Impact of the COVID-19 Pandemic on Iron Overload Assessment by MRI in Patients with Hemoglobinopathies: The E-MIOT Network Experience
Abstract
:1. Introduction
2. Materials and Methods
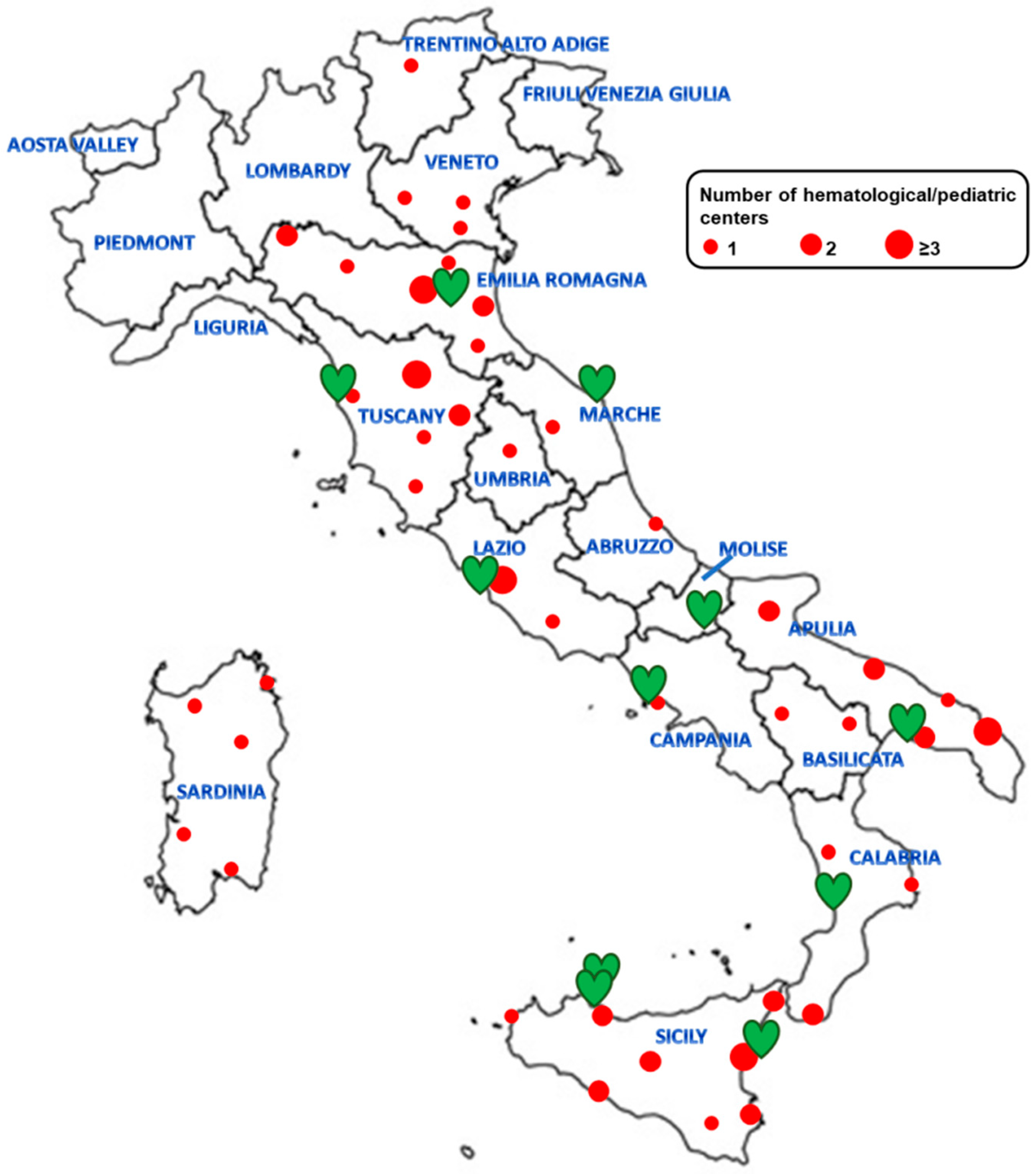
2.1. E-MIOT Network
2.2. Data Collection
2.3. Statistical Analysis
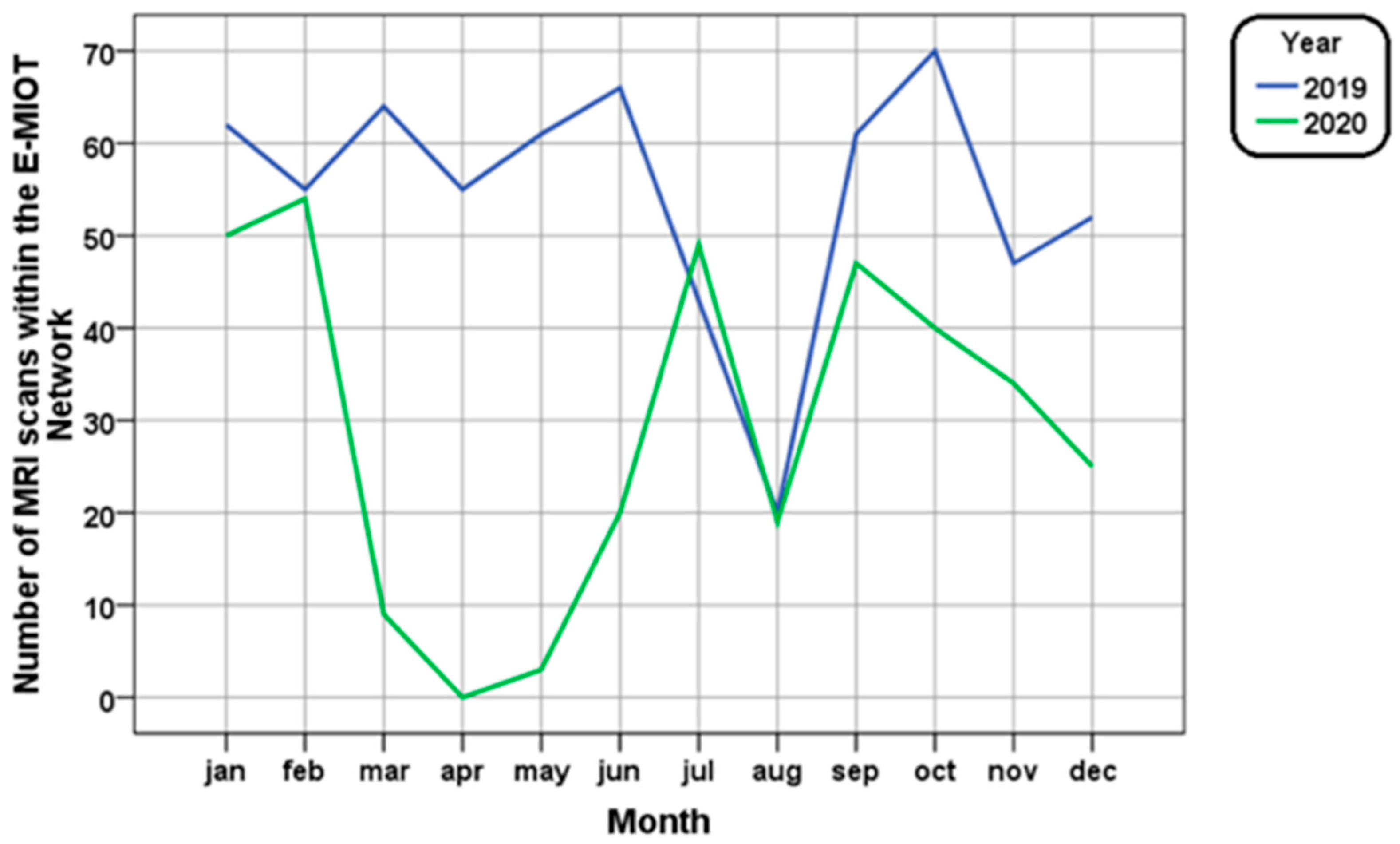
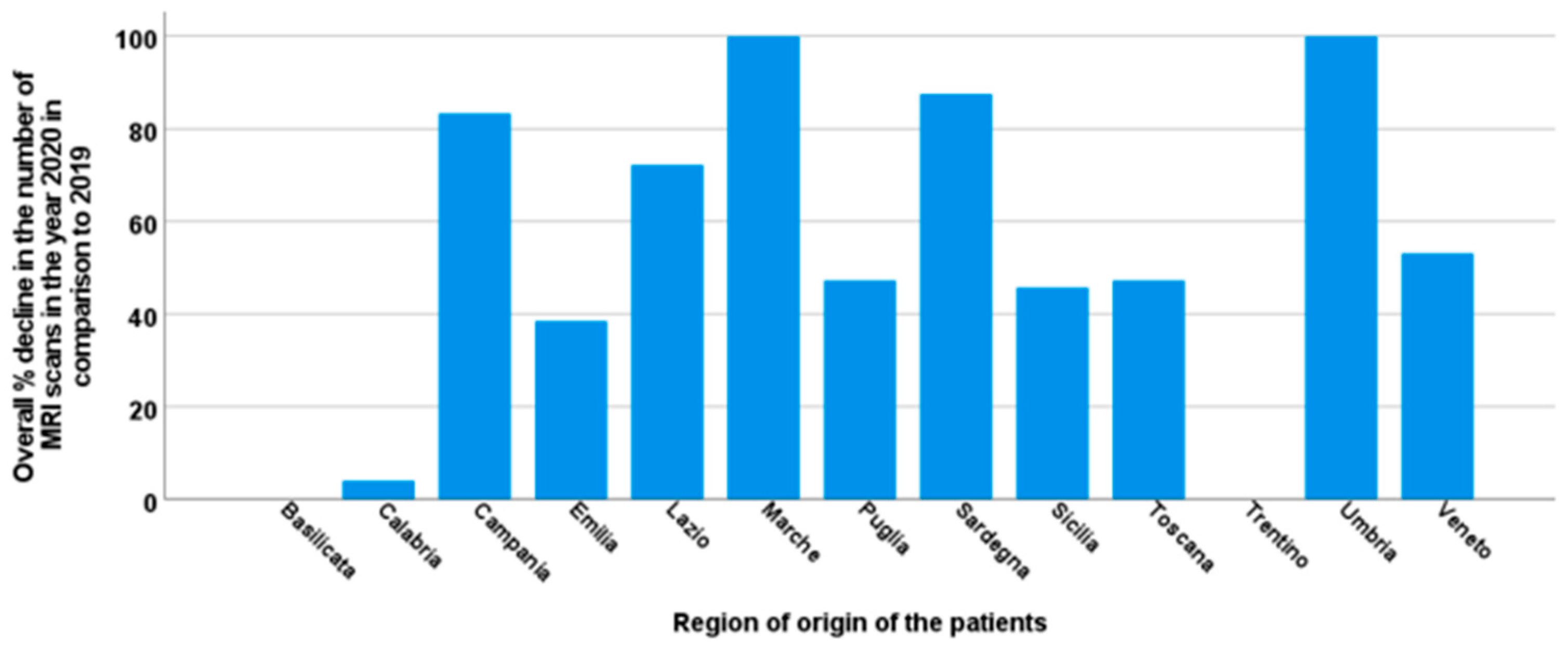
3. Results
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Weatherall, D.J. The thalassaemias. BMJ 1997, 314, 1675–1678. [Google Scholar] [CrossRef]
- Bunn, H.F. Pathogenesis and treatment of sickle cell disease. N. Engl. J. Med. 1997, 337, 762–769. [Google Scholar] [CrossRef]
- Weatherall, D.J. The inherited diseases of hemoglobin are an emerging global health burden. Blood 2010, 115, 4331–4336. [Google Scholar] [CrossRef] [PubMed]
- Origa, R. Beta-Thalassemia. Genet. Med. 2017, 19, 609–619. [Google Scholar] [CrossRef] [PubMed]
- Bou-Fakhredin, R.; Bazarbachi, A.H.; Chaya, B.; Sleiman, J.; Cappellini, M.D.; Taher, A.T. Iron Overload and Chelation Therapy in Non-Transfusion Dependent Thalassemia. Int. J. Mol. Sci. 2017, 18, 2778. [Google Scholar] [CrossRef]
- Miller, S.T.; Wright, E.; Abboud, M.; Berman, B.; Files, B.; Scher, C.D.; Styles, L.; Adams, R.J. Impact of chronic transfusion on incidence of pain and acute chest syndrome during the Stroke Prevention Trial (STOP) in sickle-cell anemia. J. Pediatr. 2001, 139, 785–789. [Google Scholar] [CrossRef] [PubMed]
- Vichinsky, E.P.; Neumayr, L.D.; Earles, A.N.; Williams, R.; Lennette, E.T.; Dean, D.; Nickerson, B.; Orringer, E.; McKie, V.; Bellevue, R.; et al. Causes and outcomes of the acute chest syndrome in sickle cell disease. National Acute Chest Syndrome Study Group. N. Engl. J. Med. 2000, 342, 1855–1865. [Google Scholar] [CrossRef]
- Rund, D.; Rachmilewitz, E. Beta-thalassemia. N. Engl. J. Med. 2005, 353, 1135–1146. [Google Scholar] [CrossRef] [PubMed]
- Tanno, T.; Miller, J.L. Iron Loading and Overloading due to Ineffective Erythropoiesis. Adv. Hematol. 2010, 2010, 358283. [Google Scholar] [CrossRef]
