Efficacy and Usability of eHealth Technologies in Stroke Survivors for Prevention of a New Stroke and Improvement of Self-Management: Phase III Randomized Control Trial
Abstract
1. Background
2. Methods/Design
2.1. Primary Objective
2.2. Secondary Objectives
- Self-management.
- Level of independence in the activities of daily living (ADL).
- Quality of life.
- Adherence to home-based rehabilitation and pharmacological and non-pharmacological treatment.
- Need for caregivers (family, care providers…).
- Recurrent stroke and the complications related to stroke.
- A number of hospital readmissions, emergency care, and outpatient visits to the hospital and primary care centers.
- Cost-effectiveness of the developed system.
- Accessibility of the developed system.
- Sustainability of the developed system.
- User’s satisfaction (by users, we mean stroke survivors, caregivers, and professionals using the developed system).
- Possible adverse events that the system can cause on the study participants.
2.3. Participants
- age 18 years to 80 years;
- having a diagnosis of first ischemic stroke within the past 6 months;
- hemiparesis with mild (91-99) or moderate (61-90) disability (Barthel Index, BI);
- with or without speech pathology but able to understand simple orders (Mississippi Aphasia Screening Test, >45);
- able to cope and to understand the guidelines to use the devices;
- the life expectancy of at least 12 months;
- no severe cognitive impairments (Montreal Cognitive Assessment, MoCA, >26);
- without medical comorbidities that could interfere with the home-rehabilitation program (for example, severe aortic stenosis, respiratory failure, severe osteoarthrosis);
- without socio-familiar dystocia (Gijon’s socio-familial evaluation scale (SFES) <14)
- without a basal functional situation >1 by a Modified Rankin Scale (MRS).
- medical comorbidities that could interfere with the home-rehabilitation program.
- refusal to sign the informed consent and participate in the study.
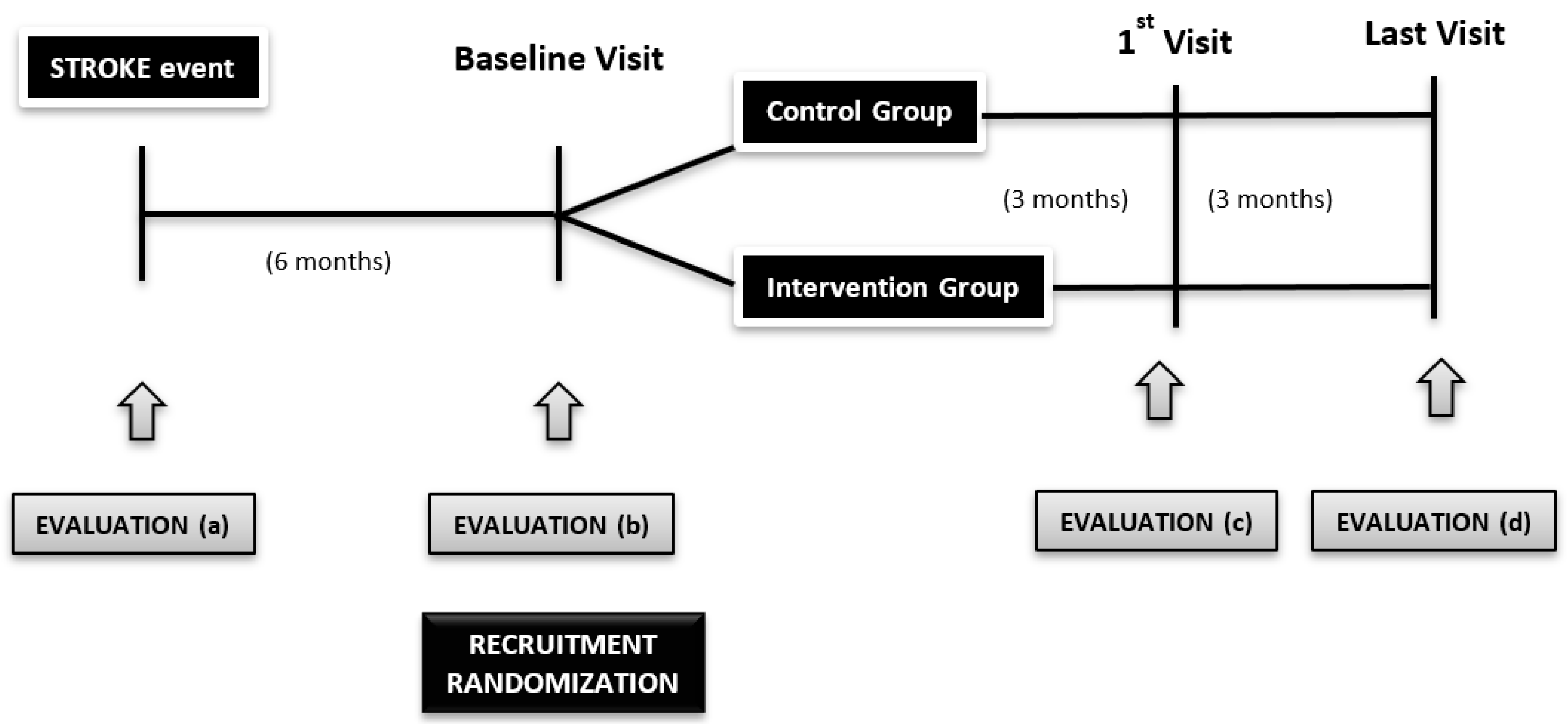
2.4. Study Design
- Wearables and connected objects: provide regular information about the evolution of certain risk factors (e.g., physical activity, blood pressure) without taking over users’ attention. The devices of the STARR system are composed of a tensiometer, a glucometer, a heart rate band, a balance, and a thermometer. All of them are commercial devices tested and with CE marking.
