Congenital Hypothyroidism 3-Year Follow-Up Project: Region 4 Midwest Genetics Collaborative Results
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Study Design
3. Results
3.1. General
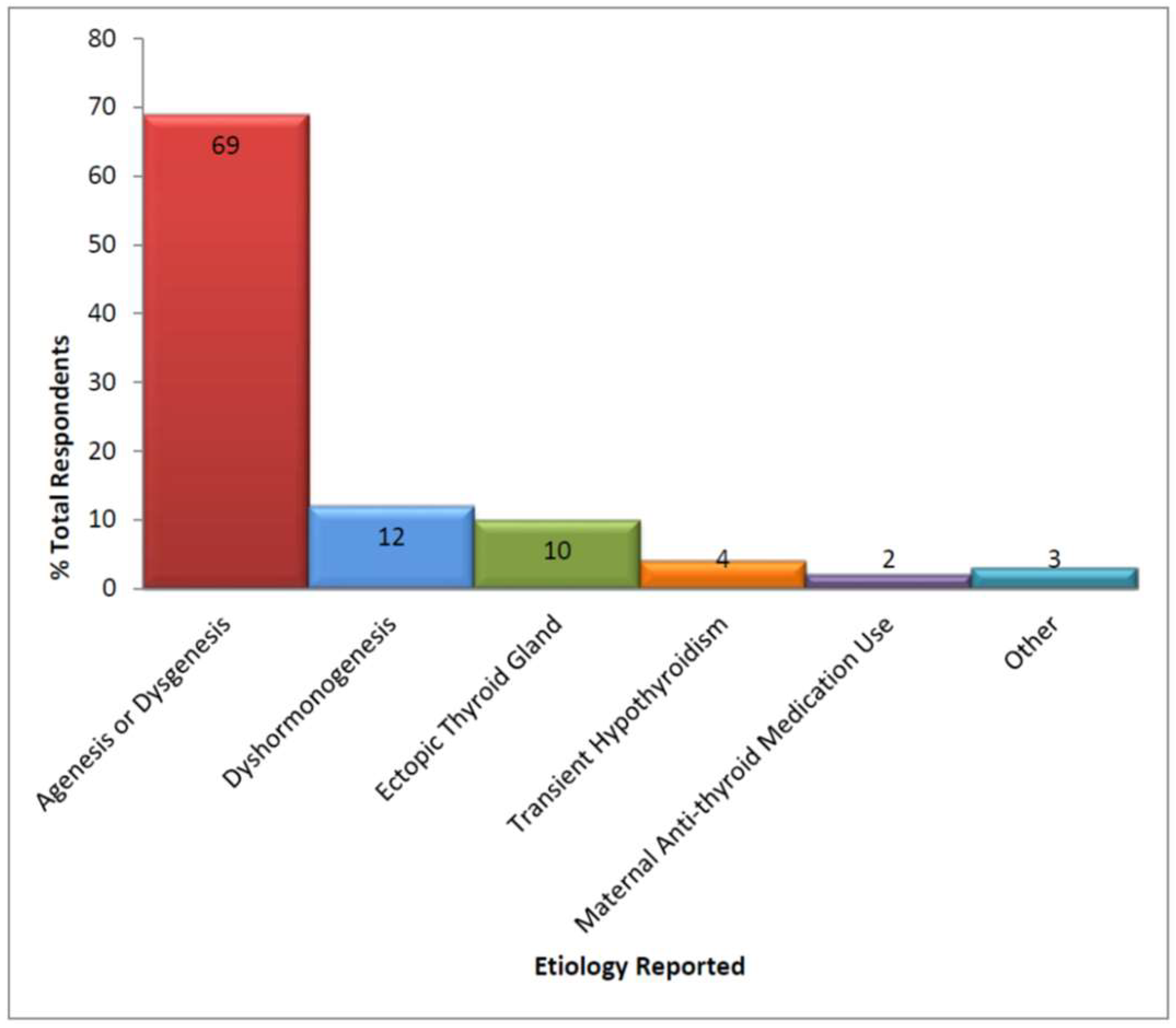
3.2. Diagnosis and Source of Care
3.3. Thyroid Management and Re-Evaluation
3.3.1. Clinician Responses
3.3.2. Parent Responses
3.4. Education and Genetic Counseling
3.4.1. Clinician Responses
3.4.2. Parent Responses
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- National Newborn Screening and Global Resource Center (NNSGRC). National Newborn Screening 2006 Incidence Report. 2006. Available online: http://genes-r-us.uthscsa.edu/sites/genes-r-us/files/resources/genetics/2006datareport.pdf (accessed on 12 June 2016).
