Non-Invasive Modalities in the Assessment of Vulnerable Coronary Atherosclerotic Plaques
Abstract
:1. Introduction
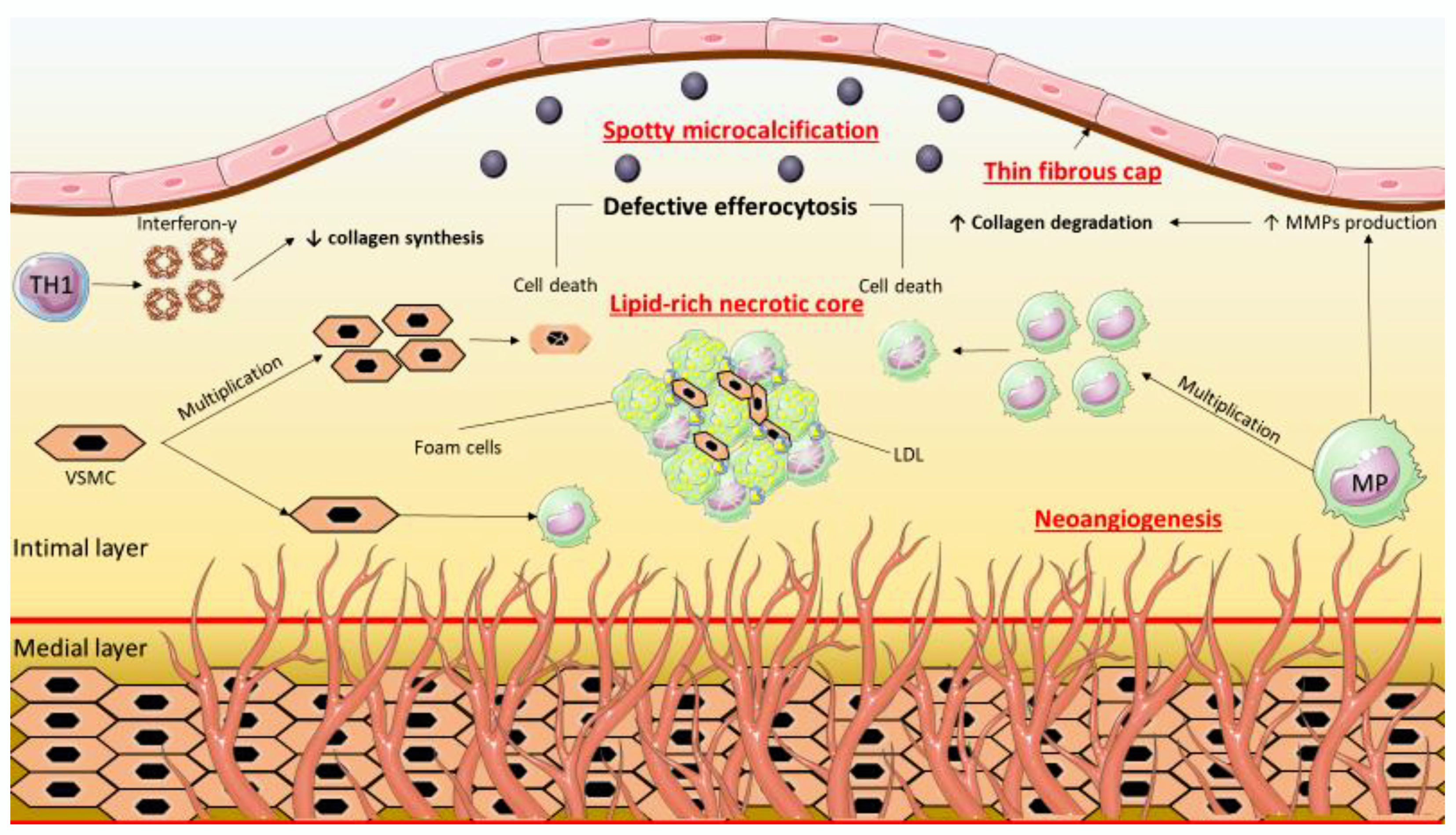
2. Features of the Vulnerable Coronary Atherosclerotic Plaque
3. Clinical Significance of Vulnerable Coronary Atherosclerotic Plaque
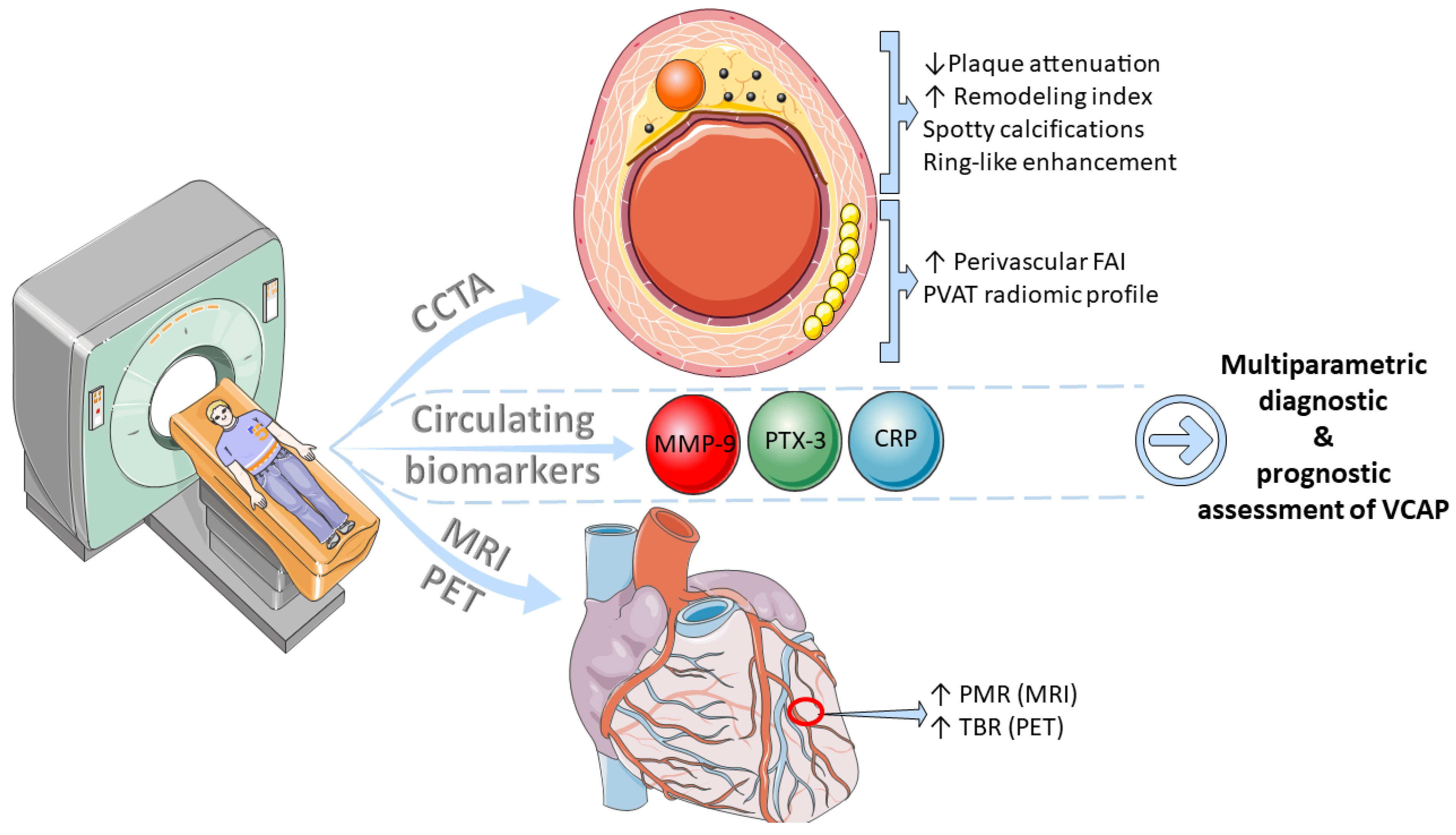
4. Non-Invasive Assessment of the Vulnerable Coronary Atherosclerotic Plaque
4.1. Circulating Biomarkers
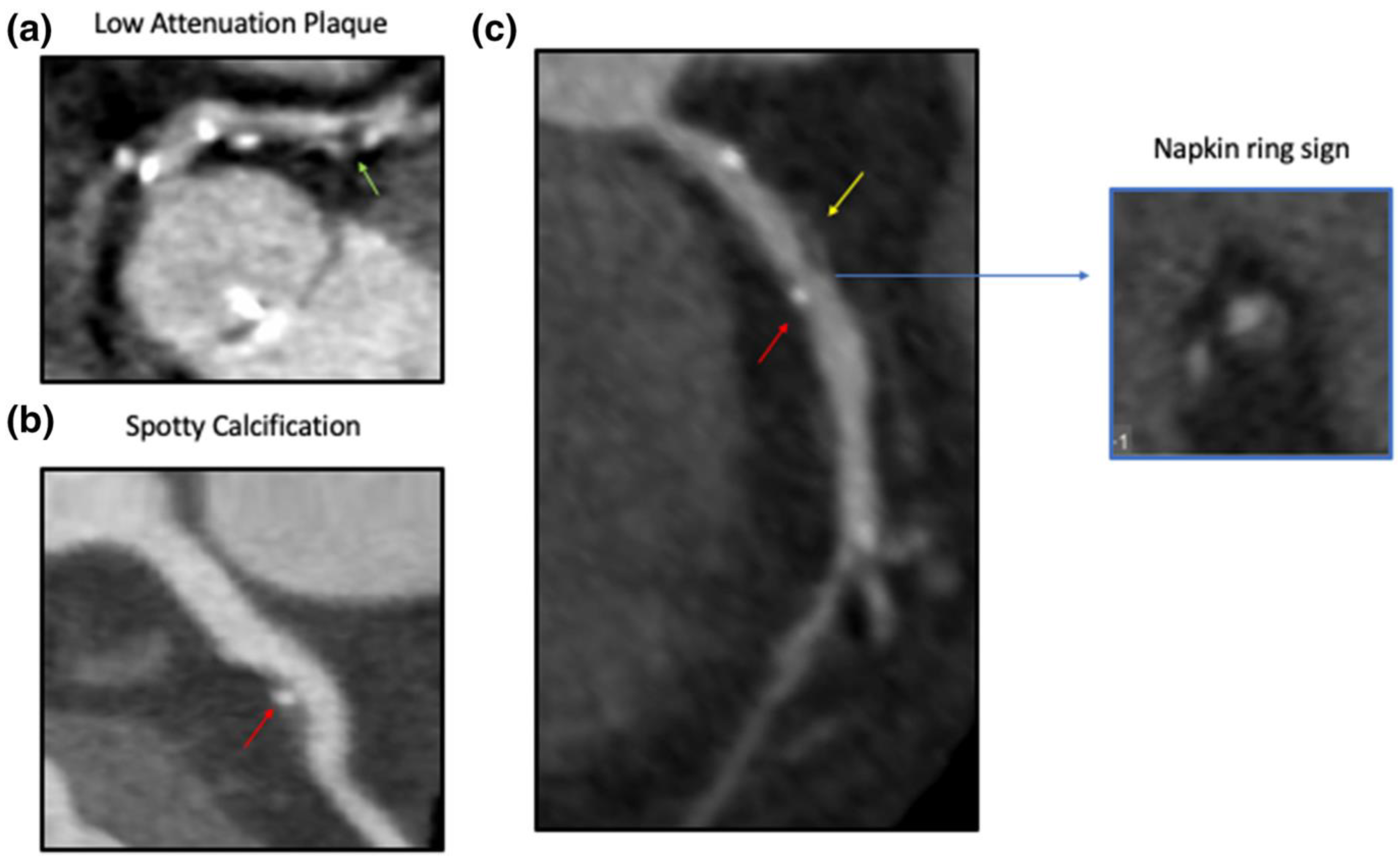
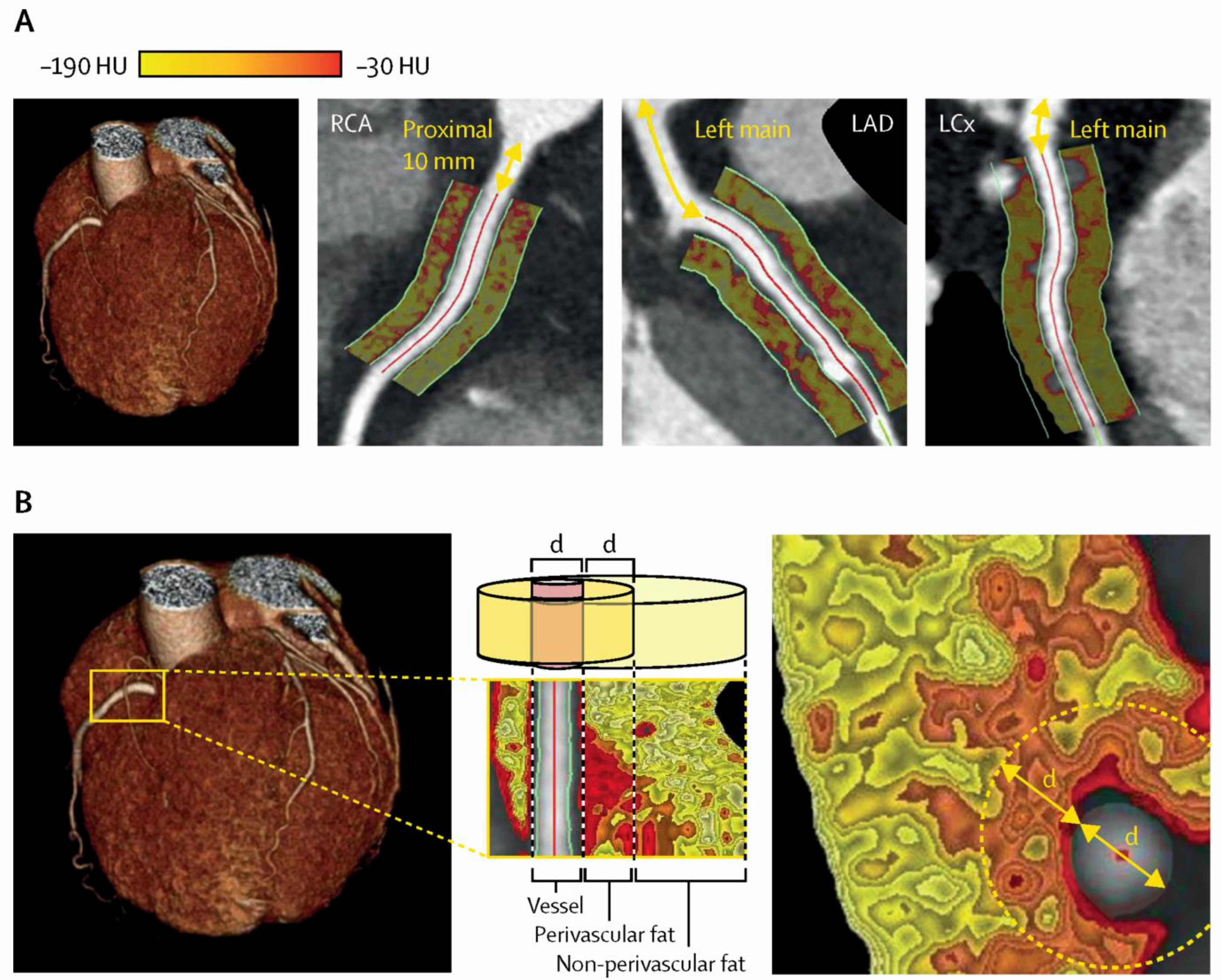
4.2. Computed Tomography Coronary Angiography
4.3. Magnetic Resonance Imaging
4.4. Nuclear Imaging
5. Non-Invasive Assessment of VCAP: Current State
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Steinberg, D.; Witztum, J.L. Oxidized low-density lipoprotein and atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2010, 30, 2311–2316. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gimbrone, M.A., Jr.; Garcia-Cardena, G. Endothelial Cell Dysfunction and the Pathobiology of Atherosclerosis. Circ. Res. 2016, 118, 620–636. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Theofilis, P.; Sagris, M.; Oikonomou, E.; Antonopoulos, A.S.; Siasos, G.; Tsioufis, C.; Tousoulis, D. Inflammatory Mechanisms Contributing to Endothelial Dysfunction. Biomedicines. 2021, 9, 781. [Google Scholar] [CrossRef] [PubMed]
- Sagris, M.; Theofilis, P.; Antonopoulos, A.S.; Tsioufis, C.; Oikonomou, E.; Antoniades, C.; Crea, F.; Kaski, J.C.; Tousoulis, D. Inflammatory Mechanisms in COVID-19 and Atherosclerosis: Current Pharmaceutical Perspectives. Int. J. Mol. Sci. 2021, 22, 6607. [Google Scholar] [CrossRef] [PubMed]
- Nowak, W.N.; Deng, J.; Ruan, X.Z.; Xu, Q. Reactive Oxygen Species Generation and Atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2017, 37, e41–e52. [Google Scholar] [CrossRef]
- Oikonomou, E.; Leopoulou, M.; Theofilis, P.; Antonopoulos, A.S.; Siasos, G.; Latsios, G.; Mystakidi, V.C.; Antoniades, C.; Tousoulis, D. A link between inflammation and thrombosis in atherosclerotic cardiovascular diseases: Clinical and therapeutic implications. Atherosclerosis 2020, 309, 16–26. [Google Scholar] [CrossRef]
- Miano, J.M.; Fisher, E.A.; Majesky, M.W. Fate and State of Vascular Smooth Muscle Cells in Atherosclerosis. Circulation 2021, 143, 2110–2116. [Google Scholar] [CrossRef]
- Gomez, D.; Owens, G.K. Smooth muscle cell phenotypic switching in atherosclerosis. Cardiovasc. Res. 2012, 95, 156–164. [Google Scholar] [CrossRef] [Green Version]
- Shankman, L.S.; Gomez, D.; Cherepanova, O.A.; Salmon, M.; Alencar, G.F.; Haskins, R.M.; Swiatlowska, P.; Newman, A.A.; Greene, E.S.; Straub, A.C.; et al. KLF4-dependent phenotypic modulation of smooth muscle cells has a key role in atherosclerotic plaque pathogenesis. Nat. Med. 2015, 21, 628–637. [Google Scholar] [CrossRef] [Green Version]
- Kolodgie, F.D.; Burke, A.P.; Farb, A.; Gold, H.K.; Yuan, J.; Narula, J.; Finn, A.V.; Virmani, R. The thin-cap fibroatheroma: A type of vulnerable plaque: The major precursor lesion to acute coronary syndromes. Curr. Opin. Cardiol. 2001, 16, 285–292. [Google Scholar] [CrossRef]
- Shi, X.; Gao, J.; Lv, Q.; Cai, H.; Wang, F.; Ye, R.; Liu, X. Calcification in Atherosclerotic Plaque Vulnerability: Friend or Foe? Front. Physiol. 2020, 11, 56. [Google Scholar] [CrossRef] [PubMed]
- Michel, J.B.; Virmani, R.; Arbustini, E.; Pasterkamp, G. Intraplaque haemorrhages as the trigger of plaque vulnerability. Eur. Heart. J. 2011, 32, 1977–1985. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stone, G.W.; Maehara, A.; Lansky, A.J.; de Bruyne, B.; Cristea, E.; Mintz, G.S.; Mehran, R.; McPherson, J.; Farhat, N.; Marso, S.P.; et al. A prospective natural-history study of coronary atherosclerosis. N. Engl. J. Med. 2011, 364, 226–235. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Fang, C.; Jiang, S.; Wang, J.; Wang, Y.; Guo, J.; Lei, F.; Sun, S.; Pei, X.; Jia, R.; et al. Optical Coherence Tomographic Features of Pancoronary Plaques in Patients with Acute Myocardial Infarction Caused by Layered Plaque Rupture Versus Layered Plaque Erosion. Am. J. Cardiol. 2022, 167, 35–42. [Google Scholar] [CrossRef]