- A decision support system (DSS): implements personalized advice, guidance, and follow-up for daily life activities of the stroke survivors by analyzing the information coming from the wearables and the connected objects, a number of predictive models, and user profiles. The DDS has a system of alarms that will guide patients in making decisions, with recommendations, such as modifying life habits, consulting with their general practitioner, or going to the emergency department. These alarms are based on clinical practice guidelines with proven evidence in the management of patients with stroke. In addition, the responsible doctor of the study participant will have access to a control panel in the health system and a mobile application, where the alarms of the patients can be managed.
- Predictive models: will be populated by risk assessment information provided by validated predictive models calculating stroke risk. The risk estimation done by these models will be complemented by information from a model of human motion analysis and guidance developed during the project using Kinect’s cameras and a created program algorithm, which has been found to be very useful for assuring continued engagement in physical activities in clinical and home settings. It will also be supported by the implementation of models of behavior change to capture individual variations and attitude changes over time. The key requirement behind the implementation of these models is to motivate self-management by encouraging self-awareness and trend-awareness in lifestyle in the sub-acute and chronic phases of stroke.
- Self-management apps (DSS user interface): tools to inform and encourage stroke survivors to self-manage their condition. The STARR system will try to determine the user’s reason for non-adherence using a mobile phone app and an online lifestyle diary. The user will then automatically receive generated messages with persuasive, tailored content. The content will be different at different stages of the initiation and maintenance of health behavior.
- Serious games: promote physical activity and rehabilitation at home with suggested activities in serious games with a screen and a mini-bike.
2.5. Reason for Withdrawal From the Trial
- Death
- Loss of follow-up
- Severe disease of the principal caregiver
- Any other problems that, in the opinion of the research team, justify treatment withdrawal.
2.6. Recruitment
2.7. Protocol
2.8. Control Group
2.9. Intervention Group
2.10. Outcome Measures
- -
- Physical function (PF) assessed by the modified BI and Lawton index.
- -
- Risk factor reduction (blood pressure, analytical profile -glycemia, HbA1c, lipids, weight, heart rate control, medication compliance).
- -
- Self-management behaviors by self-reported information on lifestyles: diet assessed by the Mediterranean diet assessment tool, exercise assessed by tracking with the wearables, smoking, and alcohol consumption.
- -
- Healthcare resource utilization by the information available in the public health system in Osakidetza, Cruces university hospital.
- -
- Knowledge of condition to assess whether there is a relationship between health literacy and control of cardiovascular risk factors, number of complications, number of recurrences, low adherence to pharmacological and non-pharmacological treatment, and self-management behaviors.
- -
- Mood and social isolation by Goldberg scale that measures anxiety and depression.
- -
- Stroke Self-management questionnaire (SSMQ).
2.11. Safety
- causes the death of the patient
- threatens the life of the patient
- requires hospitalization or prolongation of patient hospitalization
- causes disability or permanent or major disability
- results in a congenital anomaly or malformation
2.12. Follow-up Period
2.13. Sample Size
2.14. Statistical Analysis
2.15. Quality Control and Assurance
- (a)
- the physical or psychological integrity or safety of patients during the trial, or
- (b)
- the values of the study. The study sponsor will be contacted as soon as possible. In any case, all violations will be notified to the relevant authorities in accordance with current legislation.
