The First Results of Extended Newborn Screening in Slovakia—Differences between the Majority and the Roma Ethnic Group
Abstract
:1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Dluholucký, S.; Knapková, M. Newborn Screening in Slovakia—From 1985 till today. Acta Facult. Pharm. Univ. Comen. 2013, 60 (Suppl. 8), 32–37. [Google Scholar]
- Dluholucký, S.; Knapková, M. Newborn screening in Slovakia—What news. In Proceedings of the 8th ISNS European Neonatal Screening Regional Meeting, Budapest, Hungary, 4–6 November 2012. [Google Scholar]
- Dluholucký, S.; Knapková, M.; Záhorcová, M. First Results from Expanded Newborn Screening in the Slovak Republic. Acta Fac. Pharm. Univ. Comen. 2014, 1, 17–20. [Google Scholar] [CrossRef]
- Loeber, J.G.; Burgard, P.; Cornel, M.C.; Rigter, T.; Weinreich, S.S.; Rupp, K.; Hoffmann, G.F.; Vittozzi, L. Newborn screening pro-grammes in Europe: Arguments and efforts regarding harmonization. Part 1—Blood spot to screening result. J. Inherit. Metab. Dis. 2012, 35, 603–611. [Google Scholar] [CrossRef] [PubMed]
- Burgard, P.; Rupp, K.; Linder, M.; Haege, G.; Rigter, T.; Weinrich, S.S.; Loeber, J.G.; Taruscio, D.; Vittozzi, L.; Cornel, M.C.; et al. Newborn screening pro-grammes in Europe; arguments and efforts regarding harmonization. Part 2—From screening la-boratory results to treatment, follow-up and quality assurance. J. Inherit. Metab. Dis. 2012, 35, 613–625. [Google Scholar] [CrossRef] [PubMed]
- Feuchtbaum, L.; Carter, J.; Dowray, S.; Currier, R.J. Lorey F Birth prevalence of disorders detectable through newborn screening by race/ethnicity. Genet. Med. 2012, 14, 1937–1945. [Google Scholar] [CrossRef] [PubMed]
