Superconductivity in La2O2M4S6 -Type Bi-based Compounds: A Review on Element Substitution Effects
Abstract
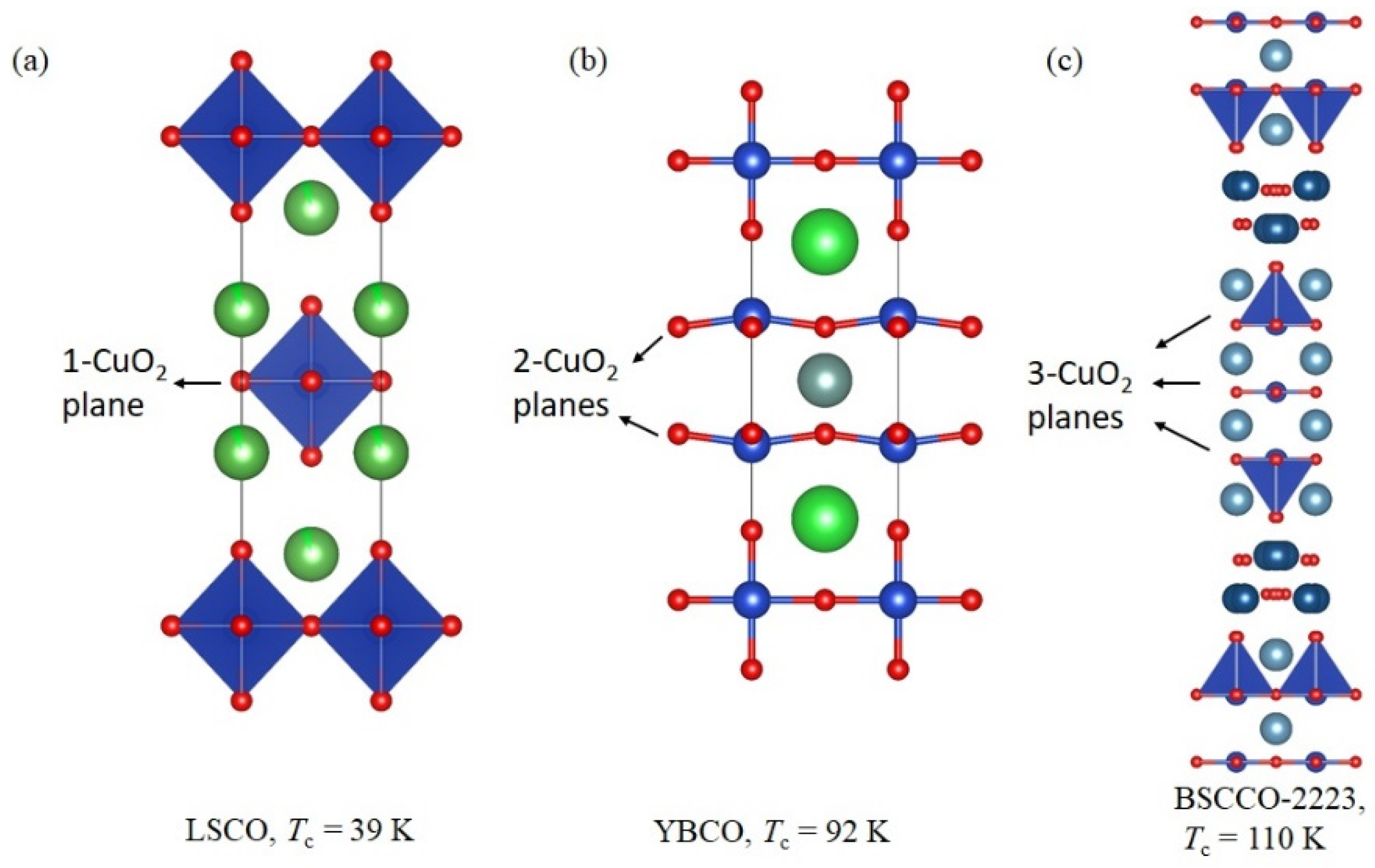
1. Introduction
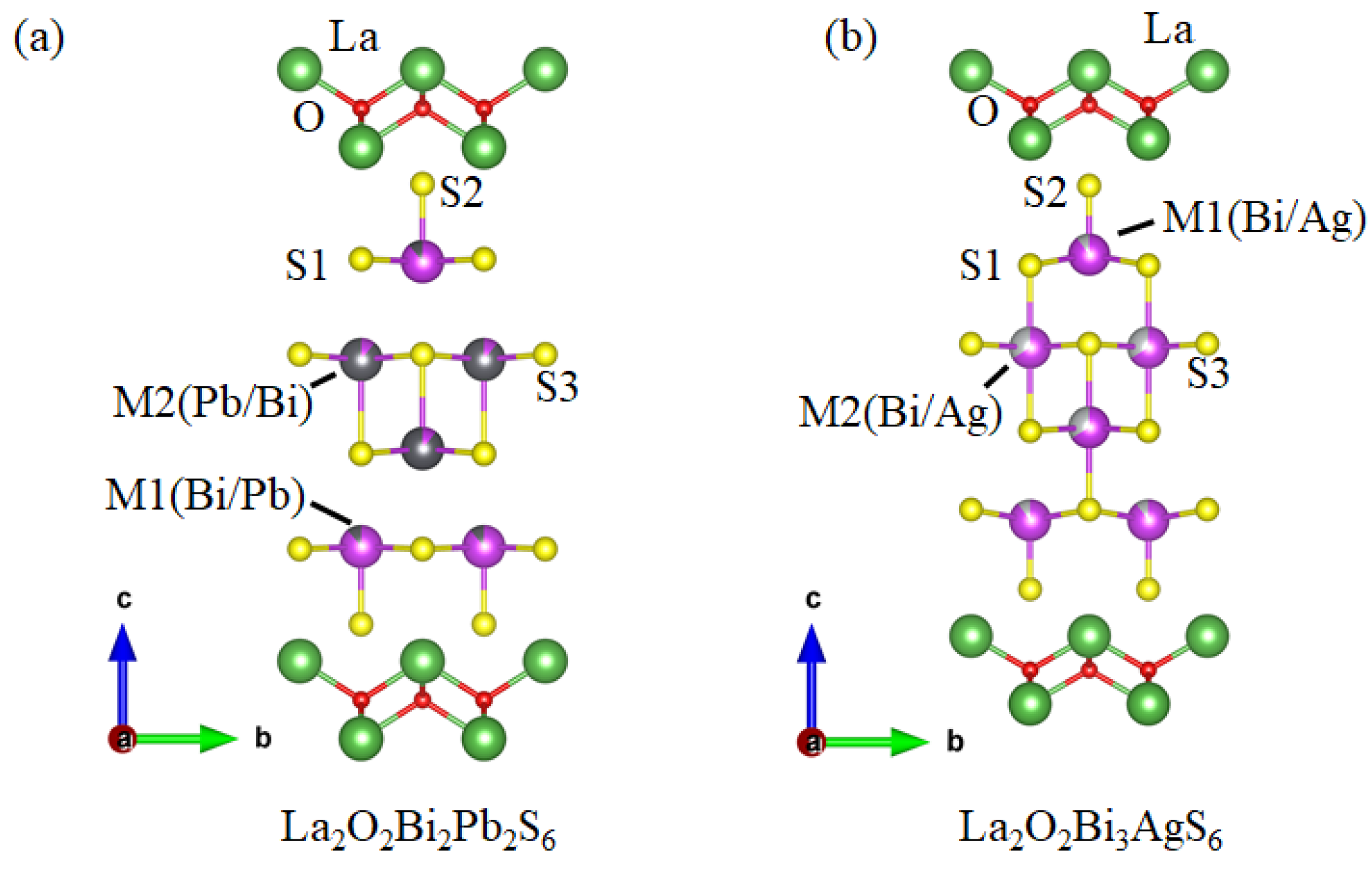
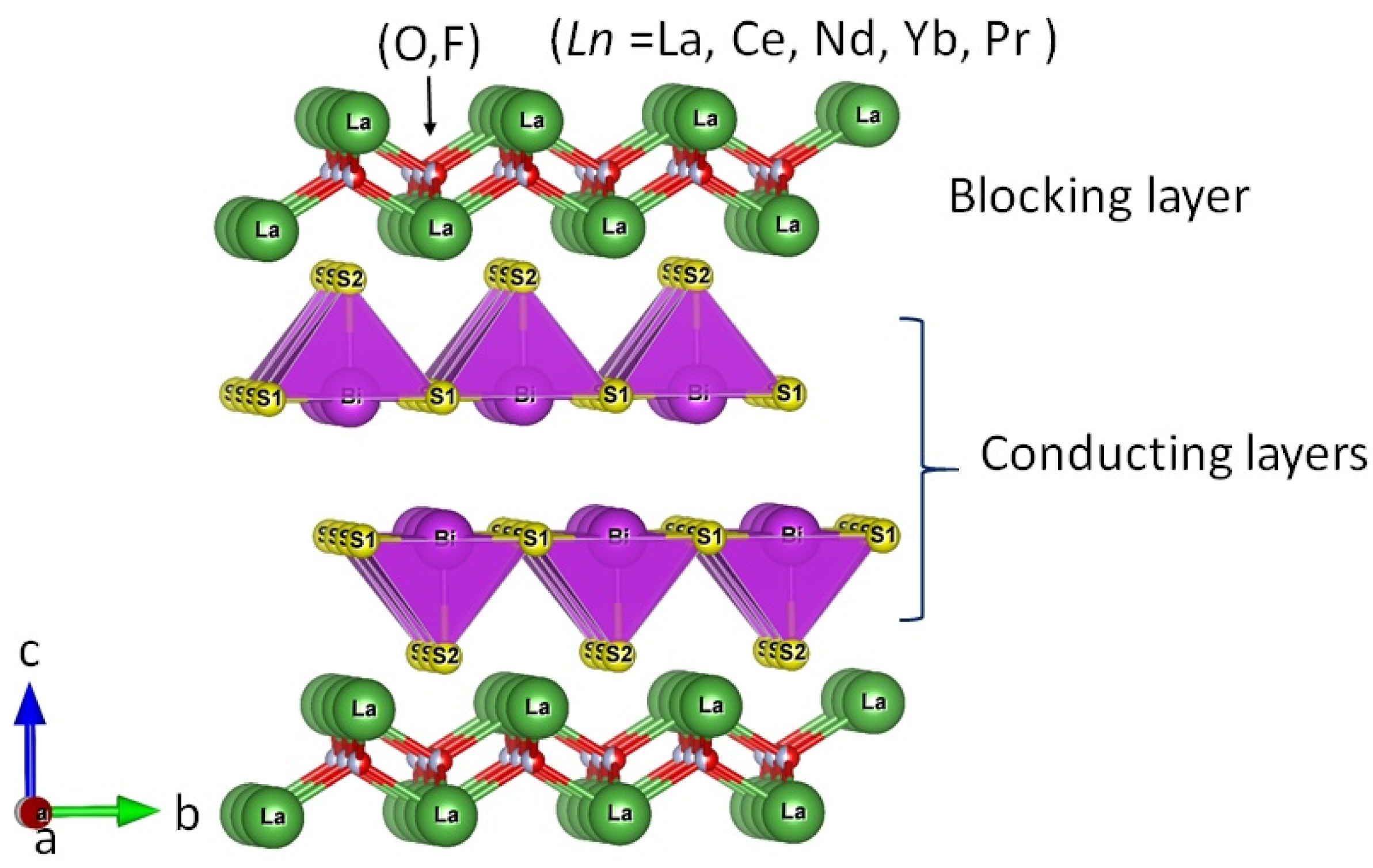
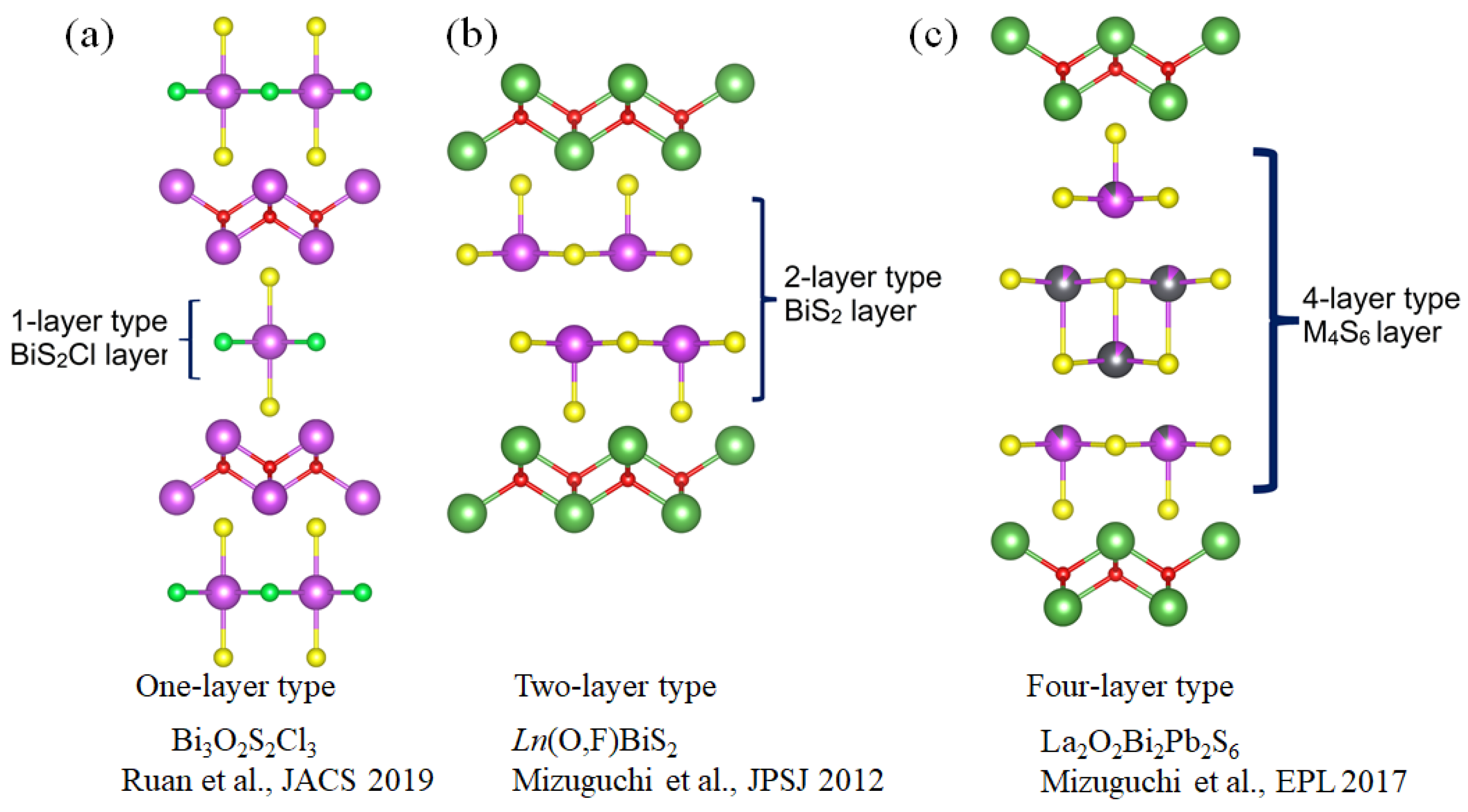
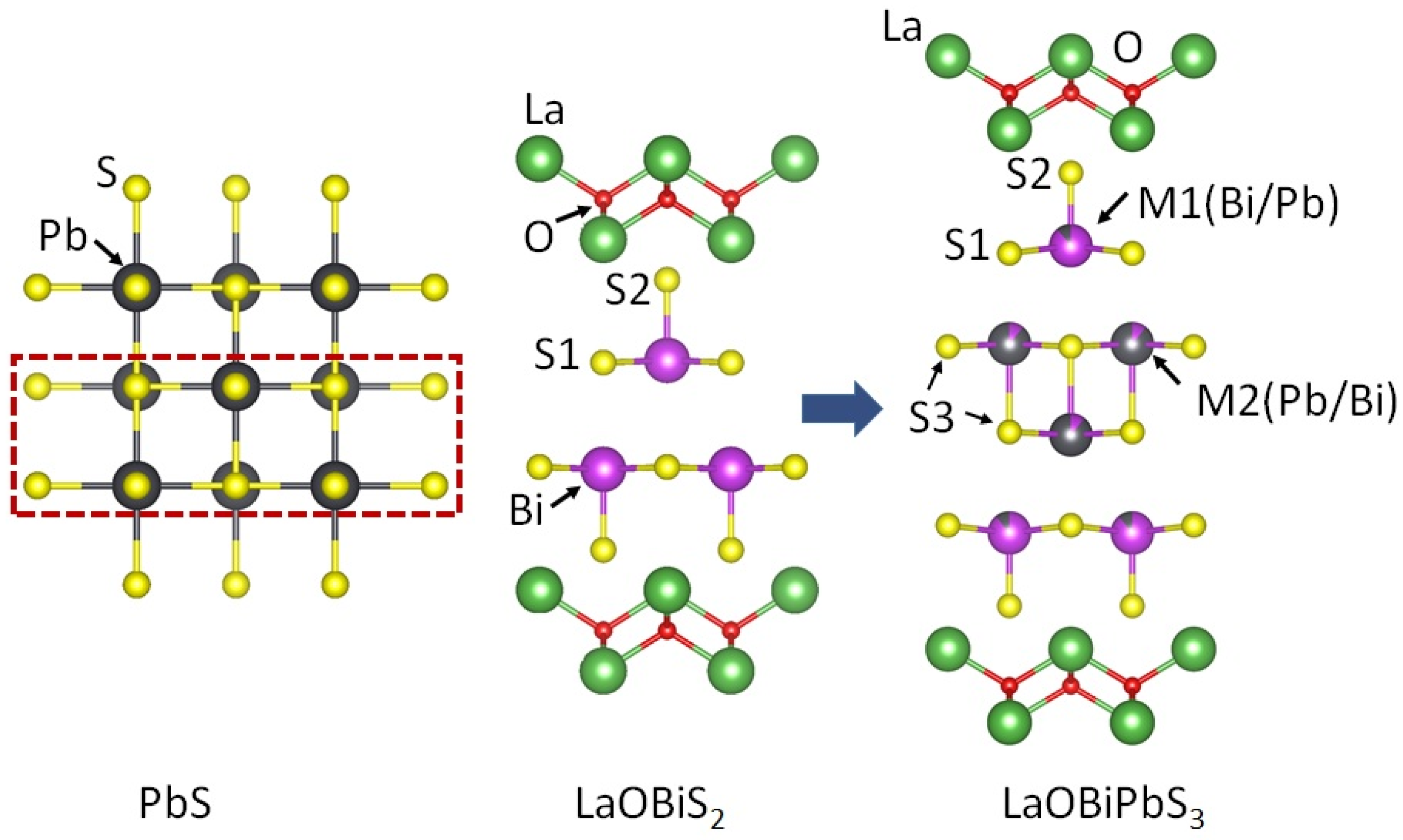
2. The Crystal Structure of La2O2M4S6 Type Four-Layer Compounds
2.1. La2O2Bi2Pb2S6
2.2. La2O2Bi3AgS6
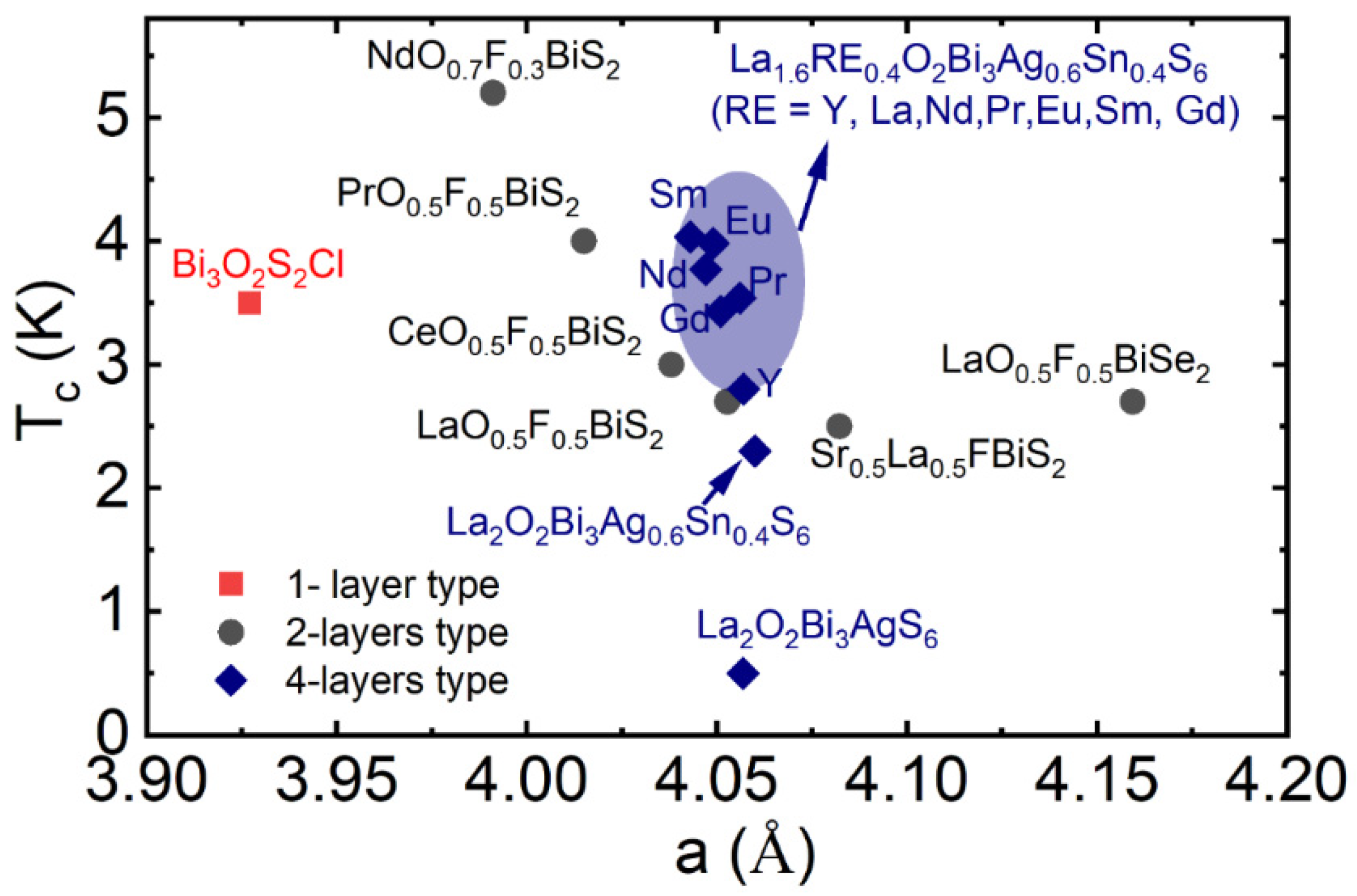
2.3. Available Elements for RE, M, and S Sites of La2O2M4S6
3. Physical Properties of La2O2M4S6 (M = Pb, Ag)
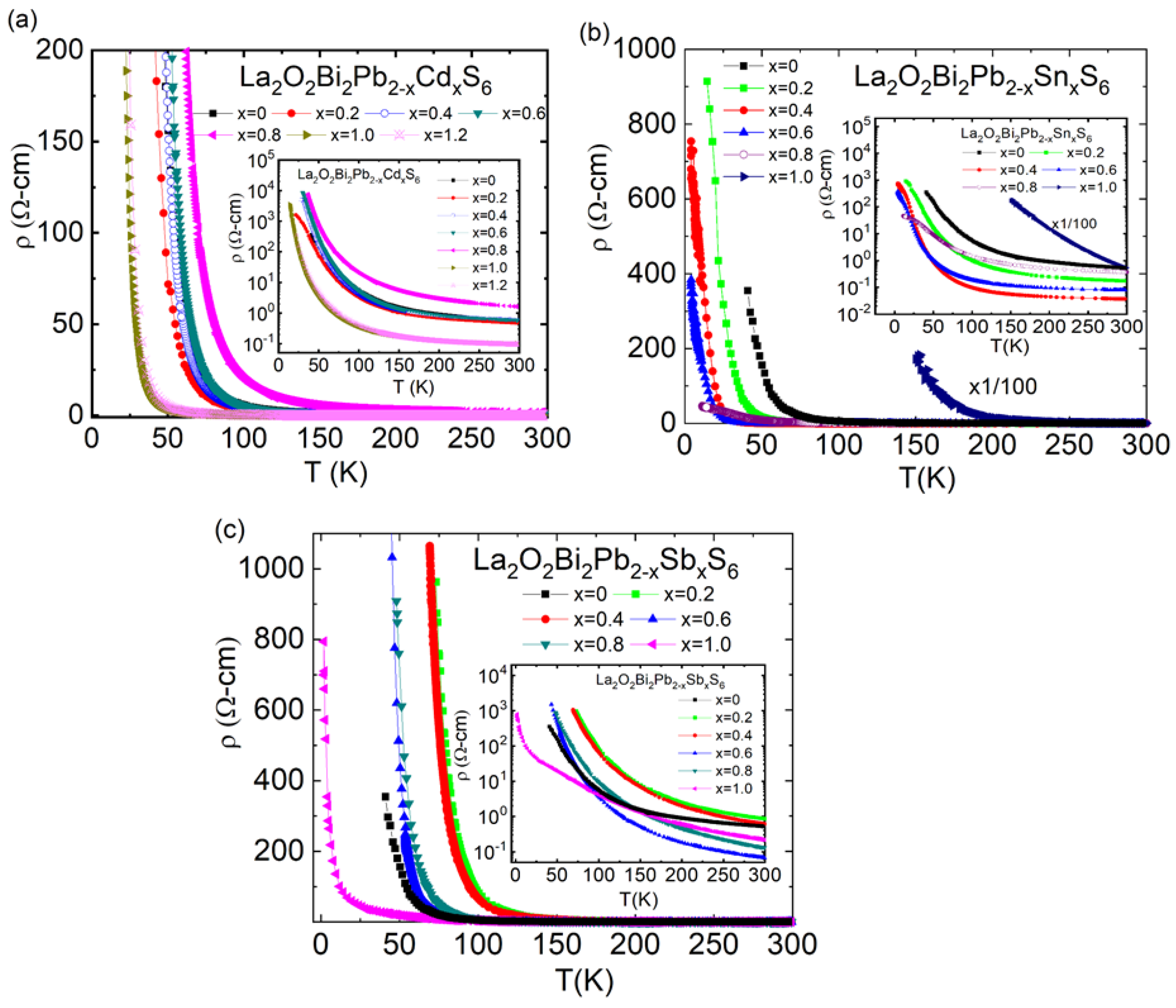
3.1. La2O2Bi2Pb2S6; Doping Effect of Cd, Sn, and Sb at Pb Site
3.2. Superconductivity in Se-Doped La2O2Bi2Pb2S6-xSex
3.3. Superconductivity in La2O2Bi3AgS6
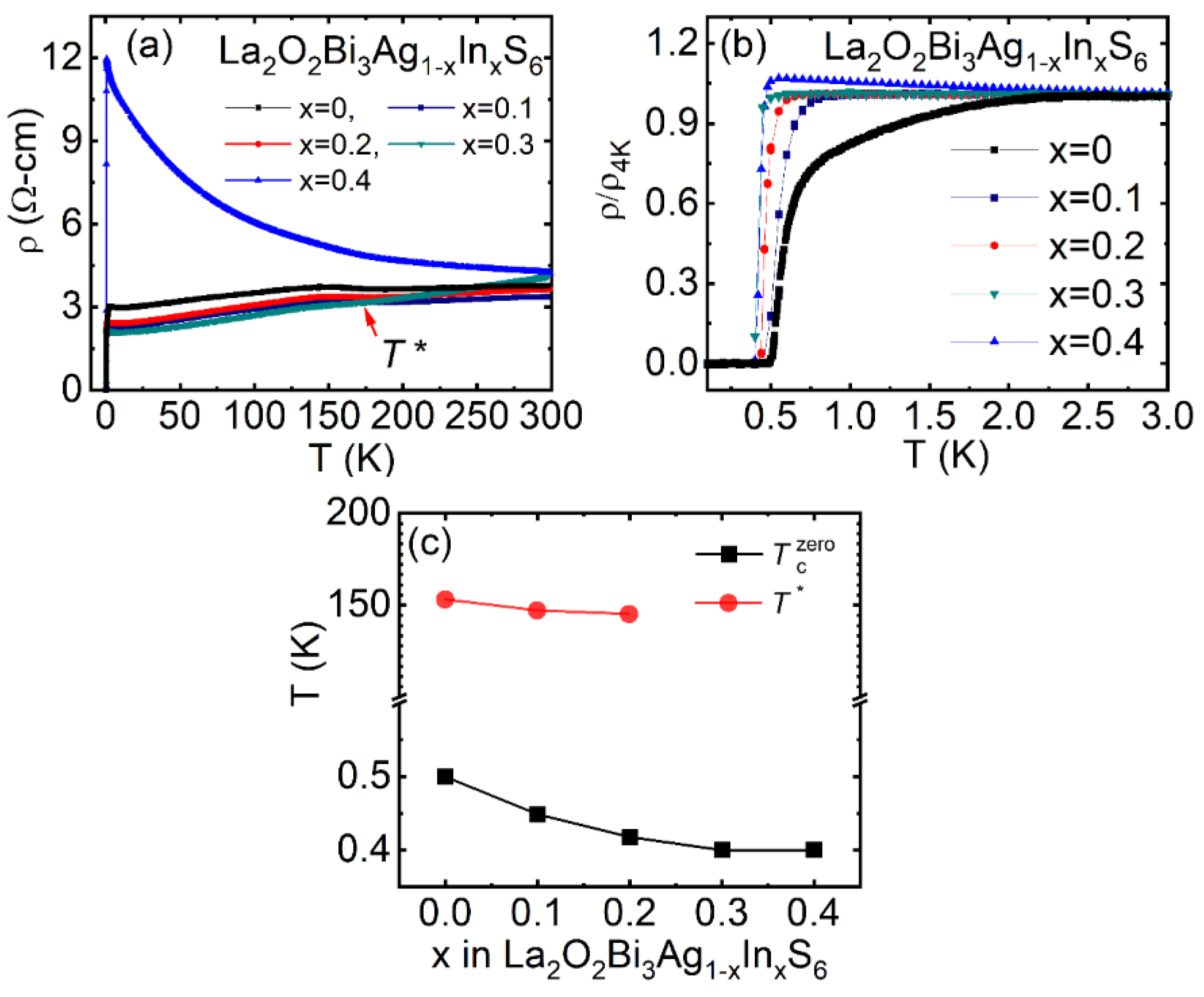
3.3.1. Effect of In Doping in La2O2Bi3Ag1-xInxS6
3.3.2. Sn-Substitution Effect in La2O2Bi3Ag1-xSnxS6
3.3.3. Se-Substitution Effect in La2O2Bi3Ag0.6Sn0.4S5.7Se0.3
3.3.4. Eu-Substitution Effect on La2-xEuxO2Bi3Ag0.6Sn0.4S6
3.3.5. Rare Earth (RE) Substitution Effects in La1.6RE0.4O2Bi3Ag0.6Sn0.4S6
4. Summary
5. Experimental Details
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bednorz, J.G.; Muller, K.A. Possible high Tc superconductivity in the Ba−La−Cu−O system. Z. Phys. B 1986, 64, 189–193. [Google Scholar] [CrossRef]
- Kamihara, Y.; Watanabe, T.; Hirano, M.; Hosono, H. Iron-Based Layered Superconductor La[O1-xFx]FeAs (x = 0.05−0.12) with Tc = 26 K. J. Am. Chem. Soc. 2008, 130, 3296–3297. [Google Scholar] [CrossRef] [PubMed]
- Mizuguchi, Y.; Fujihisa, H.; Gotoh, Y.; Suzuki, K.; Usui, H.; Kuroki, K.; Demura, S.; Takano, Y.; Izawa, H.; Miura, O. BiS2-based layered superconductor Bi4O4S3. Phys. Rev. B 2012, 86, 220510. [Google Scholar] [CrossRef]
- Mizuguchi, Y.; Demura, S.; Deguchi, K.; Takano, Y.; Fujihisa, H.; Gotoh, Y.; Izawa, H.; Miura, O. Superconductivity in Novel BiS2-Based Layered Superconductor LaO1-xFxBiS2. J. Phys. Soc. Jpn. 2012, 81, 114725. [Google Scholar] [CrossRef]
- Singh, S.K.; Kumar, A.; Gahtori, B.; Kirtan, S.; Sharma, G.; Patnaik, S.; Awana, V.P.S. Bulk Superconductivity in Bismuth Oxysulfide Bi4O4S3. J. Am. Chem. Soc. 2012, 134, 16504–16507. [Google Scholar] [CrossRef] [PubMed]
- Demura, S.; Mizuguchi, Y.; Deguchi, K.; Okazaki, H.; Hara, H.; Watanabe, T.; Denholme, S.J.; Fujioka, M.; Ozaki, T.; Fujihisa, H.; et al. New Member of BiS2-Based Superconductor NdO1-xFxBiS2. J. Phys. Soc. Jpn. 2013, 82, 033708. [Google Scholar] [CrossRef]
- Jha, R.; Kumar, A.; Singh, S.K.; Awana, V.P.S. Superconductivity at 5 K in NdO0.5F0.5BiS2. J. Appl. Phys. 2013, 113, 056102. [Google Scholar] [CrossRef]
- Jha, R.; Kumar, A.; Singh, S.K.; Awana, V.P.S. Synthesis and Superconductivity of New BiS2-Based Superconductor PrO0.5F0.5BiS2. J. Supercond. Nov. Magn. 2013, 26, 499–502. [Google Scholar] [CrossRef]
- Xing, J.; Li, S.; Ding, X.; Yang, H.; Wen, H.H. Superconductivity appears in the vicinity of semiconducting-like behavior in CeO1−xFxBiS2. Phys. Rev. B 2012, 86, 214518. [Google Scholar] [CrossRef]
