THz Pulsed Imaging in Biomedical Applications
Abstract
1. Introduction
2. THz Pulsed System/Imaging
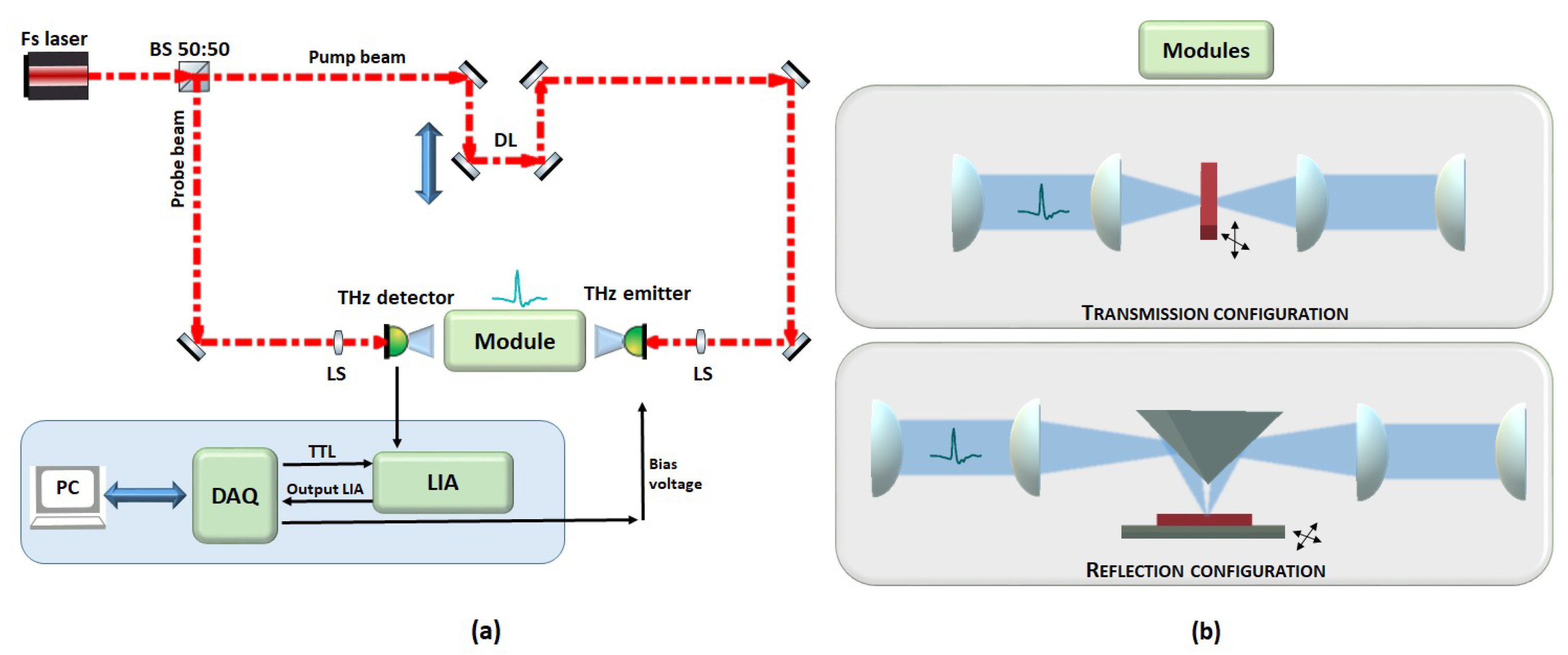
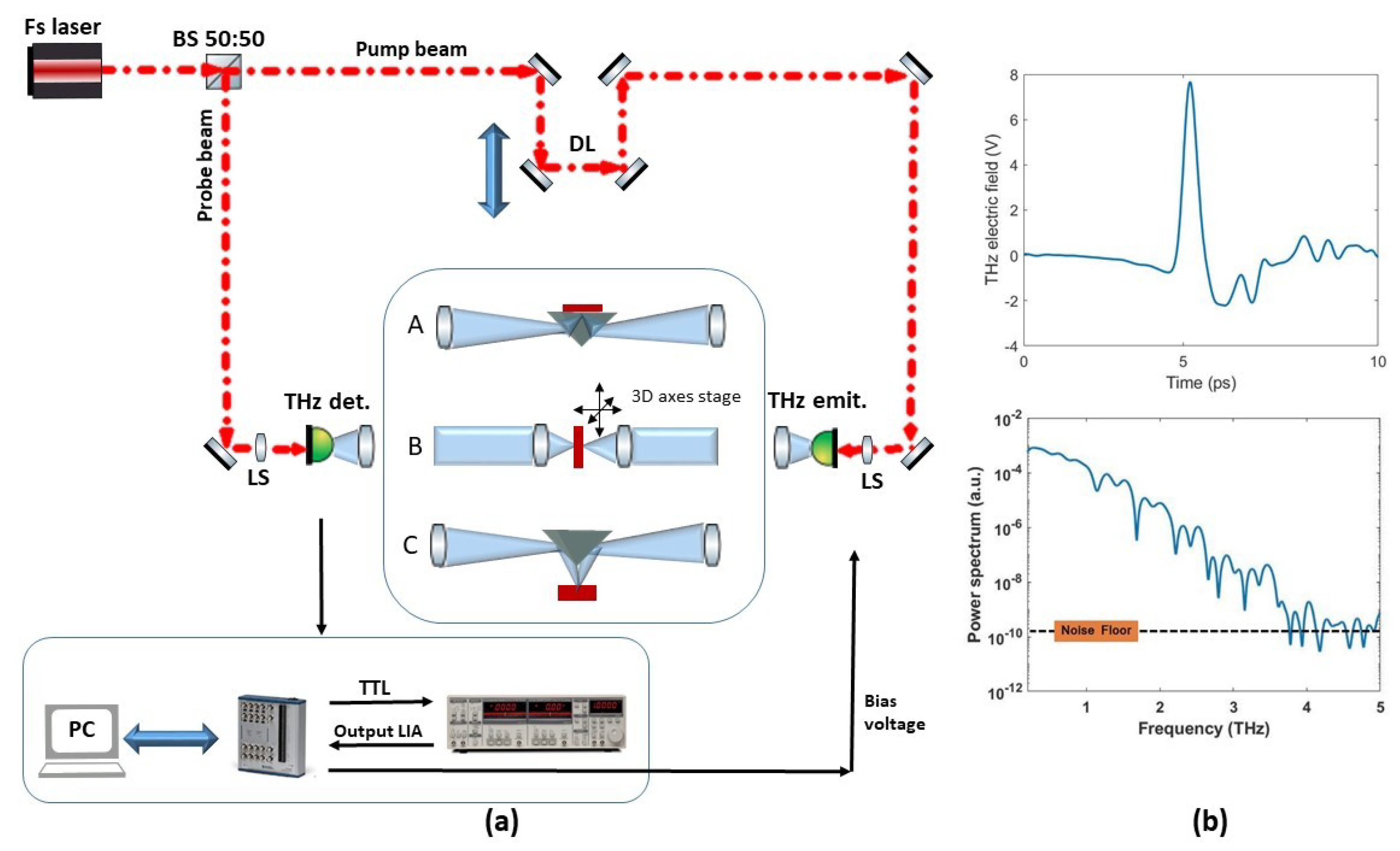
2.1. Generation and Detection of THz Pulsed Radiation
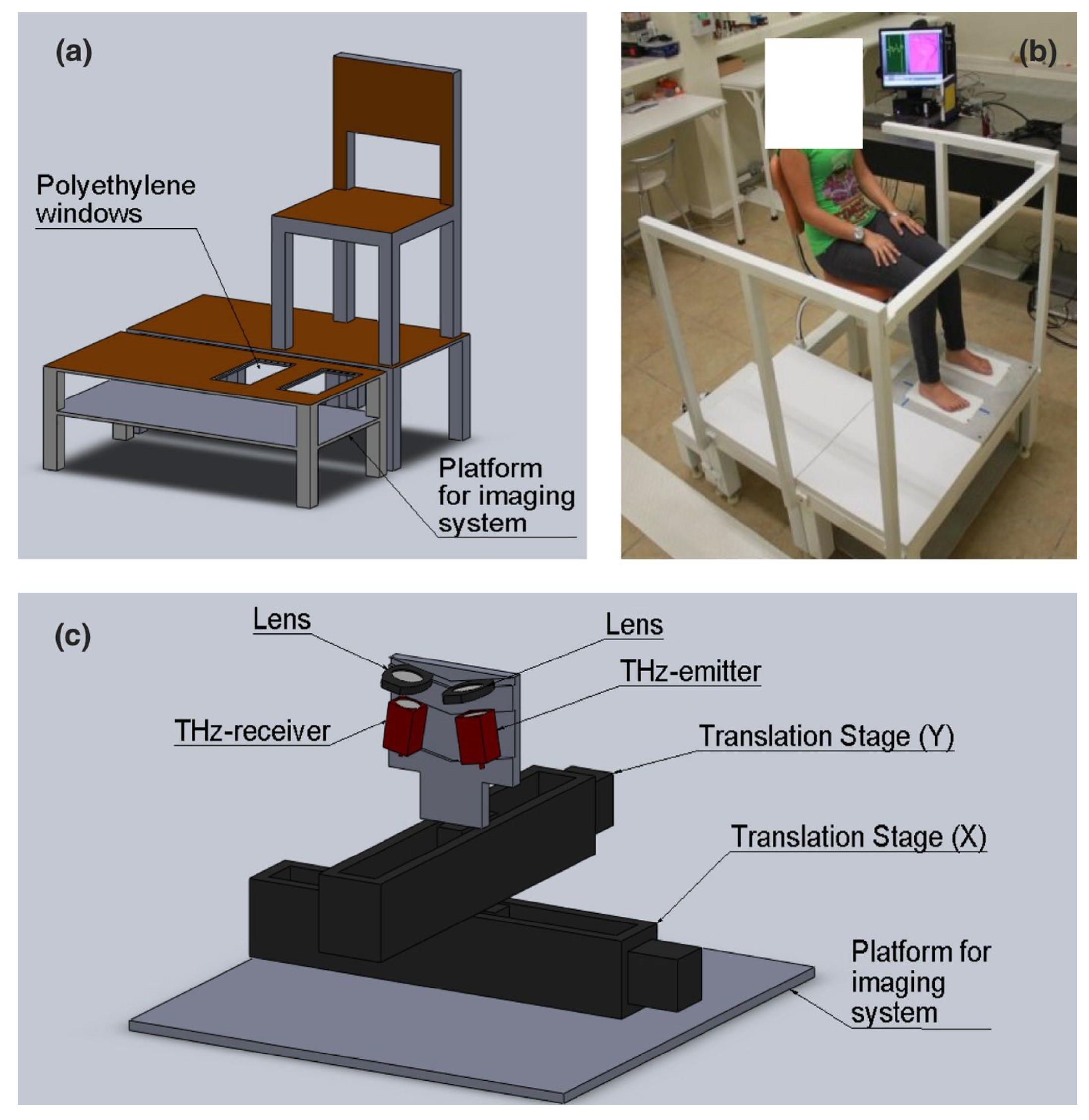
2.2. TPI Equipment
2.2.1. TPI Far-Field Systems
2.2.2. TPI Near-Field Systems
3. Basics of THz Imaging
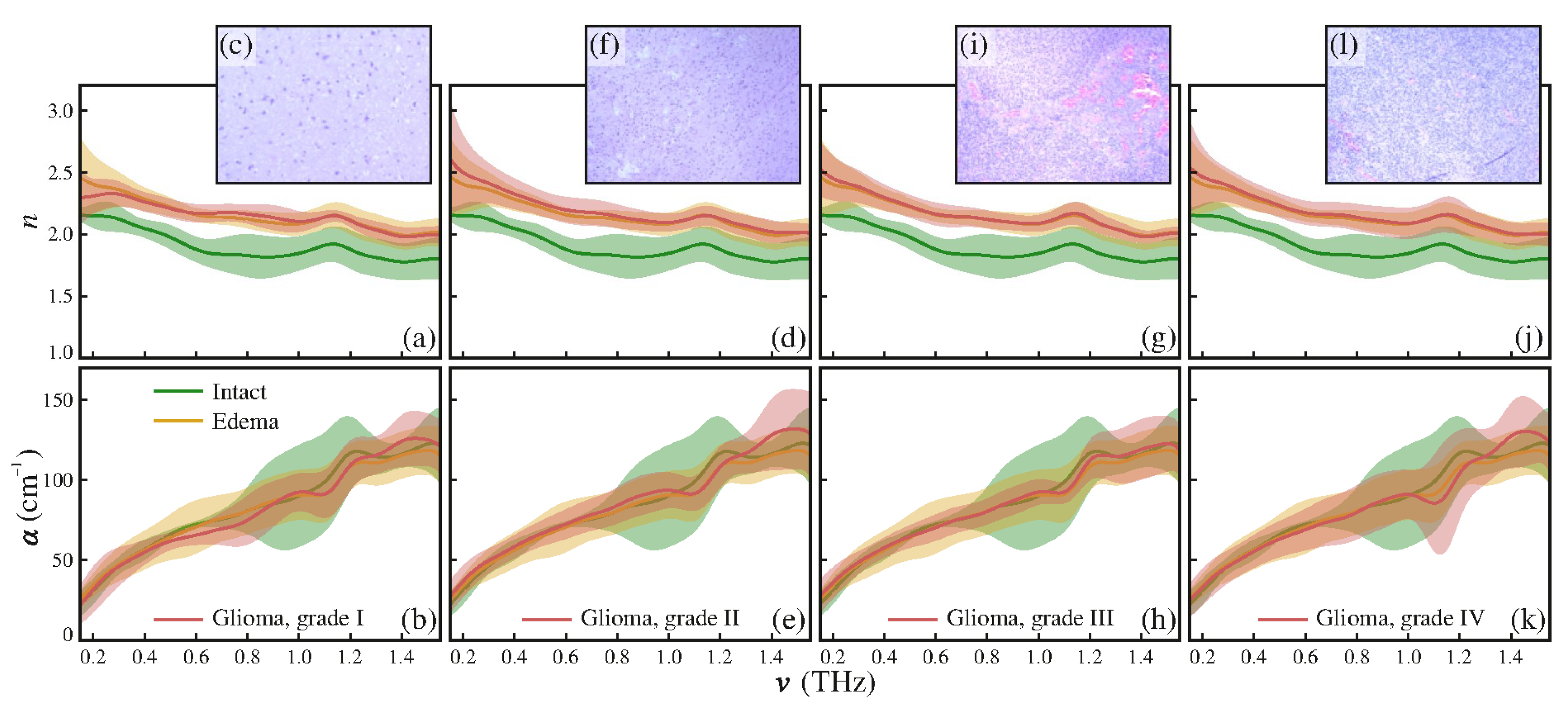
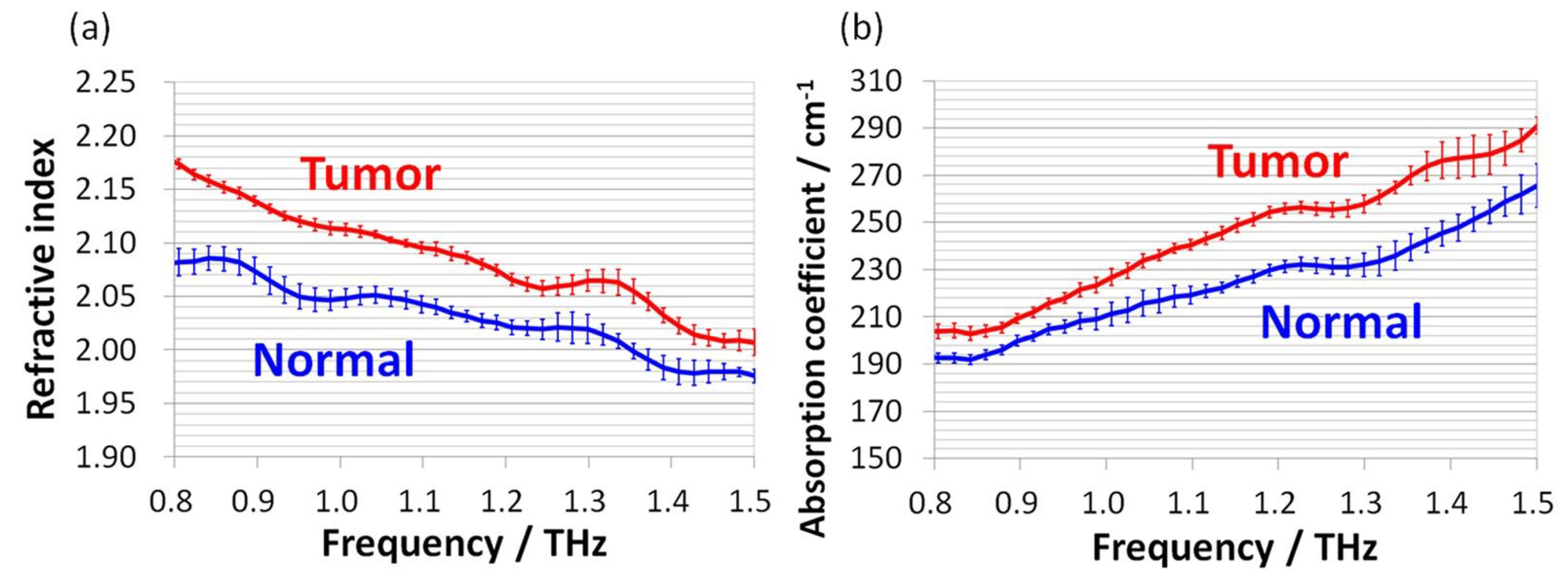
4. THz Pulsed Imaging: Uses, Advantages and Challenges for Biomedical Applications
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Cao, Q.; Zhegalova, N.; Wang, S.; Akers, W.; Berezin, M. Multispectral imaging in the extended near-infrared window based on endogenous chromophores. J. Biomed. Opt. 2013, 18, 101318. [Google Scholar] [CrossRef] [PubMed]
- Wilson, R.; Nadeau, K.; Jaworski, F.; Rowland, R.; Nguyen, J.; Crouzet, C.; Saager, R.; Choi, B.; Tromberg, B.; Durkin, A. Quantitative short-wave infrared multispectral imaging of in vivo tissue optical properties. J. Biomed. Opt. 2014, 19, 086011. [Google Scholar] [CrossRef] [PubMed]
- Bazylewski, P.; Ezugwu, S.; Fanchini, G. A review of Three-Dimensional Scanning Near-Field Optical Microscopy (3D-SNOM) and Its Applications in Nanoscale Ligth Management. Appl. Sci. 2017, 7, 973. [Google Scholar] [CrossRef]
- Weng, Q.; Panchai, V.; Lin, K.T.; Sun, L.; Kajihara, Y.; Tzalenchuk, A.; Komiyama, S. Comparison of active and passive methods for the infrared scanning near-field microscopy. Appl. Phys. Lett. 2019, 114, 153101. [Google Scholar] [CrossRef]
- Jeon, S.; Kim, J.; Lee, D.; Baik, J.W.; Kim, C. Review on pratical photoacoustic microscopy. Photoacoustics 2019, 15, 100141. [Google Scholar] [CrossRef]
- Guo, R.; Lu, G.; Qin, B.; Fe, B. Ultrasound Imaging Technologies for Breast Cancer Detection and Management: A Review. Ultrasound Med. Biol. 2018, 44, 37–70. [Google Scholar] [CrossRef]
- Xu, C.; Carney, P.S.; Boppart, S.A. Wavelength-dependent scattering in spectroscopic optical coherence tomography. Opt. Express 2005, 13, 450–462. [Google Scholar] [CrossRef]
- Xu, C.; Vinegoni, C.; Ralston, T.S.; Luo, W.; Tan, W.; Boppart, S.A. Spectroscopic spectral-domain optical coherence microscopy. Opt. Lett. 2006, 31, 1079–1081. [Google Scholar] [CrossRef]
- Kim, M.K. Principles and techniques of digital holographic microscopy. SPIE Rev. 2010, 1, 018005. [Google Scholar] [CrossRef]
- Marquet, P.; Depeursinge, C.; Magistretti, P.J. Review of quantitative phase-digital holographic microscopy: Promising novel imaging technique to resolve neuronal network activity and identify cellular biomarkers of psychiatric disorders. Neurophotonics 2014, 1, 020901. [Google Scholar] [CrossRef]
- Pitkäaho, T.; Manninen, A.; Naughton, T.J. Focus prediction in digital holographic microscopy using deep convolutional neural networks. Appl. Opt. 2019, 58, A202–A208. [Google Scholar] [CrossRef] [PubMed]
- Jermyn, M.; Mok, K.; Mercier, J.; Desroches, J.; Pichette, J.; Saint-Arnaud, K.; Bernstein, L.; Guiot, M.C.; Petrecca, K.; Leblond, F. Intraoperative brain cancer detection with Raman spectroscopy in humans. Sci. Transl. Med. 2015, 7, 274ra19. [Google Scholar] [CrossRef] [PubMed]
- Min, W.; Freudiger, C.W.; Lu, S.; Xie, X.S. Coherent Nonlinear Optical Imaging: Beyond Fluorescence Microscopy. Annu. Rev. Phys. Chem. 2011, 62, 507–530. [Google Scholar] [CrossRef] [PubMed]
- Zumbusch, A.; Langbein, W.; Borri, P. Nonlinear vibrational microscopy applied to lipid biology. Prog. Lipid. Res. 2013, 52, 615–632. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Zhang, D.; Cheng, J.X. Coherent Raman Scattering Microscopy in Biology and Medicine. Annu. Rev. Biomed. Eng. 2015, 17, 415–445. [Google Scholar] [CrossRef] [PubMed]
- D’Arco, A.; Brancati, N.; Ferrara, M.A.; Indolfi, M.; Frucci, M.; Sirleto, L. Subcellular chemical and morphological analysis by stimulated Raman scattering microscopy and image analysis techniques. BOE 2016, 7, 1853–1864. [Google Scholar]
- Cheng, J.X.; Xie, X.S. Vibrational spectroscopic imaging of living systems: An emerging platform for biology and medicine. Science 2015, 350, aaa8870. [Google Scholar] [CrossRef]
- Lu, F.K.; Calligaris, D.; Olibiyi, O.I.; Norton, I.; Yang, W.; Santagata, S.; Xie, X.S.; Golby, A.J.; Agar, N.Y.R. Label-Free Neurosurgical pathology with Stimulated Raman Imaging. Cancer Res. 2016, 76, OF1–OF12. [Google Scholar] [CrossRef] [PubMed]
- Oheim, M.; Michael, D.J.; Geisbauer, M.; Madsen, D.; Chow, R.H. Principles of two-photon excitation fluorescence microscopy and other nonlinear imaging approaches. Adv. Drug Deliv. Rev. 2006, 58, 788–808. [Google Scholar] [CrossRef]
- Müller, M.; Zumbusch, A. Coherent anti-Stokes Raman Scattering Microscopy. Chem. Phys. Chem. 2007, 8, 2156–2170. [Google Scholar] [CrossRef]
- Li, J.; Chen, Q.; Sun, J.; Zhang, J.; Ding, J.; Zuo, C. Three-dimensional tomographic microscopy technique with multi-frequency combination with partially coherent illuminations. BOE 2018, 9, 2526–2542. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Lin, P.; Tan, Y.; Cheng, J.X. Volumetric stimulated Raman scattering imaging of cleared tissues towards three-dimensional chemical histopathology. BOE 2019, 10, 4329. [Google Scholar] [CrossRef] [PubMed]
