewater analysis for COVID-19 epidemiological surveillance. Int. J. Hyg. Environ. Health 2020, 230, 113621. [Google Scholar] [CrossRef]
- Jahn, K.; Dreifuss, D.; Topolsky, I.; Kull, A.; Ganesanandamoorthy, P.; Fernandez-Cassi, X.; Bänziger, C.; Stachler, E.; Fuhrmann, L.; Jablonski, K.P. Detection of SARS-CoV-2 variants in Switzerland by genomic analysis of wastewater samples. medRxiv 2021. [Google Scholar] [CrossRef]
- Kocamemi, B.A.; Kurt, H.; Hacioglu, S.; Yarali, C.; Saatci, A.M.; Pakdemirli, B. First data-set on SARS-CoV-2 detection for Istanbul wastewaters in Turkey. medRxiv 2020. [Google Scholar] [CrossRef]
- Kocamemi, B.A.; Kurt, H.; Sait, A.; Kadi, H.; Sarac, F.; Aydin, I.; Saatci, A.M.; Pakdemirli, B. Nationwide SARS-CoV-2 Surveillance Study for Sewage and Sludges of Wastewater Treatment Plants in Turkey. medRxiv 2020. [Google Scholar] [CrossRef]
- Xu, X.; Zheng, X.; Li, S.; Lam, N.S.; Wang, Y.; Chu, D.K.; Poon, L.L.; Tun, H.M.; Peiris, M.; Deng, Y. The first case study of wastewater-based epidemiology of COVID-19 in Hong Kong. Sci. Total Environ. 2021, 790, 148000. [Google Scholar] [CrossRef]
- Or, I.B.; Yaniv, K.; Shagan, M.; Ozer, E.; Erster, O.; Mendelson, E.; Mannasse, B.; Shirazi, R.; Kramarsky-Winter, E.; Nir, O. Regressing SARS-CoV-2 sewage measurements onto COVID-19 burden in the population: A proof-of-concept for quantitative environmental surveillance. medRxiv 2020. [Google Scholar] [CrossRef]
- Yaniv, K.; Shagan, M.; Lewis, Y.E.; Kramarsky-Winter, E.; Weil, M.; Indenbaum, V.; Elul, M.; Erster, O.; Brown, A.S.; Mendelson, E. City-level SARS-CoV-2 sewage surveillance. Chemosphere 2021, 283, 131194. [Google Scholar] [CrossRef]
- Kumar, M.; Patel, A.K.; Shah, A.V.; Raval, J.; Rajpara, N.; Joshi, M.; Joshi, C.G. First proof of the capability of wastewater surveillance for COVID-19 in India through detection of genetic material of SARS-CoV-2. Sci. Total Environ. 2020, 746, 141326. [Google Scholar] [CrossRef]
- Yaqub, T.; Nawaz, M.; Shabbir, M.Z.; Ali, M.A.; Altaf, I.; Raza, S.; Shabbir, M.A.B.; Ashraf, M.A.; Aziz, S.Z.; Cheema, S.Q. A longitudinal survey for genome-based identification of SARS-CoV-2 in sewage water in selected lockdown areas of Lahore city, Pakistan; a potential approach for future smart lockdown strategy. medRxiv 2020. [Google Scholar] [CrossRef]
- Alahdal, H.M.; Ameen, F.; AlYahya, S.; Sonbol, H.; Khan, A.; Alsofayan, Y.; Alahmari, A. Municipal wastewater viral pollution in Saudi Arabia: Effect of hot climate on COVID-19 disease spreading. Environ. Sci. Pollut. Res. 2021, 1–8. [Google Scholar] [CrossRef]
- Mota, C.R.; Bressani-Ribeiro, T.; Araújo, J.C.; Leal, C.D.; Leroy-Freitas, D.; Machado, E.C.; Espinosa, M.F.; Fernandes, L.; Leão, T.L.; Chamhum-Silva, L. Assessing spatial distribution of COVID-19 prevalence in Brazil using decentralised sewage monitoring. Water Res. 2021, 202, 117388. [Google Scholar] [CrossRef] [PubMed]
- Fongaro, G.; Stoco, P.H.; Souza, D.S.M.; Grisard, E.C.; Magri, M.E.; Rogovski, P.; Schörner, M.A.; Barazzetti, F.H.; Christoff, A.P.; de Oliveira, L.F.V. The presence of SARS-CoV-2 RNA in human sewage in Santa Catarina, Brazil, November 2019. Sci. Total Environ. 2021, 778, 146198. [Google Scholar] [CrossRef] [PubMed]
