DNA Extraction with DNAzol and LAMP, Performed in a Heating Block as a Simple Procedure for Detection of Mycobacterium tuberculosis in Sputum Specimens
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Sputum Specimens
2.3. Extraction of DNA from Sputum Samples
2.4. Typical LAMP Assay
2.5. Standard Endpoint PCR Assay
2.6. Analysis of Amplification Products
2.7. Assay Settings
2.8. Statistical Analysis of Data
3. Results and Discussion
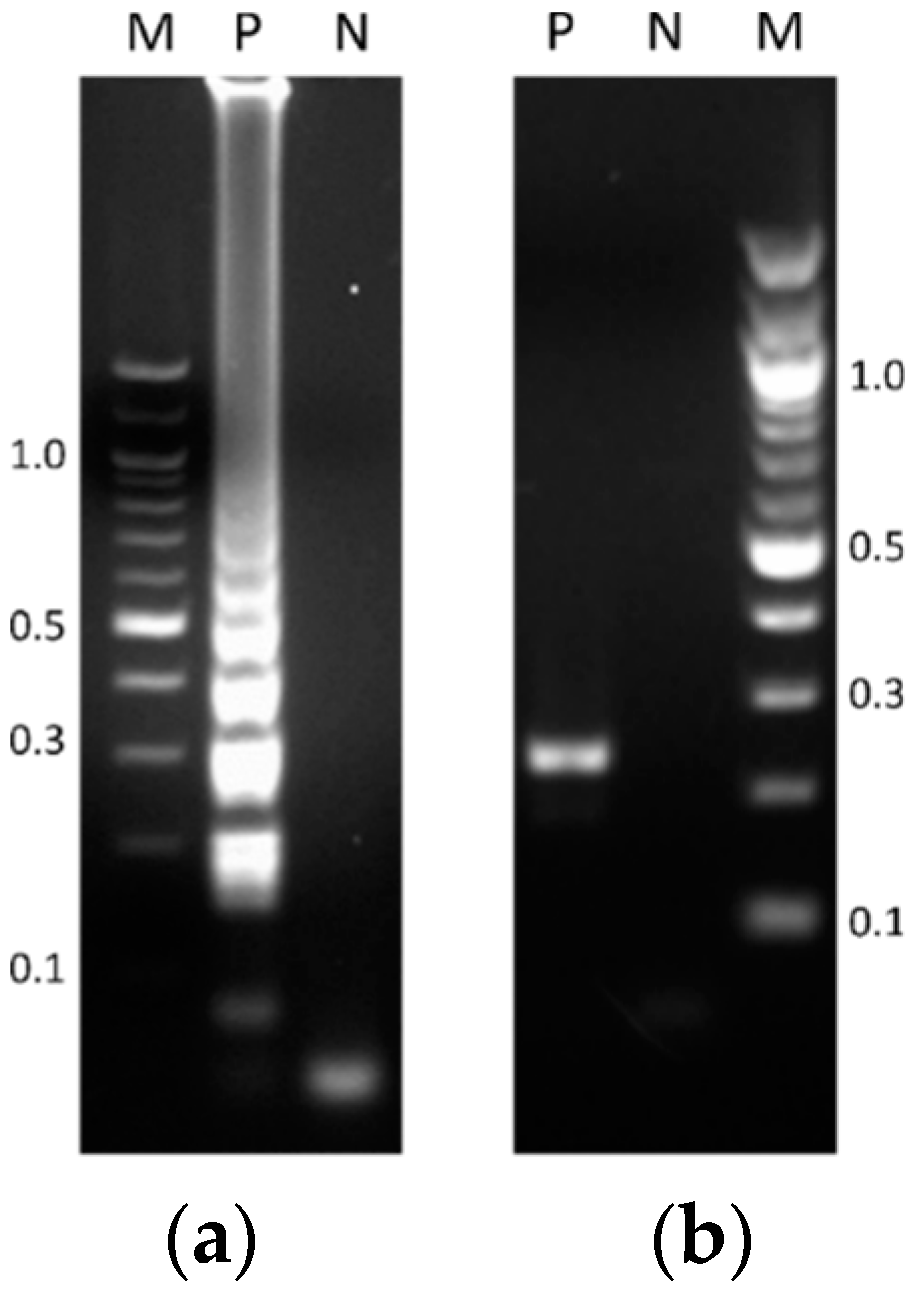
3.1. Assessment of the LAMP Assay Performed in a Heating Block
3.2. Detection of TB in Sputum Specimens by the DNAzol–LAMP Procedure
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Limitations
References
- Raviglione, M.; Sulis, G. Tuberculosis 2015: Burden, challenges and strategy for control and elimination. Infect. Dis. Rep. 2016, 8, 6570. [Google Scholar] [CrossRef] [PubMed]
- WHO (World Health Organization). Global Tuberculosis Report 2016; WHO: Geneva, Switzerland, 2016. [Google Scholar]
- Yuen, C.M.; Amanullah, F.; Dharmadhikari, A.; Nardell, E.A.; Seddon, J.A.; Vasilyeva, I.; Zhao, Y.; Keshavjee, S.; Becerra, M.C. Turning off the tap: Stopping tuberculosis transmission through active case-finding and prompt effective treatment. Lancet 2015, 386, 2334–2343. [Google Scholar] [CrossRef]
- Caulfield, A.J.; Wengenack, N.L. Diagnosis of active tuberculosis disease: From microscopy to molecular techniques. J. Clin. Tuberc. Other Mycobact. Dis. 2016, 4, 33–43. [Google Scholar] [CrossRef]
- Ou, X.; Li, Q.; Xia, H.; Pang, Y.; Wang, S.; Zhao, B.; Song, Y.; Zhou, Y.; Zheng, Y.; Zhang, Z.; et al. Diagnostic accuracy of the PURE-LAMP test for pulmonary tuberculosis at the county-level laboratory in China. PLoS ONE 2014, 9, e94544. [Google Scholar] [CrossRef] [PubMed]
- Gelaw, B.; Shiferaw, Y.; Alemayehu, M.; Bashaw, A.A. Comparison of loop-mediated isothermal amplification assay and smear microscopy with culture for the diagnostic accuracy of tuberculosis. BMC Infect. Dis. 2017, 17, 79. [Google Scholar] [CrossRef] [PubMed]
