Feasibility of Early Evaluation for the Recurrence of Bladder Cancer after Trans-Urethral Resection: A Comparison between Magnetic Resonance Imaging and Multidetector Computed Tomography
Abstract
1. Introduction
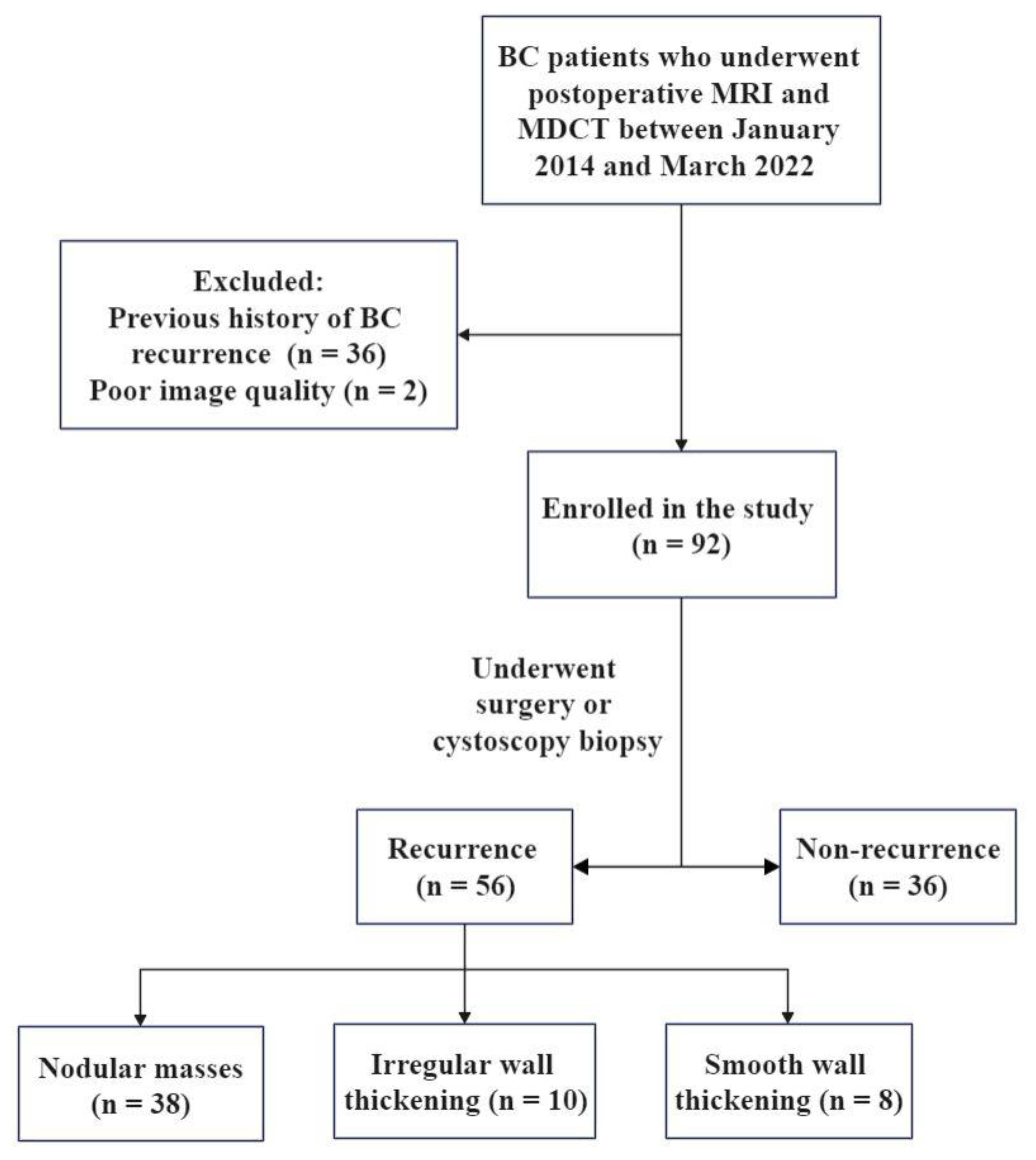
2. Materials and Methods
2.1. Subjects
2.2. Patient Preparation
2.3. Equipment and Parameters
2.4. Image Analysis
2.5. Reference Standard
2.6. Statistical Analysis
3. Results
3.1. Clinical Characteristics
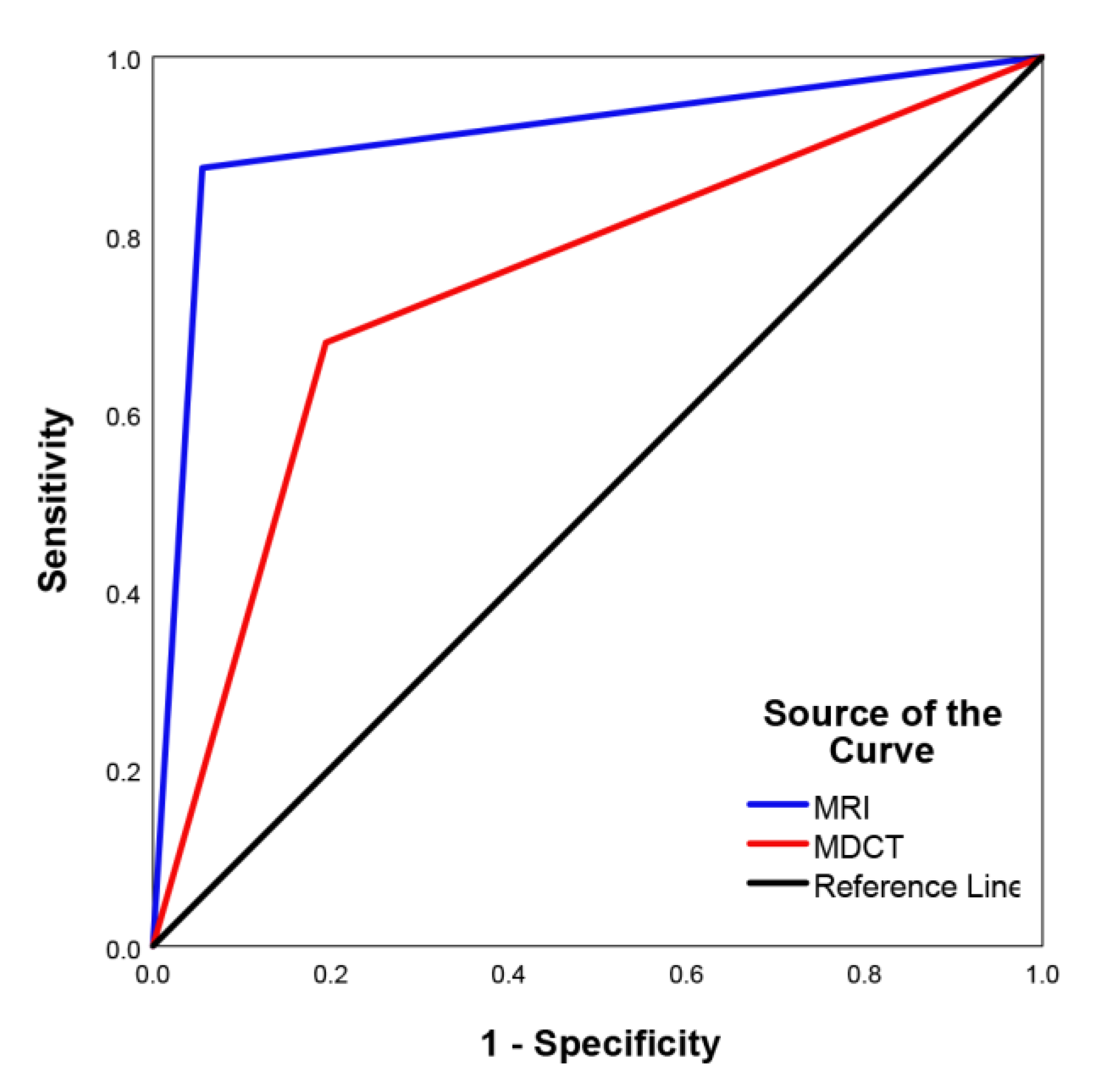
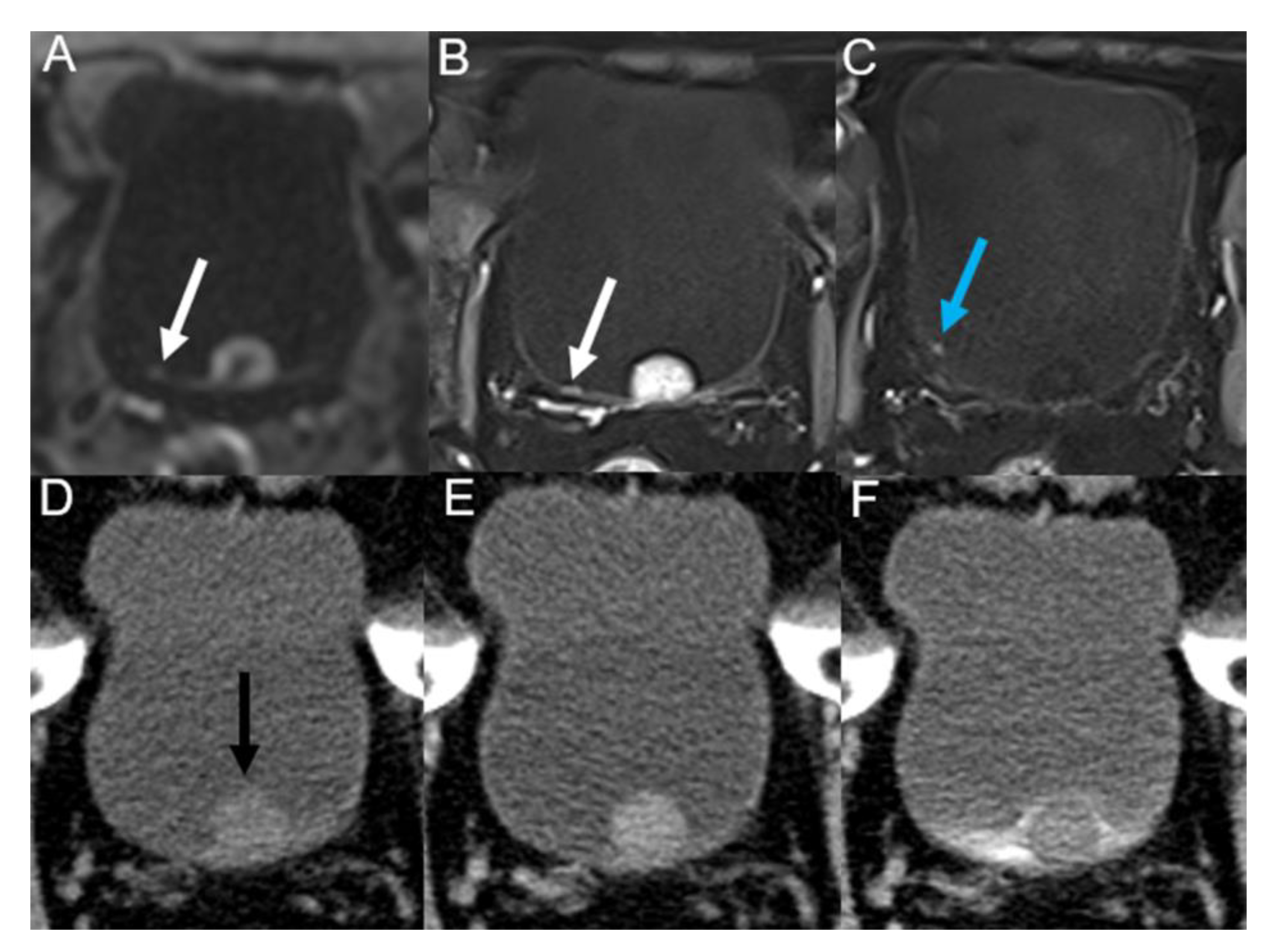
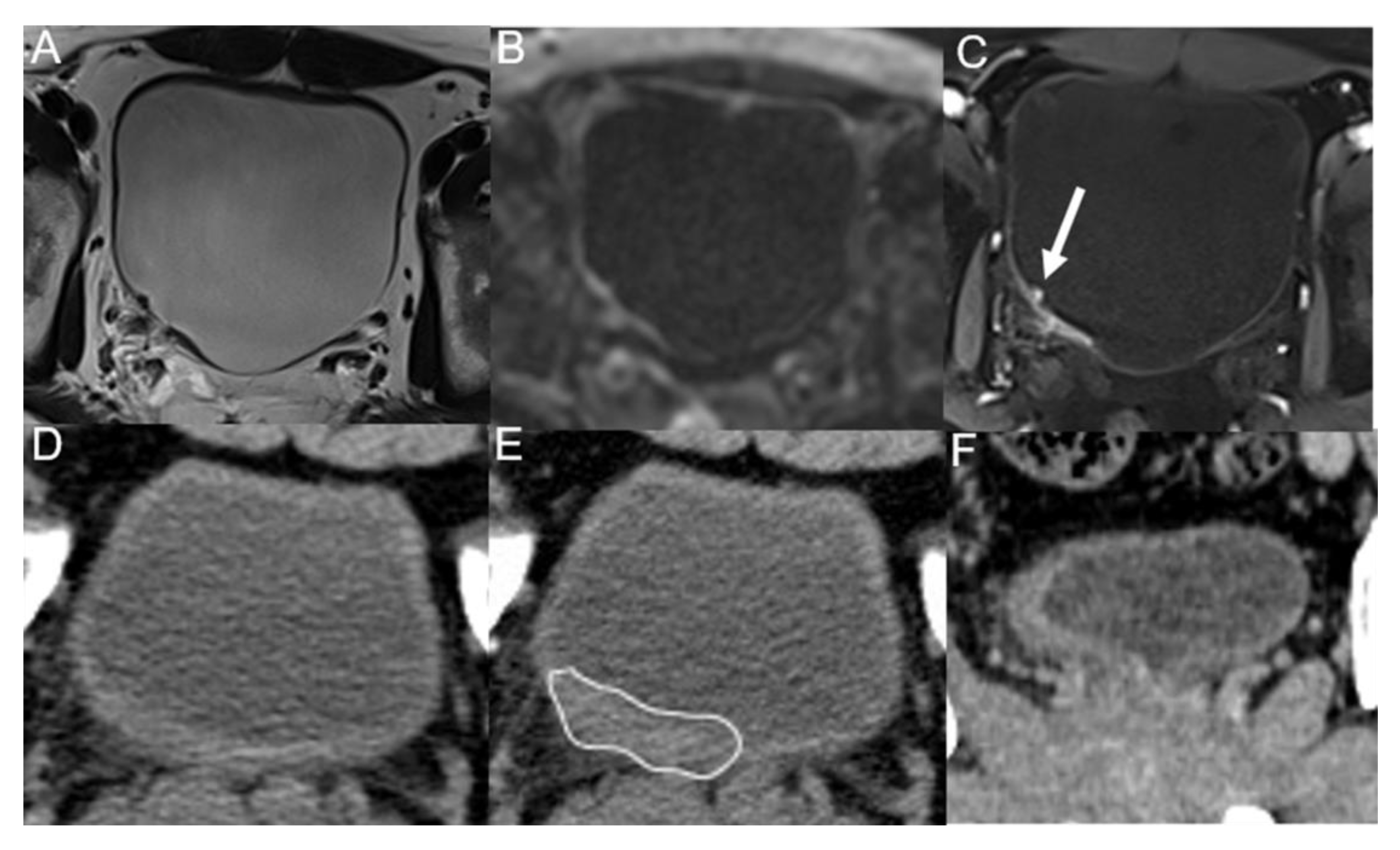
3.2. Imaging Diagnostic Performance
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sylvester, R.J.; van der Meijden, A.P.; Oosterlinck, W.; Witjes, J.A.; Bouffioux, C.; Denis, L.; Newling, D.W.; Kurth, K. Predicting recurrence and progression in individual patients with stage Ta T1 bladder cancer using EORTC risk tables: A combined analysis of 2596 patients from seven EORTC trials. Eur. Urol. 2006, 49, 465–466; discussion 467–475. [Google Scholar] [CrossRef] [PubMed]
- Witjes, J.A.; Bruins, H.M.; Cathomas, R.; Compérat, E.M.; Cowan, N.C.; Gakis, G.; Hernández, V.; Linares Espinós, E.; Lorch, A.; Neuzillet, Y.; et al. European Association of Urology Guidelines on Muscle-invasive and Metastatic Bladder Cancer: Summary of the 2020 Guidelines. Eur. Urol. 2021, 79, 82–104. [Google Scholar] [CrossRef] [PubMed]