- Ampuero, M.; Valenzuela, S.; Valiente-Echeverría, F.; Soto-Rifo, R.; Barriga, G.P.; Chnaiderman, J.; Rojas, C.; Guajardo-Leiva, S.; Díez, B.; Gaggero, A. SARS-CoV-2 Detection in Sewage in Santiago, Chile—Preliminary results. medRxiv 2020. [Google Scholar] [CrossRef]
- Carrillo-Reyes, J.; Barragán-Trinidad, M.; Buitrón, G. Surveillance of SARS-CoV-2 in sewage and wastewater treatment plants in Mexico. J. Water Process Eng. 2021, 40, 101815. [Google Scholar] [CrossRef]
- Peccia, J.; Zulli, A.; Brackney, D.E.; Grubaugh, N.D.; Kaplan, E.H.; Casanovas-Massana, A.; Ko, A.I.; Malik, A.A.; Wang, D.; Wang, M. SARS-CoV-2 RNA concentrations in primary municipal sewage sludge as a leading indicator of COVID-19 outbreak dynamics. medRxiv 2020. [Google Scholar] [CrossRef]
- Larsen, D.A.; Wigginton, K.R. Tracking COVID-19 with wastewater. Nat. Biotechnol. 2020, 38, 1151–1153. [Google Scholar] [CrossRef]
- Ahmed, W.; Angel, N.; Edson, J.; Bibby, K.; Bivins, A.; O’Brien, J.W.; Choi, P.M.; Kitajima, M.; Simpson, S.L.; Li, J. First confirmed detection of SARS-CoV-2 in untreated wastewater in Australia: A proof of concept for the wastewater surveillance of COVID-19 in the community. Sci. Total Environ. 2020, 728, 138764. [Google Scholar] [CrossRef]
- Black, J.; Aung, P.; Nolan, M.; Roney, E.; Poon, R.; Hennessy, D.; Crosbie, N.D.; Deere, D.; Jex, A.R.; John, N. Epidemiological evaluation of sewage surveillance as a tool to detect the presence of COVID-19 cases in a low case load setting. Sci. Total Environ. 2021, 786, 147469. [Google Scholar] [CrossRef]
- Aslan, A.; Shah, G.; Sittaramane, V.; Shankar, P. Sewage Monitoring in Rural Communities: A Powerful Strategy for COVID-19 Surveillance. J. Environ. Health 2020, 83, 8–10. [Google Scholar]
- Calabria de Araujo, J.; Gavazza, S.; Leao, T.L.; Florencio, L.; da Silva, H.P.; Albuquerque, J.d.O.; de Lira Borges, M.A.; de Oliveira Alves, R.B.; Rodrigues, R.H.A.; dos Santos, E.B. SARS-CoV-2 sewage surveillance in low-income countries: Potential and challenges. J. Water Health 2021, 19, 1–19. [Google Scholar] [CrossRef]
- Panchal, D.; Prakash, O.; Bobde, P.; Pal, S. SARS-CoV-2: Sewage surveillance as an early warning system and challenges in developing countries. Environ. Sci. Pollut. Res. 2021, 28, 22221–22240. [Google Scholar] [CrossRef] [PubMed]
- Michael-Kordatou, I.; Karaolia, P.; Fatta-Kassinos, D. Sewage analysis as a tool for the COVID-19 pandemic response and management: The urgent need for optimised protocols for SARS-CoV-2 detection and quantification. J. Environ. Chem. Eng. 2020, 8, 104306. [Google Scholar] [CrossRef] [PubMed]
- Sharma, D.K.; Nalavade, U.P.; Kalgutkar, K.; Gupta, N.; Deshpande, J.M. SARS-CoV-2 detection in sewage samples: Standardization of method & preliminary observations. Indian J. Med. Res. 2021, 153, 159. [Google Scholar] [PubMed]
- De Feo, G.; Antoniou, G.; Fardin, H.F.; El-Gohary, F.; Zheng, X.Y.; Reklaityte, I.; Butler, D.; Yannopoulos, S.; Angelakis, A.N. The historical development of sewers worldwide. Sustainability 2014, 6, 3936–3974. [Google Scholar] [CrossRef]