- Orringer, D.A.; Pandian, B.; Niknafs, Y.S.; Hollon, T.C.; Boyle, J.; Lewis, S.; Garrard, M.; Hervey-Jumper, S.L.; Garton, H.J.L.; Maher, C.O.; et al. Rapid intraoperative histology of unprocessed surgical specimens via fibre-laser-based stimulated Raman scattering microscopy. Nat. Biomed. Eng. 2017, 1, 0027. [Google Scholar] [CrossRef]
- Denk, W.; Strickler, J.H.; Webb, W.W. Two-photon laser scanning fluorescence microscopy. Science 1990, 248, 73–76. [Google Scholar] [CrossRef] [PubMed]
- Cicchi, R.; Kapsokalyvas, D.; Pavone, F.S. Clinical Nonlinear Laser Imaging of Human Skin: A Review. Biomed. Res. Int. 2014, 2014, 903589. [Google Scholar] [CrossRef] [PubMed]
- Campagnola, P.J.; Loew, L.M. Second-harmonic imaging microscopy for visualizing biomolecular arrays in cells, tissues and organisms. Nat. Biotechnol. 2003, 21, 1356–1360. [Google Scholar] [CrossRef]
- Miller, D.R.; Jarrett, J.W.; Hassan, A.M.; Dunn, A.K. Deep tissue imaging with multiphoton fluorescence microscopy. Curr. Opin. Biomed. Eng. 2017, 4, 32–39. [Google Scholar] [CrossRef]
- Castello, M.; Tortarolo, G.; Buttafava, M.; Deguchi, T.; Villa, F.; Koho, S.; Pesce, L.; Oneto, M.; Pelicci, S.; Lanzanò, L.; et al. A robust and versatile platform for imaging scanning microscopy enabling super-resolution FLIM. Nat. Methods 2019, 16, 175–178. [Google Scholar] [CrossRef]
- Müller, T.; Schumann, C.; Kraegeloh, A. STED Microscopy and its Applications: New Insights into Cellular Processes on the Nanoscale. Chem. Phys. Chem. 2012, 13, 1986–2000. [Google Scholar] [CrossRef]
- Huang, B.; Babcock, H.; Zhuang, X. Breaking the Diffraction Barrier: Super-Resolution Imaging of Cells. Cell 2010, 143, 1047–1058. [Google Scholar] [CrossRef]
- Pietraszewska-Bogiel, A.; Gadella, T.W.J. FRET microscopy: From principle to routine technology in cell biology. J. Microsc. 2011, 241, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Auston, D.H.; Nuss, M.C. Electrooptical generation and detection of femtosecond electrical transients. IEEE J. Quantum Electron. 1988, 24, 184–197. [Google Scholar] [CrossRef]
- Mittleman, D.; Gupta, M.; Neelamani, R.; Baraniuk, R.G.; Rudd, J.V.; Koc, M. Recent advances in terahertz imaging. Appl. Phy. B 1999, 68, 1085–1094. [Google Scholar] [CrossRef]
- Siegel, P.H. Terahertz technology in biology and medicine. IEEE Trans. Microw. Theory 2004, 52, 2438–2447. [Google Scholar] [CrossRef]
- Zhang, X.C. Terahertz wave imaging: Horizons and hurdles. Phys. Med. Biol. 2002, 47, 3667–3677. [Google Scholar] [CrossRef]
- Wallace, V.P.; Taday, P.F.; Fitzgerald, A.J.; Woodward, R.M.; Cluff, J.; Pye, R.J.; Arnone, D.D. Terahertz pulsed imaging and spectroscopy for biomedical and pharmaceutical applications. Faraday Discuss 2004, 126, 255–263. [Google Scholar] [CrossRef]
- Withayachumnankul, W.; Png, G.M.; Yin, X.; Atakaramians, S.; Jones, I.; Lin, H.; Ung, B.S.Y.; Balakrishnan, J.; Ng, B.W.-H.; Ferguson, B.; et al. T-ray sensing and imaging. Proc. IEEE 2007, 95, 1528–1558. [Google Scholar] [CrossRef]
- Mickan, S.; Abbott, D.; Munchb, J.; Zhang, X.C.; van Doornd, T. Analysis of system trade-offs for terahertz imaging. Microelectron. J. 2000, 31, 503–514. [Google Scholar] [CrossRef]
- Yamaguchi, S.; Yamaguchi, S.; Fukushi, Y.; Kubota, O.; Itsuji, T.; Ouchi, T.; Yamamoto, S. Brain tumor imaging of rat fresh tissue using terahertz spectroscopy. Sci. Rep. 2016, 6, 30124. [Google Scholar] [CrossRef]
- Bajwa, N.; Au, J.; Jarrahy, R.; Sung, S.; Fishbein, M.C.; Riopelle, D.; Ennis, D.B.; Aghaloo, T.; John, M.A.; Grundfest, W.S.; et al. Non-invasive terahertz imaging of tissue water content for flap viability assessment. BOE 2017, 8, 460–474. [Google Scholar] [CrossRef]
- Karagoz, B.; Altan, H.; Kamburoglu, K. Terahertz pulsed imaging study of dental caries. In Medical Laser Applications and Laser-tissue Interactions VII. Proceedings of the European Conference on Biomedical Optics, Optical Society of America, Munich, Germany, 21–25 June 2015; Lilge, L., Sroka, R., Eds.; SPIE: Washington, NY, USA, 2015; Volume 9542, 95420N. [Google Scholar]
- Zaytsev, I.; Dolganova, I.N.; Chernomyrdin, N.V.; Katyba, G.M.; Gavdush, A.A.; Cherkasova, O.P.; Komandin, G.A.; Shchedrina, M.A.; Khodan, A.N.; Ponomarev, D.S.; et al. The progress and perspectives of terahertz technology for diagnosis of neoplasms: A review. J. Opt. 2020, 22, 013001. [Google Scholar] [CrossRef]
- Yang, X.; Zhao, X.; Yang, K.; Liu, Y.; Liu, Y.; Fu, W.; Luo, Y. Biomedical Applications of Terahertz Spectroscopy and Imaging. Trends Biotechnol. 2016, 34, 810–824. [Google Scholar] [CrossRef] [PubMed]
- Son, J.H.; Oh, S.J.; Cheon, H. Potential clinical applications of terahertz radiation. J. Appl. Phys. 2019, 125, 190901. [Google Scholar] [CrossRef]
- Leahy-Hoppa, M.R.; Miragliotta, J.; Osiander, R.; Burnett, J.; Dikmelik, Y.; McEnnis, C.; Spicer, J.B. Ultrafast Laser-Based Spectroscopy and Sensing: Applications in LIBS, CARS, and THz Spectroscopy. Sensors 2010, 10, 4342–4372. [Google Scholar] [CrossRef] [PubMed]
- Fischer, B.; Hoffmann, M.; Helm, H.; Modjesch, G.; Uhd Jepsen, P. Chemical recognition in terahertz time-domain spectroscopy and imaging. Semicond. Sci. Technol. 2005, 20, S246. [Google Scholar] [CrossRef]
- D’Arco, A.; Di Fabrizio, M.; Dolci, V.; Marcelli, A.; Petrarca, M.; Della Ventura, G.; Lupi, S. Characterization of volatile organic compounds (VOCs) in their liquid-phase by terahertz time-domain spectroscopy. BOE 2020, 11, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Stoik, C.D.; Bohn, M.J.; Blackshire, J.L. Nondestructive evaluation of aircraft composites using transmissive terahertz time domain spectroscopy. Opt. Express 2008, 16, 17039–17051. [Google Scholar] [CrossRef] [PubMed]