- Leščišinová, M.; Kúseková, M.; Sedlák, J.; Murková, V.; Langer, P.; Pavkovceková, O. Increased incidence of congenital hypothyroidism in gypsies in East Slovakia as compared with white population. Endocr. Exper. 1989, 23, 137–141. [Google Scholar]
- Giusti, R. Elevated IRT levels in African-American infants: Implica-tions for newborn screening in an ethnically diverse population. Pediatr. Pulmonol. 2008, 43, 638–641. [Google Scholar] [CrossRef] [PubMed]
- Giusti, R.; Badgwell, A.; Iglesias, A.D. New York State Cystic Fibrosis Consortium: The Frist 2.5 Years of Experience with Cystic Fibrosis Newborn Screening in an Ethnically Diverse Population. Pediatrics 2007, 119, e460–e467. [Google Scholar] [CrossRef] [PubMed]
- Endreffy, E.; Németh, K.; Fekete, G.; Gyurkovits, K.; Stankovics, J.; Szabó, Á.; Sólyom, E.; Dolinay, T.; Raskó, I.; László, A. Molecular Genetic Diagnostic Difficulties in Two Hungarian Gypsy Samples with Cystic Fibrosis. IJHG 2002, 2, 41–44. [Google Scholar]
- Jablonský, P.M.; Dluholucký, S. Present status of SIDS in Central Slovakia. Eur. J. Pediatr. 1995, 154 (Suppl. 1), 278–279. [Google Scholar]
- Bennett, M.J.; Gray, R.G.; Isherwood, D.M.; Murphy, N.; Pollitt, R.J. The diagnosis and biochemical investigation of a patient with short chain fatty acid oxidation defect. JIMD 1985, 8 (Suppl. 2), 135–136. [Google Scholar]
- Gallant, N.M.; Leydiker, K.; Tang, H.; Feuchtbaum, L.; Lorey, F.; Puckett, R.; Deignan, J.L.; Neidich, J.; Doorani, N.; Chang, E.; et al. Biochemical, molecular, and clinical characteristics of children with short chain acyl-CoA dehydrogenase deficiency detected by newborn screening in California. Mol. Genet. MeTab. 2012, 106, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Kalaydjieva, L.; Gresham, D.; Calafell, F. Genetic studies of the Roma (Gypsies): A review. BMC Med. Genet. 2001, 2, 5–17. [Google Scholar] [CrossRef] [PubMed]
