Materials and Breakdown Phenomena: Heterogeneous Molybdenum Metallic Films
Abstract
:1. Introduction
2. Results
2.1. Molybdenum Coatings
Conductivity Experiments
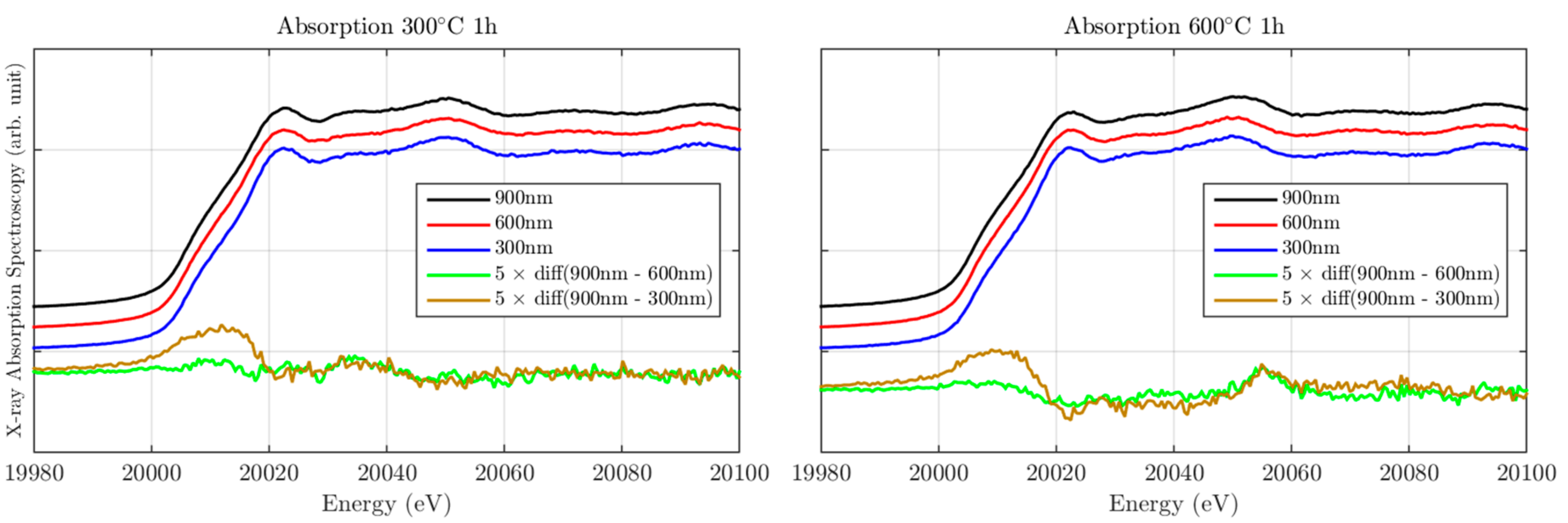
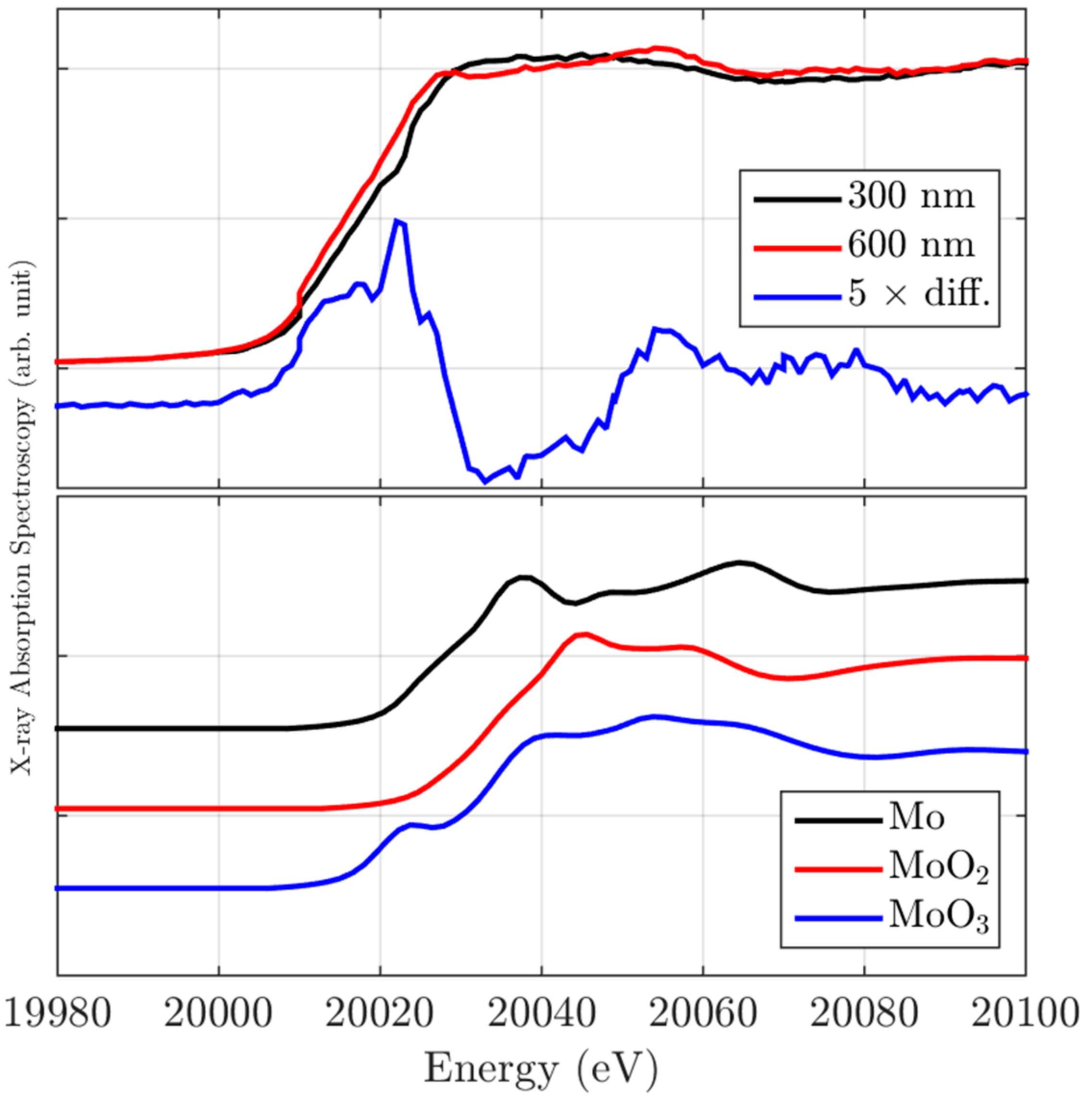
2.2. X-ray Absorption Spectroscopy Characterization
Local Electronic Properties
3. Discussion
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Grudiev, A.; Calatroni, S.; Wurnsh, W. New local field quantity describing the high gradient limit of accelerating structures. Phys. Rev. Accel. Beams 2009, 12, 102001. [Google Scholar] [CrossRef]
- Higo, T.; Higashi, Y.; Matsumoto, S.; Yokoyama, K.; Tsukuba, K.E.K.; Doebert, S.; Grudiev, A.; Riddone, G.; Wuensch, W.; et al. Advances in X-band TW accelerator structures operating in the 100 MV/m regime. In Proceedings of the International Particle Accelerator Conference, Kyoto, Japan, 23–28 May 2010; pp. 3810–3812. [Google Scholar]
- Wuensch, W. CLIC accelerating structure development. In Proceedings of the 11th European Particle Accelerator Conference, Genoa, Italy, 23–27 June 2008. [Google Scholar]
- Antoine, C.Z.; Peauger, F.; Le Pimpec, F. Electromigration occurrences and its effects on metallic surfaces submitted to high electromagnetic field: A novel approach to breakdown in accelerators. Nucl. Instrum. Methods Phys. Res. Sect. A 2011, 665, 54–69. [Google Scholar] [CrossRef]
- Bini, S.; Chimenti, V.; Marcelli, A.; Palumbo, L.; Spataro, B.; Dolgashev, V.; Tantawi, S.; Yeremian, A.D.; Higashi, Y.; Grimaldi, M.G.; et al. Development of X-band accelerating structures for high gradients. Chin. Phys. C 2012, 36, 639–647. [Google Scholar] [CrossRef]