- Pinilla-Echeverri, N.; Mehta, S.R.; Wang, J.; Lavi, S.; Schampaert, E.; Cantor, W.J.; Bainey, K.R.; Welsh, R.C.; Kassam, S.; Mehran, R.; et al. Nonculprit Lesion Plaque Morphology in Patients With ST-Segment-Elevation Myocardial Infarction: Results from the COMPLETE Trial Optical Coherence Tomography Substudys. Circ. Cardiovasc. Interv. 2020, 13, e008768. [Google Scholar] [CrossRef]
- Cao, M.; Zhao, L.; Ren, X.; Wu, T.; Yang, G.; Du, Z.; Yu, H.; Dai, J.; Li, L.; Wang, Y.; et al. Pancoronary Plaque Characteristics in STEMI Caused by Culprit Plaque Erosion Versus Rupture: 3-Vessel OCT Study. JACC Cardiovasc. Imaging 2021, 14, 1235–1245. [Google Scholar] [CrossRef]
- Fang, C.; Yin, Y.; Jiang, S.; Zhang, S.; Wang, J.; Wang, Y.; Li, L.; Wang, Y.; Guo, J.; Yu, H.; et al. Increased Vulnerability and Distinct Layered Phenotype at Culprit and Nonculprit Lesions in STEMI Versus NSTEMI. JACC Cardiovasc. Imaging 2021, 15, 672–681. [Google Scholar] [CrossRef]
- Kedhi, E.; Berta, B.; Roleder, T.; Hermanides, R.S.; Fabris, E.; AJJ, I.J.; Kauer, F.; Alfonso, F.; von Birgelen, C.; Escaned, J.; et al. Thin-cap fibroatheroma predicts clinical events in diabetic patients with normal fractional flow reserve: The COMBINE OCT-FFR trial. Eur. Heart J. 2021, 42, 4671–4679. [Google Scholar] [CrossRef]
- Mehta, S.R.; Wood, D.A.; Storey, R.F.; Mehran, R.; Bainey, K.R.; Nguyen, H.; Meeks, B.; Di Pasquale, G.; Lopez-Sendon, J.; Faxon, D.P.; et al. Complete Revascularization with Multivessel PCI for Myocardial Infarction. N. Engl. J. Med. 2019, 381, 1411–1421. [Google Scholar] [CrossRef] [Green Version]
- Stone, G.W.; Maehara, A.; Ali, Z.A.; Held, C.; Matsumura, M.; Kjoller-Hansen, L.; Botker, H.E.; Maeng, M.; Engstrom, T.; Wiseth, R.; et al. Percutaneous Coronary Intervention for Vulnerable Coronary Atherosclerotic Plaque. J. Am. Coll. Cardiol. 2020, 76, 2289–2301. [Google Scholar] [CrossRef]
- Koyama, K.; Yoneyama, K.; Mitarai, T.; Ishibashi, Y.; Takahashi, E.; Kongoji, K.; Harada, T.; Akashi, Y.J. Association between inflammatory biomarkers and thin-cap fibroatheroma detected by optical coherence tomography in patients with coronary heart disease. Arch. Med. Sci. 2015, 11, 505–512. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raffel, O.C.; Tearney, G.J.; Gauthier, D.D.; Halpern, E.F.; Bouma, B.E.; Jang, I.K. Relationship between a systemic inflammatory marker, plaque inflammation, and plaque characteristics determined by intravascular optical coherence tomography. Arterioscler. Thromb. Vasc. Biol. 2007, 27, 1820–1827. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fujii, K.; Masutani, M.; Okumura, T.; Kawasaki, D.; Akagami, T.; Ezumi, A.; Sakoda, T.; Masuyama, T.; Ohyanagi, M. Frequency and predictor of coronary thin-cap fibroatheroma in patients with acute myocardial infarction and stable angina pectoris a 3-vessel optical coherence tomography study. J. Am. Coll. Cardiol. 2008, 52, 787–788. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bouki, K.P.; Katsafados, M.G.; Chatzopoulos, D.N.; Psychari, S.N.; Toutouzas, K.P.; Charalampopoulos, A.F.; Sakkali, E.N.; Koudouri, A.A.; Liakos, G.K.; Apostolou, T.S. Inflammatory markers and plaque morphology: An optical coherence tomography study. Int. J. Cardiol. 2012, 154, 287–292. [Google Scholar] [CrossRef]
- Fracassi, F.; Niccoli, G.; Vetrugno, V.; Russo, M.; Rettura, F.; Vergni, F.; Scalone, G.; Montone, R.A.; Vergallo, R.; D’Amario, D.; et al. Optical coherence tomography and C-reactive protein in risk stratification of acute coronary syndromes. Int. J. Cardiol. 2019, 286, 7–12. [Google Scholar] [CrossRef]
- Takahashi, N.; Dohi, T.; Endo, H.; Takeuchi, M.; Doi, S.; Kato, Y.; Okai, I.; Iwata, H.; Okazaki, S.; Isoda, K.; et al. Coronary lipid-rich plaque characteristics in Japanese patients with acute coronary syndrome and stable angina: A near infrared spectroscopy and intravascular ultrasound study. Int. J. Cardiol. Heart Vasc. 2021, 33, 100747. [Google Scholar] [CrossRef]
- Koga, S.; Ikeda, S.; Yoshida, T.; Nakata, T.; Takeno, M.; Masuda, N.; Koide, Y.; Kawano, H.; Maemura, K. Elevated levels of systemic pentraxin 3 are associated with thin-cap fibroatheroma in coronary culprit lesions: Assessment by optical coherence tomography and intravascular ultrasound. JACC Cardiovasc. Interv. 2013, 6, 945–954. [Google Scholar] [CrossRef] [Green Version]