- Angelucci, E.; Muretto, P.; Nicolucci, A.; Baronciani, D.; Erer, B.; Gaziev, J.; Ripalti, M.; Sodani, P.; Tomassoni, S.; Visani, G.; et al. Effects of iron overload and hepatitis C virus positivity in determining progression of liver fibrosis in thalassemia following bone marrow transplantation. Blood 2002, 100, 17–21. [Google Scholar] [CrossRef]
- Marsella, M.; Ricchi, P. Thalassemia and hepatocellular carcinoma: Links and risks. J. Blood Med. 2019, 10, 323–334. [Google Scholar] [CrossRef]
- Wood, J.C.; Noetzl, L.; Hyderi, A.; Joukar, M.; Coates, T.; Mittelman, S. Predicting pituitary iron and endocrine dysfunction. Ann. N. Y. Acad. Sci. 2010, 1202, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Kyriakou, A.; Skordis, N. Thalassaemia and aberrations of growth and puberty. Mediterr. J. Hematol. Infect. Dis. 2009, 1, e2009003. [Google Scholar] [CrossRef] [PubMed]
- Noetzli, L.J.; Mittelman, S.D.; Watanabe, R.M.; Coates, T.D.; Wood, J.C. Pancreatic iron and glucose dysregulation in thalassemia major. Am. J. Hematol. 2012, 87, 155–160. [Google Scholar] [CrossRef]
- Noetzli, L.J.; Panigrahy, A.; Mittelman, S.D.; Hyderi, A.; Dongelyan, A.; Coates, T.D.; Wood, J.C. Pituitary iron and volume predict hypogonadism in transfusional iron overload. Am. J. Hematol. 2012, 87, 167–171. [Google Scholar] [CrossRef]
- Pepe, A.; Pistoia, L.; Gamberini, M.R.; Cuccia, L.; Peluso, A.; Messina, G.; Spasiano, A.; Allo, M.; Bisconte, M.G.; Putti, M.C.; et al. The Close Link of Pancreatic Iron with Glucose Metabolism and with Cardiac Complications in Thalassemia Major: A Large, Multicenter Observational Study. Diabetes Care 2020, 43, 2830–2839. [Google Scholar] [CrossRef]
- Wood, J.C.; Enriquez, C.; Ghugre, N.; Otto-Duessel, M.; Aguilar, M.; Nelson, M.D.; Moats, R.; Coates, T.D. Physiology and pathophysiology of iron cardiomyopathy in thalassemia. Ann. N. Y. Acad. Sci. 2005, 1054, 386–395. [Google Scholar] [CrossRef]