- Dolgashev, V.A.; Gatti, G.; Higashi, Y.; Leonardi, O.; Lewandowski, J.R.; Marcelli, A.; Rosenzweig, J.; Spataro, B.; Tantawi, S.G.; Yeremian, D.A. High power tests of an electroforming cavity operating at 11.424 GHz. J. Inst. 2016, 11, P03010. [Google Scholar]
- Gatti, G.; Marcelli, A.; Spataro, B.; Dolgashev, V.; Lewandowski, J.; Tantawi, S.G.; Yeremian, A.D.; Higashi, Y.; Rosenzweig, J.; Sarti, S.; et al. X-band accelerator structures: On going R & D at the INFN. Nucl. Instrum. Methods Phys. Res. Sect. A 2016, 829, 206–212. [Google Scholar]
- Laurent, L.; Tantawi, S.; Dolgashev, V.; Nantista, C.; Higashi, Y.; Aicheler, M.; Heikkinen, S.; Wuensch, W. Experimental study of rf pulsed heating. Phys. Rev. Accel. Beams 2011, 14, 041001. [Google Scholar] [CrossRef]
- Dolgashev, V.; Tantawi, S.; Higashi, Y.; Spataro, B. Geometric dependence of radio-frequency breakdown in normal conducting accelerating structures. Appl. Phys. Lett. 2010, 97, 171501. [Google Scholar] [CrossRef]
- Tantawi, S.; Dolgashev, V.; Higashi, Y.; Spataro, B. Research and Development for Ultra-High Gradient Accelerator Structures. AIP Conf. Proc. 2010, 1299. [Google Scholar] [CrossRef]
- Graedel, T.E.; Harper, E.M.; Nassar, N.T.; Reck, B.K. On the materials basis of modern society. Proc. Natl. Acad. Sci. USA 2015, 112, 6295–6300. [Google Scholar] [CrossRef] [PubMed]
- Darling, K.A.; Rajagopalan, M.; Komarasamy, M.; Bhatia, M.A.; Hornbuckle, B.C.; Mishra, R.S.; Solanki, K.N. Extreme creep resistance in a microstructurally stable nanocrystalline alloy. Nature 2016, 537, 378–381. [Google Scholar] [CrossRef] [PubMed]