- Kimura, S.; Sugiyama, T.; Hishikari, K.; Nakagama, S.; Nakamura, S.; Misawa, T.; Mizusawa, M.; Hayasaka, K.; Yamakami, Y.; Sagawa, Y.; et al. Relationship of systemic pentraxin-3 values with coronary plaque components on optical coherence tomography and post-percutaneous coronary intervention outcomes in patients with stable angina pectoris. Atherosclerosis 2020, 292, 127–135. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Zhao, X.; Zhou, P.; Liu, C.; Sheng, Z.; Li, J.; Zhou, J.; Chen, R.; Chen, Y.; Song, L.; et al. Plasma Pentraxin-3 Combined with Plaque Characteristics Predict Cardiovascular Risk in ST-Segment Elevated Myocardial Infarction: An Optical Coherence Tomography Study. J. Inflamm. Res. 2021, 14, 4409–4419. [Google Scholar] [CrossRef]
- Siasos, G.; Tousoulis, D.; Kioufis, S.; Oikonomou, E.; Siasou, Z.; Limperi, M.; Papavassiliou, A.G.; Stefanadis, C. Inflammatory mechanisms in atherosclerosis: The impact of matrix metalloproteinases. Curr. Top Med. Chem. 2012, 12, 1132–1148. [Google Scholar] [CrossRef]
- Funayama, H.; Yoshioka, T.; Ishikawa, S.E.; Momomura, S.I.; Kario, K. Close Association of Matrix Metalloproteinase-9 Levels with the Presence of Thin-Cap Fibroatheroma in Acute Coronary Syndrome Patients: Assessment by Optical Coherence Tomography and Intravascular Ultrasonography. Cardiovasc. Revasc. Med. 2021, 32, 5–10. [Google Scholar] [CrossRef]
- Kobayashi, N.; Takano, M.; Hata, N.; Kume, N.; Tsurumi, M.; Shirakabe, A.; Okazaki, H.; Shibuya, J.; Shiomura, R.; Nishigoori, S.; et al. Matrix Metalloproteinase-9 as a Marker for Plaque Rupture and a Predictor of Adverse Clinical Outcome in Patients with Acute Coronary Syndrome: An Optical Coherence Tomography Study. Cardiology 2016, 135, 56–65. [Google Scholar] [CrossRef] [PubMed]
- Ezhov, M.; Safarova, M.; Afanasieva, O.; Mitroshkin, M.; Matchin, Y.; Pokrovsky, S. Matrix Metalloproteinase 9 as a Predictor of Coronary Atherosclerotic Plaque Instability in Stable Coronary Heart Disease Patients with Elevated Lipoprotein(a) Levels. Biomolecules 2019, 9, 129. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, R.; Fischer, R.; Charles, P.D.; Adlam, D.; Valli, A.; Di Gleria, K.; Kharbanda, R.K.; Choudhury, R.P.; Antoniades, C.; Kessler, B.M.; et al. A novel workflow combining plaque imaging, plaque and plasma proteomics identifies biomarkers of human coronary atherosclerotic plaque disruption. Clin. Proteomics 2017, 14, 22. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kook, H.; Jang, D.H.; Kim, J.H.; Cho, J.Y.; Joo, H.J.; Cho, S.A.; Park, J.H.; Hong, S.J.; Yu, C.W.; Lim, D.S. Identification of plaque ruptures using a novel discriminative model comprising biomarkers in patients with acute coronary syndrome. Sci. Rep. 2020, 10, 20228. [Google Scholar] [CrossRef]
- Antonopoulos, A.S.; Angelopoulos, A.; Papanikolaou, P.; Simantiris, S.; Oikonomou, E.K.; Vamvakaris, K.; Koumpoura, A.; Farmaki, M.; Trivella, M.; Vlachopoulos, C.; et al. Biomarkers of Vascular Inflammation for Cardiovascular Risk Prognostication: A Meta-Analysis. JACC Cardiovasc. Imaging 2021, 15, 460–471. [Google Scholar] [CrossRef]
- Theofilis, P.; Oikonomou, E.; Vogiatzi, G.; Antonopoulos, A.S.; Siasos, G.; Iliopoulos, D.C.; Perrea, D.; Tsioufis, C.; Tousoulis, D. The impact of proangiogenic microRNA modulation on blood flow recovery following hind limb ischemia. A systematic review and meta-analysis of animal studies. Vascul. Pharmacol. 2021, 141, 106906. [Google Scholar] [CrossRef]
- Theofilis, P.; Vogiatzi, G.; Oikonomou, E.; Gazouli, M.; Siasos, G.; Katifelis, H.; Perrea, D.; Vavuranakis, M.; Iliopoulos, D.C.; Tsioufis, C.; et al. The Effect of MicroRNA-126 Mimic Administration on Vascular Perfusion Recovery in an Animal Model of Hind Limb Ischemia. Front. Mol. Biosci. 2021, 8, 724465. [Google Scholar] [CrossRef]
- Taraldsen, M.D.; Wiseth, R.; Videm, V.; Bye, A.; Madssen, E. Associations between circulating microRNAs and coronary plaque characteristics: Potential impact from physical exercise. Physiol. Genomics 2022, 54, 129–140. [Google Scholar] [CrossRef]