- Bartsocas, C.S.; Karayanni, C.; Tsipouras, P.; Baibas, E.; Bouloukos, A.; Papadatos, C. Genetic structure of the Greek gypsies. Clin. Genet. 1979, 15, 5–10. [Google Scholar] [CrossRef] [PubMed]
- Ferák, V.; Siváková, D.; Kroupová, Z. Genetic-distance, geographic distance and migration between for villages of a single region in Slovakia. J. Hum. Evol. 1980, 9, 573–581. [Google Scholar] [CrossRef]
- Genčík, A. Epidemiology and genetics of primary congenital glaucoma in Slovakia. Description of a form of primary congenital glaucoma in Gypsies with autosomal recessive inheritance and com-plete penetrance. Dev. Ophtalmol. 1989, 16, 76–115. [Google Scholar]
- Pollitt, R.J. Introducing new screens: Why we are all doing different things? JIMD 2007, 30, 423–429. [Google Scholar] [CrossRef] [PubMed]
- Plass, A.M.C.; van El, C.G.; Pieters, T.; Cornel, M.C. Neonatal Screening for Treatable and Untreatable Disorders: Prospective Parents´ Opinions. Pediatrics 2009, 125, e99–e106. [Google Scholar] [CrossRef] [PubMed]
- Engel, A.G.; Rebouche, C.J. Carnitine Metabolism and Inborn Errors. J. Inherit. Metab. Dis. 1984, 7, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.L.; Huang, X.W.; Yang, J.B.; Zhou, X.L.; Huang, X.L.; Yang, R.L. Screening and diagnosis of chil-dren with primary carnitine deficiency in Zheijang Province. China HK J. Pediatr. 2013, 18, 167–173. [Google Scholar]
- Van Maldegen, B.T.; Duran, M.; Wanders, R.J.A.; Niezen-Koning, K.E.; Hogeveen, M.; Ijlst, L.; Waterham, H.R.; Wijburg, F.A. Clinical, Biochemical, and Genetic Heterogeneity in Short-Chain Acyl-Coen-zyme A Dehydrogenase Deficiency. JAMA 2006, 296, 943–951. [Google Scholar] [CrossRef] [PubMed]
| IEM Reg | Abb. |
|---|---|