- Heimbeck, M.S.; Ng, W.R.; Golish, D.R.; Gehm, M.E.; Everitt, H.O. Terahertz digital holographic imaging of voids within visibly opaque dielectrics. IEEE Trans. Terahertz Sci. Technol. 2015, 5, 110–116. [Google Scholar] [CrossRef]
- Giuliano, B.M.; Gavdush, A.A.; Müller, B.; Zaytsev, K.I.; Grassi, T.; Ivlev, A.V.; Palumbo, M.E.; Baratta, G.A.; Scirè, C.; Komandin, G.A.; et al. Broadband spectrsocpy of astrophysical ice analogues I. Direct measurement of the complex refractive index of CO ice using terahertz time-domain spectroscopy. Astron. Astrophys. 2019, 629, A112. [Google Scholar] [CrossRef]
- Federici, J.F.; Schulkin, B.; Huang, F.; Gary, D.; Barat, R.; Oliveira, F.; Zimdars, D. THz imaging and sensing for security applications—Explosives, weapons and drugs. Semicond. Sci. Technol. 2005, 20, S266. [Google Scholar] [CrossRef]
- Ergün, S.; Sönmez, S. Terahertz Technology for Military Applications. J. Assoc. Inf. Sci. Technol. 2015, 3, 13–16. [Google Scholar] [CrossRef]
- Liu, H.B.; Zhong, H.; Karpowicz, N.; Chen, Y.; Zhang, X.C. Terahertz spectroscopy and imaging for defense and security applications. Proc. IEEE 2007, 95, 1514–1527. [Google Scholar] [CrossRef]
- Wang, K.; Sun, D.W.; Pu, H. Emerging non-destructive terahertz spectroscopic imaging technique: Principle and applications in the agri-food industry. Trend Food Sci. Technol. 2017, 67, 93–105. [Google Scholar] [CrossRef]
- Cosentino, A. Terahertz and cultural heritage science: Examination of art and archeology. Technologies 2016, 4, 6. [Google Scholar] [CrossRef]
- Lee, Y.S. Principles of Terahertz Science and Technology; Springer: Berlin/Heidelberg, Germany, 2009. [Google Scholar]
- Zhang, X.C.; Xu, J. Introduction to THz Wave Photonics; Springer: Berlin/Heidelberg, Germany, 2010. [Google Scholar]
- Manti, L.; D’Arco, A. Cooperative biological effects between ionizing radiation and other physical and chemical agents. Mutat. Res. 2010, 704, 115–122. [Google Scholar] [CrossRef]
- Titova, L.V.; Rodriguez-Juarez, R.; Woycicki, R.; Hegmann, F.A.; Kovalchuk, O. Intense THz pulses down-regulate genes associated with skin cancer and psoriasis: A new therapeutic avenue? Sci. Rep. 2013, 3, 2363. [Google Scholar] [CrossRef] [PubMed]
- Fëdorov, V.I.; Serdyukov, D.S.; Cherkasova, O.P.; Popova, S.S.; Nemova, E.F. The influence of terahertz radiation on the cell’s genetic apparatus. J. Opt. Technol. 2017, 84, 509–514. [Google Scholar] [CrossRef]
- Naftaly, M. (Ed.) Terahertz Metrology; Artech House: London, UK, 2015. [Google Scholar]
- Chevalier, P.; Amirzhan, A.; Wang, F.; Piccardo, M.; Johnson, S.G.; Capasso, F.; Everitt, H.O. Widely tunable compact terahertz gas lasers. Science 2019, 366, 856–860. [Google Scholar] [CrossRef]
- Otsuji, T. Trends in the research of modern terahertz detectors: Plasmon detectors. IEEE Trans. Terahertz Sci. Technol. 2015, 5, 1110–1120. [Google Scholar]
- Hu, B.B.; Nuss, M.C. Imaging with terahertz waves. Opt. Lett. 1995, 20, 1716–1718. [Google Scholar] [CrossRef]
- Walther, M.; Fischer, B.M.; Ortner, A.; Bitzer, A.; Thoman, A.; Helm, H. Chemical sensing and imaging with pulsed terahertz radiation. Anal. Bioanal. Chem. 2010, 397, 1009–1017. [Google Scholar] [CrossRef] [PubMed]
- Pickwell, E.; Wallace, V.P. Biomedical applications of terahertz technology. J. Phys. D Appl. Phys. 2006, 39, R301–R310. [Google Scholar] [CrossRef]
- Auston, D.H. Picosecond optolectronic switching and gating in silicon. Appl. Phys. Lett. 1975, 26, 101–103. [Google Scholar] [CrossRef]
- Grischkowsky, D.; Keiding, S.; van Exter, M.; Fattinger, C.H. Far-infrared time-domain spectroscopy with terahertz beams of dielectrics and semiconductors. J. Opt. Soc. Am. B 1990, 7, 2006–2015. [Google Scholar] [CrossRef]
- Tani, M.; Herrmann, M.; Sakai, K. Generation and detection of terahertz pulsed radiation with photoconductive antennas and its application to imaging. Meas. Sci. Technol. 2002, 13, 1739–1745. [Google Scholar] [CrossRef]
- Zhang, X.C.; Ma, X.F.; Jin, Y.; Lu, T.M.; Boden, E.P.; Phelp, P.D.; Stewart, K.R.; Yakymyshyn, C.P. Terahertz optical rectification from a nonlinear organic crystal. Appl. Phys. Lett. 1992, 61, 3080–3082. [Google Scholar] [CrossRef]
- Wu, Q.; Zhang, X.C. Terahertz broadband GaP electro-optic sensor. Appl. Phys. Lett. 1997, 70, 1784. [Google Scholar] [CrossRef]
- Winnewisser, C.; Jepsen, P.; Schall, M.; Schyja, V.; Helm, H. Electro-optic detection of THz radiation in LiaTO3, LiNbO3 and ZnTe. Appl. Phys. Lett. 1997, 70, 3069. [Google Scholar] [CrossRef]
- Kim, K.Y.; Taylor, A.J.; Glownia, J.H.; Rodriguez, G. Coherent control of terahertz supercontinuum generation in ultrafast laser-gas interactions. Nat. Photonics 2008, 2, 605–609. [Google Scholar] [CrossRef]
- Thomson, M.D.; Blank, V.; Roskos, H.G. Terahertz white-ligth pulses from an air plasma photo-induced by incommensurate two-color optical fields. Opt. Express 2010, 18, 23173–23182. [Google Scholar] [CrossRef]
- Dai, J.; Liu, J.; Zhang, X.C. Terahertz wave air photonics: Terahertz wave generation and detection with laser-induced gas plasma. IEEE J. Sel. Top. Quantum Electron. 2011, 17, 183–190. [Google Scholar] [CrossRef]