- Meloni, A.; Restaino, G.; Borsellino, Z.; Caruso, V.; Spasiano, A.; Zuccarelli, A.; Valeri, G.; Toia, P.; Salvatori, C.; Positano, V.; et al. Different patterns of myocardial iron distribution by whole-heart T2* magnetic resonance as risk markers for heart complications in thalassemia major. Int. J. Cardiol. 2014, 177, 1012–1019. [Google Scholar] [CrossRef]
- Modell, B.; Khan, M.; Darlison, M.; Westwood, M.A.; Ingram, D.; Pennell, D.J. Improved survival of thalassaemia major in the UK and relation to T2* cardiovascular magnetic resonance. J. Cardiovasc. Magn. Reson. 2008, 10, 42. [Google Scholar] [CrossRef] [PubMed]
- Pepe, A.; Pistoia, L.; Gamberini, M.R.; Cuccia, L.; Lisi, R.; Cecinati, V.; Maggio, A.; Sorrentino, F.; Filosa, A.; Rosso, R.; et al. National networking in rare diseases and reduction of cardiac burden in thalassemia major. Eur. Heart J. 2022, 43, 2482–2492. [Google Scholar] [CrossRef]
- Coates, T.D.; Wood, J.C. How we manage iron overload in sickle cell patients. Br. J. Haematol. 2017, 177, 703–716. [Google Scholar] [CrossRef] [PubMed]
- Pepe, A.; Meloni, A.; Pistoia, L.; Cuccia, L.; Gamberini, M.R.; Lisi, R.; D’Ascola, D.G.; Rosso, R.; Allo, M.; Spasiano, A.; et al. MRI multicentre prospective survey in thalassaemia major patients treated with deferasirox versus deferiprone and desferrioxamine. Br. J. Haematol. 2018, 183, 783–795. [Google Scholar] [CrossRef] [PubMed]
- Borrelli, E.; Grosso, D.; Vella, G.; Sacconi, R.; Querques, L.; Zucchiatti, I.; Prascina, F.; Bandello, F.; Querques, G. Impact of COVID-19 on outpatient visits and intravitreal treatments in a referral retina unit: Let’s be ready for a plausible “rebound effect”. Graefes Arch. Clin. Exp. Ophthalmol. 2020, 258, 2655–2660. [Google Scholar] [CrossRef]
- Parravano, M.; Borrelli, E.; Costanzo, E.; Sacconi, R.; Varano, M.; Querques, G. Protect Healthcare Workers and Patients from COVID-19: The Experience of Two Tertiary Ophthalmology Care Referral Centers in Italy. Ophthalmol. Ther. 2020, 9, 231–234. [Google Scholar] [CrossRef]
- Ricci, G.; Pallotta, G.; Sirignano, A.; Amenta, F.; Nittari, G. Consequences of COVID-19 Outbreak in Italy: Medical Responsibilities and Governmental Measures. Front. Public Health 2020, 8, 588852. [Google Scholar] [CrossRef] [PubMed]