- Bini, S.; Spataro, B.; Marcelli, A.; Sarti, S.; Dolgashev, V.A.; Tantawi, S.; Yeremian, A.D.; Higashi, Y.; Grimaldi, M.G.; Romano, L.; et al. Molybdenum sputtering film characterization for high gradient accelerating structures. Chin. Phys. C 2013, 37, 097005. [Google Scholar] [CrossRef]
- Xu, Y.; Spataro, B.; Sarti, S.; Dolgashev, V.A.; Tantawi, S.; Yeremian, A.D.; Higashi, Y.; Grimaldi, M.G.; Romano, L.; Ruffino, F.; et al. Structural and morphological characterization of Mo coatings for high gradient accelerating structures. In Proceedings of the 15th International Conference on X-ray Absorption Fine Structure (XAFS15), Beijing, China, 22–28 July 2012. [Google Scholar]
- Marcelli, A.; Spataro, B.; Sarti, S.; Dolgashev, V.A.; Tantawi, S.; Yeremian, D.A.; Higashi, Y.; Parodi, R.; Notargiacomo, A.; Xu, J.; et al. Characterization of thick conducting molybdenum films: Enhanced conductivity via thermal annealing. Surf. Coat. Technol. 2015, 261, 391–397. [Google Scholar] [CrossRef]
- Fowler, R.H.; Nordheim, L. Electron Emission in Intense Electric Fields. R. Soc. Publ. 1928, 119, 173–181. [Google Scholar] [CrossRef]
- Furuta, F.; Nakanishi, T.; Okumi, S.; Gotou, T.; Yamamoto, M.; Miyamoto, M.; Kuwahara, M.; Yamamoto, N.; Naniwa, K.; Yasui, K.; et al. Reduction of field emission dark current for high-field gradient electron gun by using a molybdenum cathode and titanium anode. Nucl. Instrum. Methods Phys. Res. Sect. A 2005, 538, 33–44. [Google Scholar] [CrossRef]
- Chimenti, P.; Caliendo, C.; Spataro, B. A Procedure to Obtain a Very Low Mo films with Oxygen Contamination. SPARC 2012. Available online: http://www.lnf.infn.it/acceleratori/sparc/TECHNOTES/RF/SPARC_RF_12_004.pdf (accessed on 28 November 2013).