2.16. Limitations of the Study
3. Discussion
Author Contributions
Funding
Conflicts of Interest
Ethics Approval and Consent to Participate
Abbreviations
| STARR | The Decision Support and self-management system for stroke survivors |
| DSS | Decision Support System |
| BI | Barthel Index |
| MoCA | Montreal Cognitive Assessment |
| SFES | Gijon’s Socio-Familial Evaluation Scale |
| MRS | Modified Rankin Scale |
| RCT | Research Clinical Trial |
| RMD | Rehabilitation Medical Doctor |
| PF | Physical Function |
| SSMQ | Stroke Self-Management Questionnaire |
References
- Gustavsson, A.; Svensson, M.; Jacobi, F.; Allgulander, C.; Alonso, J.; Beghi, E.; Dodel, R.; Ekman, M.; Faravelli, C.; Fratiglioni, L.; et al. Cost of disorders of the brain in Europe 2010. Eur. Neuropsychopharmacol. 2011, 21, 718–779. [Google Scholar] [CrossRef] [PubMed]
- Truelsen, T.; Begg, S.; Mathers, C. The Global Burden of Cerebrovascular Disease; WHO Press: Geneva, Switzerland, 2006; Available online: http://www.who.int/healthinfo/statistics/bod_cerebrovasculardiseasestroke.pdf (accessed on 12 June 2019).
- Kernan, W.N.; Ovbiagele, B.; Black, H.R.; Bravata, D.M.; Chimowitz, M.I.; Ezekowitz, M.D.; Fang, M.C.; Fisher, M.; Furie, K.L.; Heck, D.V.; et al. Guidelines for the prevention of stroke in patients with stroke and transient ischemic attack: A guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2014, 45, 2160–2236. [Google Scholar] [CrossRef] [PubMed]
- Di Carlo, A. Human and economic burden of stroke. Age Ageing 2009, 38, 4–5. [Google Scholar] [CrossRef] [PubMed]
- WHO. The World Health Report 2002. Reducing Risks, Promoting Healthy Life; WHO Press: Geneva, Switzerland, 2002; Available online: http://www.who.int/whr/2002/en/whr02_en.pdf?ua=1 (accessed on 12 June 2019).
- Truelsen, T.; Piechowski-Jóźwiak, B.; Bonita, R.; Mathers, C.; Bogousslavsky, J.; Boysen, G. Stroke incidence and prevalence in Europe: A review of available data. Eur. J. Neurol. 2006, 13, 581–598. [Google Scholar] [CrossRef] [PubMed]
- Paul, S.L.; Sturm, J.W.; Dewey, H.M.; Donnan, G.A.; Macdonell, R.A.; Thrift, A.G. Long-term outcome in the North East Melbourne stroke incidence study: Predictors of quality of life at 5 years after stroke. Stroke 2005, 36, 2082–2086. [Google Scholar] [CrossRef] [PubMed]
- WHO. GLOBAL STATUS REPORT on Noncommunicable Diseases; WHO Press: Geneva, Switzerland, 2014; Available online: http://apps.who.int/iris/bitstream/10665/148114/1/9789241564854_eng.pdf?ua=1 (accessed on 12 June 2019).
- Samsa, G.P.; Bian, J.; Lipscomb, J.; Matchar, D.B. Epidemiology of recurrent cerebral infarction: A medicare claims-based comparison of first and recurrent strokes on 2-year survival and cost. Stroke 1999, 30, 338–349. [Google Scholar] [CrossRef] [PubMed]
- Jorgensen, H.S.; Nakayama, H.; Reith, J.; Raaschou, H.O.; Olsen, T.S. Stroke recurrence: Predictors, severity, and prognosis. The Copenhagen stroke study. Neurology 1997, 48, 891–895. [Google Scholar] [CrossRef] [PubMed]
- Williams, G.R. Incidence and characteristics of total stroke in the United States. BMC Neurol. 2001, 1, 2. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention, National Center for Health Statistics. Underlying Cause of Death 1999–2013 on CDC WONDER Online Database, Released 2015. Data Are from the Multiple Cause of Death Files, 1999–2013, as Compiled from Data Provided by the 57 Vital Statistics Jurisdictions through the Vital Statistics Cooperative Program. Available online: http://wonder.cdc.gov/ucd-icd10.html (accessed on 12 June 2019).