- Pundziute, G.; Schuijf, J.D.; Jukema, J.W.; Decramer, I.; Sarno, G.; Vanhoenacker, P.K.; Boersma, E.; Reiber, J.H.; Schalij, M.J.; Wijns, W.; et al. Evaluation of plaque characteristics in acute coronary syndromes: Non-invasive assessment with multi-slice computed tomography and invasive evaluation with intravascular ultrasound radiofrequency data analysis. Eur. Heart J. 2008, 29, 2373–2381. [Google Scholar] [CrossRef] [Green Version]
- Daghem, M.; Newby, D.E. Detecting unstable plaques in humans using cardiac CT: Can it guide treatments? Br. J. Pharmacol. 2021, 178, 2204–2217. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van Velzen, J.E.; de Graaf, F.R.; de Graaf, M.A.; Schuijf, J.D.; Kroft, L.J.; de Roos, A.; Reiber, J.H.; Bax, J.J.; Jukema, J.W.; Boersma, E.; et al. Comprehensive assessment of spotty calcifications on computed tomography angiography: Comparison to plaque characteristics on intravascular ultrasound with radiofrequency backscatter analysis. J. Nucl. Cardiol. 2011, 18, 893–903. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pundziute, G.; Schuijf, J.D.; Jukema, J.W.; Decramer, I.; Sarno, G.; Vanhoenacker, P.K.; Reiber, J.H.; Schalij, M.J.; Wijns, W.; Bax, J.J. Head-to-head comparison of coronary plaque evaluation between multislice computed tomography and intravascular ultrasound radiofrequency data analysis. JACC Cardiovasc. Interv. 2008, 1, 176–182. [Google Scholar] [CrossRef] [Green Version]
- Kashiwagi, M.; Tanaka, A.; Kitabata, H.; Tsujioka, H.; Kataiwa, H.; Komukai, K.; Tanimoto, T.; Takemoto, K.; Takarada, S.; Kubo, T.; et al. Feasibility of noninvasive assessment of thin-cap fibroatheroma by multidetector computed tomography. JACC Cardiovasc. Imaging 2009, 2, 1412–1419. [Google Scholar] [CrossRef] [PubMed]
- Ito, T.; Terashima, M.; Kaneda, H.; Nasu, K.; Matsuo, H.; Ehara, M.; Kinoshita, Y.; Kimura, M.; Tanaka, N.; Habara, M.; et al. Comparison of in vivo assessment of vulnerable plaque by 64-slice multislice computed tomography versus optical coherence tomography. Am. J. Cardiol. 2011, 107, 1270–1277. [Google Scholar] [CrossRef] [PubMed]
- Otsuka, K.; Fukuda, S.; Tanaka, A.; Nakanishi, K.; Taguchi, H.; Yoshikawa, J.; Shimada, K.; Yoshiyama, M. Napkin-ring sign on coronary CT angiography for the prediction of acute coronary syndrome. JACC Cardiovasc. Imaging 2013, 6, 448–457. [Google Scholar] [CrossRef] [Green Version]
- Tomizawa, N.; Yamamoto, K.; Inoh, S.; Nojo, T.; Nakamura, S. Accuracy of computed tomography angiography to identify thin-cap fibroatheroma detected by optical coherence tomography. J. Cardiovasc. Comput. Tomogr. 2017, 11, 129–134. [Google Scholar] [CrossRef]
- Obaid, D.R.; Calvert, P.A.; Gopalan, D.; Parker, R.A.; Hoole, S.P.; West, N.E.; Goddard, M.; Rudd, J.H.; Bennett, M.R. Atherosclerotic plaque composition and classification identified by coronary computed tomography: Assessment of computed tomography-generated plaque maps compared with virtual histology intravascular ultrasound and histology. Circ. Cardiovasc. Imaging 2013, 6, 655–664. [Google Scholar] [CrossRef]
- Obaid, D.R.; Calvert, P.A.; Brown, A.; Gopalan, D.; West, N.E.J.; Rudd, J.H.F.; Bennett, M.R. Coronary CT angiography features of ruptured and high-risk atherosclerotic plaques: Correlation with intra-vascular ultrasound. J. Cardiovasc. Comput. Tomogr. 2017, 11, 455–461. [Google Scholar] [CrossRef] [Green Version]
- Ito, T.; Nasu, K.; Terashima, M.; Ehara, M.; Kinoshita, Y.; Ito, T.; Kimura, M.; Tanaka, N.; Habara, M.; Tsuchikane, E.; et al. The impact of epicardial fat volume on coronary plaque vulnerability: Insight from optical coherence tomography analysis. Eur. Heart J. Cardiovasc. Imaging 2012, 13, 408–415. [Google Scholar] [CrossRef] [Green Version]
- Yuan, M.; Wu, H.; Li, R.; Yu, L.; Zhang, J. Epicardial adipose tissue characteristics and CT high-risk plaque features: Correlation with coronary thin-cap fibroatheroma determined by intravascular ultrasound. Int. J. Cardiovasc. Imaging 2020, 36, 2281–2289. [Google Scholar] [CrossRef] [PubMed]
- Soeda, T.; Uemura, S.; Morikawa, Y.; Ishigami, K.; Okayama, S.; Hee, S.J.; Nishida, T.; Onoue, K.; Somekawa, S.; Takeda, Y.; et al. Diagnostic accuracy of dual-source computed tomography in the characterization of coronary atherosclerotic plaques: Comparison with intravascular optical coherence tomography. Int. J. Cardiol. 2011, 148, 313–318. [Google Scholar] [CrossRef] [PubMed]