- Cudahy, P.; Shenoi, S.V. Diagnostics for pulmonary tuberculosis. Postgrad. Med. J. 2016, 92, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Notomi, T.; Okayama, H.; Masubuchi, H.; Yonekawa, T.; Watanabe, K.; Amino, N.; Hase, T. Loop-mediated isothermal amplification of DNA. Nucleic Acids Res. 2000, 28, e63. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Soto, P.; Mvoulouga, P.O.; Akue, J.P.; Abán, J.L.; Santiago, B.V.; Sánchez, M.C.; Muro, A. Development of a highly sensitive loop-mediated isothermal amplification (LAMP) method for the detection of Loa loa. PLoS ONE 2014, 9, e94664. [Google Scholar] [CrossRef] [PubMed]
- Aryan, E.; Makvandi, M.; Farajzadeh, A.; Huygen, K.; Bifani, P.; Mousavi, S.L.; Fateh, A.; Jelodar, A.; Gouya, M.M.; Romano, M. A novel and more sensitive loop-mediated isothermal amplification assay targeting IS6110 for detection of Mycobacterium tuberculosis complex. Microbiol. Res. 2010, 165, 211–220. [Google Scholar] [CrossRef] [PubMed]
- WHO (World Health Organization). The Use of a Commercial Loop-Mediated Isothermal Amplification Assay (TB-LAMP) for the Detection of Tuberculosis: Expert Group Meeting Report; WHO: Geneva, Switzerland, 2013. [Google Scholar]
- PAHO (Pan American Health Organization). Health in the Americas: 2012 Edition; Regional Outlook and Country Profiles; PAHO/WHO: Washington, DC, USA, 2012. [Google Scholar]
- Oren, E.; Fiero, M.H.; Barrett, E.; Anderson, B.; Nuῆez, M.; Gonzalez-Salazar, F. Detection of latent tuberculosis infection among migrant farmworkers along the US-Mexico border. BMC Infect. Dis. 2016, 16, 630. [Google Scholar] [CrossRef] [PubMed]
- Goto, M.; Honda, E.; Ogura, A.; Nomoto, A.; Hanaki, K. Colorimetric detection of loop-mediated isothermal amplification reaction by using hydroxy naphthol blue. BioTechniques 2009, 46, 167–172. [Google Scholar] [CrossRef] [PubMed]
- GraphPad QuickCalcs. Available online: https://www.graphpad.com/quickcalcs/kappa1/ (accessed on 22 October 2018).