- Xylinas, E.; Kent, M.; Kluth, L.; Pycha, A.; Comploj, E.; Svatek, R.S.; Lotan, Y.; Trinh, Q.D.; Karakiewicz, P.I.; Holmang, S.; et al. Accuracy of the EORTC risk tables and of the CUETO scoring model to predict outcomes in non-muscle-invasive urothelial carcinoma of the bladder. Br. J. Cancer 2013, 109, 1460–1466. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Wang, H.; Guo, Y.; Zhang, X.; Li, B.; Du, P.; Liu, Y.; Lu, H. Study Progress of Noninvasive Imaging and Radiomics for Decoding the Phenotypes and Recurrence Risk of Bladder Cancer. Front. Oncol. 2021, 11, 704039. [Google Scholar] [CrossRef] [PubMed]
- Barak, V.; Itzkovich, D.; Einarsson, R.; Gofrit, O.; Pode, D. Non-invasive Detection of Bladder Cancer by UBC Rapid Test, Ultrasonography and Cytology. Anticancer Res. 2020, 40, 3967–3972. [Google Scholar] [CrossRef] [PubMed]
- Messer, J.; Shariat, S.F.; Brien, J.C.; Herman, M.P.; Ng, C.K.; Scherr, D.S.; Scoll, B.; Uzzo, R.G.; Wille, M.; Eggener, S.E.; et al. Urinary cytology has a poor performance for predicting invasive or high-grade upper-tract urothelial carcinoma. BJU Int. 2011, 108, 701–705. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.J.; Wong, Y.C.; Ng, K.F.; Chuang, C.K.; Lee, S.Y.; Wan, Y.L. Tumor characteristics of urothelial carcinoma on multidetector computerized tomography urography. J. Urol. 2010, 183, 2154–2160. [Google Scholar] [CrossRef] [PubMed]