- Yazici, D.; Huang, K.; White, B.D.; Chang, A.H.; Friedman, A.J.; Maple, M.B. Superconductivity of F-substituted LnOBiS2 (Ln=La, Ce, Pr, Nd, Yb) compounds. Philos. Mag. 2012, 93, 673–680. [Google Scholar] [CrossRef]
- Yazici, D.; Huang, K.; White, B.D.; Jeon, I.; Burnett, V.W.; Friedman, A.J.; Lum, I.K.; Nallaiyan, M.; Spagna, S.; Maple, M.B. Superconductivity induced by electron doping in La1−xMxOBiS2(M= Ti, Zr, Hf, Th). Phys. Rev. B 2013, 87, 174512. [Google Scholar] [CrossRef]
- Krzton-Maziopa, A.; Guguchia, Z.; Pomjakushina, E.; Pomjakushin, V.; Khasanov, R.; Luetkens, H.; Biswas, P.; Amato, A.; Keller, H.; Conder, K. Superconductivity in a new layered bismuth oxyselenide: LaO0.5F0.5BiSe2. J. Phys.-Condens. Matter 2014, 26, 215702. [Google Scholar] [CrossRef] [PubMed]
- Mizuguchi, Y.; Omachi, A.; Goto, Y.; Kamihara, Y.; Matoba, M.; Hiroi, T.; Kajitani, J.; Miura, O. Enhancement of thermoelectric properties by Se substitution in layered bismuth-chalcogenide LaOBiS2-xSex. J. Appl. Phys. 2014, 116, 163915. [Google Scholar] [CrossRef]
- Lin, X.; Ni, X.; Chen, B.; Xu, X.; Yang, X.; Dai, J.; Li, Y.; Yang, X.; Luo, Y.; Tao, Q.; et al. Superconductivity induced by La doping in Sr1−xLaxFBiS2. Phys. Rev. B 2013, 87, 020504. [Google Scholar] [CrossRef]
- Zhai, H.F.; Tang, Z.T.; Jiang, H.; Xu, K.; Zhang, K.; Zhang, P.; Bao, J.K.; Sun, Y.L.; Jiao, W.H.; Nowik, I.; et al. Possible charge-density wave, superconductivity, and f-electron valence instability in EuBiS2F. Phys. Rev. B 2014, 90, 064518. [Google Scholar] [CrossRef]
- Kotegawa, H.; Tomita, Y.; Tou, H.; Izawa, H.; Mizuguchi, Y.; Miura, O.; Demura, S.; Deguchi, K.; Takano, Y. Pressure Study of BiS2-Based Superconductors Bi4O4S3 and La(O,F)BiS2. J. Phys. Soc. Jpn. 2012, 81, 103702. [Google Scholar] [CrossRef]
- Wolowiec, C.T.; Yazici, D.; White, B.D.; Huang, K.; Maple, M.B. Pressure-induced enhancement of superconductivity and suppression of semiconducting behavior in LnO0.5F0.5BiS2 (Ln=La,Ce) compounds. Phys. Rev. B 2013, 88, 064503. [Google Scholar] [CrossRef]
- Wolowiec, C.T.; White, B.D.; Jeon, I.; Yazici, D.; Huang, K.; Maple, M.B. Enhancement of superconductivity near the pressure-induced semiconductor–metal transition in the BiS2-based superconductors LnO0.5F0.5BiS2 (Ln = La, Ce, Pr, Nd). J. Phys. Condens. Matter 2013, 25, 422201. [Google Scholar] [CrossRef]
- Jha, R.; Tiwari, B.; Awana, V.P.S. Impact of Hydrostatic Pressure on Superconductivity of Sr0.5La0.5FBiS2. J. Phys. Soc. Jpn. 2014, 83, 063707. [Google Scholar] [CrossRef]
- Jha, R.; Tiwari, B.; Awana, V.P.S. Appearance of bulk superconductivity under hydrostatic pressure in Sr0.5RE0.5FBiS2 (RE = Ce, Nd, Pr, and Sm) compounds. J. Appl. Phys. 2015, 117, 013901. [Google Scholar] [CrossRef]
- Mizuguchi, Y.; Miura, A.; Kajitani, J.; Hiroi, T.; Miura, O.; Tadanaga, K.; Kumada, N.; Magome, E.; Moriyoshi, C.; Kuroiwa, Y. In-plane chemical pressure essential for superconductivity in BiCh2-based (Ch: S, Se) layered structure. Sci. Rep. 2015, 5, 14968. [Google Scholar] [CrossRef] [PubMed]