- Estadísticas de Código Abierto Para la Salud Pública. Available online: http://www.openepi.com/DiagnosticTest/DiagnosticTest.htm (accessed on 22 October 2018).
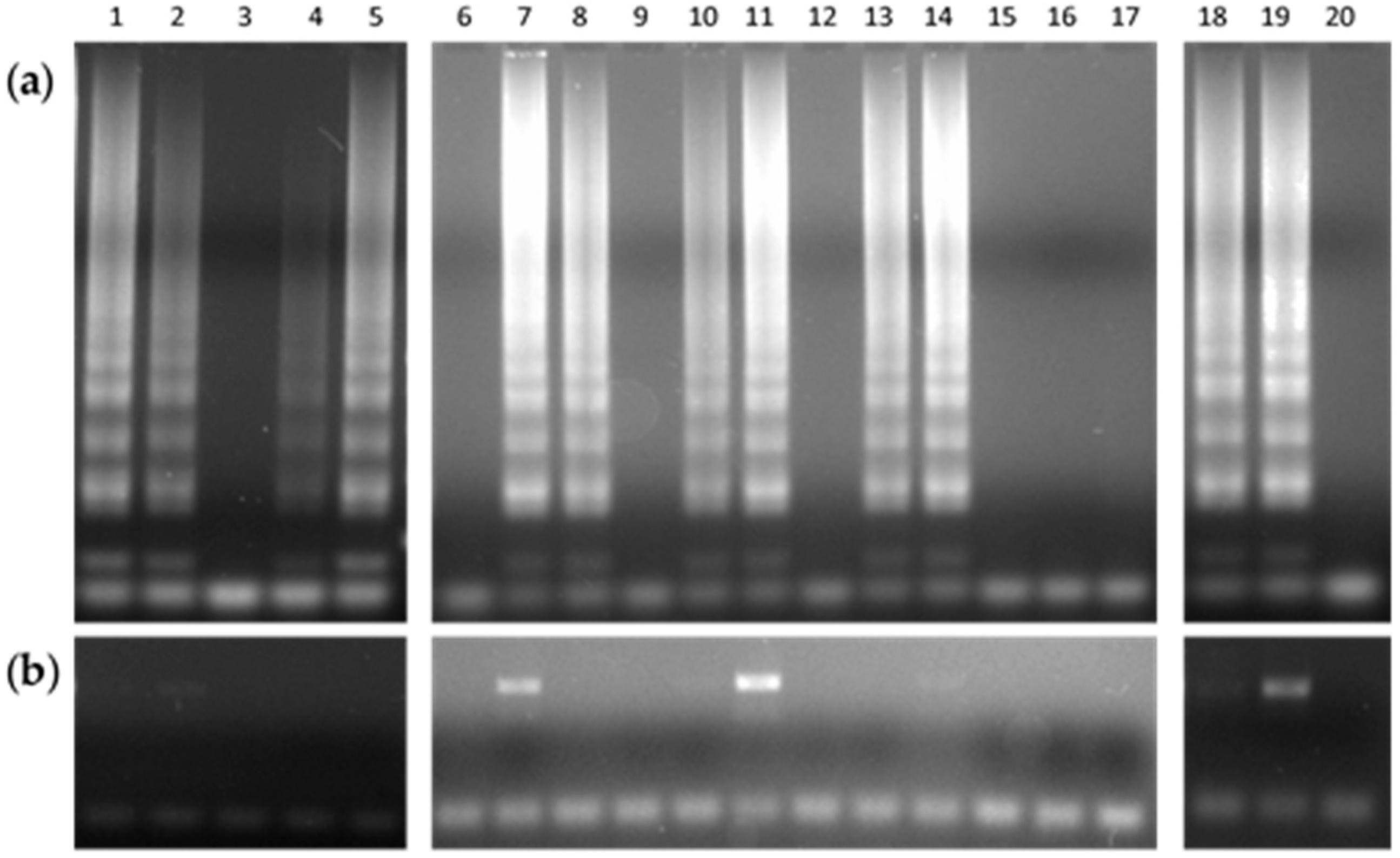
| Sample | LAMP | PCR | Culture |
|---|---|---|---|
| 01 | + | - | + |
| 02 | + | + | + |
| 03 | - | - | + |
| 04 | + | - | + |
| 05 | + | - | - |
| 06 | - | - | - |
| 07 | + | + | + |
| 08 | + | - | - |
| 09 | - | - | - |
| 10 | + | - | + |
| 11 | + | + | + |
| 12 | - | - | - |
| 13 | + | - | - |
| 14 | + | - | + |
| 15 | - | - | - |
| 16 | - | - | - |
| 17 | - | - | - |
| 18 | + | - | - |
| 19 | + | + | + |
| 20 | - | - | - |
| LAMP | PCR | Culture | ||
|---|---|---|---|---|
| Positive | Negative | Positive | Negative | |
| Positive | 4 | 8 | 8 | 4 |
| Negative | 0 | 8 | 1 | 7 |
| Agreements | 60% | 75% | ||
| κ-Coefficient | 0.29 ± 0.14 | 0.51 ± 0.18 | ||
| Strength | Fair | Moderate | ||
| Sensitivity | 88.9% |
|---|---|
| Specificity | 63.6% |
| PPV | 66.7% |
| NPV | 87.5% |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodríguez-García, Á.; Mares, R.E.; Muñoz, P.L.A.; Meléndez-López, S.G.; Licea-Navarro, A.F.; Ramos, M.A. DNA Extraction with DNAzol and LAMP, Performed in a Heating Block as a Simple Procedure for Detection of Mycobacterium tuberculosis in Sputum Specimens. Methods Protoc. 2018, 1, 37. https://doi.org/10.3390/mps1040037