- Sadow, C.A.; Wheeler, S.C.; Kim, J.; Ohno-Machado, L.; Silverman, S.G. Positive predictive value of CT urography in the evaluation of upper tract urothelial cancer. AJR Am. J. Roentgenol. 2010, 195, W337–W343. [Google Scholar] [CrossRef] [PubMed]
- Azam, A.; Rottenberg, G. Multimodality Imaging Findings Postcystectomy: Postoperative Anatomy, Surgical Complications, and Surveillance Imaging. Semin. Ultrasound CT MR 2020, 41, 392–401. [Google Scholar] [CrossRef] [PubMed]
- Panebianco, V.; Narumi, Y.; Altun, E.; Bochner, B.H.; Efstathiou, J.A.; Hafeez, S.; Huddart, R.; Kennish, S.; Lerner, S.; Montironi, R.; et al. Multiparametric Magnetic Resonance Imaging for Bladder Cancer: Development of VI-RADS (Vesical Imaging-Reporting And Data System). Eur. Urol. 2018, 74, 294–306. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.J.; Pui, M.H.; Guo, Y.; Yang, D.; Pan, B.T.; Zhou, X.H. Diffusion-weighted MRI in bladder carcinoma: The differentiation between tumor recurrence and benign changes after resection. Abdom. Radiol. 2014, 39, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Liu, L.; Ding, L.; Zhang, Z.; Zhang, M. Quantitative Assessment of Bladder Cancer Reflects Grade and Recurrence: Comparing of Three Methods of Positioning Region of Interest for ADC Measurements at Diffusion-weighted MR Imaging. Acad. Radiol. 2019, 26, 1148–1153. [Google Scholar] [CrossRef] [PubMed]