- Remuzzi, A.; Remuzzi, G. COVID-19 and Italy: What next? Lancet 2020, 395, 1225–1228. [Google Scholar] [CrossRef]
- Ramazzotti, A.; Pepe, A.; Positano, V.; Rossi, G.; De Marchi, D.; Brizi, M.G.; Luciani, A.; Midiri, M.; Sallustio, G.; Valeri, G.; et al. Multicenter validation of the magnetic resonance T2* technique for segmental and global quantification of myocardial iron. J. Magn. Reson. Imaging 2009, 30, 62–68. [Google Scholar] [CrossRef]
- Meloni, A.; De Marchi, D.; Pistoia, L.; Grassedonio, E.; Peritore, G.; Preziosi, P.; Restaino, G.; Righi, R.; Riva, A.; Renne, S.; et al. Multicenter validation of the magnetic resonance T2* technique for quantification of pancreatic iron. Eur. Radiol. 2019, 29, 2246–2252. [Google Scholar] [CrossRef]
- Positano, V.; Salani, B.; Pepe, A.; Santarelli, M.F.; De Marchi, D.; Ramazzotti, A.; Favilli, B.; Cracolici, E.; Midiri, M.; Cianciulli, P.; et al. Improved T2* assessment in liver iron overload by magnetic resonance imaging. Magn. Reson. Imaging 2009, 27, 188–197. [Google Scholar] [CrossRef]
- Restaino, G.; Meloni, A.; Positano, V.; Missere, M.; Rossi, G.; Calandriello, L.; Keilberg, P.; Mattioni, O.; Maggio, A.; Lombardi, M.; et al. Regional and global pancreatic T*(2) MRI for iron overload assessment in a large cohort of healthy subjects: Normal values and correlation with age and gender. Magn. Reson. Med. 2011, 65, 764–769. [Google Scholar] [CrossRef]
- Pepe, A.; Positano, V.; Santarelli, F.; Sorrentino, F.; Cracolici, E.; De Marchi, D.; Maggio, A.; Midiri, M.; Landini, L.; Lombardi, M. Multislice multiecho T2* cardiovascular magnetic resonance for detection of the heterogeneous distribution of myocardial iron overload. J. Magn. Reson. Imaging 2006, 23, 662–668. [Google Scholar] [CrossRef] [PubMed]
- Meloni, A.; Positano, V.; Pepe, A.; Rossi, G.; Dell’Amico, M.; Salvatori, C.; Keilberg, P.; Filosa, A.; Sallustio, G.; Midiri, M.; et al. Preferential patterns of myocardial iron overload by multislice multiecho T*2 CMR in thalassemia major patients. Magn. Reson. Med. 2010, 64, 211–219. [Google Scholar] [CrossRef]