- Planken, P.C.M.; Nuss, M.C.; Knox, W.H.; Miller, D.A.B.; Goossen, K.W. THz pulses from the creation of polarized electron-hole pairs in biased quantum wells. Appl. Phys. Lett. 1992, 61, 2009–2011. [Google Scholar] [CrossRef]
- Sun, G.; Xu, G.; Ding, Y.J.; Zhao, H.; Liu, G.; Zhang, J.; Tansu, N. Efficient Terahertz Generation Within InGaN/GaN Multiple Quantum Wells. IEEE J. Sel. Top. Quantum Electron. 2011, 17, 48–53. [Google Scholar] [CrossRef]
- Roskos, H.G.; Nuss, M.C.; Shah, J.; Leo, K.D.; Miller, A.; Fox, A.M.; Schmitt-Rink, S.; Köhler, K. Coherent submillimeter-wave emission from change oscillations in a double-well potential. Appl. Phys. Lett. 1992, 68, 2216–2219. [Google Scholar] [CrossRef] [PubMed]
- Huber, R.; Brodshelm, A.; Tauser, F.; Leitenstorfer, A. Generation and field-resolved detection of femtosecond electromagnetic pulses tunable up to 41 THz. Appl. Phys. Lett. 2000, 76, 3191–3193. [Google Scholar] [CrossRef]
- Kono, S.; Tani, M.; Sakai, K. Ultrabroadband Photoconductive Detection: Comparison with Free-Space Electro-Optic Sampling. Appl. Phys. Lett. 2001, 79, 898–900. [Google Scholar] [CrossRef]
- Houver, S.; Huber, L.; Savoini, M.; Abreu, E.; Johnson, S.L. 2D THz spectroscopic investigation of ballistic conduction-band electron dynamics in InSb. Opt. Express 2019, 27, 10854. [Google Scholar] [CrossRef]
- Curcio, A.; Dolci, V.; Lupi, S.; Petrarca, M. Terahertz-based retrieval of the spectral phase and amplitude of ultrashort laser pulses. Opt. Lett. 2018, 43, 783. [Google Scholar] [CrossRef]
- Liu, B.; Bromberger, H.; Cartella, A.; Gebert, T.; Först, M.; Cavalleri, A. Generation of narrowband, high-intensity,carrier-envelope phase-stable pulses tunable between 4 and 18 THz. Opt. Lett. 2017, 42, 129. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, X.; Li, S.; Gu, J.; Li, Y.; Tian, Z.; Ouyang, C.; He, M.; Hanand, J.; Zhang, W. Broadband THz-TDS System based on DSTMS emitter and LTGInGaAs/InAlAs Photoconductive Antenna Detector. Sci. Rep. 2016, 6, 26949. [Google Scholar] [CrossRef]
- Jazbinšek, M.; Puc, U.; Abina, A.; Zidansek, A. Organic crystals for THz photonics. Appl. Sci. 2019, 9, 882. [Google Scholar] [CrossRef]
- Hebling, J.; Yeh, K.L.; Hoffmann, M.C.; Bartal, B.; Nelson, K.A. Generation of High-Power Terahertz Pulses by Tilted-Pulse-Front Excitation and Their Application Possibilities. J. Opt. Soc. Am. B 2008, 25, B6–B19. [Google Scholar] [CrossRef]
- Hamster, H.; Sullivan, A.; Gordon, S.; White, W.; Falcone, R.W. Subpicosecond Electromagnetic Pulses from Intense Laser-Plasma Interaction. Phys. Rev. Lett. 1993, 71, 2725. [Google Scholar] [CrossRef] [PubMed]
- Hamster, H.; Sullivan, A.; Gordon, S.; Falcone, R.W. Short-Pulse Terahertz Radiation from High-Intensity-Laser- Produced Plasmas. Phys. Rev. E 1994, 49, 671. [Google Scholar] [CrossRef]
- Löffler, T.; Jacob, F.; Roskos, H.G. Generation of terahertz pulses by photoionization of electrically biased air. Appl. Phys. Lett. 2000, 77, 453–455. [Google Scholar] [CrossRef]
- Cook, D.J.; Hochstrasser, R.M. Intense terahertz pulses by four-wave rectification in air. Opt. Lett. 2000, 25, 1210–1212. [Google Scholar] [CrossRef]
- Schmuttenmaer, C.A. Exploring dynamics in the far-infrared with terahertz spectroscopy. Chem. Rev. 2004, 104, 1759–1780. [Google Scholar] [CrossRef]
- Greene, B.I.; Federici, J.F.; Dykaar, D.R.; Levi, A.F.J.; Pfeiffer, L. Picosecond pump and probe spectroscopy utilizing freely propagating terahertz radiation. Opt. Lett. 1991, 16, 48–49. [Google Scholar] [CrossRef]
- Gouider, F.; Vasilyev, Y.B.; Bugár, M.; Könemann, J.; Buckle, P.D.; Nachtwei, G. Terahertz photoresponse of AlInSb/InSb/AlInSb quantum well structures. Phys. Rev. B 2010, 81, 155304. [Google Scholar] [CrossRef]
- Oda, N. Uncooled bolometer-type terahertz focal plane array and camera for real-time imaging. C. R. Phys. 2010, 11, 496–509. [Google Scholar] [CrossRef]
- Dean, P.; Shaukat, M.U.; Khanna, S.P.; Lachab, M.; Burnett, A.; Davies, A.G.; Linfield, E.H.; Chakraborty, S. Absorption–sensitive diffuse reflection imaging of concealed powders using a terahertz quantum cascade laser. Opt. Express 2008, 16, 5997–6007. [Google Scholar] [CrossRef] [PubMed]
- Golay, M.J.E. The theoretical and pratical sensitivity of the pneumatic infrared detector. Rev. Sci. Instrum. 1949, 20, 816–820. [Google Scholar] [CrossRef] [PubMed]
- Karpowicz, N.; Zhong, H.; Xu, J.; Lin, K.I.; Hwang, J.S.; Zhang, X.C. Comparison between pulsed terahertz time-domain imaging and continuous wave terahertz imaging. Semicond. Sci. Technol. 2005, 20, S293–S299. [Google Scholar] [CrossRef]
- Cox, J.A.; Higashi, R.; Nusseibeh, F.; Newstrom-Peitso, K.; Zins, C. Uncooled MEMS-based detector arrays for THz imaging applications. In Terahertz Physics, Devices, and Systems III: Advanced Applications in Industry and Defense, Proceedings of the SPIE Defense, Security, and Sensing, Orlando, FL, USA, 13–17 April 2009; SPIE: Washington, NY, USA; Volume 7311, 73110R.
- Kašalynas, I.; Adam, A.J.L.; Klaassen, O.; Hovenier, N.J.; Pandraud, G.; Iordanov, V.P.; Sarro, P.M. Some properties of a room temperature THz detection array. In Advanced Optical Materials, Technologies, and Devices, Proceedings of the SPIE Advanced Optical Materials, Technologies, and Devices, Vilnius, Lithuania, 27–30 August 2006; SPIE: Washington, NY, USA; Volume 6596, 65960J.