- Rosenkrantz, A.B.; Ego-Osuala, I.O.; Khalef, V.; Deng, F.M.; Taneja, S.S.; Huang, W.C. Investigation of Multisequence Magnetic Resonance Imaging for Detection of Recurrent Tumor After Transurethral Resection for Bladder Cancer. J. Comput. Assist. Tomogr. 2016, 40, 201–205. [Google Scholar] [CrossRef] [PubMed]
- Zattoni, F.; Incerti, E.; Colicchia, M.; Castellucci, P.; Panareo, S.; Picchio, M.; Fallanca, F.; Briganti, A.; Moschini, M.; Gallina, A.; et al. Comparison between the diagnostic accuracies of 18F-fluorodeoxyglucose positron emission tomography/computed tomography and conventional imaging in recurrent urothelial carcinomas: A retrospective, multicenter study. Abdom. Radiol. 2018, 43, 2391–2399. [Google Scholar] [CrossRef] [PubMed]
- Park, J.J.; Park, B.K. The utility of CT and MRI in detecting male urethral recurrence after radical cystectomy. Abdom. Radiol. 2017, 42, 2521–2526. [Google Scholar] [CrossRef] [PubMed]
- El-Assmy, A.; Abou-El-Ghar, M.E.; Refaie, H.F.; Mosbah, A.; El-Diasty, T. Diffusion-weighted magnetic resonance imaging in follow-up of superficial urinary bladder carcinoma after transurethral resection: Initial experience. BJU Int. 2012, 110, E622–E627. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, Y.; Yoshida, S.; Tanaka, H.; Inoue, M.; Ito, M.; Kijima, T.; Yokoyama, M.; Ishioka, J.; Matsuoka, Y.; Saito, K.; et al. Potential Utility of Diffusion-Weighted Magnetic Resonance Imaging in Diagnosis of Residual Bladder Cancer before Second Transurethral Resection. Urol. Int. 2017, 98, 298–303. [Google Scholar] [CrossRef]
- Caglic, I.; Panebianco, V.; Vargas, H.A.; Bura, V.; Woo, S.; Pecoraro, M.; Cipollari, S.; Sala, E.; Barrett, T. MRI of Bladder Cancer: Local and Nodal Staging. J. Magn. Reson. Imaging 2020, 52, 649–667. [Google Scholar] [CrossRef]
- Shinagare, A.B.; Sadow, C.A.; Silverman, S.G. Surveillance of patients with bladder cancer following cystectomy: Yield of CT urography. Abdom. Radiol. 2013, 38, 1415–1421. [Google Scholar] [CrossRef] [PubMed]

| Parameter | T1WI | T2WI | DWI | DCE |
|---|---|---|---|---|
| plane | axial | axial, sagittal and coronal | axial and sagittal (or coronal) | axial, sagittal and coronal |
| echo time (ms) | 2.78/1.36 (in/opp) | 85 | 50 | 1.31 |
| repetition time (ms) | 4.48 | 4280 | 7000 | 3.16 |
| field of view (mm2) | 380 × 308 | 240 × 240 | 240 × 211 | 280 × 280 |
| matrix | 182 × 320 | 275 × 320 | 100 × 88 | 224 × 320 |
| slice thickness (mm) | 3.5 | 3.5 | 3.5 | 3.5 |
| slice gap (mm) | 0.4 | 0.4 | 0.4 | 0.4 |
| b-value (s/mm2) | 50, 600, 1200 | |||
| average | 1 | 1 | 2, 3, 4 | 1 |