- Positano, V.; Pepe, A.; Santarelli, M.F.; Scattini, B.; De Marchi, D.; Ramazzotti, A.; Forni, G.; Borgna-Pignatti, C.; Lai, M.E.; Midiri, M.; et al. Standardized T2* map of normal human heart in vivo to correct T2* segmental artefacts. NMR Biomed. 2007, 20, 578–590. [Google Scholar] [CrossRef] [PubMed]
- Meloni, A.; Luciani, A.; Positano, V.; De Marchi, D.; Valeri, G.; Restaino, G.; Cracolici, E.; Caruso, V.; Dell’amico, M.C.; Favilli, B.; et al. Single region of interest versus multislice T2* MRI approach for the quantification of hepatic iron overload. J. Magn. Reson. Imaging 2011, 33, 348–355. [Google Scholar] [CrossRef] [PubMed]
- Meloni, A.; Rienhoff, H.Y., Jr.; Jones, A.; Pepe, A.; Lombardi, M.; Wood, J.C. The use of appropriate calibration curves corrects for systematic differences in liver R2* values measured using different software packages. Br. J. Haematol. 2013, 161, 888–891. [Google Scholar] [CrossRef]
- Wood, J.C.; Enriquez, C.; Ghugre, N.; Tyzka, J.M.; Carson, S.; Nelson, M.D.; Coates, T.D. MRI R2 and R2* mapping accurately estimates hepatic iron concentration in transfusion-dependent thalassemia and sickle cell disease patients. Blood 2005, 106, 1460–1465. [Google Scholar] [CrossRef]
- Meloni, A.; De Marchi, D.; Positano, V.; Neri, M.G.; Mangione, M.; Keilberg, P.; Lendini, M.; Cirotto, C.; Pepe, A. Accurate estimate of pancreatic T2* values: How to deal with fat infiltration. Abdom. Imaging 2015, 40, 3129–3136. [Google Scholar] [CrossRef]
- Cerqueira, M.D.; Weissman, N.J.; Dilsizian, V.; Jacobs, A.K.; Kaul, S.; Laskey, W.K.; Pennell, D.J.; Rumberger, J.A.; Ryan, T.; Verani, M.S. Standardized myocardial segmentation and nomenclature for tomographic imaging of the heart: A statement for healthcare professionals from the Cardiac Imaging Committee of the Council on Clinical Cardiology of the American Heart Association. Circulation 2002, 105, 539–542. [Google Scholar]