- National Newborn Screening and Global Resource Center (NNSGRC). Conditions Screened by US Programs: National Newborn Screening Status Report. 2011. Available online: http://genes-r-us.uthscsa.edu/sites/genes-r-us/files/nbsdisorders.pdf (accessed on 12 June 2016).
- Rose, S.R.; Brown, R.S. Update of newborn screening and therapy for congenital hypothyroidism. Pediatrics 2006, 117, 2290–2303. [Google Scholar] [CrossRef] [PubMed]
- Léger, J.; Olivieri, A.; Donaldson, M.; Torresani, T.; Krude, H.; van Vliet, G.; Polak, M.; Butler, G.; ESPE-PES-SLEP-JSPE-APEG-APPES-ISPAE; Congenital Hypothyroidism Consensus Conference Group. European Society for Paediatric Endocrinology consensus guidelines on screening, diagnosis, and management of congenital hypothyroidism. J. Clin. Endocrinol. Metab. 2014, 99, 363–384. [Google Scholar] [CrossRef]
- Kemper, A.R.; Ouyang, L.; Grosse, S.D. Discontinuation of thyroid hormone treatment among children in the United States with congenital hypothyroidism: Findings from health insurance claims data. BMC Pediatr. 2010, 10. [Google Scholar] [CrossRef] [PubMed]
- Korzeniewski, S.J.; Grigorescu, V.; Kleyn, M.; Young, W.I.; Birbeck, G.; Todem, D.; Romero, R.; Paneth, N. Transient hypothyroidism at 3-year follow-up among cases of congenital hypothyroidism detected by newborn screening. J. Pediatr. 2013, 162, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Wintergerst, K.; Gembel, G.; Kreipe, T.; Zeller, P.; Eugster, E.; Young, B.; Andruszewski, K.; Kleyn, M.; Cunningham, T.; Fawbush, S.; et al. Congenital Hypothyroidism Long-Term Follow-up Project: Navigating the Rough Waters of a Multi-Center, Multi-State Public Health Project. J. Genet. Couns. 2015, 24, 464–472. [Google Scholar] [CrossRef] [PubMed]
- Szinnai, G. Genetics of normal and abnormal thyroid development in humans. Best Pract. Res. Clin. Endocrinol. Metab. 2014, 28, 133–150. [Google Scholar] [CrossRef] [PubMed]
- Arnold, C.L.; Davis, T.C.; Frempong, J.O.; Humiston, S.G.; Bocchini, A.; Kennen, E.M.; Lloyd-Puryear, M. Assessment of newborn screening parent education materials. Pediatrics 2006, 117 Pt 2, S320–S325. [Google Scholar] [CrossRef]
- Fant, K.; Clark, S.; Kemper, A. Completeness and complexity of information available to parents from newborn-screening programs. Pediatrics 2005, 115, 1268–1272. [Google Scholar] [CrossRef] [PubMed]
- Rastogi, M.; Varma, S.; Pediatric Endocrine Society. Educational Materials. Fact Sheets. 2018. Available online: https://www.pedsendo.org (accessed on 18 February 2018).
| State | First-Tier Screening Method | Live Births | CH Diagnosis | Birth Prevalence |
|---|---|---|---|---|
| Michigan | TSH | 125,172 | 85 | 1:1473 |
| Minnesota | TSH | 73,675 | 46 | 1:1602 |
| Wisconsin | TSH | 72,757 | 43 | 1:1692 |
| Illinois | TSH | 180,530 | 94 | 1:1920 |
| Ohio | TSH | 150,784 | 75 | 1:2010 |
| Indiana | TSH | 89,719 | 42 | 1:2136 |
| Kentucky | T4/TSH | 58,507 | 24 | 1:2438 |
| Total | - | 751,144 | 409 | 1:1836 |
| Laboratory Studies | n | % | Imaging Studies | N | % |
|---|---|---|---|---|---|
| TSH | 162 | 97 | Thyroid ultrasound | 40 | 24 |
| Free thyroxine (T4) | 139 | 84 | Thyroid technetium scan | 50 | 29 |
| Total T4 | 28 | 16 | Thyroid uptake scan (I123) | 3 | 2 |
| Free triiodothyronine (T3) | 8 | 5 | Bone age X-ray | 4 | 2 |
| Total T3 | 5 | 3 | Brain MRI/CT | 1 | <1 |
| T3 Uptake | 1 | <1 | |||
| Thyroglobulin level | 4 | 2 | |||
| TBII/TSH receptor antibody | 4 | 3 | |||
| Anti-thyroid peroxidase and/or Anti-thyroglobulin antibodies | 3 | 2 | |||
| Maternal thyroid testing | 0 | -- |
| n = 138 | % | |
|---|---|---|