- Sunderland, D. ‘A monument to defective administration’? The London Commissions of Sewers in the early nineteenth century. Urban Hist. 1999, 26, 349–372. [Google Scholar] [CrossRef]
- Cook, G.C. Construction of London’s Victorian sewers: The vital role of Joseph Bazalgette. Postgrad. Med. J. 2001, 77, 802. [Google Scholar] [CrossRef]
- Goldman, J.A. Building New York’s Sewers: Developing Mechanisms of Urban Management; Purdue University Press: West Lafayette, IN, USA, 1997. [Google Scholar]
- Eggimann, S.; Truffer, B.; Maurer, M. To connect or not to connect? Modelling the optimal degree of centralisation for wastewater infrastructures. Water Res. 2015, 84, 218–231. [Google Scholar] [CrossRef]
- Spennemann, D.H.R.; Parker, M. Hitting the ‘Pause’ Button: What does COVID tell us about the future of heritage sounds? Noise Mapp. 2020, 7, 265–275. [Google Scholar] [CrossRef]
- Storen, R.; Corrigan, N. COVID-19: A chronology of state and territory government announcements (up until 30 June 2020). In Parliamentary Library Research Paper Series 2020–21; Parliamentary Library, Commonwealth of Australia: Canberra, Australia, 2020. [Google Scholar]
- Siddik, M.N.A. Economic stimulus for COVID-19 pandemic and its determinants: Evidence from cross-country analysis. Heliyon 2020, 6, e05634. [Google Scholar] [CrossRef]
- Elgin, C.; Basbug, G.; Yalaman, A. Economic policy responses to a pandemic: Developing the COVID-19 economic stimulus index. Covid Econ. 2020, 1, 40–53. [Google Scholar]
- Wong, J.; Wong, N. The economics and accounting for COVID-19 wage subsidy and other government grants. Pac. Account. Rev. 2021, 33, 199–211. [Google Scholar] [CrossRef]
- Risch, E.; Gutierrez, O.; Roux, P.; Boutin, C.; Corominas, L. Life cycle assessment of urban wastewater systems: Quantifying the relative contribution of sewer systems. Water Res. 2015, 77, 35–48. [Google Scholar] [CrossRef]
- Monteiro, H.; Fernandez, J.E.; Freire, F. Comparative life-cycle energy analysis of a new and an existing house: The significance of occupant’s habits, building systems and embodied energy. Sustain. Cities Soc. 2016, 26, 507–518. [Google Scholar] [CrossRef]
- Wuyts, W.; Miatto, A.; Sedlitzky, R.; Tanikawa, H. Extending or ending the life of residential buildings in Japan: A social circular economy approach to the problem of short-lived constructions. J. Clean. Prod. 2019, 231, 660–670. [Google Scholar] [CrossRef]
- Öberg, G.; Merlinsky, M.G.; LaValle, A.; Morales, M.; Tobias, M.M. The notion of sewage as waste: A study of infrastructure change and institutional inertia in Buenos Aires, Argentina and Vancouver, Canada. Ecol. Soc. 2014, 19. [Google Scholar] [CrossRef]
- Otterpohl, R.; Braun, U.; Oldenburg, M. Innovative technologies for decentralised wastewater management in urban and peri-urban areas. Ber. -Wassergute Und Abfallwirtsch. Tech. Univ. Munch. Berichtsh. 2002, 173, 109–126. [Google Scholar] [CrossRef]
- Kjerstadius, H.; Haghighatafshar, S.; Davidsson, Å. Potential for nutrient recovery and biogas production from blackwater, food waste and greywater in urban source control systems. Environ. Technol. 2015, 36, 1707–1720. [Google Scholar] [CrossRef] [PubMed]