- Angelucci, E.; Brittenham, G.M.; McLaren, C.E.; Ripalti, M.; Baronciani, D.; Giardini, C.; Galimberti, M.; Polchi, P.; Lucarelli, G. Hepatic iron concentration and total body iron stores in thalassemia major. N. Engl. J. Med. 2000, 343, 327–331. [Google Scholar] [CrossRef]
- Anderson, L.J.; Holden, S.; Davis, B.; Prescott, E.; Charrier, C.C.; Bunce, N.H.; Firmin, D.N.; Wonke, B.; Porter, J.; Walker, J.M.; et al. Cardiovascular T2-star (T2*) magnetic resonance for the early diagnosis of myocardial iron overload. Eur. Heart J. 2001, 22, 2171–2179. [Google Scholar] [CrossRef]
- Fersia, O.; Bryant, S.; Nicholson, R.; McMeeken, K.; Brown, C.; Donaldson, B.; Jardine, A.; Grierson, V.; Whalen, V.; Mackay, A. The impact of the COVID-19 pandemic on cardiology services. Open Heart 2020, 7, e001359. [Google Scholar] [CrossRef]
- Aydemir, D.; Ulusu, N.N. People with blood disorders can be more vulnerable during COVID-19 pandemic: A hypothesis paper. Transfus. Apher. Sci. 2021, 60, 103080. [Google Scholar] [CrossRef] [PubMed]
- Karimi, M.; Haghpanah, S.; Azarkeivan, A.; Zahedi, Z.; Zarei, T.; Akhavan Tavakoli, M.; Bazrafshan, A.; Shirkavand, A.; De Sanctis, V. Prevalence and mortality in beta-thalassaemias due to outbreak of novel coronavirus disease (COVID-19): The nationwide Iranian experience. Br. J. Haematol. 2020, 190, e137–e140. [Google Scholar] [CrossRef] [PubMed]
- Motta, I.; Migone De Amicis, M.; Pinto, V.M.; Balocco, M.; Longo, F.; Bonetti, F.; Gianesin, B.; Graziadei, G.; Cappellini, M.D.; De Franceschi, L.; et al. SARS-CoV-2 infection in beta thalassemia: Preliminary data from the Italian experience. Am. J. Hematol. 2020, 95, E198–E199. [Google Scholar] [CrossRef]
- Minniti, C.P.; Zaidi, A.U.; Nouraie, M.; Manwani, D.; Crouch, G.D.; Crouch, A.S.; Callaghan, M.U.; Carpenter, S.; Jacobs, C.; Han, J.; et al. Clinical predictors of poor outcomes in patients with sickle cell disease and COVID-19 infection. Blood Adv. 2021, 5, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Pennell, D.J.; Udelson, J.E.; Arai, A.E.; Bozkurt, B.; Cohen, A.R.; Galanello, R.; Hoffman, T.M.; Kiernan, M.S.; Lerakis, S.; Piga, A.; et al. Cardiovascular function and treatment in beta-thalassemia major: A consensus statement from the American Heart Association. Circulation 2013, 128, 281–308. [Google Scholar] [CrossRef] [PubMed]