- Chapman, B.; Bogle, V. Adherence to medication and self-management in stroke patients. Br. J. Nurs. 2014, 23, 158–166. [Google Scholar] [CrossRef] [PubMed]
- Delamater, A. Improving Patient Adherence. Clin. Diabetes 2006, 2, 71–77. [Google Scholar] [CrossRef]
- Gurol-Urganci, I.; de Jongh, T.; Vodopivec-Jamsek, V.; Car, J.; Atun, R. Mobile phone messaging for communicating results of medical investigations. Cochrane Database Syst. Rev. 2012, 6, CD007456. [Google Scholar] [CrossRef] [PubMed]
- Pal, K.; Eastwood, S.V.; Michie, S.; Farmer, A.J.; Barnard, M.L.; Peacock, R.; Wood, B.; Inniss, J.D.; Murray, E. Computer-based diabetes self-management interventions for adults with type 2 diabetes mellitus. Cochrane Database Syst. Rev. 2013, 3, CD008776. [Google Scholar]
- Fisher, L.; Dickinson, P. New technologies to advance self-management support in diabetes. Diabetes Care 2011, 34, 240–243. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Morrissey, E.C.; Glynn, L.G.; Casey, M.; Walsh, J.C.; Molloy, G.J. New self-management technologies for treatment of hypertension: General practitioners’ perspectives. Fam. Pract. 2017, 35, 318–322. [Google Scholar] [CrossRef] [PubMed]
- Spassova, L.; Vittore, D.; Droste, D.W.; Rösch, N. Randomised controlled trial to evaluate the efficacy and usability of a computerised phone-based lifestyle coaching system for primary and secondary prevention of stroke. BMC Neurol. 2016, 16, 22. [Google Scholar] [CrossRef] [PubMed]
- Vanacker, P.; Standaert, D.; Libbrecht, N.; Vansteenkiste, I.; Bernard, D.; Yperzeele, L.; Vanhooren, G. An individualized coaching program for patients with acute ischemic stroke: Feasibility study. Clin. Neurol. Neurosurg. 2017, 154, 89–93. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Feng, W.; Chhatbar, P.Y.; Liu, Y.; Ji, X.; Ovbiagele, B. Mobile health as a viable strategy to enhance stroke risk factor control: A systematic review and meta-analysis. J. Neurol. Sci. 2017, 378, 140–145. [Google Scholar] [CrossRef] [PubMed]
- Bernhardt, J.; Hayward, K.; Kwakkel, G.; Ward, N.; Wolf, S.; Borschmann, K.; Krakauer, J.; Boyd, L.; Carmichael, T.; Corbett, D.; et al. Agreed definitions and shared vision for new standards in stroke recovery research: The Stroke Recovery and Rehabilitation Roundtable taskforce. Int. J. Stroke. 2017, 12, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Fryer, C.E.; Luker, J.A.; McDonnell, M.N.; Hillier, S.L. Self-management programmes for quality of life in people with stroke. Cochrane Database Syst. Rev. 2016, 8, CD010442. [Google Scholar] [CrossRef] [PubMed]
- Pei-Chun, T.; Ping-Keung, Y.; John Jen, T.; Meei-Fang, L. Needs of family caregivers of stroke patients: A longitudinal study of caregivers’ perspectives. Patient Prefer Adher. 2015, 9, 449–457. [Google Scholar]
| Stroke Survivors | Variables/Scales | Event Visit (a) Month 0 | Baseline Visit (b) Month 6 | First Visit (c) Month 9 | Final Visit (d) Month 12 |
|---|---|---|---|---|---|
| Inclusion | Age | x | x | ||
| Stroke characteristics and type | x | x | |||
| Modified Barthel Index | x | x | x | ||
| Modified Rankin Scale | x | x | x | ||
| Mississippi Aphasia Screening Test [MAST] | x | x | x | ||
| Montreal Cognitive Assessment [MoCA] | x | x | x | ||
| Gijon’s social-familial evaluation scale | x | x | |||
| Comorbidities | x | x | |||
| Charlson Comorbidity Index | x | x | |||
| Socio Demographic | Gender, ethnic group, deprivation index, hand dominance, education level, type of job, hobbies | x | x | ||
| Clinical/ Neurological | Cardiovascular risk factors | x | x | x | x |
| Neurological physical examination: Medical Research Council Scale [MRC] | x | x | x | x | |
| Mississippi Aphasia Screening Test [MAST] | x | x | x | ||
| Montreal Cognitive Assessment [MoCA] | x | x | x | ||
| Functional Ambulation Categories [FAC] | x | x | x | x | |
| 10 m walking test/6 min walking test | x | x | x | x | |
| Berg Balance Scale [BBS] | x | x | x | x | |