- Semenov, A.; Cojocari, O.; Hübers, H.-W.; Song, F.; Klushin, A.; Müller, A.-S. Application of Zero-Bias Optical Schottky-Diode Detectors for Monitoring Short-Pulse and Weak Terahertz Radiation. IEEE Electron Device Lett. 2010, 31, 674–676. [Google Scholar] [CrossRef]
- Maestrini, A.; Thomas, B.; Wang, H.; Jung, C.; Treuttel, J.; Jin, Y.; Chattopadhyay, G.; Mehdi, I.; Beaudin, G. Schottky diode-based terahertz frequency multipliers and mixers. C. R. Phys. 2010, 11, 480–495. [Google Scholar] [CrossRef]
- Nazarov, M.M.; Makarova, S.A.; Shkurinov, A.P.; Okhotnikov, O.G. The use of combination of nonlinear optical materials to control terahertz pulse generation and detection. Appl. Phys. Lett. 2008, 92, 021114–021117. [Google Scholar] [CrossRef]
- Zhang, Y.; Hosono, S.; Nagai, N.; Song, S.-H.; Hirakawa, K. Fast and sensitive bolometric terahertz detection at room temperature through thermomechanical transduction. J. Appl. Phys. 2019, 125, 151602. [Google Scholar] [CrossRef]
- Rezvani, J.; Di Gioacchino, D.; Gatti, C.; Poccia, N.; Ligi, C.; Tocci, S.; Cestelli Guidi, M.; Cibella, S.; Lupi, S.; Marcelli, A. Tunable Vortex Dynamics in Proximity Junction Arrays: A Possible Accurate and Sensitive 2D THz Detector. In Proceedings of the LIV Zakopane School of Physics, Breaking Frontiers, Zakopane, Poland, 21–25 May 2019; Volume 137, pp. 17–20. [Google Scholar]
- Knap, W.; Dyakonov, M.I. Field effect transistors for terahertz applications. In Handbook of Terahertz Technologiey for Imaging, Sensing and Communications; Volume in Woodhead Publishing Series in Electronics and Optical Materials; Saeedkia, D., Ed.; Elsevier: Amsterdam, The Nederland, 2013; pp. 121–155. [Google Scholar]
- Fan, K.; Suen, J.Y.; Liu, X.; Padilla, W.J. All-dielectric metasurface absorbers for uncooled terahertz imaging. Optica 2017, 4, 601–604. [Google Scholar] [CrossRef]
- Guerboukha, H.; Nallappan, K.; Skorobogatiy, M. Toward real-time terahertz imaging. Adv. Opt. Phot. 2018, 10, 843–938. [Google Scholar] [CrossRef]
- Cho, H.; Lee, S.H.; Nam-Gung, C.; Oh, S.J.; Oh, J.H.; Park, H.; Ahn, C.B. Fast terahertz reflection tomography using block-based compressed sensing. Opt. Express 2011, 19, 16401–16409. [Google Scholar] [CrossRef]
- Hwang, B.M.; Lee, S.H.; Lim, W.T.; Ahn, C.B.; Son, J.H.; Park, H. A fast spatial-domain terahertz imaging using block-based compressed sensing. J. Infrared Millim. Terahertz Waves 2011, 32, 1328–1336. [Google Scholar] [CrossRef]
- Hong, H.J.; Park, J.; Park, H.; Son, J.H.; Ahn, C.B. Pre- and post-processing for tomographic reconstruction of terahertz time-domain spectroscopy. Opt. Express 2013, 21, 19943–19950. [Google Scholar] [CrossRef] [PubMed]
- Wallace, V.P.; Macpherson, E.; Zeitler, J.A.; Reid, C. Three-dimensional imaging of optically opaque materials using nonionizing terahertz radiation. J. Opt. Soc. Am. Opt. Image Sci. Vis. 2008, 25, 3120–3133. [Google Scholar] [CrossRef] [PubMed]
- Castro-Camus, E.; Alfaro, M. Photoconductive devices for terahertz pulsed spectroscopy: A review. Photon. Res. 2016, 4, A36. [Google Scholar] [CrossRef]
- Chan, W.L.; Deibel, J.; Mittleman, D.M. Imaging with terahertz radiation. Rep. Prog. Phys. 2007, 70, 1325–1379. [Google Scholar] [CrossRef]
- Adam, A.J.L. Review of Near-Field Terahertz Measurement Methods and Their Applications. J. Infrared Millim. Terahertz Waves 2011, 32, 976–1019. [Google Scholar] [CrossRef]
- Goodman, J.W. Introduction to Fourier Optics, 2nd ed.; McGraw-Hill: New York, NY, USA, 1996. [Google Scholar]
- Abbe, E. Beiträge zur theroie des mikroskops und der mikroskopischen wahrnehmung. Arch. Mikrosk. Anat. 1873, 9, 413–468. [Google Scholar] [CrossRef]
- Born, M.; Wolf, E. Principles of Optics, 7th ed.; Cambridge University Press: Cambridge, UK, 1999. [Google Scholar]
- Pawley, J.B. Handbook of Biological Confocal Microscopy; Springer: Berlin/Heidelberg, Germany, 2006. [Google Scholar]
- Yuan, T.; Xu, Z.; Zhang, X.C. Development of terahertz wave microscopes. Infrared Phys. Technol. 2004, 45, 417–425. [Google Scholar] [CrossRef]
- Blanchard, F.; Doi, A.; Tanaka, T.; Tanaka, K. Real-time, subwavelength terahertz imaging. Annu. Rev. Mater. Res. 2013, 43, 237–259. [Google Scholar] [CrossRef]
- Hunsche, S.; Koch, M.; Brener, I.; Nuss, M.C. THz near-field imaging. Opt. Commun. 1998, 150, 22–26. [Google Scholar] [CrossRef]
- Flammini, M.; Bonsi, C.; Ciano, C.; Giliberti, V.; Pontecorvo, E.; Italia, P.; DelRe, E.; Ortolani, M. Confocal terahertz imaging of ancient manuscripts. J. Infrared Millim. Terahertz Waves 2017, 38, 435–442. [Google Scholar] [CrossRef]
- Salhi, M.A.; Pupeza, I.; Koch, M. Confocal THz laser microscope. J. Infrared Millm. Terahertz Waves 2010, 31, 358–366. [Google Scholar] [CrossRef]
- Saeedkia, D. Handbook of Terahertz Technology for Imaging, Sensing and Communications; Volume in Woodhead Publishing Series in Electronics and Optical Materials; Saeedkia, D., Ed.; Elsevier: Amsterdam, The Nederland, 2013. [Google Scholar]
- Adam, A.J.; Brok, J.M.; Seo, M.A.; Ahn, K.J.; Kim, D.S.; Kang, J.H.; Park, Q.H.; Nagel, M.; Planken, P.C. Advanced terahertz electric near-field measurements at sub-wavelength diameter metallic apertures. Opt. Express 2008, 16, 7407–7417. [Google Scholar] [CrossRef] [PubMed]
- Mitrofanov, O.; Brener, I.; Harel, R.; Wynn, J.D.; Pfeiffer, L.N.; West, K.W.; Federici, J. Terahertz near-field microscopy based on a collection mode detector. Appl. Phys. Lett. 2000, 77, 3496. [Google Scholar] [CrossRef]
- Iwami, K.; Ono, T.; Esashi, M. A New Approach to Terahertz Local Spectroscopy Using Microfabricated Scanning Near-Field Probe. Jpn. J. Appl. Phys. 2008, 47, 8095–8097. [Google Scholar] [CrossRef]
- Cao, H.; Agrawal, A.; Nahata, A. Controlling the transmission resonance lineshape of a single subwavelength aperture. Opt. Express 2005, 13, 763–769. [Google Scholar] [CrossRef]
- Keilmann, F.; Hillenbrand, R. Near-field microscopy by elastic light scattering from a tip. Philos. Trans. Math. Phys. Eng. Sci. 2004, 362, 787–805. [Google Scholar] [CrossRef]
- Adam, A.J.L.; Brok, J.M.; Planken, P.C.M.; Seo, M.A.; Kim, D.S. THz near-field measurements of metal structure. C. R. Phys. 2008, 9, 161–168. [Google Scholar] [CrossRef]
- Löffler, T.; Siebert, K.; Czasch, S.; Bauer, T.; Roskos, H.G. Visualization and classification in biomedical terahertz pulsed imaging. Phys. Med. Biol. 2002, 47, 3847–3852. [Google Scholar] [CrossRef]
- Fitzgerald, A.J.; Wallace, V.P.; Pinder, S.E.; Purushotham, A.D.; O’Kelly, P.; Ashworth, P.C. Classification of terahertz-pulsed imaging data from excised breast tissue. J. Biomed. Opt. 2012, 17, 016005. [Google Scholar] [CrossRef]
- Ushakov, A.; Chizhov, P.; Bukin, V.; Savel’ev, A.; Garnov, S. Broadband in-line terahertz 2D imaging: Comparative study with time-of-ligth, cross-correlation, and Fourier transform data processing. J. Opt. Soc. Am. B 2018, 35, 1159–1164. [Google Scholar] [CrossRef]
- Wan, M.; Healy, J.J.; Sheridan, J.T. Terahertz phase imaging and biomedical applications. Opt. Laser Technol. 2020, 122, 105859. [Google Scholar] [CrossRef]
- Dressel, M.; Grüner, G. Electrodynamics of Solids; Cambridge University Press: Cambridge, UK, 2002. [Google Scholar]