- Kajitani, J.; Hiroi, T.; Omachi, A.; Miura, O.; Mizuguchi, Y. Chemical Pressure Effect on Superconductivity of BiS2-Based Ce1−xNdxO1−yFyBiS2 and Nd1−zSmzO1−yFyBiS2. J. Phys. Soc. Jpn. 2015, 84, 044712. [Google Scholar] [CrossRef]
- Paris, E.; Joseph, B.; Iadecola, A.; Sugimoto, T.; Olivi, L.; Demura, S.; Mizuguchi, Y.; Takano, Y.; Mizokawa, T.; Saini, N.L. Determination of local atomic displacements in CeO1−xFxBiS2 system. J. Phys. Condens. Matter 2014, 26, 435701. [Google Scholar]
- Mizuguchi, Y.; Paris, E.; Sugimoto, T.; Iadecola, A.; Kajitani, J.; Miura, O.; Mizokawa, T.; Saini, N.L. The effect of RE substitution in layered REO0.5F0.5BiS2: Chemical pressure, local disorder and superconductivity. Phys. Chem. Chem. Phys. 2015, 17, 22090–22096. [Google Scholar] [CrossRef] [PubMed]
- Athauda, A.; Yang, J.; Lee, S.; Mizuguchi, Y.; Deguchi, K.; Takano, Y.; Miura, O.; Louca, D. In-plane charge fluctuations in bismuth-sulfide superconductors. Phys. Rev. B 2015, 91, 144112. [Google Scholar] [CrossRef]
- Nagasaka, K.; Nishida, A.; Jha, R.; Kajitani, J.; Miura, O.; Higashinaka, R.; Matsuda, T.D.; Aoki, Y.; Miura, A.; Moriyoshi, C.; et al. Intrinsic Phase Diagram of Superconductivity in the BiCh2-Based System Without In-Plane Disorder. J. Phys. Soc. Jpn. 2017, 86, 074701. [Google Scholar] [CrossRef]
- Mizuguchi, Y. Material Development and Physical Properties of BiS2-Based Layered Compounds. J. Phys. Soc. Jpn. 2019, 88, 041001. [Google Scholar] [CrossRef]
- Jinno, G.; Jha, R.; Yamada, A.; Higashinaka, R.; Matsuda, T.D.; Aoki, Y.; Nagao, M.; Miura, O.; Mizuguchi, Y. Bulk Superconductivity Induced by In-Plane Chemical Pressure Effect in Eu0.5La0.5FBiS2−xSex. J. Phys. Soc. Jpn. 2016, 85, 124708. [Google Scholar] [CrossRef]
- Tanaka, M.; Yamaki, T.; Matsushita, Y.; Fujioka, M.; Denholme, S.J.; Yamaguchi, T.; Takeya, H.; Takano, Y. Site selectivity on chalcogen atoms in superconducting La(O,F)BiSSe. Appl. Phys. Lett. 2015, 106, 112601. [Google Scholar] [CrossRef]
- Hiroi, T.; Kajitani, J.; Omachi, A.; Miura, O.; Mizuguchi, Y. Evolution of Superconductivity in BiS2-Based Superconductor LaO0.5F0.5Bi(S1−xSex)2. J. Phys. Soc. Jpn. 2015, 84, 024723. [Google Scholar] [CrossRef]
- Tanaka, M.; Nagao, M.; Matsushita, Y.; Fujioka, M.; Denholme, S.J.; Yamaguchi, T.; Takeya, H.; Takano, Y. First single crystal growth and structural analysis of superconducting layered bismuth oxyselenide; La(O,F)BiSe2. J. Solid State Chem. 2014, 219, 168. [Google Scholar] [CrossRef]
- Nagao, M.; Tanaka, M.; Watauchi, S.; Tanaka, I.; Takano, Y. Superconducting Anisotropies of F-Substituted LaOBiSe2 Single Crystals. J. Phys. Soc. Jpn. 2014, 83, 114709. [Google Scholar] [CrossRef]
- Feng, Y.; Ding, H.C.; Du, Y.; Wan, X.; Wang, B.; Savrasov, S.Y.; Duan, C.G. Electron-phonon superconductivity in LaO0.5F0.5BiSe2. J. Appl. Phys. 2014, 115, 233901. [Google Scholar] [CrossRef]
- Wang, G.; Wang, D.; Shi, X.; Peng, Y. First-principles study of the electronic structure and thermoelectric properties of LaOBiCh2 (Ch = S, Se). Mod. Phys. Lett. B 2017, 31, 1750265. [Google Scholar] [CrossRef]