| Within 3 months | 134 | 97 |
| 2–4 weeks after discontinuation | 40 | 29 |
| 4–8 weeks after discontinuation | 83 | 60 |
| 8–12 weeks after discontinuation | 11 | 8 |
| Greater than 3 months | 4 | 3 |
| 3–6 months after discontinuation | 4 | 3 |
| 6–12 months after discontinuation | - |
| Source | CH Education * (n = 182) | Genetic Counseling * (n = 160) |
|---|---|---|
| n (valid %) | n (valid %) | |
| Face-to-face education in office | 170 (93) | 65 (41) |
| Printed literature | 105 (58) | 40 (25) |
| Internet references | 22 (12) | 3 (2) |
| Information sent by state agencies | 18 (10) | 1 (<1) |
| No standard form of education provided | 6 (3) | 72 (45) |
| Referral to pediatric endocrinologist | 3 (2) | 3 (2) |
| Referral to genetic specialist for counseling | - | 14 (9) |
| Source | Satisfied a (n = 48) | Not Satisfied a (n = 26) | χ2 |
|---|---|---|---|
| n (valid %) | n (valid %) | ||
| Pediatrician talked to parent | 30 (63) | 9 (35) | 5.26 * |
| Pediatric endocrinologist talked to parent | 42 (88) | 22 (85) | 0.12 |
| Genetic doctor/counselor talked to parent | 2 (4) | 1 (4) | <0.01 |
| Printed materials on CH | 29 (60) | 7 (27) | 7.57 ** |
| Internet sites were referred to me | 8 (17) | 4 (15) | 0.02 |
| Information received in the mail | 8 (17) | 2 (8) | 1.16 |
| I looked up information on my own via the Internet | 20 (42) | 11 (42) | <0.01 |
| No one explained the diagnosis or gave me information | 1 (1) |
| When to Re-Evaluate | When Re-Evaluation Can Be Waived |
|---|---|
| Definitive etiology not determined by diagnostic testing | Thyroid dysgenesis demonstrated by imaging (with exception of DUOX2 mutations or Pendred syndrome) |
| Initial treatment started for pre-term infants or during illness | Dyshormonogenesis confirmed by molecular genetic testing |
| Evidence of positive thyroid autoantibodies at diagnosis | TSH elevations above normal reference range for age after first year of life |
| No dosage increase in L-T4 needed since infancy as indicated by rise in TSH above normal range for age | |
| Negative molecular investigation for enzyme defect or whom no testing has been performed |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wintergerst, K.A.; Eugster, E.; Andruszewski, K.; Kleyn, M.; Vanderburg, N.; Sockalosky, J.; Menon, R.; Linard, S.; Kingery, S.; Rose, S.R.; et al. Congenital Hypothyroidism 3-Year Follow-Up Project: Region 4 Midwest Genetics Collaborative Results. Int. J. Neonatal Screen. 2018, 4, 18. https://doi.org/10.3390/ijns4020018
Wintergerst KA, Eugster E, Andruszewski K, Kleyn M, Vanderburg N, Sockalosky J, Menon R, Linard S, Kingery S, Rose SR, et al. Congenital Hypothyroidism 3-Year Follow-Up Project: Region 4 Midwest Genetics Collaborative Results. International Journal of Neonatal Screening. 2018; 4(2):18. https://doi.org/10.3390/ijns4020018
Chicago/Turabian StyleWintergerst, Kupper A., Erica Eugster, Karen Andruszewski, Mary Kleyn, Nancy Vanderburg, Joe Sockalosky, Ram Menon, Sharon Linard, Suzanne Kingery, Susan R. Rose, and et al. 2018. "Congenital Hypothyroidism 3-Year Follow-Up Project: Region 4 Midwest Genetics Collaborative Results" International Journal of Neonatal Screening 4, no. 2: 18. https://doi.org/10.3390/ijns4020018
APA StyleWintergerst, K. A., Eugster, E., Andruszewski, K., Kleyn, M., Vanderburg, N., Sockalosky, J., Menon, R., Linard, S., Kingery, S., Rose, S. R., Moore, J., Gembel, G., & Gorman, L. (2018). Congenital Hypothyroidism 3-Year Follow-Up Project: Region 4 Midwest Genetics Collaborative Results. International Journal of Neonatal Screening, 4(2), 18. https://doi.org/10.3390/ijns4020018