- Kužel, P.; Němec, H.; Kadlec, F. Gouy shift correction for highly accurate refractive index retrieval in time-domain terahertz spectroscopy. Opt. Express 2010, 18, 15338–15348. [Google Scholar] [CrossRef] [PubMed]
- Hegmann, F.A.; Ostroverkhova, O.; Cooke, D.G. Probing organic semiconductors with terahertz pulses. In Photophysics of Molecular Materials; Wiley: Hoboken, NJ, USA, 2000; pp. 367–428. [Google Scholar]
- Llioyd-Hughes, J.; Jeon, T.I. A review of the terahertz conductivity of bulk and nano-materials. J. Infrared Millim. Terahertz Waves 2012, 33, 871–925. [Google Scholar] [CrossRef]
- Guillet, J.P.; Recur, B.; Frederique, L.; Bousquet, B.; Canioni, L.; Manek-Hönninger, I.; Desbarats, P.; Mounaix, P. Review of Terahertz Tomography Techniques. J. Infrared. Millim. Terahertz Waves 2014, 35, 382–411. [Google Scholar] [CrossRef]
- Mittleman, D.M. Twenty years of terahertz imaging. Opt. Express 2018, 26, 9417–9431. [Google Scholar] [CrossRef]
- Ferguson, B.; Wang, S.; Gray, D.; Abbot, D.; Zhang, X.-C. T-ray computed tomography. Opt. Lett. 2002, 27, 1312–1314. [Google Scholar] [CrossRef]
- Thrane, L.; Jacobsen, R.H.; Jepsen, P.U.; Keiding, S.R. THz reflection spectroscopy of liquid water. Chem. Phys. Lett. 1995, 240, 330–333. [Google Scholar] [CrossRef]
- Cheon, H.; Yang, H.J.; Son, J.-H. Toward Clinical Cancer Imaging Using Terahertz Spectroscopy. IEEE J. Sel. Top. Quantum Electron. 2017, 23, 8600109. [Google Scholar] [CrossRef]
- Zaytsev, K.I.; Gavdush, A.; Chernomyrdin, N.; Yurchenko, S. Highly accurate in vivo terahertz spectroscopy of healthy skin: Variation of refractive index and absorption coefficient along the human body. IEEE Trans. Terahertz Sci. Technol. 2015, 5, 817–827. [Google Scholar] [CrossRef]
- Parrott, E.; Sy, S.; Blu, T.; Wallace, V.; Pickwell-MacPherson, E. Terahertz pulsed imaging in vivo: Measurements and processing methods. J. Biomed. Opt. 2011, 16, 106010. [Google Scholar] [CrossRef] [PubMed]
- Kolesniko, A.; Kolesnikova, E.; Popov, A.; Nazarov, M.; Shkurinov, A.; Tuchin, V. In vitro terahertz monitoring of muscle tissue dehydration under the action of hyperosmotic agents. Quantum Electron. 2014, 44, 633. [Google Scholar] [CrossRef]
- Smolyanskaya, O.; Schelkanova, I.; Kulya, M.; Odlyanitskiy, E.; Goryachev, I.; Tcypkin, A.; Grachev, Y.; Toropova, Y.; Tuchin, V. Glycerol dehydration of native and diabetic animal tissues studied by THz-TDS and NMR methods. BOE 2018, 9, 1198–1215. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, A.J.; Berry, E.; Zinov’ev, N.N.; Homer-Vanniasinkam, S.; Miles, R.E.; Chamberlain, J.M.; Smith, M.A. Catalogue of human tissue optical properties at terahertz frequencies. J. Biol. Phys. 2003, 29, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Kan, W.C.; Lee, W.S.; Cheung, W.H.; Wallace, V.P.; Pickwell-Macpherson, E. Terahertz pulsed imaging of knee cartilage. BOE 2010, 1, 967–974. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sun, Y.; Fischer, B.; Pickwell-MacPherson, E. Effects of formalin fixing on the terahertz properties of biological tissues. J. Biomed. Opt. 2009, 14, 064017. [Google Scholar] [CrossRef]
- Fan, S.; Ung, B.; Parrott, E.P.J.; Pickwell-MacPherson, E. Gelatin embedding: A novel way to preserve biological samples for terahertz imaging and spectroscopy. Phys. Med. Biol. 2015, 60, 2703–2713. [Google Scholar] [CrossRef]
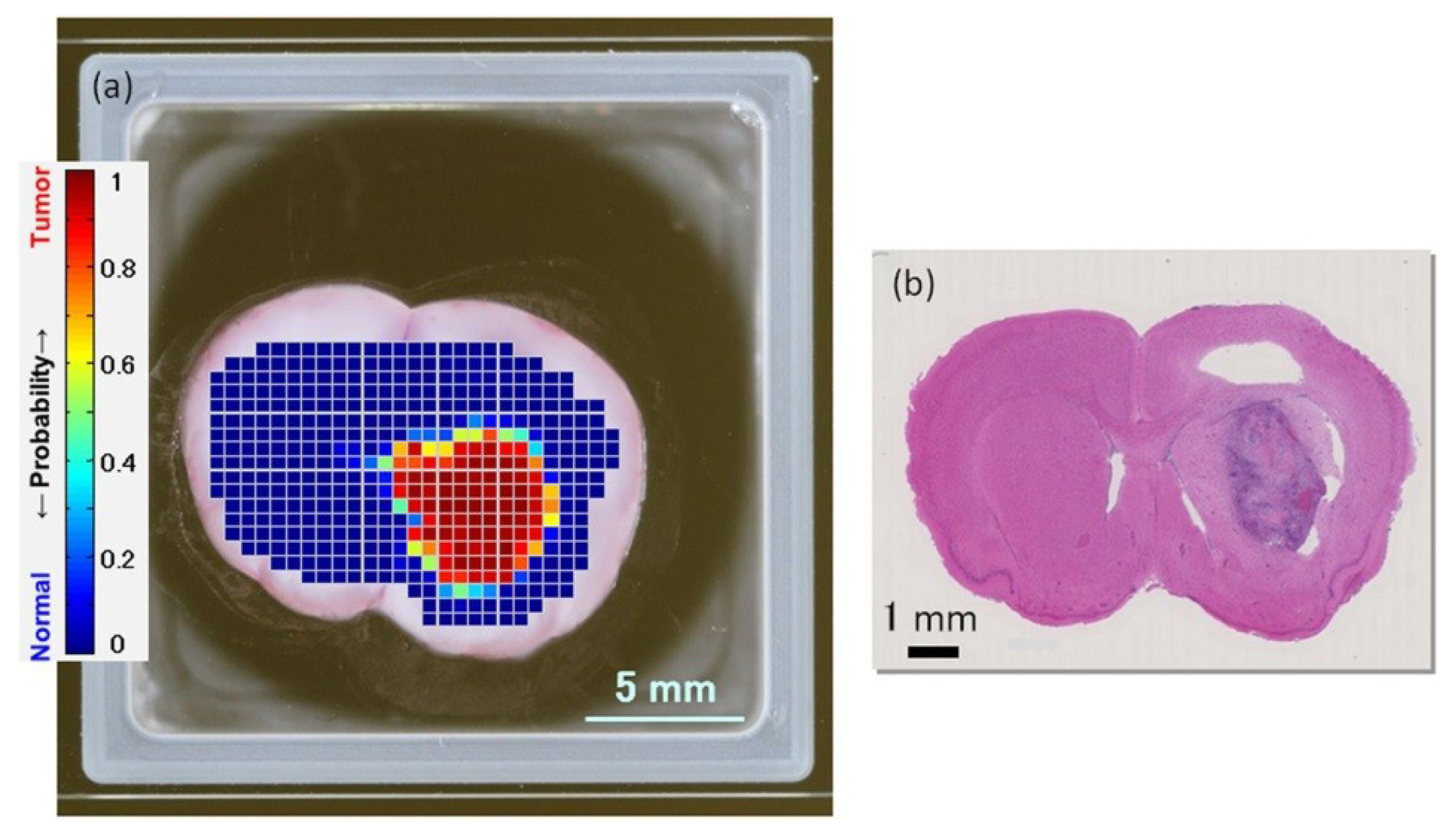
- Gavdush, A.A.; Chernomyrdin, N.V.; Malakhov, K.M.; Beshplav, S.-I.T.; Dolganova, I.N.; Kosyrkova, A.V.; Nikitin, P.V.; Musina, G.R.; Katyba, G.M.; Reshetov, I.V.; et al. Terahertz spectroscopy of gelatin-embedded human brain gliomas of different grades: A road toward intraoperative THz diagnosis. J. Biomed. Opt. 2019, 24, 027001. [Google Scholar] [CrossRef]
- Png, G.M.; Choi, J.W.; Ng, B.W.; Mickan, S.P.; Abbott, D.; Zhang, X.C. The impact of hydration changes in fresh bio-tissue on THz spectroscopic measurements. Phys. Med. Biol. 2008, 53, 3501. [Google Scholar] [CrossRef]
- Sim, Y.; Park, J.; Ahn, K.-M.; Park, C.; Son, J.-H. Terahertz imaging of excised oral cancer at frozen temperature. BOE 2013, 4, 1413–1421. [Google Scholar] [CrossRef]
- He, Y.; Ung, B.-Y.; Parrott, E.; Ahuja, A.; Pickwell-MacPherson, E. Freeze-thaw hysteresis effects in terahertz imaging of biomedical tissues. BOE 2016, 7, 4711–4717. [Google Scholar] [CrossRef] [PubMed]
- Meng, K.; Chen, T.N.; Chen, T.; Zhu, T.G.; Liu, Q.; Li, Z.; Li, F.; Zhong, S.-C.; Li, Z.-R.; Feng, H.; et al. Terahertz pulsed spectroscopy of paraffin embedded brain glioma. J. Biomed. Opt. 2014, 19, 077001. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.J.; Kim, S.H.; Jeong, K.; Park, Y.; Huh, Y.M.; Son, J.H.; Suh, J.S. Measurement depth enhancement in terahertz imaging of biological tissues. Opt. Express 2013, 21, 21299–21305. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Notake, T.; Tang, M.; Nawata, K.; Ito, H.; Minamide, H. Terahertz-wave water concentration and distribution measurement in thin biotissue based on a novel sample preparation. Phys. Med. Biol. 2011, 56, 4517. [Google Scholar] [CrossRef] [PubMed]
- Wallace, V.P.; Fitzgerald, A.J.; Pickwell, E.; Pye, R.J.; Taday, P.F.; Flanagan, N.; Ha, T. Terahertz pulsed spectroscopy of human basal cell carcinoma. Appl. Spectrosc. 2006, 60, 1127–1133. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Sy, M.Y.; Wang, Y.X.J.; Ahuja, A.T.; Zhang, Y.T.; Pickwell-MacPherson, E. A promising diagnostic method: Terahertz pulsed imaging and spectroscopy. World J. Radiol. 2011, 3, 55–65. [Google Scholar] [CrossRef]
- Yeo, W.G.; Gurel, O.; Hitchcock, C.L.; Park, S.; Sertel, K.; Nahar, N.K. Evaluation of cancer tissue morphology via THz spectroscopic imaging: Human lung and small intestine malignancies. Infrared Phys. Technol. 2019, 97, 411–416. [Google Scholar] [CrossRef]
- Wahaia, F.; Kasălynas, I.; Minkevičius, L.; Carvalho Silva, C.D.; Urbanowicz, A.; Valušis, G. Terahertz spectroscopy and imaging for gastic cancer diagnosis. J. Spectr. Imaging 2020, 9, 1–8. [Google Scholar]
- El-Shenawee, M.; Vohra, N.; Bowman, T.; Bailey, K. Cancer detection in excised breast tumors using terahertz imaging and spectroscopy. Biomed. Spectrosc. Imaging 2019, 8, 1–9. [Google Scholar] [CrossRef]
- Cao, Y.; Huang, P.; Chen, J.; Ge, W.; Hou, D.; Zhang, G. Qualitative and quantitative detection of liver injury with terahertz time-domain spectroscopy. BOE 2020, 11, 982–993. [Google Scholar] [CrossRef]
- Cao, Y.; Chen, J.; Huang, P.; Ge, W.; Hou, D.; Zhang, G. Inspecting human colon adenocarcinoma cell lines by using terahertz time-domain reflection spectroscopy. Spectrochim. Acta 2019, 211, 356–363. [Google Scholar] [CrossRef] [PubMed]
- Kuzikova, A.V.; Grigorev, R.O.; Kurasova, A.P.; Demchenko, P.S.; Senyuk, A.V.; Zakharenko, A.A.; Belolipetskaya, J.R.; Khamid, A.H.; Khodzitsky, M.K. Study of refractive index of human stomach cancer tissue in THz frequency range. In Proceedings of the 6th International School and Conference “Saint Petersburg OPEN 2019”: Optoelectronics, Photonics, Engineering and Nanostructure, Saint Petersburg, Russia, 22–25 April 2019; Volume 1410. 012070. [Google Scholar]