- Kujawa-Roeleveld, K.; Zeeman, G. Anaerobic Treatment in Decentralised and Source-Separation-Based Sanitation Concepts. Rev. Environ. Sci. Bio/Technol. 2006, 5, 115–139. [Google Scholar] [CrossRef]
- Skambraks, A.-K.; Kjerstadius, H.; Meier, M.; Davidsson, Å.; Wuttke, M.; Giese, T. Source separation sewage systems as a trend in urban wastewater management: Drivers for the implementation of pilot areas in Northern Europe. Sustain. Cities Soc. 2017, 28, 287–296. [Google Scholar] [CrossRef]
- Water Services Association of Australia. Sewerage Code of Australia; WSA 02-2002; Water Services Association of Australia: Melbourne, Australia; Sydney, Australia, 2002. [Google Scholar]
- Water Services Association of Australia. Gravity Sewerage Code of Australia; WSA 02-2014; Water Services Association of Australia: Melbourne, Australia; Sydney, Australia, 2014. [Google Scholar]
- Water Services Association of Australia. Pressure Sewerage Code of Australia; WSA 07-2007; Water Services Association of Australia: Melbourne, Australia; Sydney, Australia, 2007. [Google Scholar]
- Mandel, A.; Veetil, V. The economic cost of COVID lockdowns: An out-of-equilibrium analysis. Econ. Disasters Clim. Chang. 2020, 4, 431–451. [Google Scholar] [CrossRef] [PubMed]
- Magli, A.C.; d’Onofrio, A.; Manfredi, P. Deteriorated Covid19 control due to delayed lockdown resulting from strategic interactions between Governments and oppositions. medRxiv 2020. [Google Scholar] [CrossRef]
- Lasaulce, S.; Varma, V.S.; Morarescu, C.; Siying, L. How efficient are the lockdown measures taken for mitigating the Covid-19 epidemic? medRxiv 2020. [Google Scholar] [CrossRef]
- López-Valcárcel, B.G.; Vallejo-Torres, L. The costs of COVID-19 and the cost-effectiveness of testing. Appl. Econ. Anal. 2021, 29. [Google Scholar] [CrossRef]
- Balmford, B.; Annan, J.D.; Hargreaves, J.C.; Altoè, M.; Bateman, I.J. Cross-country comparisons of COVID-19: Policy, politics and the price of life. Environ. Resour. Econ. 2020, 76, 525–551. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Spennemann, D.H.R. Preparing for COVID-2x: Urban Planning Needs to Regard Urological Wastewater as an Invaluable Communal Public Health Asset and Not as a Burden. Urban Sci. 2021, 5, 75. https://doi.org/10.3390/urbansci5040075
Spennemann DHR. Preparing for COVID-2x: Urban Planning Needs to Regard Urological Wastewater as an Invaluable Communal Public Health Asset and Not as a Burden. Urban Science. 2021; 5(4):75. https://doi.org/10.3390/urbansci5040075
Chicago/Turabian StyleSpennemann, Dirk H. R. 2021. "Preparing for COVID-2x: Urban Planning Needs to Regard Urological Wastewater as an Invaluable Communal Public Health Asset and Not as a Burden" Urban Science 5, no. 4: 75. https://doi.org/10.3390/urbansci5040075
APA StyleSpennemann, D. H. R. (2021). Preparing for COVID-2x: Urban Planning Needs to Regard Urological Wastewater as an Invaluable Communal Public Health Asset and Not as a Burden. Urban Science, 5(4), 75. https://doi.org/10.3390/urbansci5040075