Rodríguez-García Á, Mares RE, Muñoz PLA, Meléndez-López SG, Licea-Navarro AF, Ramos MA. DNA Extraction with DNAzol and LAMP, Performed in a Heating Block as a Simple Procedure for Detection of Mycobacterium tuberculosis in Sputum Specimens. Methods and Protocols. 2018; 1(4):37. https://doi.org/10.3390/mps1040037
Chicago/Turabian StyleRodríguez-García, Álvaro, Rosa E. Mares, Patricia L. A. Muñoz, Samuel G. Meléndez-López, Alexei F. Licea-Navarro, and Marco A. Ramos. 2018. "DNA Extraction with DNAzol and LAMP, Performed in a Heating Block as a Simple Procedure for Detection of Mycobacterium tuberculosis in Sputum Specimens" Methods and Protocols 1, no. 4: 37. https://doi.org/10.3390/mps1040037
APA StyleRodríguez-García, Á., Mares, R. E., Muñoz, P. L. A., Meléndez-López, S. G., Licea-Navarro, A. F., & Ramos, M. A. (2018). DNA Extraction with DNAzol and LAMP, Performed in a Heating Block as a Simple Procedure for Detection of Mycobacterium tuberculosis in Sputum Specimens. Methods and Protocols, 1(4), 37. https://doi.org/10.3390/mps1040037