- Globus, T.; Moskaluk, C.; Pramoonjago, P.; Gelmont, B.; Moyer, A.; Bykhovski, A.; Ferrance, J. Sub-terahertz vibrational spectroscopy of ovarian cancer and normal control tissue for molecular diagnostic technology. Cancer Biomark. 2019, 24, 405–419. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, S.; Fukushi, Y.; Kubota, O.; Itsuji, T.; Ouchi, T.; Yamamoto, S. Origin and quantification of differences between normal and tumor tissues observed by terahertz spectroscopy. Phys. Med. Biol. 2016, 68, 6808–6820. [Google Scholar] [CrossRef] [PubMed]
- Chau, D.Y.S.; Dennis, A.R.; Lin, H.; Zeitler, J.A.; Tunnacliffe, A. Determination of Water Content in Dehydrated Mammalian Cells Using Terahertz Pulsed Imaging: A Feasibility Study. Curr. Pharm. Biotechnol. 2015, 17, 200–207. [Google Scholar] [CrossRef]
- Smolyanskaya, O.A.; Chernomyrdin, N.V.; Konovko, A.A.; Zaytsev, K.I.; Ozheredov, I.A.; Cherkasova, O.P.; Nazarov, M.M.; Guillet, J.P.; Kozlov, S.A.; Kistenev, Y.V.; et al. Terahertz biophotonics as a tool for studies of dielectric and spectral properties of biological tissues and liquids. Prog. Quantum Electron. 2018, 62, 1–77. [Google Scholar] [CrossRef]
- Brun, M.A.; Formanek, F.; Yasuda, A.; Sekine, M.; Ando, N.; Eishii, Y. Terahertz imaging applied to cancer diagnosis. Phys. Med. Biol. 2010, 55, 4615–4623. [Google Scholar] [CrossRef]
- Chen, H.; Chen, T.H.; Tseng, T.F.; Lu, J.T.; Kuo, C.C.; Fu, S.C.; Lee, W.J.; Tsai, Y.F.; Huang, Y.Y.; Chuang, E.Y.; et al. High-sensitivity in vivo THz transmission imaging of early human breast cancer in a subcutaneous xenograft mouse model. Opt. Express 2011, 19, 21552–21562. [Google Scholar] [CrossRef]
- Miura, Y.; Kamataki, A.; Uzuki, M.; Sasaki, T.; Nishizawa, J.I.; Sawai, T. Terahertz-wave spectroscopy for precise histopathological imaging of tumor and non-tumor lesions in paraffin sections. J. Exp. Med. 2011, 223, 291–296. [Google Scholar] [CrossRef]
- Knobloch, P.; Schildknecht, C.; Kleine-Ostmann, T.; Koch, M.; Hoffmann, S.; Hofmann, M.; Rehberg, E.; Sperling, M.; Donhuijsen, K.; Hein, G.; et al. Medical THz imaging: An investigation of histo-pathological samples. Phys. Med. Biol. 2002, 47, 3875. [Google Scholar] [CrossRef]
- Doradla, P.; Alavi, K.; Joseph, C.; Giles, R. Single-channel prototype terahertz endoscopic system. J. Biomed. Opt. 2014, 19, 080501. [Google Scholar] [CrossRef]
- Reid, C.B.; Fitzgerald, A.; Reese, G.; Goldin, R.; Tekkis, P.; O’Kelly, P.S.; Pickwell-MacPherson, E.; Gibson, A.P.; Wallace, V.P. Terahertz pulsed imaging of freshly excised human colonic tissues. Phys. Med. Biol. 2011, 56, 4333–4353. [Google Scholar] [CrossRef] [PubMed]
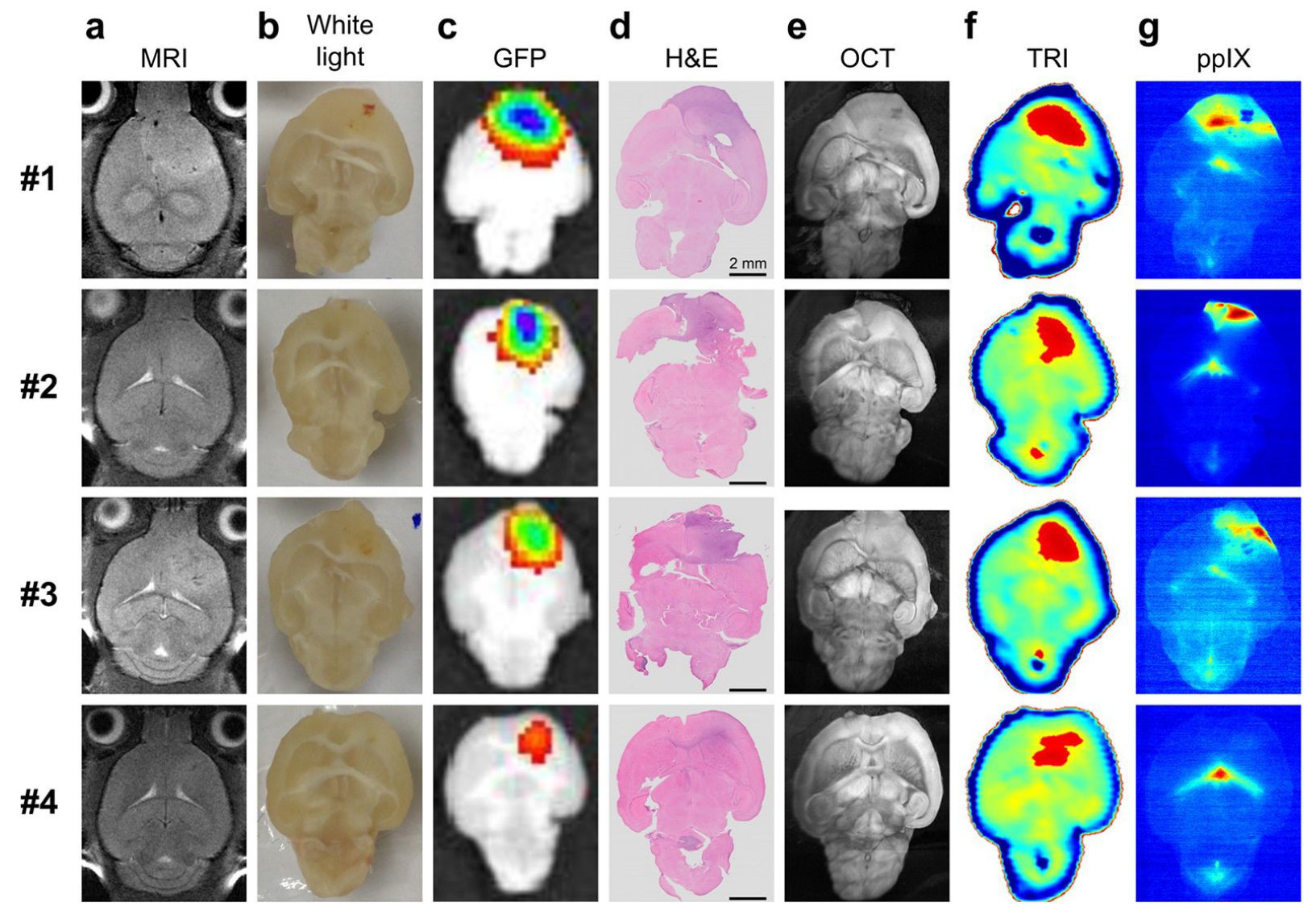
- Oh, S.J.; Kim, S.H.; Ji, Y.B.; Jeong, K.; Park, Y.; Yang, J.; Park, D.W.; Noh, S.K.; Kang, S.G.; Huh, Y.M.; et al. Study of freshly excised brain tissues using terahertz imaging. BOE 2014, 5, 2837–2842. [Google Scholar] [CrossRef] [PubMed]
- Zhong, S. Progress in terahertz nondestructive testing: A review. Front. Mech. Eng. 2019, 14, 273–281. [Google Scholar] [CrossRef]
- Bowman, T.C.; El-Shenawee, M.; Campbell, L.K. Terahertz Imaging of Excised Breast Tumor Tissue on Paraffin Sections. IEEE Trans. Antennas Propag. 2015, 63, 2088–2097. [Google Scholar] [CrossRef]
- Danciu, M.; Alexa-Stratulat, T.; Stefanescu, C.; Dodi, G.; Tamba, B.I.; Mihai, C.T.; Stanciu, G.D.; Luca, A.; Spiridon, I.A.; Ungureanu, L.B.; et al. Terahertz Spectroscopy and Imaging: A Cutting-Edge Method for Diagnosing Digestive Cancers. Materials 2019, 12, 1519. [Google Scholar] [CrossRef] [PubMed]
- Wallace, V.P.; MacPherson, E.; Fitzgerald, A.J.; Lo, T.; Provenzano, E.; Pinder, S.; Purushotham, A. Terahertz pulsed imaging and spectroscopy of breast tumors. In Proceedings of the Optical Methods in the Life Sciences, Boston, MA, USA, 1–3 October 2006. [Google Scholar]
- Woodward, R.M.; Wallace, V.P.; Pye, R.J.; Cole, B.E.; Arnone, D.D.; Linfield, E.H.; Pepper, M. Terahertz pulse imaging of ex vivo basal cell carcinoma. J. Investig. Dermatol. 2003, 120, 72–78. [Google Scholar] [CrossRef]
- Woodward, R.M.; Cole, B.E.; Wallace, V.P.; Pye, R.J.; Arnone, D.D.; Linfield, E.H.; Pepper, M. Terahertz pulse imaging in reflection geometry of human skin cancer and skin tissue. Phys. Med. Biol. 2002, 47, 3853. [Google Scholar] [CrossRef]
- Wallace, V.P.; Fitzegerald, A.J.; Shankar, S.; Flanagan, N.; Pye, R.; Cluff, J.; Arnone, D.D. Dermatological Surgery and Lasers. Terahertz pulsed imaging of basal cell carcinoma ex vivo and in vivo. Br. J. Dermatol. 2004, 151, 424–432. [Google Scholar] [CrossRef]
- Reese, G.; Reid, C.; Goldin, R.; Tran-Dang, M.A.; Fitzgerald, A.; Tekkis, P.; Wallace, V.P. Using terahertz pulsed imaging (TPI) to identify colonic pathology. In Proceedings of the 33rd International Conference on Infrared, Millimeter and Terahertz Waves, IEEE, Pasadena, CA, USA, 15–19 September 2008. [Google Scholar]
- Wahaia, F.; Valusis, G.; Bernardo, L.M.; Almeida, A.; Moreira, J.A.; Lopes, P.C.; Macutkevic, J.; Kasalynas, I.; Seliuta, D.; Adomavicius, R.; et al. Detection of colon cancer by terahertz techniques. J. Mol. Struct. 2011, 1006, 77–82. [Google Scholar] [CrossRef]
- Fitzgerald, A.J.; Wallace, V.P.; Jimenez-Linan, M.; Bobrow, L.; Pye, R.J.; Purushotham, A.D.; Arnone, D.D. Terahertz pulsed imaging of human breast tumors. Radiology 2006, 239, 533–540. [Google Scholar] [CrossRef]
- Bowman, T.C. Experimental Terahertz Imaging and Spectroscopy for Ex-vivo Breast Cancer Tissue; Master of Science in Electrical Engineering (Graduate), University of Arkansas: Fayetteville, AR, USA, August 2014. [Google Scholar]
- Ashworth, P.C.; Pickwell-MacPherson, E.; Provenzano, E.; Pinder, S.E.; Purushotham, A.D.; Pepper, M.; Wallace, V.P. Terahertz pulsed spectroscopy of freshly excised human breast cancer. Opt. Express 2009, 17, 12444–12454. [Google Scholar] [CrossRef] [PubMed]
- Bowman, T.; Wu, Y.; Gauch, J.; Campbell, L.K.; El-Shenawee, M. Terahertz Imaging of Three-Dimensional Dehydrated Breast Cancer Tumors. J. Infrared Millim. Terahertz Waves 2017, 38, 766–786. [Google Scholar] [CrossRef]
- Eadie, L.H.; Reid, C.B.; Fitzgerald, A.J.; Wallace, V.P. Optimizing multi-dimensional terahertz imaging analysis for colon cancer diagnosis. Expert Syst. Appl. 2013, 40, 2043–2205. [Google Scholar] [CrossRef]
- Santaolalla, A.; Sheikh, M.; Van Hemelrijck, M.; Portieri, A.; Coolen, A.C.C. Improved resection margins in breast-conserving surgery using Terahertz Pulsed imaging data. arXiv 2018, arXiv:1805.01349. [Google Scholar]
- Zaytsev, K.I.; Chernomyrdin, N.V.; Kudrin, K.C.; Gavdush, A.A.; Nosova, P.A.; Yurchenko, S.O.; Reshetov, I.V. In vivo terahertz pulsed spectroscopy of dysplastic and non-dysplastic skin nevi. J. Phys. Conf. Ser. 2016, 735, 012076. [Google Scholar] [CrossRef]
- Zaytsev, K.I.; Kudrin, K.G.; Karasik, V.E.; Reshetov, I.V.; Yurchenko, S.O. In vivo terahertz spectroscopy of pigmentary skin nevi: Pilot study of non-invasive early diagnosis of dysplasia. Appl. Phys. Lett. 2015, 106, 053702. [Google Scholar] [CrossRef]
- Grootendorst, M.R.; Fitzgerald, A.J.; Brouwer, S.G.; de Koning, S.A.; Portieri, A.; Van Hemelrijck, M.; Young, M.R.; Owen, J.; Cariati, M.; Pepper, M.; et al. Use of a handheld terahertz pulsed imaging device to differentiate benign and malignant breast tissue. BOE 2017, 8, 2932–2945. [Google Scholar] [CrossRef]
- Matsuura, Y.; Takeda, E. Hollow optical fibers loaded with an inner dielectric film for terahertz broadband spectroscopy. JOSA 2008, 25, 1949–1954. [Google Scholar] [CrossRef]