- Schmid, U.; Seidel, H. Effect of substrate properties and thermal annealing on the resistivity of molybdenum thin films. Thin Solid Films 2005, 489, 310–319. [Google Scholar] [CrossRef]
- Rafaja, D.; Köstenbauer, H.; Mühle, U.; Löffler, C.; Schreiber, G.; Kathrein, M.; Winkler, J. Effect of the deposition process and substrate temperature on the microstructure defects and electrical conductivity of molybdenum thin films. Thin Solid Films 2013, 528, 42–48. [Google Scholar] [CrossRef]
- Glebovsky, V.G.; Markaryans, E.A. Thin film metallization by magnetron sputtering from highly pure molybdenum targets. J. Alloy. Compd. 1993, 190, 157–160. [Google Scholar] [CrossRef]
- Descoeudres, A.; Ramsvik, T.; Calatroni, S.; Taborelli, M.; Wuensch, W. Dc breakdown conditioning and breakdown rate of metals and metallic alloys under ultrahigh vacuum. Phys. Rev. Spec. Top. Accel. Beams 2009, 12, 032001. [Google Scholar] [CrossRef]
- Sondheimer, E.H. The mean free path of electrons in metals. Adv. Phys. 2001, 50, 499–537. [Google Scholar] [CrossRef]
- Harrison, K.W.; Corolewski, C.D.; McCluskey, M.D.; Lindemuth, J.; Ha, S.; Norton, M.G. Electronic transport in molybdenum dioxide thin films. J. Mater. Sci. Mater. Electron. 2015, 26, 9717–9720. [Google Scholar] [CrossRef]
- Alonzo-Medina, G.M.; González-González, A.; Sacedón, J.L.; Oliva, A.I. Understanding the thermal annealing process on metallic thin films. IOP Conf. Ser. Mater. Sci. Eng. 2013, 45, 012013. [Google Scholar] [CrossRef]
- Tosoratti, N.; Fastampa, R.; Giura, M.; Lenzi, V.; Sarti, S.; Silva, E. Two techniques for broadband measurement of the surface impedance of high critical temperature superconducting thin films. Int. J. Mod. Phys. B 2000, 14, 2926–2931. [Google Scholar] [CrossRef]
- Ganchev, S.; Qaddoumi, N.; Bakhtiari, S.; Zoughi, R. Calibration and measurement of dielectric properties of finite thickness composite sheets with open-ended coaxial sensors. IEEE Trans. Instrum. Meas. 1995, 44, 1023–1029. [Google Scholar] [CrossRef]