- Liu, J.; Li, S.; Li, Y.; Zhu, X.; Wen, H.H. Pressure-tuned enhancement of superconductivity and change of ground state properties in LaO0.5F0.5BiSe2 single crystals. Phys. Rev. B 2014, 90, 094507. [Google Scholar] [CrossRef]
- Shao, J.; Liu, Z.; Yao, X.; Zhang, L.; Pi, L.; Tan, S.; Zhang, C.; Zhang, Y. Superconducting properties of BiSe2-based LaO1−xFxBiSe2 single crystals. EPL 2014, 107, 37006. [Google Scholar] [CrossRef][Green Version]
- Fujioka, M.; Tanaka, M.; Denholme, S.J.; Yamaki, T.; Takeya, H.; Yamaguchi, T.; Takano, Y. Pressure-induced phase transition for single-crystalline LaO0.5F0.5BiSe2. EPL 2014, 108, 47007. [Google Scholar] [CrossRef]
- Hoshi, K.; Goto, Y.; Mizuguchi, Y. Selenium isotope effect in the layered bismuth chalcogenide superconductor LaO0.6F0.4Bi(S,Se)2. Phys. Rev. B 2018, 97, 094509. [Google Scholar] [CrossRef]
- Jha, R.; Awana, V.P.S. Anomalous Impact of Hydrostatic Pressure on Superconductivity of Polycrystalline LaO0.5F0.5BiSe2. J. Supercond. Nov. Magn. 2015, 28, 2229–2233. [Google Scholar] [CrossRef][Green Version]
- Nishida, A.; Miura, O.; Lee, C.H.; Mizuguchi, Y. High thermoelectric performance and low thermal conductivity of densified LaOBiSSe. Appl. Phys. Express 2015, 8, 111801. [Google Scholar] [CrossRef]
- Mizuguchi, Y.; Hiroi, T.; Miura, O. Superconductivity phase diagram of Se-substituted CeO0.5F0.5Bi(S1-xSex)2. J. Phys. Conf. Ser. 2016, 683, 012001. [Google Scholar] [CrossRef]
- Hoshi, K.; Kimata, M.; Goto, Y.; Matsuda, T.D.; Mizuguchi, Y. Two-Fold-Symmetric Magnetoresistance in Single Crystals of Tetragonal BiCh2-Based Superconductor LaO0.5F0.5BiSSe. J. Phys. Soc. Jpn. 2019, 88, 033704. [Google Scholar] [CrossRef]
- Mizuguchi, Y.; Miura, A.; Nishida, A.; Miura, O.; Tadanaga, K.; Kumada, N.; Lee, C.H.; Magome, E.; Moriyoshi, C.; Kuroiwa, Y. Compositional and temperature evolution of crystal structure of new thermoelectric compound LaOBiS2−xSex. J. Appl. Phys. 2016, 119, 155103. [Google Scholar] [CrossRef]
- Lee, C.H.; Nishida, A.; Hasegawa, T.; Nishiate, H.; Kunioka, H.; Ohira-Kawamura, S.; Nakamura, M.; Nakajima, K.; Mizuguchi, Y. Effect of rattling motion without cage structure on lattice thermal conductivity in LaOBiS2−xSex. Appl. Phys. Lett. 2018, 112, 023903. [Google Scholar] [CrossRef]
- Kase, N.; Terui, Y.; Nakano, T.; Takeda, N. Superconducting gap symmetry of the BiS2-based superconductor LaO0.5F0.5BiSSe elucidated through specific heat measurements. Phys. Rev. B 2017, 96, 214506. [Google Scholar] [CrossRef]
- Nagao, M.; Demura, S.; Deguchi, K.; Miura, A.; Watauchi, S.; Takei, T.; Takano, Y.; Kumada, N.; Tanaka, I. Structural Analysis and Superconducting Properties of F-Substituted NdOBiS2 Single Crystals. J. Phys. Soc. Jpn. 2013, 82, 113701. [Google Scholar] [CrossRef]
- Liu, J.; Fang, D.; Wang, Z.; Xing, J.; Du, Z.; Zhu, X.; Yang, H.; Wen, H.H. Giant superconducting fluctuation and anomalous semiconducting normal state in NdO1−xFxBi1−yS2 single crystals. EPL 2014, 106, 67002. [Google Scholar] [CrossRef]