| phenylketonuria/hyperphenylalaninaemia | PKU/HPA |
| maple syrup urine disease | MSUD |
| isovaleric acidemia | IVA |
| glutaric acidemia type I | GA-I |
| medium chain acyl-CoA Dehydrogenase Deficiency | MCADD |
| very long chain acyl-CoA Dehydrogenase Deficiency | VLCADD |
| long chain hydrxoyacyl-CoA Dehydrogenase Deficiency | LCHADD |
| carnitine palmitoyl tranferase 1 deficiency | CPT-1 |
| carnitine palmitoyl tranferase 2 deficienc | CPT-2 |
| carnitine-acylcarnitine translocase deficiency | CACT |
| IEM pil. Study | |
| tyrosinemia types I and II | Tyr I, II |
| hypermethioninaemia | Met |
| propionic acidaemia | PA |
| methylmalonic acidaemia | MMA |
| citrullinaemia | Cit |
| argininaemia | Arg |
| short chain acyl CoA Dehydrogenase Deficiency | SCADD |
| 3-methyl crotonyl CoA carboxylase deficiency | 3-MCC |
| 3-hydroxymethylglutaryl CoA dehydrogenase deficiency | HMG |
| carnitine uptake defect | CUD |
| Data | Total Pop. | Major | Roma | OR |
|---|---|---|---|---|
| Absol.n. | 165,648 | 140,327 | 25,321 | |
| M/R% | 100 | 84.70% | 14.30% | |
| IEMabs. | 192 | 84 | 108 | |
| incid. | 862 | 1670 | 234 | |
| prev.104 | 11.59 | 5.98 | 42.65 | OR:7.13 |
| CH | 87 | 68 | 19 | |
| incid. | 1904 | 2063 | 1333 | |
| prev.104 | 5.25 | 4.84 | 6.56 | OR:1.35 |
| CAH | 10 | 10 | 0 | |
| incid. | 16,565 | 16,565 | <25,321 | |
| prev.104 | 0.6 | 0.71 | 0 | |
| CF | 24 | 23 | 1 | |
| incid. | 6902 | 6101 | 25,321 | |
| prev.104 | 1.45 | 1.64 | 0.39 | |
| total abs | 313 | 185 | 128 | |
| tot.incid. | 529 | 758 | 197 | |
| prev.104 | 18.9 | 13.2 | 50.5 | OR:3.83 |
| Prev.104 | Major | Roma | Total |
|---|---|---|---|
| IEM | 5.98 | 42.65 | 11.59 |
| CH | 4.84 | 6.56 | 5.25 |
| CAH | 0.71 | 0 | 0.6 |
| CF | 1.64 | 0.39 | 1.45 |
| Total | 13.2 | 50.5 | 18.9 |