- Meloni, A.; Positano, V.; Ruffo, G.B.; Spasiano, A.; D’Ascola, D.G.; Peluso, A.; Keilberg, P.; Restaino, G.; Valeri, G.; Renne, S.; et al. Improvement of heart iron with preserved patterns of iron store by CMR-guided chelation therapy. Eur. Heart J. Cardiovasc. Imaging 2015, 16, 325–334. [Google Scholar] [CrossRef]
| Characteristic | Year 2019 (N = 656) | Year 2020 (N = 350) | p-Value |
|---|---|---|---|
| Females, N (%) | 353 (53.8) | 184 (52.6) | 0.707 |
| Mean age (years) | 40.6 ± 11.9 | 39.3 ± 12.1 | 0.104 |
| Main disease, N (%) | |||
| sickle cell anemia | 7 (1.1) | 5 (1.4) | 0.448 |
| sickle beta thalassemia | 15 (2.3) | 8 (2.3) | |
| thalassemia major | 521 (79.4) | 290 (82.9) | |
| thalassemia intermedia | 113 (17.2) | 47 (13.4) | |
| MRI scan, N (%) | <0.0001 | ||
| first | 184 (28.0) | 52 (14.9) | |
| follow-up | 472 (72.0) | 298 (85.1) | |
| MRI LIC (mg/d dw) | 5.68 ± 7.14 | 6.40 ± 10.54 | 0.326 |
| Global pancreas T2* (ms) | 15.16 ± 11.14 | 14.87 ± 10.58 | 0.907 |
| Global heart T2* (ms) | 37.81 ± 8.06 | 37.57 ± 8.50 | 0.931 |
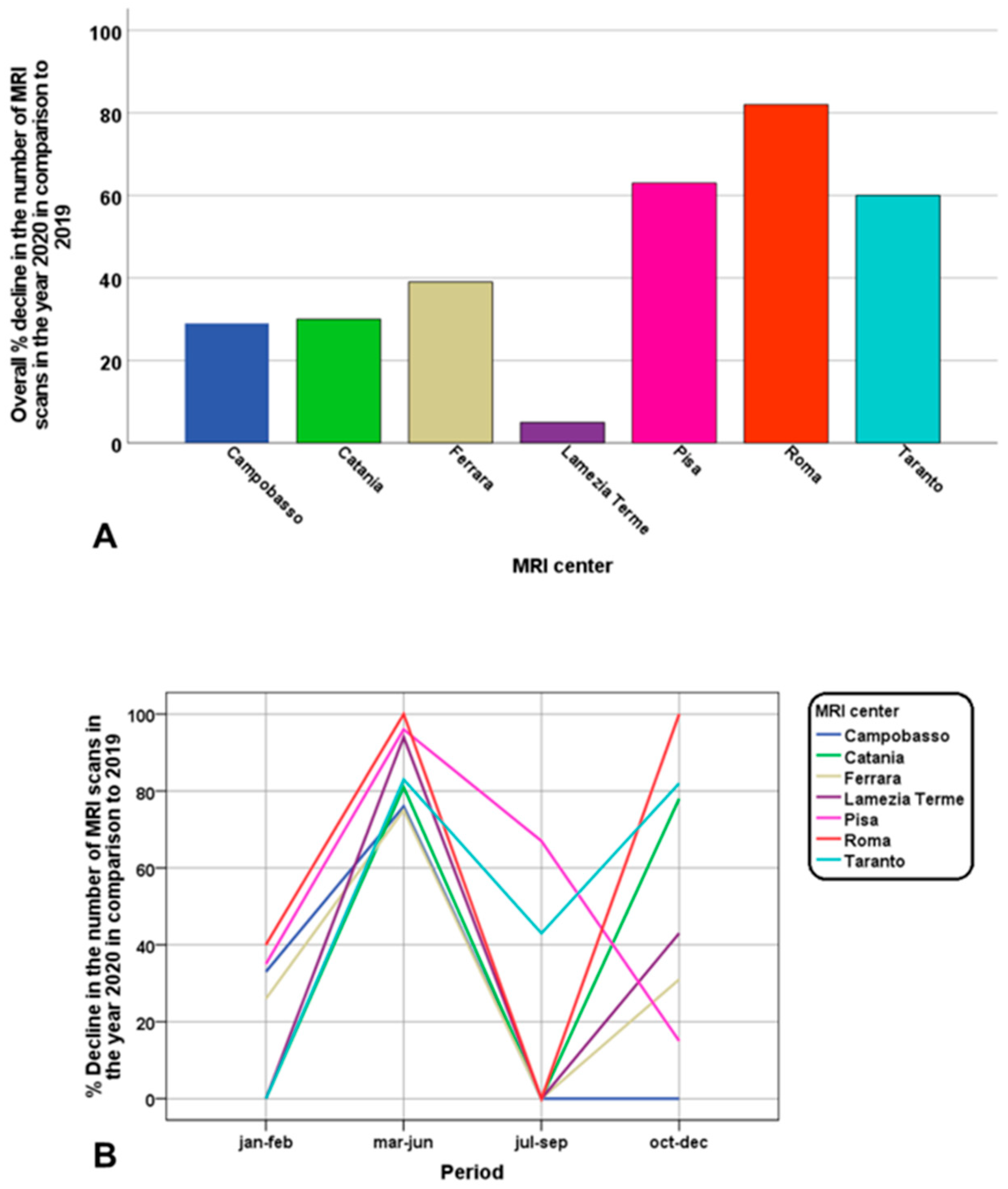
| MRI Site | Period | Number of MRI Scans in 2019 | Number of MRI Scans in 2020 |
|---|---|---|---|
| Campobasso | Entire year | 56 | 40 |
| January–February | 9 | 6 | |
| March–June | 25 | 6 | |
| July–September | 8 | 12 | |
| October–December | 14 | 16 | |
| Catania | Entire year | 74 | 52 |
| January–February | 17 | 21 | |
| March–June | 16 | 3 | |
| July–September | 18 | 23 | |
| October–December | 23 | 5 | |
| Ferrara | Entire year | 202 | 124 |
| January–February | 42 | 31 | |
| March–June | 69 | 17 | |
| July–September | 43 | 43 | |
| October–December | 48 | 33 | |
| Lamezia Terme | Entire year | 43 | 41 |
| January–February | 3 | 12 | |
| March–June | 17 | 1 | |
| July–September | 2 | 16 | |
| October–December | 21 | 12 | |