- Nagao, M. Growth and characterization of R(O,F)BiS2 (R = La, Ce, Pr, Nd) superconducting single crystals. Nov. Supercond. Mater. 2015, 1, 64–74. [Google Scholar] [CrossRef]
- Sun, Y.L.; Ablimit, A.; Zhai, H.F.; Bao, J.K.; Tang, Z.T.; Wang, X.B.; Wang, N.L.; Feng, C.M.; Cao, G.H. Design and Synthesis of a New Layered Thermoelectric Material LaPbBiS3O. Inorg. Chem. 2014, 53, 11125–11129. [Google Scholar] [CrossRef]
- Mizuguchi, Y.; Hijikata, Y.; Abe, T.; Moriyoshi, C.; Kuroiwa, Y.; Goto, Y.; Miura, A.; Lee, S.; Torii, S.; Kamiyama, T.; et al. Crystal structure, site selectivity, and electronic structure of layered chalcogenide LaOBiPbS3. EPL 2017, 119, 26002. [Google Scholar] [CrossRef][Green Version]
- Hijikata, Y.; Abe, T.; Moriyoshi, C.; Kuroiwa, Y.; Goto, Y.; Miura, A.; Tadanaga, K.; Wang, Y.; Miura, O.; Mizuguchi, Y. Synthesis, Crystal Structure, and Physical Properties of New Layered Oxychalcogenide La2O2Bi3AgS6. J. Phys. Soc. Jpn. 2017, 86, 124802. [Google Scholar] [CrossRef]
- Jha, R.; Goto, Y.; Higashinaka, R.; Matsuda, T.D.; Aoki, Y.; Mizuguchi, Y. Superconductivity in Layered Oxychalcogenide La2O2Bi3AgS6. J. Phys. Soc. Jpn. 2018, 87, 083704. [Google Scholar] [CrossRef]
- Wu, M.K.; Ashburn, J.R.; Torng, C.J.; Hor, P.H.; Meng, R.L.; Gao, L.; Huang, Z.J.; Wang, Y.Q.; Chu, C.W. Superconductivity at 93 K in a new mixed-phase Y-Ba-Cu-O compound system at ambient pressure. Phys. Rev. Lett. 1987, 58, 908. [Google Scholar] [CrossRef] [PubMed]
- Cava, R.J.; Van Dover, R.B.; Batlogg, B.; Rietman, E.A. Bulk superconductivity at 36 K in La1.8Sr0.2CuO4. Phys. Rev. Lett. 1987, 58, 408. [Google Scholar] [CrossRef]
- PELIKÁN, P. The relation between structure and superconductive properties of high-temperature superconductors. Chem. Papers 1990, 44, 721–736. [Google Scholar]
- Hazen, R.M.; Prewitt, C.T.; Angel, R.J.; Ross, N.L.; Finger, L.W.; Hadidiacos, C.G.; Veblen, D.R.; Heaney, P.J.; Hor, P.H.; Meng, R.L.; et al. Superconductivity in the high-Tc Bi-Ca-Sr-Cu-O system: Phase identification. Phys. Rev. Lett. 1988, 60, 1174. [Google Scholar] [CrossRef]
- Maeda, H.; Tanaka, Y.; Fukitoki, M.; Asano, T. A New High-Tc Oxide Superconductor without a Rare Earth Element. Jpn. J. Appl. Phys. 1988, 27, L209. [Google Scholar] [CrossRef]
- Eisaki, H.; Kaneko, N.; Feng, D.L.; Damascelli, A.; Mang, P.K.; Shen, K.M.; Shen, Z.X.; Greven, M. Effect of chemical inhomogeneity in bismuth-based copper oxide superconductors. Phys. Rev. B 2004, 69, 064512. [Google Scholar] [CrossRef]
- Brandt, B.; Ginzburg, N.I. Critical Fields of the Crystalline Modifications Bi II and Bi III. SoV. Phys. JETP 1963, 17, 326. [Google Scholar]
- Stromberg, H.D.; Stephens, D.R. Effects of pressure on the electrical resistance of certain metals. J. Phys. Chem. Solids 1964, 25, 1015. [Google Scholar] [CrossRef]
- Brandt, N.B.; Ginzburg, N.I. Superconductivity at high pressures. Comtemp. Phys. 1969, 10, 355. [Google Scholar] [CrossRef]