- Yuan, M.; Wu, H.; Li, R.; Yu, M.; Dai, X.; Zhang, J. The value of quantified plaque analysis by dual-source coronary CT angiography to detect vulnerable plaques: A comparison study with intravascular ultrasound. Quant. Imaging Med. Surg. 2020, 10, 668–677. [Google Scholar] [CrossRef] [PubMed]
- Antonopoulos, A.S.; Sanna, F.; Sabharwal, N.; Thomas, S.; Oikonomou, E.K.; Herdman, L.; Margaritis, M.; Shirodaria, C.; Kampoli, A.M.; Akoumianakis, I.; et al. Detecting human coronary inflammation by imaging perivascular fat. Sci. Transl. Med. 2017, 9, eaal265. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oikonomou, E.K.; Marwan, M.; Desai, M.Y.; Mancio, J.; Alashi, A.; Hutt Centeno, E.; Thomas, S.; Herdman, L.; Kotanidis, C.P.; Thomas, K.E.; et al. Non-invasive detection of coronary inflammation using computed tomography and prediction of residual cardiovascular risk (the CRISP CT study): A post-hoc analysis of prospective outcome data. Lancet 2018, 392, 929–939. [Google Scholar] [CrossRef] [Green Version]
- Oikonomou, E.K.; Desai, M.Y.; Marwan, M.; Kotanidis, C.P.; Antonopoulos, A.S.; Schottlander, D.; Channon, K.M.; Neubauer, S.; Achenbach, S.; Antoniades, C. Perivascular Fat Attenuation Index Stratifies Cardiac Risk Associated with High-Risk Plaques in the CRISP-CT Study. J. Am. Coll. Cardiol. 2020, 76, 755–757. [Google Scholar] [CrossRef]
- Dai, X.; Yu, L.; Lu, Z.; Shen, C.; Tao, X.; Zhang, J. Serial change of perivascular fat attenuation index after statin treatment: Insights from a coronary CT angiography follow-up study. Int. J. Cardiol. 2020, 319, 144–149. [Google Scholar] [CrossRef]
- Baritussio, A.; Vacirca, F.; Ocagli, H.; Tona, F.; Pergola, V.; Motta, R.; Marcolongo, R.; Lorenzoni, G.; Gregori, D.; Iliceto, S.; et al. Assessment of Coronary Inflammation by Pericoronary Fat Attenuation Index in Clinically Suspected Myocarditis with Infarct-Like Presentation. J. Clin. Med. 2021, 10, 4200. [Google Scholar] [CrossRef]
- Bao, W.; Yang, M.; Xu, Z.; Yan, F.; Yang, Q.; Li, X.; Yang, W. Coronary Inflammation Assessed by Perivascular Fat Attenuation Index in Patients with Psoriasis: A Propensity Score-Matched Study. Dermatology 2021, 238, 1–9. [Google Scholar] [CrossRef]
- Elnabawi, Y.A.; Oikonomou, E.K.; Dey, A.K.; Mancio, J.; Rodante, J.A.; Aksentijevich, M.; Choi, H.; Keel, A.; Erb-Alvarez, J.; Teague, H.L.; et al. Association of Biologic Therapy with Coronary Inflammation in Patients With Psoriasis as Assessed by Perivascular Fat Attenuation Index. JAMA Cardiol. 2019, 4, 885–891. [Google Scholar] [CrossRef]
- Chen, Q.; Pan, T.; Yin, X.; Xu, H.; Gao, X.; Tao, X.; Zhou, L.; Xie, G.; Kong, X.; Huang, X.; et al. CT texture analysis of vulnerable plaques on optical coherence tomography. Eur. J. Radiol. 2021, 136, 109551. [Google Scholar] [CrossRef] [PubMed]
- Kolossvary, M.; Karady, J.; Kikuchi, Y.; Ivanov, A.; Schlett, C.L.; Lu, M.T.; Foldyna, B.; Merkely, B.; Aerts, H.J.; Hoffmann, U.; et al. Radiomics versus Visual and Histogram-based Assessment to Identify Atheromatous Lesions at Coronary CT Angiography: An ex Vivo Study. Radiology 2019, 293, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Kolossvary, M.; Park, J.; Bang, J.I.; Zhang, J.; Lee, J.M.; Paeng, J.C.; Merkely, B.; Narula, J.; Kubo, T.; Akasaka, T.; et al. Identification of invasive and radionuclide imaging markers of coronary plaque vulnerability using radiomic analysis of coronary computed tomography angiography. Eur. Heart J. Cardiovasc. Imaging. 2019, 20, 1250–1258. [Google Scholar] [CrossRef] [PubMed]
- Oikonomou, E.K.; Williams, M.C.; Kotanidis, C.P.; Desai, M.Y.; Marwan, M.; Antonopoulos, A.S.; Thomas, K.E.; Thomas, S.; Akoumianakis, I.; Fan, L.M.; et al. A novel machine learning-derived radiotranscriptomic signature of perivascular fat improves cardiac risk prediction using coronary CT angiography. Eur. Heart J. 2019, 40, 3529–3543. [Google Scholar] [CrossRef]
- Oikonomou, E.K.; Antonopoulos, A.S.; Schottlander, D.; Marwan, M.; Mathers, C.; Tomlins, P.; Siddique, M.; Kluner, L.V.; Shirodaria, C.; Mavrogiannis, M.C.; et al. Standardized measurement of coronary inflammation using cardiovascular computed tomography: Integration in clinical care as a prognostic medical device. Cardiovasc. Res. 2021, 117, 2677–2690. [Google Scholar] [CrossRef]