- Doradla, P.; Joseph, C.S.; Kumar, J.; Giles, R.H. Characterization of bending loss in hollow flexible terahertz waveguides. Opt. Express 2012, 20, 19176–19184. [Google Scholar] [CrossRef]
- Doradla, P.; Joseph, C.S.; Kumar, J.; Giles, R.H. Propagation loss optimization in metal/dielectric coated hollow flexible terahertz waveguides. Proc. SPIE 2012, 8261, 82610P1–82610P10. [Google Scholar]
- Ji, Y.B.; Lee, E.S.; Kim, S.-H.; Son, J.H.; Jeon, T.I. A miniaturized fiber-coupled terahertz endoscope system. Opt. Express 2009, 17, 17082–17087. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, R.C.; Woods, R.E. Digital Image Processing, 3rd ed.; Pearson Prentice Hall: Upper Saddle River, NJ, USA, 2008; pp. 120–144. [Google Scholar]
- Pratt, W.K. Digital Image Processing, 4th ed.; John Wiley & Sons: Hoboken, NJ, USA, 2007; pp. 288–291. [Google Scholar]
- Canny, J.F. A Computational Approach to Edge Detection. IEEE Trans. Pattern Anal. Mach. Intell. 1986, 8, 679–698. [Google Scholar] [CrossRef] [PubMed]
- Ji, Y.B.; Moon, I.-S.; Bark, H.S.; Kim, S.H.; Park, D.W.; Noh, S.K.; Huh, Y.-M.; Suh, J.-S.; Oh, S.J.; Jeon, T.-I. Terahertz otoscope and potential for diagnosis otitis media. BOE 2016, 7, 1201. [Google Scholar] [PubMed]
- Hernandez-Cardoso, G.G.; Rojas-Landeros, S.C.; Alfaro-Gomez, M.; Hernandez-Serrano, A.I.; Salas-Gutierrez, I.; Lemus-Bedolla, E.; Castillo-Guzman, A.R.; Lopez-Lemus, H.L.; Castro-Camus, E. Terahertz imaging for early screening of diabetic foot syndrome: A proof of concept. Sci. Rep. 2017, 7, 42124. [Google Scholar] [CrossRef] [PubMed]
- Jung, E.; Lim, M.H.; Moon, K.W.; Do, Y.W.; Lee, S.S.; Han, H.W.; Choi, H.J.; Cho, K.S.; Kim, K.R. Terahertz pulse imaging of micro-metastatic lymph nodes in early-stage cervical cancer patients. J. Opt. Soc. 2011, 15, 155–160. [Google Scholar] [CrossRef]
- Ji, Y.B.; Oh, S.J.; Kang, S.G.; Heo, J.; Kim, S.H.; Choi, Y.; Song, S.; Son, S.H.; Lee, J.H.; Haam, S.J.; et al. Terahertz reflectometry imaging for low and high grade gliomas. Sci. Rep. 2016, 6, 36040. [Google Scholar] [CrossRef]
- Kashanian, H.A.; Ghaary, H.B.; Bagherzadeh, N.C. Gastric Cancer Diagnosis Using Terahertz Imaging. Majlesi J. Multimed. Process. 2015, 4, 1–7. [Google Scholar]
- Ji, Y.B.; Kim, S.-H.; Jeong, K.; Choi, Y.; Son, J.-H.; Park, D.W.; Noh, S.K.; Jeon, T.-I.; Huh, Y.-M.; Seungjoo, H.; et al. Terahertz spectroscopic imaging and properties of gastrointestinal tract in a rat model. BOE 2014, 5, 4162–4170. [Google Scholar] [CrossRef]
- Ji, Y.B.; Park, C.H.; Kim, H.; Kim, S.H.; Lee, G.M.; Noh, S.K.; Jeon, T.-I.; Son, J.-H.; Huh, Y.-M.; Seungjoo, H.; et al. Feasibility of terahertz reflectometry for discrimination of human early gastric cancers. BOE 2015, 6, 1398–1406. [Google Scholar] [CrossRef]
- Sim, Y.C.; Ahn, K.-M.; Park, J.Y.; Park, C.; Son, J.-H. Temperature-Dependent Terahertz Imaging of Excised Oral Malignant Melanoma. IEEE J. Biomed. Health Inform. 2013, 17, 779. [Google Scholar] [CrossRef]
- Taylor, Z.D.; Singh, R.S.; Bennett, D.B.; Tewari, P.; Kealey, C.P.; Bajwa, N.; Culjat, M.O.; Hubschman, J.; Brown, E.R.; Grundfest, W.S. THz Medical Imaging: In vivo Hydration Sensing. IEEE Trans. Terahertz Sci. Technol. 2011, 1, 201–219. [Google Scholar] [CrossRef]
- Wilmink, G.J.; Grundt, J.E. Current State of Research on Biological Effects of Terahertz Radiation. J. Infrared Millim. Terahertz Waves 2011, 32, 1074–1122. [Google Scholar] [CrossRef]
- Humphreys, K.; Loughran, J.P.; Gradziel, M.; Lanigan, W.; Ward, T.; Murphy, J.A.; O’Sullivan, C. Medical applications of terahertz imaging: A review of current technology and potential applications in biomedical engineering. In Proceedings of the 26th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, San Francisco, CA, USA, 1–5 September 2004. [Google Scholar]
- Arnone, D.D.; Ciesla, C.M.; Corchia, A.; Egusa, S.; Pepper, M.; Chamberlain, J.M.; Bezant, C.; Linfield, E.H.; Clothier, R.; Khammo, N. Applications of terahertz (THz) technology to medical imaging. In Proceedings of the Terahertz Spectroscopy and Applications II, Munich, Germany, 16–18 June 1999; Volume 3828. [Google Scholar]
- Zinov’ev, N.N.; Fitzgerald, A.F.; Strafford, S.M.; Wood, D.J.; Carmichael, F.A.; Miles, R.E.; Smith, M.A.; Chamberlain, J.M. Identification of tooth decay using terahertz imaging and spectroscopy. In Proceedings of the Twenty Seventh International Conference on Infrared and Millimeter Waves, San Diego, CA, USA, 22–26 September 2002. [Google Scholar]
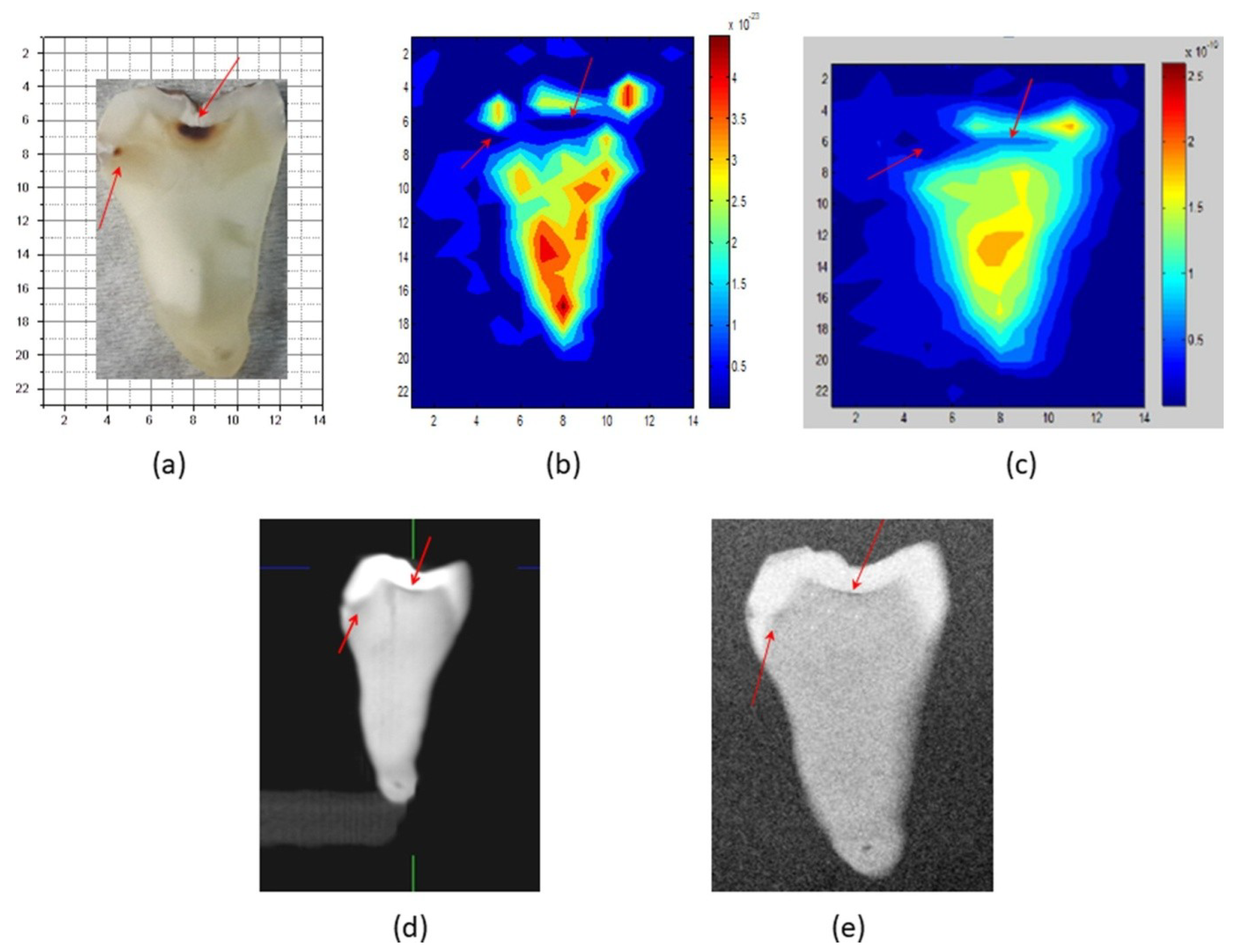
- Crawley, D.A.; Longbottom, C.; Cole, B.E.; Ciesla, C.M.; Arnone, D.; Wallace, V.P.; Pepper, M. Terahertz pulse imaging: A pilot study of potential applications in dentistry. Caries Res. 2003, 37, 352–359. [Google Scholar] [CrossRef]
- Crawley, D.; Longbottom, C.; Wallace, V.P.; Cole, B.; Arnone, D.; Pepper, M. Three-dimensional terahertz pulse imaging of dental tissue. J. Biomed. Opt. 2003, 8, 303–307. [Google Scholar] [CrossRef]
- Kamburoğlu, K.; Karagöz, B.; Altan, H.; Özen, D. An ex vivo comparative study of occlusal and proximal caries using terahertz and X-ray imaging. Dentomaxillofac. Radiol. 2019, 48, 20180250. [Google Scholar] [CrossRef]
- Kamburoğlu, K.; Kurt, H.; Kolsuz, E.; Öztaş, B.; Tatar, I.; Çelik, H.H. Occlusal caries depth measurements obtained by five different imaging modalities. J. Digit. Imaging 2011, 24, 804–813. [Google Scholar] [CrossRef]
- Torres, M.G.; Santos, A.S.; Neves, F.S.; Arriaga, M.L.; Campos, P.S.F.; Crusoé-Rebello, I. Assessment of enamel-dentin caries lesions detection using bitewing PSP digital images. J. Appl. Oral Sci. 2011, 19, 462–468. [Google Scholar] [CrossRef][Green Version]

| Detector | NEP (W/ Hz1/2) | THz Coverage (THz) | Bandwidth (Hz) |
|---|---|---|---|
| Cryogenic Bolometer | 10−14–10−12 | 0.1–30 | 102–103 |
| Golay cell | 10−10 | 0.01–20 1 0.01–700 2 | 10–102 |
| Pyroelectric | 10−10–10−9 | 0.1–10 | 10–108 |
| Schottky diode | 10−14–10−11 | 0.1–2 | 102–106 |
| Tumor | Sample Status | References |
|---|---|---|
| Liver | Ex vivo | [173,174] |
| Brain-cervical | Ex vivo | [39,156,157,177,205,206] |
| Breast | Ex vivo | [132,163,172,179,181,187,188,189,190,192,195] |
| Oral-gastric-intestinal | Ex vivo | [154,176,161,162,164,165,166,180,185,186,207,208,209,210] |
| Skin | Ex vivo In vivo In vitro | [159,182,183,184,193,194] |
| Ovarian | Ex vivo | [167] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
D’Arco, A.; Di Fabrizio, M.; Dolci, V.; Petrarca, M.; Lupi, S. THz Pulsed Imaging in Biomedical Applications. Condens. Matter 2020, 5, 25. https://doi.org/10.3390/condmat5020025
D’Arco A, Di Fabrizio M, Dolci V, Petrarca M, Lupi S. THz Pulsed Imaging in Biomedical Applications. Condensed Matter. 2020; 5(2):25. https://doi.org/10.3390/condmat5020025
Chicago/Turabian StyleD’Arco, Annalisa, Marta Di Fabrizio, Valerio Dolci, Massimo Petrarca, and Stefano Lupi. 2020. "THz Pulsed Imaging in Biomedical Applications" Condensed Matter 5, no. 2: 25. https://doi.org/10.3390/condmat5020025
APA StyleD’Arco, A., Di Fabrizio, M., Dolci, V., Petrarca, M., & Lupi, S. (2020). THz Pulsed Imaging in Biomedical Applications. Condensed Matter, 5(2), 25. https://doi.org/10.3390/condmat5020025