- Weitzel, B.; Micklitz, H. Superconductivity in granular systems built from well-defined rhombohedral Bi-clusters: Evidence for Bi-surface superconductivity. Phys. ReV. Lett. 1991, 66, 385. [Google Scholar] [CrossRef]
- Liu, L.Y.; Xing, Y.T.; Merino, I.L.C.; Micklitz, H.; Franceschini, D.F.; Baggio-Saitovitch, E.; Bell, D.C.; Solorzano, I.G. Superconductivity in Bi/Ni bilayer system: Clear role of superconducting phases found at Bi/Ni interface. Phys. Rev. Mater. 2018, 2, 014601. [Google Scholar] [CrossRef]
- Chao, S.P. Superconductivity in a Bi/Ni bilayer. Phys. Rev. B 2019, 99, 064504. [Google Scholar] [CrossRef]
- Tian, M.; Wang, J.; Kumar, N.; Han, T.; Kobayashi, Y.; Liu, Y.; Mallouk, T.E.; Chan, M.H.W. Observation of Superconductivity in Granular Bi Nanowires Fabricated by Electrodeposition. Nano Lett. 2006, 6, 2773–2780. [Google Scholar] [CrossRef] [PubMed]
- Kumar, J.; Kumar, A.; Vajpayee, A.; Gahtori, B.; Sharma, D.; Ahluwalia, P.K.; Auluck, S.; Awana, V.P.S. Physical property and electronic structure characterization of bulk superconducting Bi3Ni. Supercond. Sci. Technol. 2011, 24, 085002. [Google Scholar] [CrossRef]
- Jha, R.; Avila, M.A.; Ribeiro, R.A. Hydrostatic pressure effect on the superconducting properties of BaBi3 and SrBi3 single crystals. Supercond. Sci. Technol. 2017, 30, 025015. [Google Scholar] [CrossRef]
- Fang, Y.; Yazici, D.; White, B.D.; Maple, M.B. Enhancement of superconductivity in La1−xSmxO0.5F0.5BiS2. Phys. Rev. B 2015, 91, 064510. [Google Scholar] [CrossRef]
- Jeon, I.; Yazici, D.; White, B.D.; Friedman, A.J.; Maple, M.B. Effect of yttrium substitution on the superconducting properties of La1−xYxO0.5F0.5BiS2. Phys. Rev. B 2014, 90, 054510. [Google Scholar] [CrossRef]
- Thakur, G.S.; Selvan, G.K.; Haque, Z.; Gupta, L.C.; Samal, S.L.; Arumugam, S.; Ganguli, A.K. Synthesis and Properties of SmO0.5F0.5BiS2 and Enhancement in Tc in La1–ySmyO0.5F0.5BiS2. Inorg.Chem. 2015, 54, 1076–1081. [Google Scholar] [CrossRef]
- Mizuguchi, Y.; Hoshi, K.; Goto, Y.; Miura, A.; Tadanaga, K.; Moriyoshi, C.; Kuroiwa, Y. Evolution of Anisotropic Displacement Parameters and Superconductivity with Chemical Pressure in BiS2-Based REO0.5F0.5BiS2 (RE = La, Ce, Pr, and Nd). J. Phys. Soc. Jpn. 2018, 87, 023704. [Google Scholar] [CrossRef]
- Paris, E.; Mizuguchi, Y.; Hacisalihoglu, M.Y.; Hiroi, T.; Joseph, B.; Aquilanti, G.; Miura, O.; Mizokawa, T.; Saini, N.L. Role of the local structure in superconductivity of LaO0.5F0.5BiS2-x Sex system. J. Phys.-Condes. Matter 2017, 29, 145603. [Google Scholar] [CrossRef] [PubMed]
- Jha, R.; Goto, Y.; Matsuda, T.D.; Aoki, Y.; Nagao, M.; Tanaka, I.; Mizuguchi, Y. Bulk superconductivity in a four-layer-type Bi-based compound La2O2Bi3Ag0.6Sn0.4S5.7Se0.3. Sci. Rep. 2019, 9, 13346. [Google Scholar] [CrossRef] [PubMed]
- Jha, R.; Goto, Y.; Higashinaka, R.; Miura, A.; Moriyoshi, C.; Kuroiwa, Y.; Mizuguchi, Y. Improvement of superconducting properties by chemical pressure effect in Eu-doped La2-xEuxO2Bi3Ag0.6Sn0.4S6. arXiv 2019, arXiv:1908.09311. [Google Scholar]