| total acquisition time | 16 s | 5 min 2 s | 7 min 28 s | 5 min 45 s |
| Variables | Total | Recurrence | Non-Recurrence |
|---|---|---|---|
| Case, n (%) | 92 | 56 (60.9) | 36 (39.1) |
| Age, mean ± SD (range), years | 66.5 ± 12.1 (29–89) | 68.0 ± 11.6 (43–89) | 64.2 ± 12.6 (29–83) |
| Gender, n (%) | |||
| Male | 77 (83.7) | 47 (83.9) | 30 (83.3) |
| Female | 15 (16.3) | 9 (16.1) | 6 (16.7) |
| Follow-up time, median (IQR), months | 25.5 (11–50.5) | 35 (18.8–65.2) | 15 (5–27.5) |
| Tumor size, n (%) | |||
| ≥1 cm | 49 (53.3) | 37 (66.1) | 12 (33.3) |
| <1 cm | 43 (46.7) | 19 (33.9) | 24 (66.7) |
| Lesion multiplicity | |||
| Solitary | 58 (63.0) | 30 (53.6) | 28 (77.8) |
| Multiple | 34 (37.0) | 26 (46.4) | 8 (22.2) |
| Intravesical perfusion therapy | |||
| Yes | 62 (67.4) | 35 (62.5) | 27 (75.0) |
| No | 30 (32.6) | 21 (37.5) | 9 (25.0) |
| MRI | MDCT | |
|---|---|---|
| Sensitivity | 87.5% (49/56) | 67.9% (38/56) |
| Specificity | 94.4% (34/36) | 80.6% (29/36) |
| False-positive rate | 5.6% (2/36) | 19.4% (7/36) |
| False-negative rate | 12.5% (7/56) | 32.1% (18/56) |
| Accuracy | 90.2% (83/92) | 72.8% (67/92) |
| x2 | 1.78 | 4.00 |
| p | 0.18 | 0.04 |
| Morphological Classification | MRI | MDCT |
|---|---|---|
| Nodular masses | 100% (38/38) | 92.1% (35/38) |
| Irregular wall thickening | 90.0% (9/10) | 30.0% (3/10) |