| Frenchay Arm Test [FAT] | x | x | x | x | |
| Asworth Modified Scale for Spasticity | x | x | x | x | |
| Fatigue Severity Scale [FSS], | x | x | x | x | |
| Line Bisection Test | x | x | x | x | |
| Disphagia Sensitivity Campimetry | x | x | x | x | |
| Pain Analogic visual scale for pain Analgesic treatment consumption | x | x | x | x | |
| Depression and Anxiety: Golberg Scale | x | x | x | x | |
| Stress | x | x | x | x | |
| Weight, Height, BMI, waist size, waist-to-hip ratio | x | x | x | x | |
| Blood pressure, heart rate, glycemia | x | x | x | x | |
| Need of upper limb orthoses, lower limb orthoses, and canes and wheelchair use in outdoor activities | x | x | x | x | |
| Health Literacy | Test of Functional Health Literacy in Adults Stroke Patient Education Retention | x | x | x | |
| Usability | System Usability Scale | x | x | ||
| Life Style | Mediterranean Diet Assessment Tool Physical activity/Exercise Toxic consumption | x | x | x | x |
| Blood Test | Lipidic profile (total cholesterol, high-density lipoprotein (HDL), low-density lipoprotein (LDL), cholesterol), glycemia, proteins, albumin, HbA1c, Apoprotein B, and Apoprotein A1 | x | x | x | |
| Activities of Daily Living | Modified Barthel Index (BI) Lawton Index | x x | x x | x x | |
| Quality of life | SF-36 Stroke Impact Scale (SIS) | x x | x x | x x | |
| Self-Management | The Southampton Stroke Self-Management Questionnaire (SSSMQ) | x | x | x | |
| Satisfaction Questionnaire | x | x | |||
| Quebec User Evaluation of Satisfaction with Assistive Technology | x | x | |||
| Adherence | Post-stroke checklist | x | x | ||
| Non-pharmacological | |||||
| Pharmacological | x | x | |||
| Complications | Stroke recurrences Number of readmissions Number of consultations to the emergency department Number of visits/telephone calls to a general doctor Number of visits to specialist Number of secondary complications due to stroke | x | x |
| Caregivers | Variables/Scales | Event Visit | Baseline Visit | First Visit | Final Visit |
|---|---|---|---|---|---|
| Health Literacy | Test of Functional Health Literacy in Adults Stroke Patient Education Retention | x | x | x | |
| Quality of Life | SF-36 | x | x | x | |
| Burn-Out | Caregiver Strain Index | x | x | ||
| Self-Management | Family needs of stroke patient questionnaire | x | x | x | |
| Satisfaction Questionnaire | Satisfaction Questionnaire | x | x | ||
| Usability | System Usability Scale | x | x |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ortiz-Fernández, L.; Sagastagoya Zabala, J.; Gutiérrez-Ruiz, A.; Imaz-Ayo, N.; Alava-Menica, A.; Arana-Arri, E. Efficacy and Usability of eHealth Technologies in Stroke Survivors for Prevention of a New Stroke and Improvement of Self-Management: Phase III Randomized Control Trial. Methods Protoc. 2019, 2, 50. https://doi.org/10.3390/mps2020050
Ortiz-Fernández L, Sagastagoya Zabala J, Gutiérrez-Ruiz A, Imaz-Ayo N, Alava-Menica A, Arana-Arri E. Efficacy and Usability of eHealth Technologies in Stroke Survivors for Prevention of a New Stroke and Improvement of Self-Management: Phase III Randomized Control Trial. Methods and Protocols. 2019; 2(2):50. https://doi.org/10.3390/mps2020050
Chicago/Turabian StyleOrtiz-Fernández, Leire, Joana Sagastagoya Zabala, Agustín Gutiérrez-Ruiz, Natale Imaz-Ayo, Ander Alava-Menica, and Eunate Arana-Arri. 2019. "Efficacy and Usability of eHealth Technologies in Stroke Survivors for Prevention of a New Stroke and Improvement of Self-Management: Phase III Randomized Control Trial" Methods and Protocols 2, no. 2: 50. https://doi.org/10.3390/mps2020050
APA StyleOrtiz-Fernández, L., Sagastagoya Zabala, J., Gutiérrez-Ruiz, A., Imaz-Ayo, N., Alava-Menica, A., & Arana-Arri, E. (2019). Efficacy and Usability of eHealth Technologies in Stroke Survivors for Prevention of a New Stroke and Improvement of Self-Management: Phase III Randomized Control Trial. Methods and Protocols, 2(2), 50. https://doi.org/10.3390/mps2020050