- Kim, G.C.; Cheon, M.; Choi, W.; Ahmad, D.; Kwon, Y.S.; Ko, R.; Kim, Y.C. Superconductivity in Oxychalcogenide LaREO2Bi3Ag0.6Sn0.4S6 (RE = Pr and Nd). J. Supercond. Nov. Magn. 2019, 33, 625. [Google Scholar] [CrossRef]
- Ruan, B.B.; Zhao, K.; Mu, Q.G.; Pan, B.J.; Liu, T.; Yang, H.X.; Li, J.Q.; Chen, G.F.; Ren, Z.A. Superconductivity in Bi3O2S2Cl with Bi–Cl Planar Layers. J. Am. Chem. Soc. 2019, 141, 3404. [Google Scholar] [CrossRef]
- Jha, R.; Goto, Y.; Matsuda, T.D.; Aoki, Y.; Mizuguchi, Y. Superconductivity in Se-doped La2O2Bi2Pb2S6-xSex with a Bi2Pb2Ch4-type thick conducting layer. arXiv 2019, arXiv:1912.11981. [Google Scholar]
- Jha, R.; Goto, Y.; Matsuda, T.D.; Aoki, Y.; Mizuguchi, Y. Effect of Indium doping on the superconductivity of layered oxychalcogenide La2O2Bi3Ag1-xInxS6. In Journal of Physics: Conference Series; IOP Publishing: Bristol, UK, 2019; Volume 1293, p. 012001. [Google Scholar]
- Jerome, D.; Berthier, C.; Molinie, P.; Rouxel, J. Layer compounds. Charge density waves in transitions metal compounds. Electronic properties of transition metal dichalcogenides: Connection between structural instabilities and superconductivity. J. Physique Colloq. 1976, 37, C4-125–C4-135. [Google Scholar]
- Li, L.; Deng, X.; Wang, Z.; Liu, Y.; Abeykoon, M.; Dooryhee, E.; Tomic, A.; Huang, Y.; Warren, J.B.; Bozin, E.S.; et al. Superconducting order from disorder in 2H-TaSe2−xSx. npj Quantum Mater. 2017, 2, 11. [Google Scholar] [CrossRef]
- Goto, Y.; Sogabe, R.; Mizuguchi, Y. Bulk Superconductivity Induced by Se Substitution in BiCh2-Based Layered Compounds Eu0.5Ce0.5FBiS2−xSex. J. Phys. Soc. Jpn. 2017, 86, 104712. [Google Scholar] [CrossRef]
- Pallecchi, I.; Lamura, G.; Putti, M.; Kajitani, J.; Mizuguchi, Y.; Miura, O.; Demura, S.; Deguchi, K.; Takano, Y. Effect of high-pressure annealing on the normal-state transport of LaO0.5F0.5BiS2. Phys. Rev. B 2014, 89, 214513. [Google Scholar] [CrossRef]
- Nishida, A.; Nishiate, H.; Lee, C.H.; Miura, O.; Mizuguchi, Y. Electronic Origins of Large Thermoelectric Power Factor of LaOBiS2−xSex. J. Phys. Soc. Jpn. 2016, 85, 074702. [Google Scholar] [CrossRef]
- Kurematsu, K.; Ochi, M.; Usui, H.; Kurok, K. First-principles Study of LaOPbBiS3 and Its Analogous Compounds as Thermoelectric Materials. J. Phys. Soc. Jpn. 2020, 89, 024702. [Google Scholar] [CrossRef]
- Morice, C.; Artacho, E.; Dutton, S.E.; Kim, H.J.; Saxena, S.S. Electronic and magnetic properties of superconducting LnO1−xFxBiS2 (Ln = La, Ce, Pr, and Nd) from first principles. J. Phys.-Condes. Matter 2016, 28, 345504. [Google Scholar] [CrossRef]
- Mejía-Salazar, J.R.; Perea, J.D.; Castillo, R.; Diosa J., E.; Baca, E. Hybrid Superconducting-Ferromagnetic [Bi2Sr2(Ca,Y)2Cu3O10]0.99(La2/3Ba1/3MnO3)0.01 Composite Thick Films. Materials 2019, 12, 861. [Google Scholar] [CrossRef]
- Rouco, V.; Córdoba, R.; De Teresa, J.M.; Rodríguez, L.A.; Navau, C.; Del-Valle, N.; Via, G.; Sánchez, A.; Monton, C.; Kronast, F.; et al. Competition between Superconductor-Ferromagnetic stray magnetic fields in YBa2Cu3O7−x films pierced with Co nano-rods. Sci. Rep. 2017, 7, 5663. [Google Scholar] [CrossRef]