- Zheng, J.; El Naqa, I.; Rowold, F.E.; Pilgram, T.K.; Woodard, P.K.; Saffitz, J.E.; Tang, D. Quantitative assessment of coronary artery plaque vulnerability by high-resolution magnetic resonance imaging and computational biomechanics: A pilot study ex vivo. Magn. Reson. Med. 2005, 54, 1360–1368. [Google Scholar] [CrossRef] [Green Version]
- Tang, D.; Yang, C.; Zheng, J.; Woodard, P.K.; Saffitz, J.E.; Petruccelli, J.D.; Sicard, G.A.; Yuan, C. Local maximal stress hypothesis and computational plaque vulnerability index for atherosclerotic plaque assessment. Ann. Biomed. Eng. 2005, 33, 1789–1801. [Google Scholar] [CrossRef]
- Huang, X.; Yang, C.; Zheng, J.; Bach, R.; Muccigrosso, D.; Woodard, P.K.; Tang, D. Higher critical plaque wall stress in patients who died of coronary artery disease compared with those who died of other causes: A 3D FSI study based on ex vivo MRI of coronary plaques. J. Biomech. 2014, 47, 432–437. [Google Scholar] [CrossRef] [Green Version]
- Hassan, M.; Said, K.; Rizk, H.; ElMogy, F.; Donya, M.; Houseni, M.; Yacoub, M. Segmental peri-coronary epicardial adipose tissue volume and coronary plaque characteristics. Eur. Heart J. Cardiovasc. Imaging 2016, 17, 1169–1177. [Google Scholar] [CrossRef] [Green Version]
- Noguchi, T.; Kawasaki, T.; Tanaka, A.; Yasuda, S.; Goto, Y.; Ishihara, M.; Nishimura, K.; Miyamoto, Y.; Node, K.; Koga, N. High-intensity signals in coronary plaques on noncontrast T1-weighted magnetic resonance imaging as a novel determinant of coronary events. J. Am. Coll. Cardiol. 2014, 63, 989–999. [Google Scholar] [CrossRef] [Green Version]
- Hoshi, T.; Sato, A.; Akiyama, D.; Hiraya, D.; Sakai, S.; Shindo, M.; Mori, K.; Minami, M.; Aonuma, K. Coronary high-intensity plaque on T1-weighted magnetic resonance imaging and its association with myocardial injury after percutaneous coronary intervention. Eur. Heart J. 2015, 36, 1913–1922. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matsumoto, K.; Ehara, S.; Hasegawa, T.; Sakaguchi, M.; Otsuka, K.; Yoshikawa, J.; Shimada, K. Localization of Coronary High-Intensity Signals on T1-Weighted MR Imaging: Relation to Plaque Morphology and Clinical Severity of Angina Pectoris. JACC Cardiovasc. Imaging 2015, 8, 1143–1152. [Google Scholar] [CrossRef] [Green Version]
- Matsumoto, K.; Ehara, S.; Hasegawa, T.; Nishimura, S.; Shimada, K. The signal intensity of coronary culprit lesions on T1-weighted magnetic resonance imaging is directly correlated with the accumulation of vulnerable morphologies. Int. J. Cardiol. 2017, 231, 284–286. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Noguchi, T.; Tanaka, A.; Kawasaki, T.; Goto, Y.; Morita, Y.; Asaumi, Y.; Nakao, K.; Fujiwara, R.; Nishimura, K.; Miyamoto, Y.; et al. Effect of Intensive Statin Therapy on Coronary High-Intensity Plaques Detected by Noncontrast T1-Weighted Imaging: The AQUAMARINE Pilot Study. J. Am. Coll. Cardiol. 2015, 66, 245–256. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, T.; Zhao, X.; Liu, X.; Gao, J.; Zhao, S.; Li, X.; Zhou, W.; Cai, Z.; Zhang, W.; Yang, L. Evaluation of the early enhancement of coronary atherosclerotic plaque by contrast-enhanced MR angiography. Eur. J. Radiol. 2011, 80, 136–142. [Google Scholar] [CrossRef] [PubMed]
- Jansen, C.H.P.; Perera, D.; Wiethoff, A.J.; Phinikaridou, A.; Razavi, R.M.; Rinaldi, A.; Marber, M.S.; Greil, G.F.; Nagel, E.; Maintz, D.; et al. Contrast-enhanced magnetic resonance imaging for the detection of ruptured coronary plaques in patients with acute myocardial infarction. PLoS ONE 2017, 12, e0188292. [Google Scholar] [CrossRef] [Green Version]
- Engel, L.C.; Landmesser, U.; Gigengack, K.; Wurster, T.; Manes, C.; Girke, G.; Jaguszewski, M.; Skurk, C.; Leistner, D.M.; Lauten, A.; et al. Novel Approach for In Vivo Detection of Vulnerable Coronary Plaques Using Molecular 3-T CMR Imaging with an Albumin-Binding Probe. JACC Cardiovasc. Imaging 2019, 12, 297–306. [Google Scholar] [CrossRef]
- Engel, L.C.; Landmesser, U.; Abdelwahed, Y.S.; Jaguszewski, M.; Gigengack, K.; Wurster, T.H.; Skurk, C.; Manes, C.; Schuster, A.; Noutsias, M.; et al. Comprehensive multimodality characterization of hemodynamically significant and non-significant coronary lesions using invasive and noninvasive measures. PLoS ONE 2020, 15, e0228292. [Google Scholar] [CrossRef]