- Madhavi, V.; Kondaiah, P.; Rayudu, S.S.; Hussain, O.M.; Uthanna, S. Properties of MoO3 films by thermal oxidation: Annealing induced phase transition. Mater. Express 2013, 3, 135–143. [Google Scholar] [CrossRef]
- Kirkpatrick, S. Percolation and Conduction. Rev. Mod. Phys. 1973, 45, 574–588. [Google Scholar] [CrossRef]
- Bianconi, A.; Marcelli, A. Surface XANES. In Synchrotron Radiation Research. Advances in Surface Science; Bachrach, R.Z., Ed.; Plenum Press: New York, NY, USA, 1992; Volume 1. [Google Scholar]
- Dent, J.; Cibin, G.; Ramos, S.; Parry, S.A.; Gianolio, D.; Smith, A.D.; Scott, S.M.; Varandas, L.; Patel, S.; Pearson, M.R.; et al. Performance of B18, the Core EXAFS Bending Magnet beamline at Diamond. J. Phys. Conf. Ser. 2013, 430, 012023. [Google Scholar] [CrossRef]
- Muecke, U.P.; Graf, S.; Rhyner, U.; Gauckler, L.J. Microstructure and electrical conductivity of nanocrystalline nickel and nickel oxide/gadolinia-doped ceria thin films. Acta Mater. 2008, 56, 677–687. [Google Scholar] [CrossRef]
- Xu, W.; Liu, Y.; Zhao, L.; An, P.; Lin, Y.; Marcelli, A.; Wu, Z. Evidence of an interlayer charge transfer route in BiCu1−xSeO. J. Mater. Chem. A 2013, 1, 12154–12158. [Google Scholar] [CrossRef]
- Rehr, J.J.; Kas, J.J.; Vila, F.D.; Prange, M.P.; Jorissen, K. Parameter-free calculations of X-ray spectra with FEFF9. Phys. Chem. Chem. Phys. 2010, 12, 5503–5513. [Google Scholar] [CrossRef]
- Greiner, M.T.; Chai, L.; Helander, M.G.; Tang, W.; Lu, Z. Transition metal oxide work functions: The influence of cation oxidation state and oxygen vacancies. Adv. Funct. Mater. 2012, 22, 4557–4568. [Google Scholar] [CrossRef]
- Lagotzky, S.; Barday, R.; Jankowiak, A.; Kamps, T.; Klimm, C.; Knobloch, J.; Müller, G.; Senkovskiy, B.; Siewert, F. Prevention of electron field emission from molybdenum substrates for photocathodes by the native oxide laye. Eur. Phys. J. Appl. Phys. 2015, 70. [Google Scholar] [CrossRef]
- Why We Need Compact Linear Collider. Available online: http://clic-study.org (accessed on 6 April 2017).