- Zhang, G.; Samuely, T.; Xu, Z.; Jochum, J.K.; Volodin, A.; Zhou, S.; May, P.W.; Onufriienko, O.; Kacmarcík, J.; Steele, J.A.; et al. Superconducting Ferromagnetic Nanodiamond. ACS Nano 2017, 11, 5358–5366. [Google Scholar] [CrossRef]
- Izumi, F.; Momma, K. Three-Dimensional Visualization in Powder Diffraction. Solid State Phenom. 2007, 130, 15. [Google Scholar] [CrossRef]
- Momma, K.; Izumi, F. VESTA: A three-dimensional visualization system for electronic and structural analysis. J. Appl. Crystallogr. 2008, 41, 653. [Google Scholar] [CrossRef]

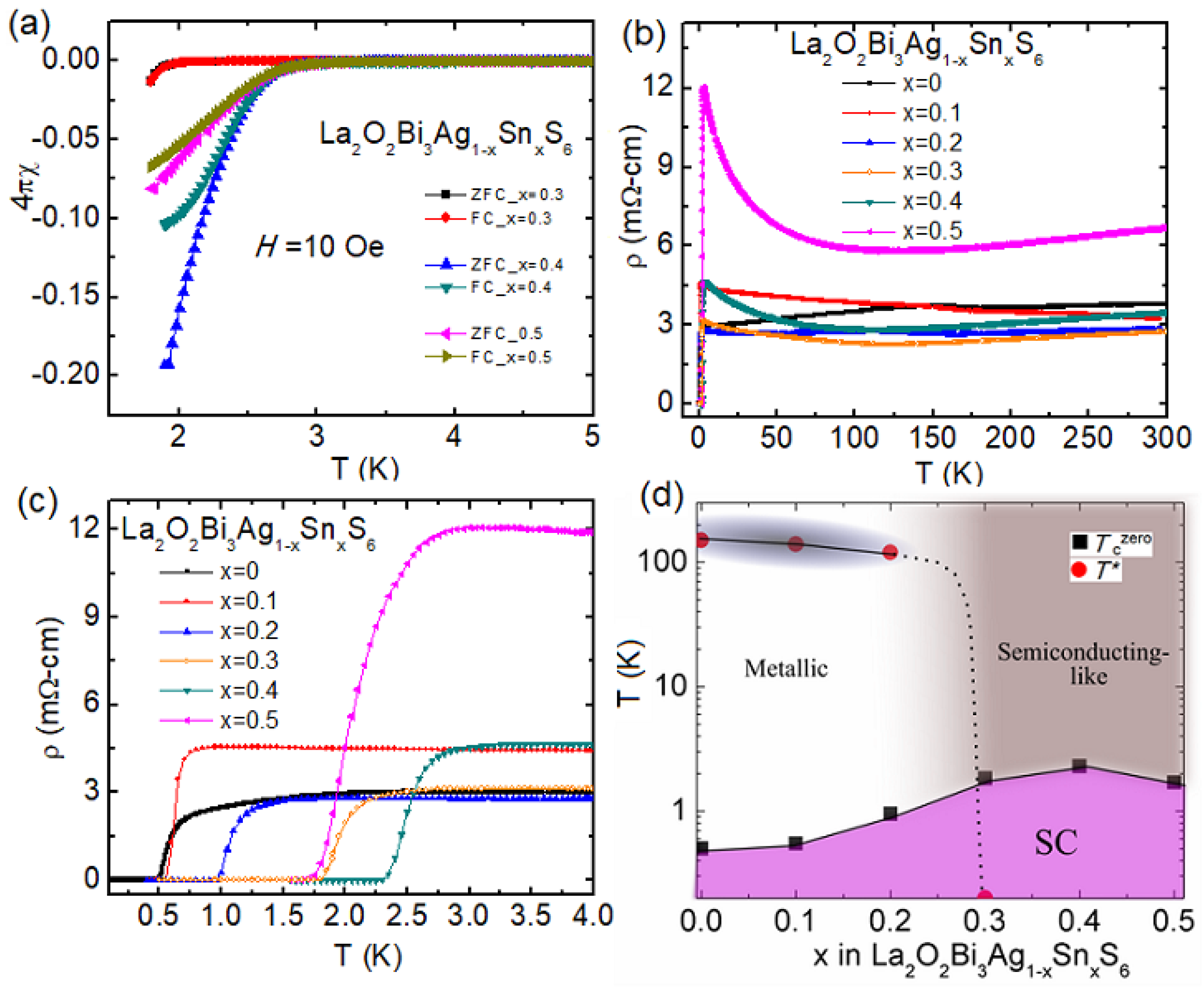
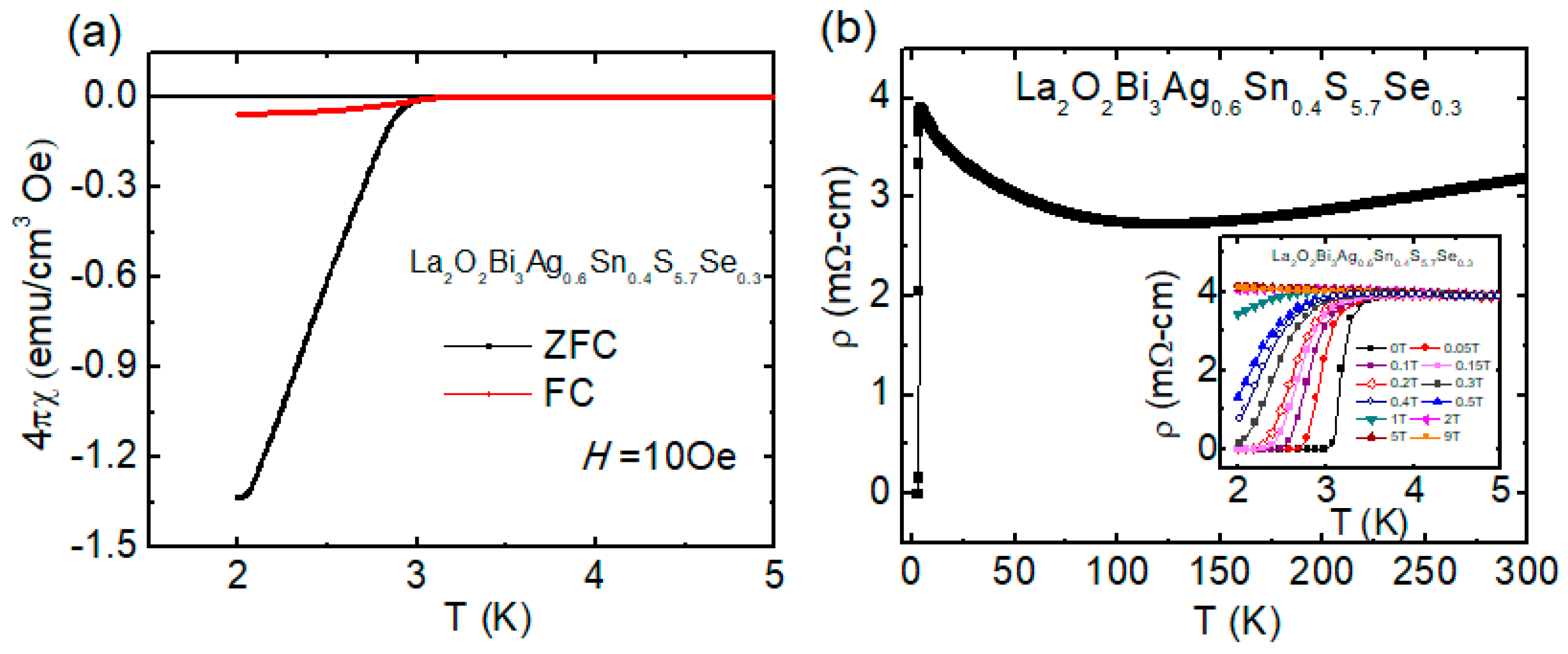
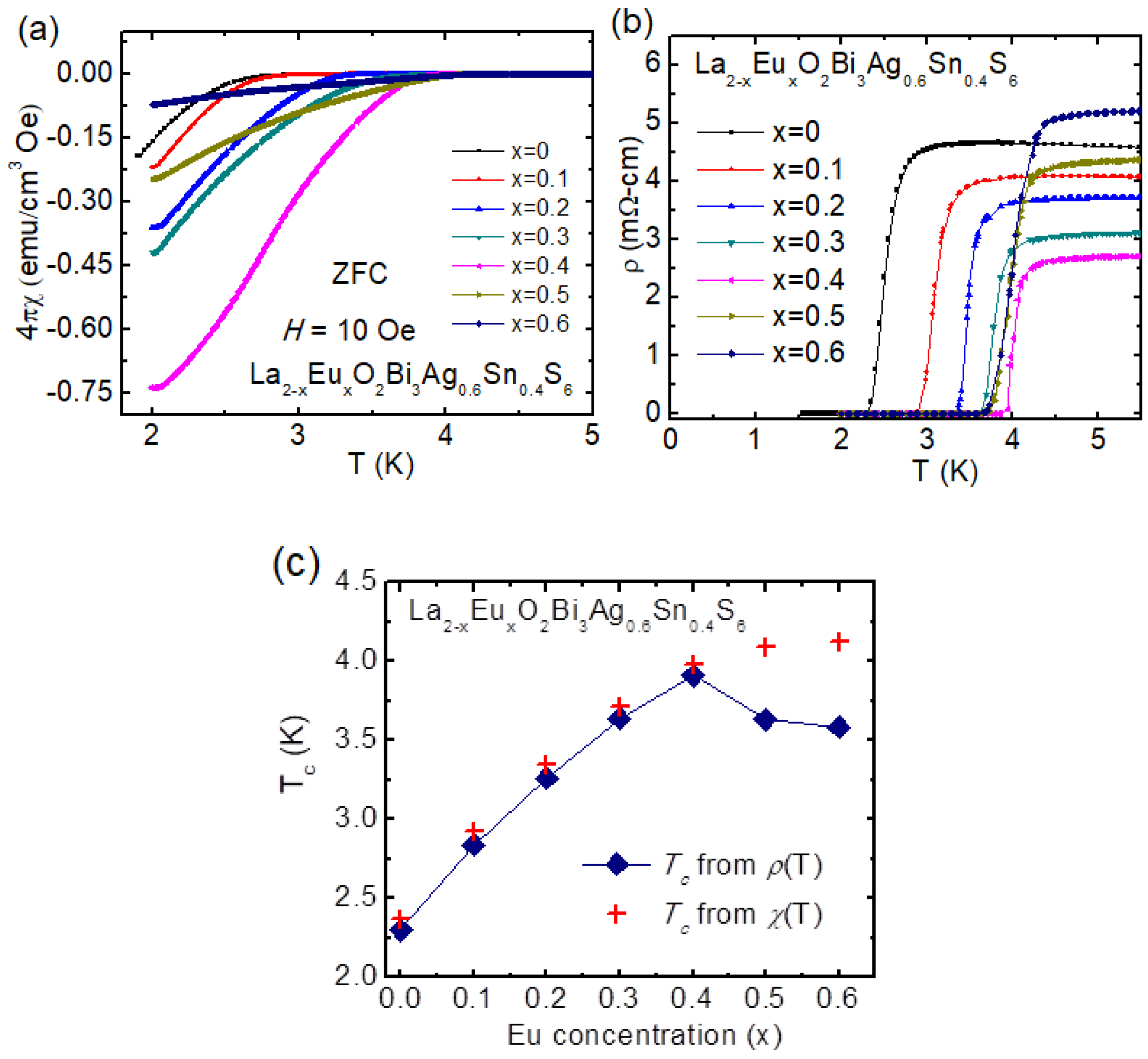
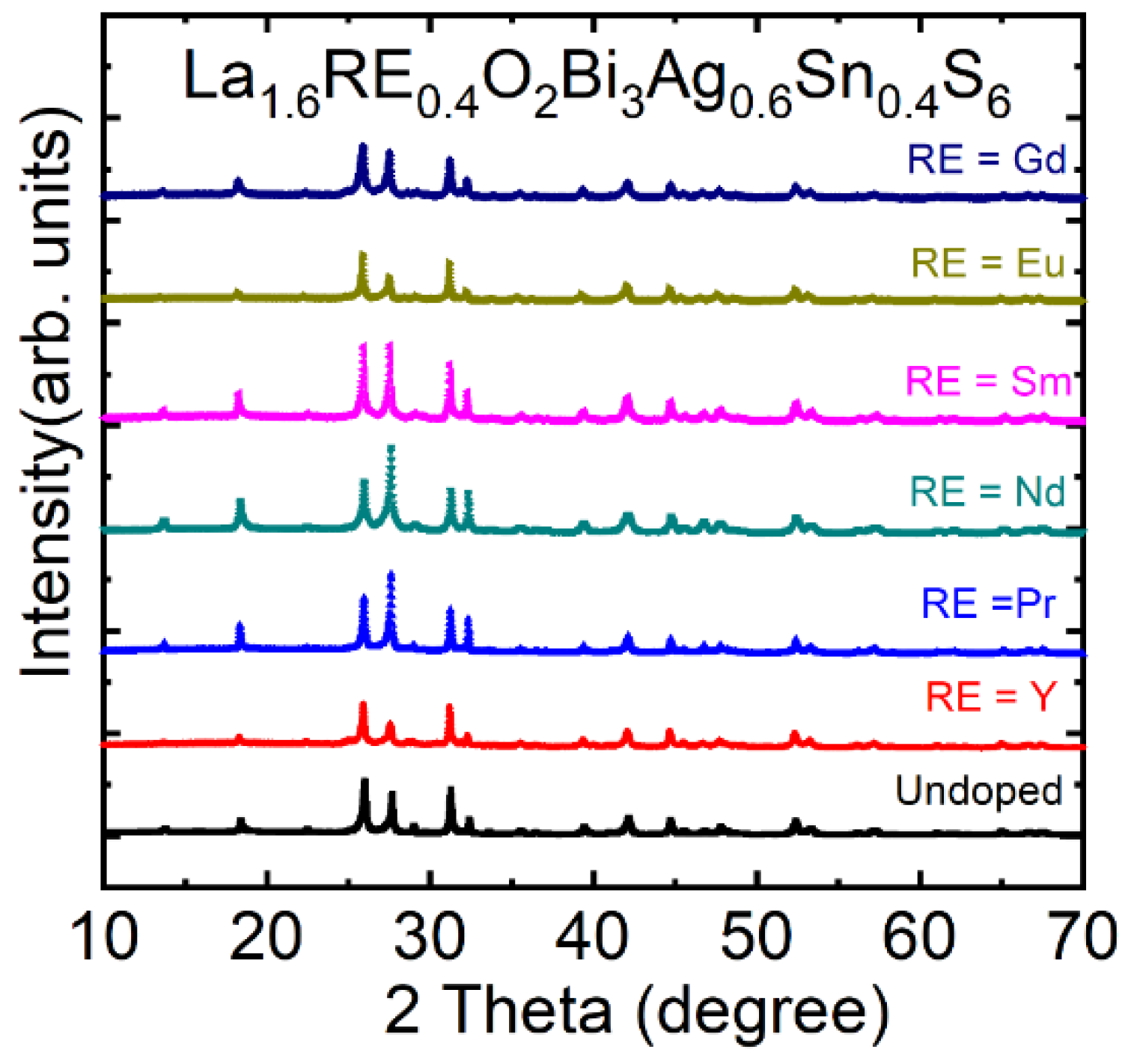
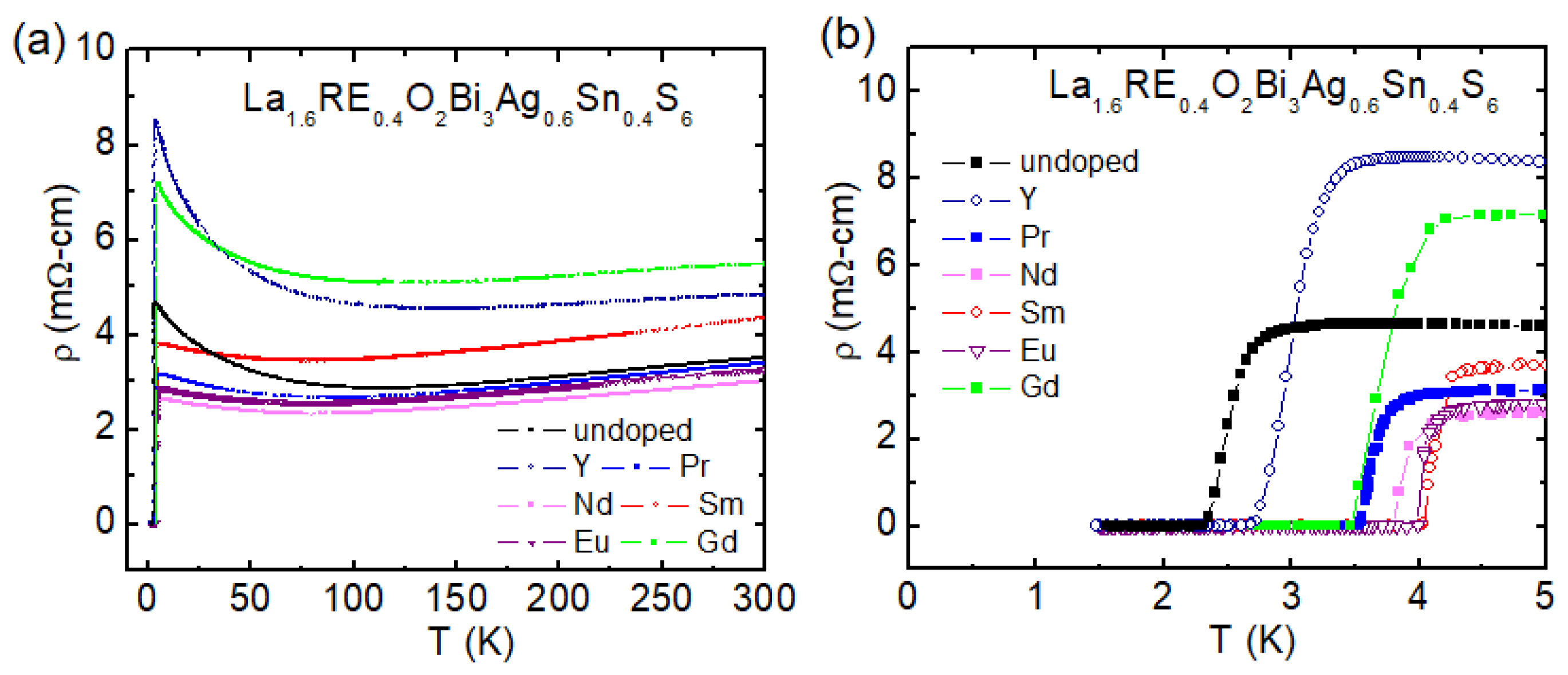
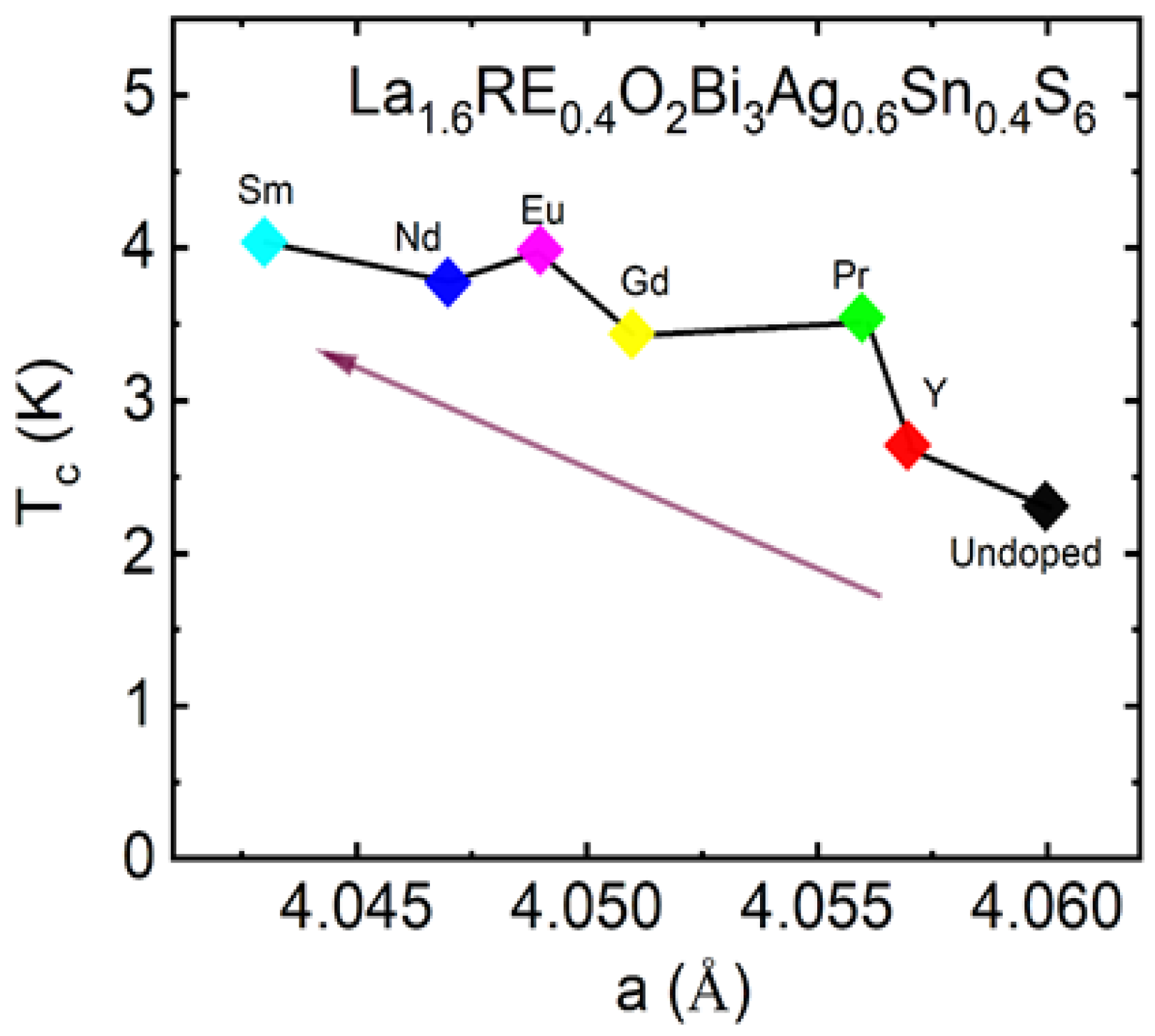
| Materials | Tc (K) | Ref. |
|---|---|---|
| La2O2Bi3AgS6 | 0.5 | [52] |
| La2O2Bi3Ag0.9In0.1S6 | 0.45 | [78] |
| La2O2Bi3Ag0.8In0.2S6 | 0.42 | |
| La2O2Bi3Ag0.7In0.3S6 | 0.4 | |
| La2O2Bi3Ag0.6In0.4S6 | 0.4 | |
| La2O2Bi3Ag0.9Sn0.1S6 | 0.55 | [73] |
| La2O2Bi3Ag0.8Sn0.2S6 | 0.95 | |
| La2O2Bi3Ag0.7Sn0.3S6 | 1.83 | |
| La2O2Bi3Ag0.6Sn0.4S6 | 2.5 | |
| La2O2Bi3Ag0.5Sn0.5S6 | 1.7 | |
| La2O2Bi3Ag0.6Sn0.4S5.7Se0.3 | 3.0 | |
| La1.9Eu0.1O2Bi3Ag0.6Sn0.4S6 | 2.84 | [74] |
| La1.8Eu0.2O2Bi3Ag0.6Sn0.4S6 | 3.26 | |
| La1.7Eu0.3O2Bi3Ag0.6Sn0.4S6 | 3.64 | |
| La1.6Eu0.4O2Bi3Ag0.6Sn0.4S6 | 4.0 | |
| La1.5Eu0.5O2Bi3Ag0.6Sn0.4S6 | 3.63 | |
| La1.4Eu0.6O2Bi3Ag0.6Sn0.4S6 | 3.58 | |
| La1.9Sm0.1O2Bi3Ag0.6Sn0.4S6 | 2.89 | To be published |
| La1.8Sm0.2O2Bi3Ag0.6Sn0.4S6 | 3.345 | |
| La1.7Sm0.3O2Bi3Ag0.6Sn0.4S6 | 3.795 | |
| La1.6Sm0.4O2Bi3Ag0.6Sn0.4S6 | 4.1 | |
| La1.5Sm0.5O2Bi3Ag0.6Sn0.4S6 | 3.867 | |
| La1.4Sm0.6O2Bi3Ag0.6Sn0.4S6 | 3.59 | |
| La1.6Y0.4O2Bi3Ag0.6Sn0.4S6 | 2.68 | [74] |
| La1.6Pr0.4O2Bi3Ag0.6Sn0.4S6 | 3.54 | |
| La1.6Nd0.4O2Bi3Ag0.6Sn0.4S6 | 3.8 | |
| La1.6Gd0.4O2Bi3Ag0.6Sn0.4S6 | 3.46 | |
| LaPrO2Bi3Ag0.6Sn0.4S6 | 3.5 | [75] |
| LaNdO2Bi3Ag0.6Sn0.4S6 | 3.4 | |
| La2O2Bi2PbS5.5Se0.5 | 1.15 | [77] |
| La2O2Bi2PbS5.0Se1.0 | 1.9 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jha, R.; Mizuguchi, Y. Superconductivity in La2O2M4S6 -Type Bi-based Compounds: A Review on Element Substitution Effects. Condens. Matter 2020, 5, 27. https://doi.org/10.3390/condmat5020027
Jha R, Mizuguchi Y. Superconductivity in La2O2M4S6 -Type Bi-based Compounds: A Review on Element Substitution Effects. Condensed Matter. 2020; 5(2):27. https://doi.org/10.3390/condmat5020027
Chicago/Turabian StyleJha, Rajveer, and Yoshikazu Mizuguchi. 2020. "Superconductivity in La2O2M4S6 -Type Bi-based Compounds: A Review on Element Substitution Effects" Condensed Matter 5, no. 2: 27. https://doi.org/10.3390/condmat5020027
APA StyleJha, R., & Mizuguchi, Y. (2020). Superconductivity in La2O2M4S6 -Type Bi-based Compounds: A Review on Element Substitution Effects. Condensed Matter, 5(2), 27. https://doi.org/10.3390/condmat5020027