| Disorder | Major | Roma | Total |
|---|---|---|---|
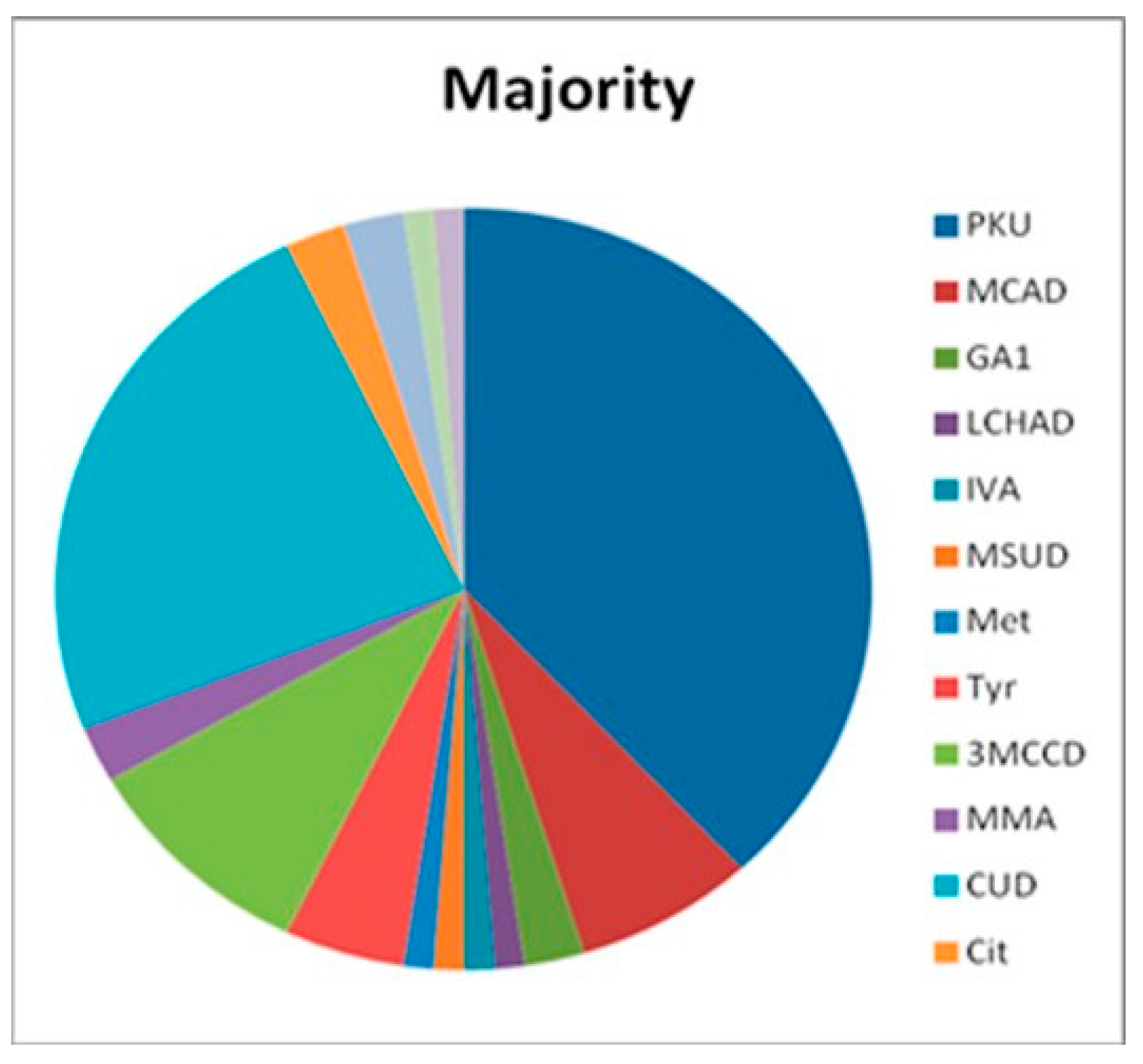
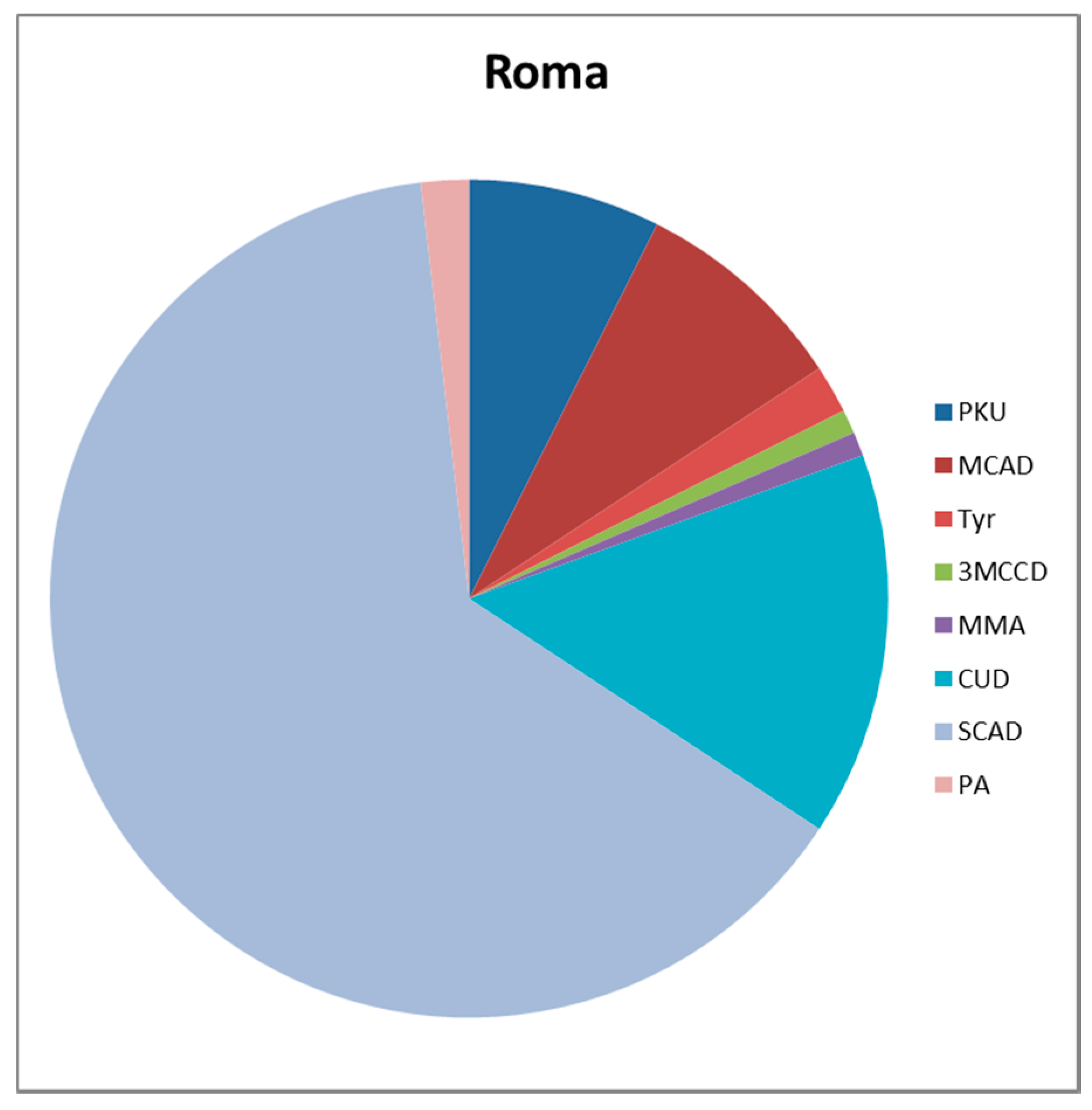
| PKU | 32 | 8 | 40 |
| MCADD | 6 | 9 | 15 |
| GA-1 | 2 | 0 | 2 |
| LCHADD | 1 | 0 | 1 |
| IVA | 1 | 0 | 1 |
| MSUD | 1 | 0 | 1 |
| Met | 1 | 0 | 1 |
| Tyr | 4 | 2 | 6 |
| 3MCCD | 8 | 1 | 9 |
| MMA | 2 | 1 | 3 |
| CUD | 20 | 16 | 36 |
| Cit | 2 | 0 | 2 |
| SCADD | 2 | 69 | 71 |
| PA | 0 | 2 | 2 |
| NKH * | 1 | 0 | 1 |
| Gal | 1 | 0 | 1 |
| Total | 84 | 108 | 192 |
| IEM | Majority | Roma | Total |
|---|---|---|---|
| PKU | 2.28 | 3.016 | 2.41 |
| MCADD | 0.42 | 3.035 | 0.9 |
| CUD | 1.43 | 6.32 | 2.17 |
| SCADD | 0.14 | 27.25 | 4.29 |
| Total | 5.99 | 42.65 | 11.59 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dluholucký, S.; Knapková, M. The First Results of Extended Newborn Screening in Slovakia—Differences between the Majority and the Roma Ethnic Group. Int. J. Neonatal Screen. 2017, 3, 25. https://doi.org/10.3390/ijns3030025
Dluholucký S, Knapková M. The First Results of Extended Newborn Screening in Slovakia—Differences between the Majority and the Roma Ethnic Group. International Journal of Neonatal Screening. 2017; 3(3):25. https://doi.org/10.3390/ijns3030025
Chicago/Turabian StyleDluholucký, Svetozár, and Mária Knapková. 2017. "The First Results of Extended Newborn Screening in Slovakia—Differences between the Majority and the Roma Ethnic Group" International Journal of Neonatal Screening 3, no. 3: 25. https://doi.org/10.3390/ijns3030025
APA StyleDluholucký, S., & Knapková, M. (2017). The First Results of Extended Newborn Screening in Slovakia—Differences between the Majority and the Roma Ethnic Group. International Journal of Neonatal Screening, 3(3), 25. https://doi.org/10.3390/ijns3030025