- Saam, T.; Rominger, A.; Wolpers, S.; Nikolaou, K.; Rist, C.; Greif, M.; Cumming, P.; Becker, A.; Foerster, S.; Reiser, M.F.; et al. Association of inflammation of the left anterior descending coronary artery with cardiovascular risk factors, plaque burden and pericardial fat volume: A PET/CT study. Eur. J. Nucl. Med. Mol. Imaging 2010, 37, 1203–1212. [Google Scholar] [CrossRef]
- Wykrzykowska, J.; Lehman, S.; Williams, G.; Parker, J.A.; Palmer, M.R.; Varkey, S.; Kolodny, G.; Laham, R. Imaging of inflamed and vulnerable plaque in coronary arteries with 18F-FDG PET/CT in patients with suppression of myocardial uptake using a low-carbohydrate, high-fat preparation. J. Nucl. Med. 2009, 50, 563–568. [Google Scholar] [CrossRef] [Green Version]
- Demeure, F.; Hanin, F.X.; Bol, A.; Vincent, M.F.; Pouleur, A.C.; Gerber, B.; Pasquet, A.; Jamar, F.; Vanoverschelde, J.L.; Vancraeynest, D. A randomized trial on the optimization of 18F-FDG myocardial uptake suppression: Implications for vulnerable coronary plaque imaging. J. Nucl. Med. 2014, 55, 1629–1635. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dilsizian, V.; Bacharach, S.L.; Beanlands, R.S.; Bergmann, S.R.; Delbeke, D.; Dorbala, S.; Gropler, R.J.; Knuuti, J.; Schelbert, H.R.; Travin, M.I. ASNC imaging guidelines/SNMMI procedure standard for positron emission tomography (PET) nuclear cardiology procedures. J. Nucl. Cardiol. 2016, 23, 1187–1226. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rubeaux, M.; Joshi, N.V.; Dweck, M.R.; Fletcher, A.; Motwani, M.; Thomson, L.E.; Germano, G.; Dey, D.; Li, D.; Berman, D.S.; et al. Motion Correction of 18F-NaF PET for Imaging Coronary Atherosclerotic Plaques. J. Nucl. Med. 2016, 57, 54–59. [Google Scholar] [CrossRef] [Green Version]
- Lassen, M.L.; Kwiecinski, J.; Dey, D.; Cadet, S.; Germano, G.; Berman, D.S.; Adamson, P.D.; Moss, A.J.; Dweck, M.R.; Newby, D.E.; et al. Triple-gated motion and blood pool clearance corrections improve reproducibility of coronary (18)F-NaF PET. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 2610–2620. [Google Scholar] [CrossRef] [PubMed]
- Dweck, M.R.; Chow, M.W.; Joshi, N.V.; Williams, M.C.; Jones, C.; Fletcher, A.M.; Richardson, H.; White, A.; McKillop, G.; van Beek, E.J.; et al. Coronary arterial 18F-sodium fluoride uptake: A novel marker of plaque biology. J. Am. Coll. Cardiol. 2012, 59, 1539–1548. [Google Scholar] [CrossRef] [Green Version]
- Raggi, P.; Senior, P.; Shahbaz, S.; Kaul, P.; Hung, R.; Coulden, R.; Yeung, R.; Abele, J. (18)F-Sodium Fluoride Imaging of Coronary Atherosclerosis in Ambulatory Patients with Diabetes Mellitus. Arterioscler. Thromb. Vasc. Biol. 2019, 39, 276–284. [Google Scholar] [CrossRef] [Green Version]
- Moss, A.J.; Doris, M.K.; Andrews, J.P.M.; Bing, R.; Daghem, M.; van Beek, E.J.R.; Forsyth, L.; Shah, A.S.V.; Williams, M.C.; Sellers, S.; et al. Molecular Coronary Plaque Imaging Using (18)F-Fluoride. Circ. Cardiovasc. Imaging 2019, 12, e008574. [Google Scholar] [CrossRef] [Green Version]
- Kwiecinski, J.; Cadet, S.; Daghem, M.; Lassen, M.L.; Dey, D.; Dweck, M.R.; Berman, D.S.; Newby, D.E.; Slomka, P.J. Whole-vessel coronary (18)F-sodium fluoride PET for assessment of the global coronary microcalcification burden. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 1736–1745. [Google Scholar] [CrossRef]
- Mojtahedi, A.; Alavi, A.; Thamake, S.; Amerinia, R.; Ranganathan, D.; Tworowska, I.; Delpassand, E.S. Assessment of vulnerable atherosclerotic and fibrotic plaques in coronary arteries using (68)Ga-DOTATATE PET/CT. Am. J. Nucl. Med. Mol. Imaging 2015, 5, 65–71. [Google Scholar]
- Bala, G.; Blykers, A.; Xavier, C.; Descamps, B.; Broisat, A.; Ghezzi, C.; Fagret, D.; Van Camp, G.; Caveliers, V.; Vanhove, C.; et al. Targeting of vascular cell adhesion molecule-1 by 18F-labelled nanobodies for PET/CT imaging of inflamed atherosclerotic plaques. Eur. Heart. J. Cardiovasc. Imaging 2016, 17, 1001–1008. [Google Scholar] [CrossRef] [Green Version]
- Buchler, A.; Munch, M.; Farber, G.; Zhao, X.; Al-Haddad, R.; Farber, E.; Rotstein, B.H. Selective Imaging of Matrix Metalloproteinase-13 to Detect Extracellular Matrix Remodeling in Atherosclerotic Lesions. Mol. Imaging Biol. 2022, 24, 93–103. [Google Scholar] [CrossRef] [PubMed]