| Pisa | Entire year | 168 | 62 |
| January–February | 26 | 17 | |
| March–June | 69 | 3 | |
| July–September | 39 | 13 | |
| October–December | 34 | 29 | |
| Roma | Entire year | 65 | 12 |
| January–February | 20 | 12 | |
| March–June | 38 | 0 | |
| July–September | 0 | 0 | |
| October–December | 7 | 0 | |
| Taranto | Entire year | 48 | 19 |
| January–February | 1 | 5 | |
| March–June | 12 | 2 | |
| July–September | 14 | 8 | |
| October–December | 22 | 4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meloni, A.; Pistoia, L.; Lupi, A.; Righi, R.; Vallone, A.; Missere, M.; Renne, S.; Fina, P.; Riva, A.; Gamberini, M.R.; et al. Impact of the COVID-19 Pandemic on Iron Overload Assessment by MRI in Patients with Hemoglobinopathies: The E-MIOT Network Experience. Tomography 2023, 9, 1711-1722. https://doi.org/10.3390/tomography9050136
Meloni A, Pistoia L, Lupi A, Righi R, Vallone A, Missere M, Renne S, Fina P, Riva A, Gamberini MR, et al. Impact of the COVID-19 Pandemic on Iron Overload Assessment by MRI in Patients with Hemoglobinopathies: The E-MIOT Network Experience. Tomography. 2023; 9(5):1711-1722. https://doi.org/10.3390/tomography9050136
Chicago/Turabian StyleMeloni, Antonella, Laura Pistoia, Amalia Lupi, Riccardo Righi, Antonino Vallone, Massimiliano Missere, Stefania Renne, Priscilla Fina, Ada Riva, Maria Rita Gamberini, and et al. 2023. "Impact of the COVID-19 Pandemic on Iron Overload Assessment by MRI in Patients with Hemoglobinopathies: The E-MIOT Network Experience" Tomography 9, no. 5: 1711-1722. https://doi.org/10.3390/tomography9050136
APA StyleMeloni, A., Pistoia, L., Lupi, A., Righi, R., Vallone, A., Missere, M., Renne, S., Fina, P., Riva, A., Gamberini, M. R., Cecinati, V., Sorrentino, F., Rosso, R., Messina, G., Ricchi, P., Positano, V., Mavrogeni, S., Quaia, E., Cademartiri, F., & Pepe, A. (2023). Impact of the COVID-19 Pandemic on Iron Overload Assessment by MRI in Patients with Hemoglobinopathies: The E-MIOT Network Experience. Tomography, 9(5), 1711-1722. https://doi.org/10.3390/tomography9050136