- Castorina, G.; Marcelli, A.; Monforte, F.; Sarti, S.; Spataro, B. An analytical model for evaluating the properties of metallic coatings in RF structures. Condens. Matter 2016, 1, 12. [Google Scholar] [CrossRef]
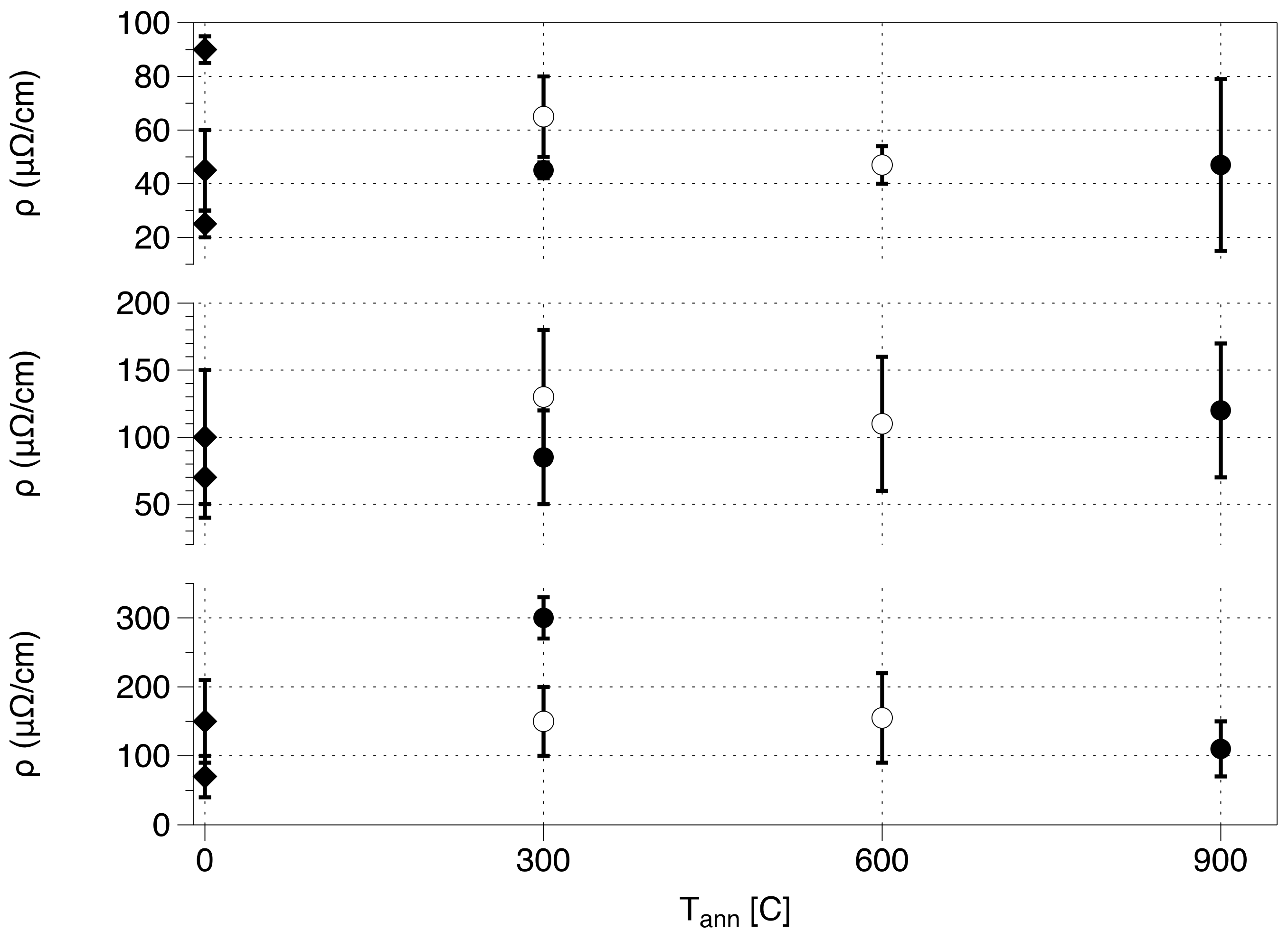
| N | D (nm) | Tann (°C) | Holding Time (h) | ρ (μΩ∙cm) | δρ 1 |
|---|---|---|---|---|---|
| 1 | 300 | 0 | 0 | 25 | 5 |
| 2 | 600 | 0 | 0 | 70 | 30 |
| 3 | 900 | 0 | 0 | 70 | 30 |
| 4 | 300 | 300 | 2 | 45 | 3 |
| 5 | 600 | 300 | 2 | 85 | 35 |
| 6 | 900 | 300 | 2 | 300 | 30 |
| 7 | 300 | 0 | 0 | 45 | 15 |
| 8 | 300 | 0 | 0 | 90 | 5 |
| 9 | 300 | 300 | 1 | 65 | 15 |
| 10 | 300 | 600 | 1 | 47 | 7 |
| 11 | 300 | 900 | 2 | 47 | 32 |
| 12 | 600 | 0 | 0 | 100 | 50 |
| 13 | 600 | 300 | 1 | 130 | 50 |
| 14 | 600 | 600 | 1 | 110 | 50 |
| 15 | 600 | 900 | 2 | 120 | 50 |
| 16 | 900 | 0 | 0 | 150 | 60 |
| 17 | 900 | 300 | 1 | 150 | 50 |
| 18 | 900 | 600 | 1 | 155 | 65 |
| 19 | 900 | 900 | 2 | 110 | 40 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marcelli, A.; Spataro, B.; Castorina, G.; Xu, W.; Sarti, S.; Monforte, F.; Cibin, G. Materials and Breakdown Phenomena: Heterogeneous Molybdenum Metallic Films. Condens. Matter 2017, 2, 18. https://doi.org/10.3390/condmat2020018
Marcelli A, Spataro B, Castorina G, Xu W, Sarti S, Monforte F, Cibin G. Materials and Breakdown Phenomena: Heterogeneous Molybdenum Metallic Films. Condensed Matter. 2017; 2(2):18. https://doi.org/10.3390/condmat2020018
Chicago/Turabian StyleMarcelli, Augusto, Bruno Spataro, Giovanni Castorina, Wei Xu, Stefano Sarti, Francesca Monforte, and Giannantonio Cibin. 2017. "Materials and Breakdown Phenomena: Heterogeneous Molybdenum Metallic Films" Condensed Matter 2, no. 2: 18. https://doi.org/10.3390/condmat2020018
APA StyleMarcelli, A., Spataro, B., Castorina, G., Xu, W., Sarti, S., Monforte, F., & Cibin, G. (2017). Materials and Breakdown Phenomena: Heterogeneous Molybdenum Metallic Films. Condensed Matter, 2(2), 18. https://doi.org/10.3390/condmat2020018