- Weiberg, D.; Thackeray, J.T.; Daum, G.; Sohns, J.M.; Kropf, S.; Wester, H.J.; Ross, T.L.; Bengel, F.M.; Derlin, T. Clinical Molecular Imaging of Chemokine Receptor CXCR4 Expression in Atherosclerotic Plaque Using (68)Ga-Pentixafor PET: Correlation with Cardiovascular Risk Factors and Calcified Plaque Burden. J. Nucl. Med. 2018, 59, 266–272. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tzolos, E.; Bing, R.; Andrews, J.; Macaskill, M.; Tavares, A.; MacNaught, G.; Clarke, T.; Williams, M.C.; Van Beek, E.J.R.; Koglin, N.; et al. In vivo coronary artery thrombus imaging with 18F-GP1 PET-CT. Eur. Heart J. 2021, 42, ehab724.0261. [Google Scholar] [CrossRef]
- Mayer, J.; Wurster, T.H.; Schaeffter, T.; Landmesser, U.; Morguet, A.; Bigalke, B.; Hamm, B.; Brenner, W.; Makowski, M.R.; Kolbitsch, C. Imaging coronary plaques using 3D motion-compensated [(18)F]NaF PET/MR. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 2455–2465. [Google Scholar] [CrossRef]
- Wurster, T.H.; Landmesser, U.; Abdelwahed, Y.S.; Skurk, C.; Morguet, A.; Leistner, D.M.; Frohlich, G.; Haghikia, A.; Engel, L.C.; Schuster, A.; et al. Simultaneous [18F]fluoride and gadobutrol enhanced coronary positron emission tomography/magnetic resonance imaging for in vivo plaque characterization. Eur. Heart J. Cardiovasc. Imaging 2022, 2022, jeab276. [Google Scholar] [CrossRef] [PubMed]
| Biomarkers | CCTA | cMRI | PET | |
|---|---|---|---|---|
| Vulnerable plaque characteristics | ↑ hsCRP ↑ PTX-3 ↑ MMP-9 | ↓ Plaque attenuation ↑ Remodeling index ↑ perivascular FAI Ring-like enhancement Spotty calcifications | ↑ PMR ↑ CNR | ↑ TBR ↑ CMA |
| Clinical correlates | ||||
| Correlation with OCT/IVUS-derived plaque characteristics | + | +++/++++ | +++ | ++ |
| Gaps in evidence | Ideal sampling site (Peripheral vein, lesion location) | Radiomic-based approaches | Definite cutoffs | Ideal radiotracer PET/CT or PET/MRI Lack of definite cutoffs Limitation of myocardial uptake in FDG-PET Motion correction |
| Modality features | ||||
| Accessibility | ++++ | +++ | ++ | + |
| Cost | + | ++ | +++ | ++++ |
| Side-effects | - | Radiation exposure Anaphylactic reaction to IVCM CI-AKI | Anaphylactic reaction to IVCM Nephrogenic systemic fibrosis | Anaphylactic reaction |
| Biomarkers | ||
| CRP | ↑ levels associated with |
|
| Pentraxin-3 | ↑ levels associated with |
|
| MMP-9 | ↑ levels associated with |
|
| CCTA features | ||
| Spotty calcifications | Associated with |
|
| ↓ Plaque attenuation ± Positive remodeling ± Ring-like enhancement | Associated with |
|
| Necrotic core/fibrous plaque ratio | Associated with |
|
| Perivascular FAI | ↑ values (≥−70.1 HU) associated with |
|
| PVAT radiomic profile | Associated with | ↑ prediction of incident MACE |
| cMRI features | ||
| PMR | ↑ values (≥1.4) associated with |
|
| CNR | ↑ values associated with |
|
| Nuclear imaging features | ||
| TBR | ↑ values (>1.25) associated with |
|
| CMA | ↑ values (>0) associated with |
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Theofilis, P.; Sagris, M.; Antonopoulos, A.S.; Oikonomou, E.; Tsioufis, K.; Tousoulis, D. Non-Invasive Modalities in the Assessment of Vulnerable Coronary Atherosclerotic Plaques. Tomography 2022, 8, 1742-1758. https://doi.org/10.3390/tomography8040147
Theofilis P, Sagris M, Antonopoulos AS, Oikonomou E, Tsioufis K, Tousoulis D. Non-Invasive Modalities in the Assessment of Vulnerable Coronary Atherosclerotic Plaques. Tomography. 2022; 8(4):1742-1758. https://doi.org/10.3390/tomography8040147
Chicago/Turabian StyleTheofilis, Panagiotis, Marios Sagris, Alexios S. Antonopoulos, Evangelos Oikonomou, Konstantinos Tsioufis, and Dimitris Tousoulis. 2022. "Non-Invasive Modalities in the Assessment of Vulnerable Coronary Atherosclerotic Plaques" Tomography 8, no. 4: 1742-1758. https://doi.org/10.3390/tomography8040147
APA StyleTheofilis, P., Sagris, M., Antonopoulos, A. S., Oikonomou, E., Tsioufis, K., & Tousoulis, D. (2022). Non-Invasive Modalities in the Assessment of Vulnerable Coronary Atherosclerotic Plaques. Tomography, 8(4), 1742-1758. https://doi.org/10.3390/tomography8040147