| Smooth wall thickening | 25.0% (2/8) | 0% (0/8) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Zhang, W.; Xiao, W.; Chen, S.; Wei, Y.; Luo, M. Feasibility of Early Evaluation for the Recurrence of Bladder Cancer after Trans-Urethral Resection: A Comparison between Magnetic Resonance Imaging and Multidetector Computed Tomography. Tomography 2023, 9, 25-35. https://doi.org/10.3390/tomography9010003
Wang Y, Zhang W, Xiao W, Chen S, Wei Y, Luo M. Feasibility of Early Evaluation for the Recurrence of Bladder Cancer after Trans-Urethral Resection: A Comparison between Magnetic Resonance Imaging and Multidetector Computed Tomography. Tomography. 2023; 9(1):25-35. https://doi.org/10.3390/tomography9010003
Chicago/Turabian StyleWang, Yiqian, Wei Zhang, Weixiong Xiao, Shaobin Chen, Yongbao Wei, and Min Luo. 2023. "Feasibility of Early Evaluation for the Recurrence of Bladder Cancer after Trans-Urethral Resection: A Comparison between Magnetic Resonance Imaging and Multidetector Computed Tomography" Tomography 9, no. 1: 25-35. https://doi.org/10.3390/tomography9010003
APA StyleWang, Y., Zhang, W., Xiao, W., Chen, S., Wei, Y., & Luo, M. (2023). Feasibility of Early Evaluation for the Recurrence of Bladder Cancer after Trans-Urethral Resection: A Comparison between Magnetic Resonance Imaging and Multidetector Computed Tomography. Tomography, 9(1), 25-35. https://doi.org/10.3390/tomography9010003





