Feasibility and Safety of a Novel Leg Exercise Apparatus for Venous Thromboembolism Prophylaxis after Total Joint Arthroplasty of the Lower Extremities—A Pilot Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Leg Exercise Apparatus
2.3. Primary Outcomes
2.4. Secondary Outcome
2.5. Intervention
2.6. Perioperative Management and Assessment
2.7. Cancellation Criteria
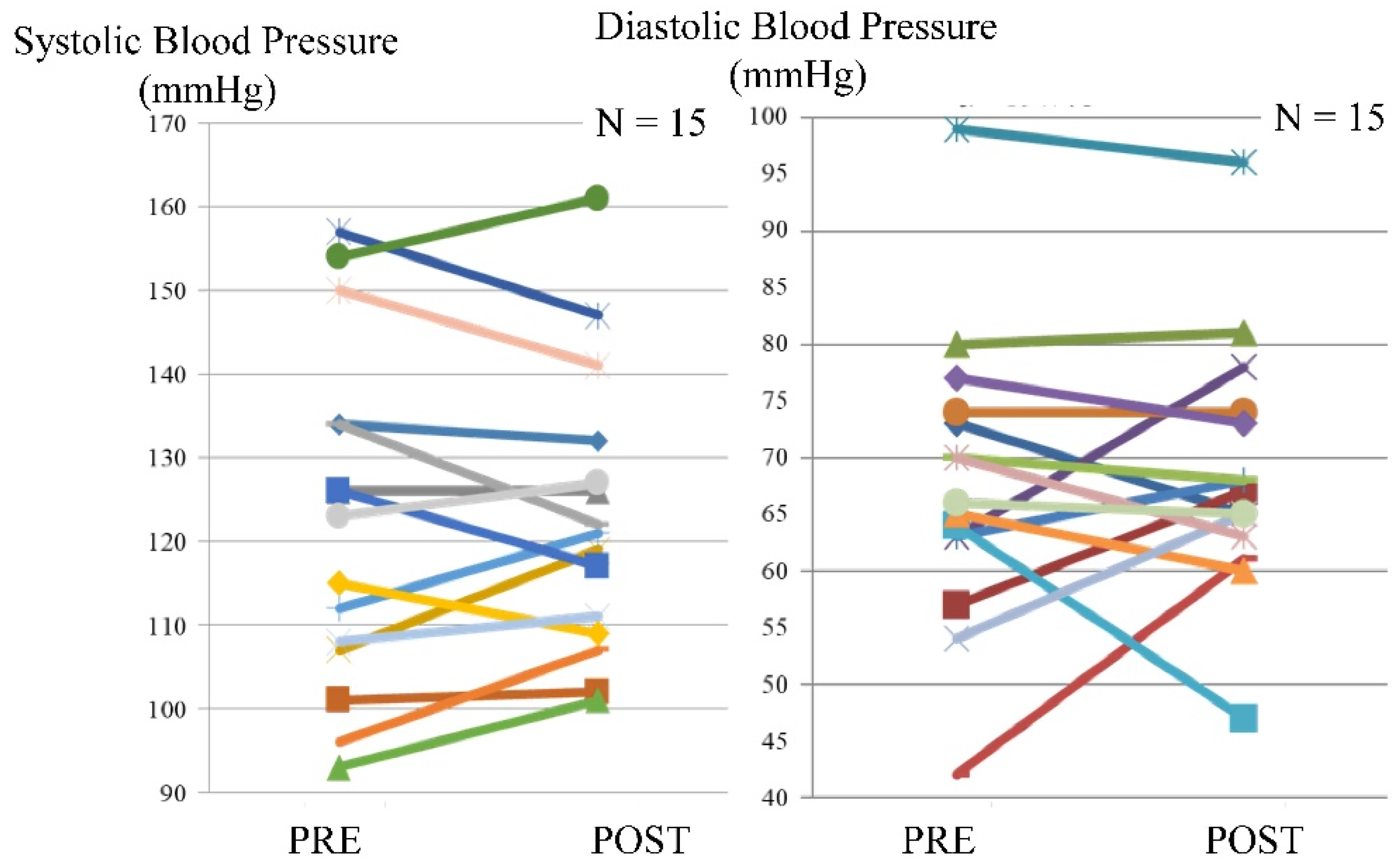
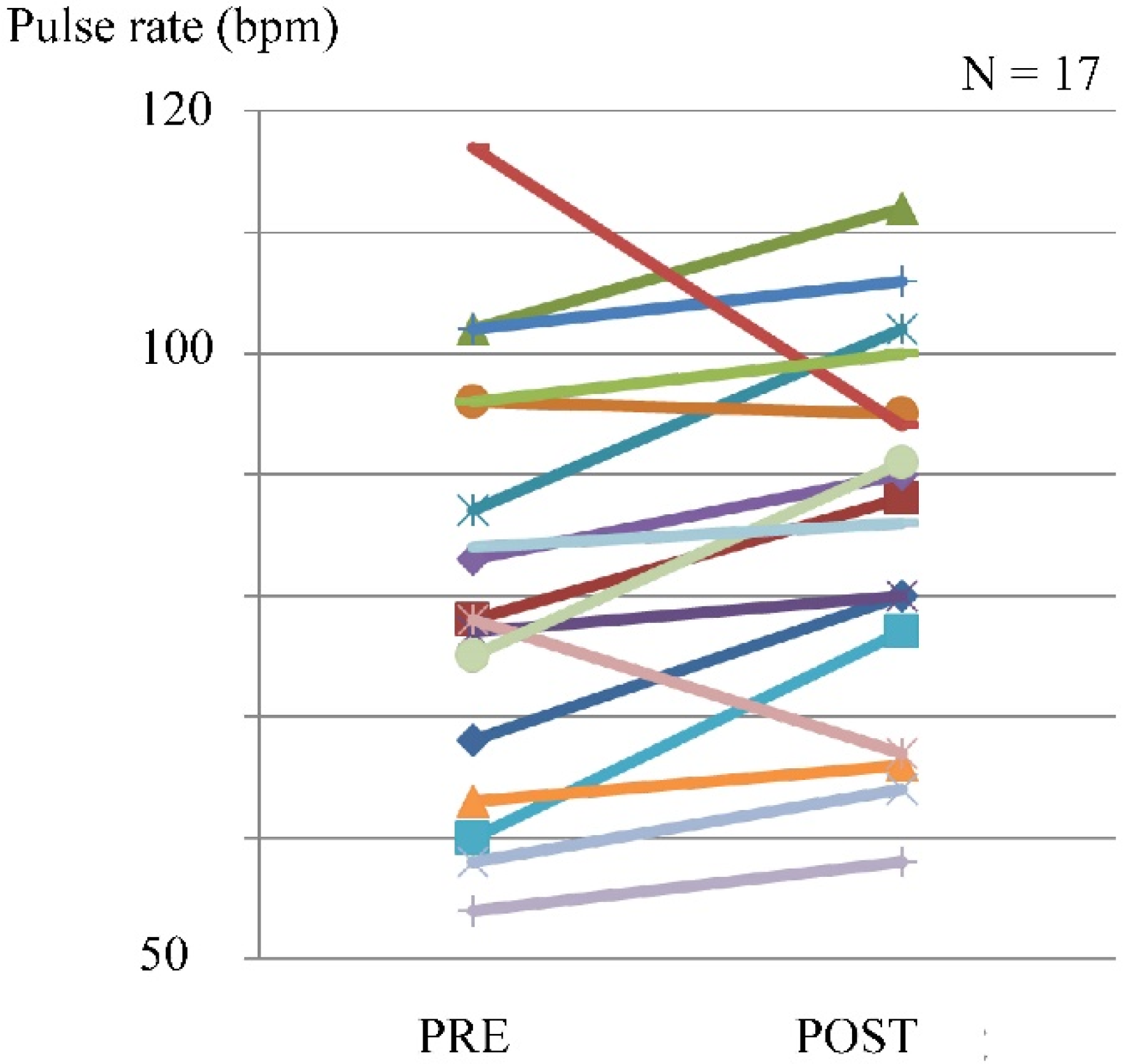
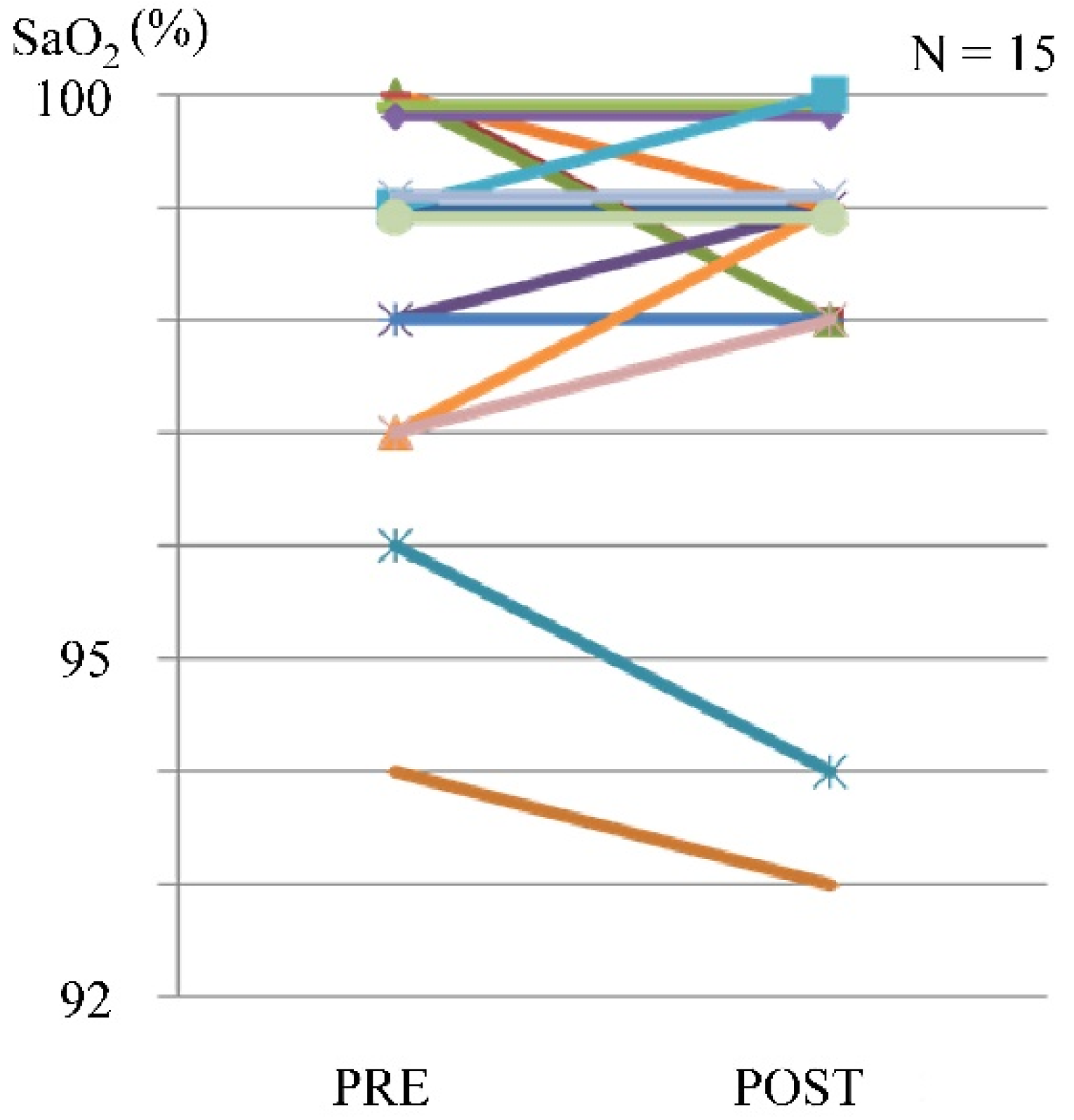
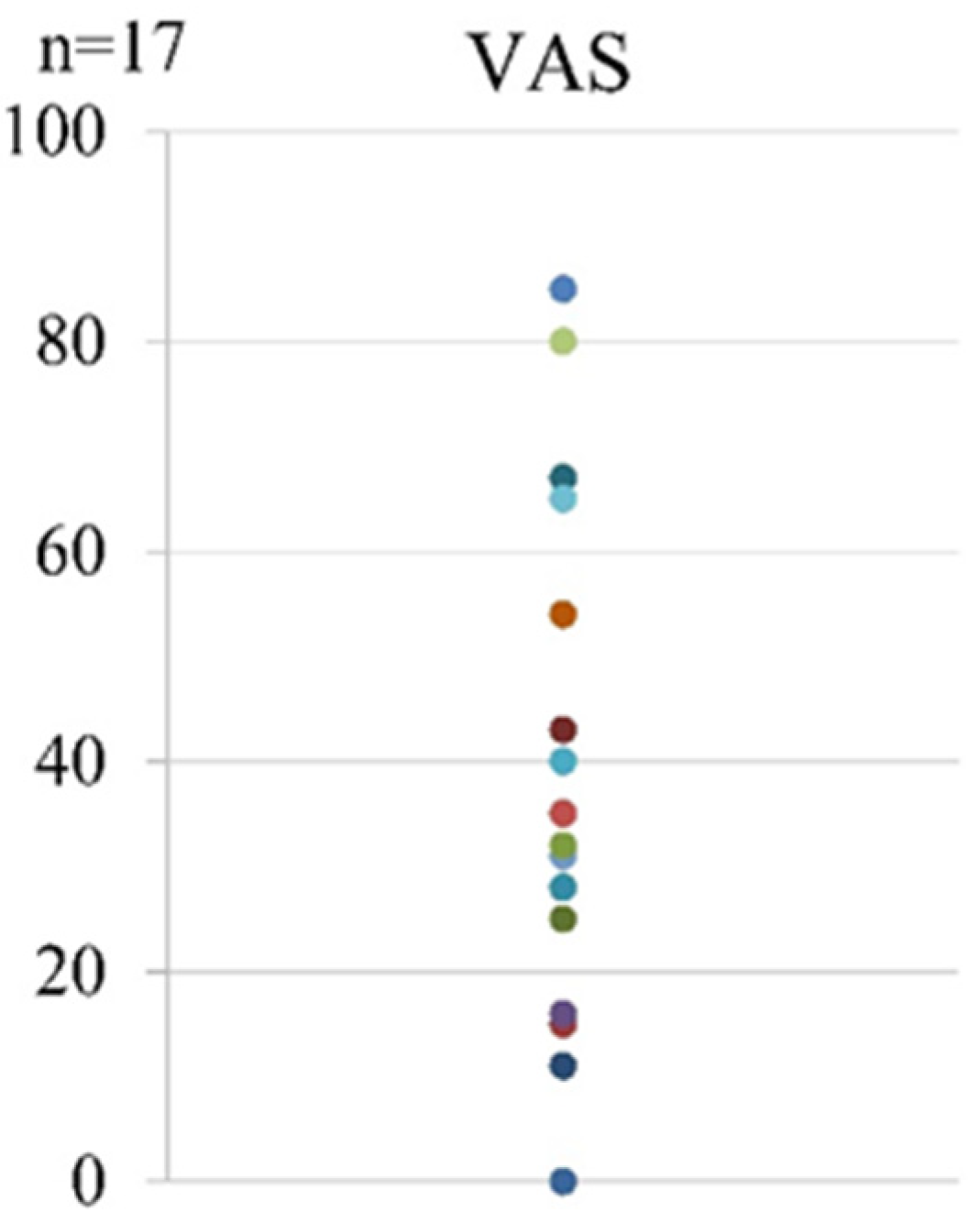
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ortel, T.L.; Neumann, I.; Ageno, W.; Beyth, R.; Clark, N.P.; Cuker, A.; Hutten, B.A.; Jaff, M.R.; Manja, V.; Schulman, S.; et al. American Society of Hematology 2020 guidelines for management of venous thromboembolism: Treatment of deep vein thrombosis and pulmonary embolism. Blood Adv. 2020, 4, 4693–4738. [Google Scholar] [CrossRef] [PubMed]
- Lichota, A.; Szewczyk, E.M.; Gwozdzinski, K. Factors affecting the formation and treatment of thrombosis by natural and synthetic compounds. Int. J. Mol. Sci. 2020, 21, 7975. [Google Scholar] [CrossRef] [PubMed]
- Santana, D.C.; Emara, A.K.; Orr, M.N.; Klika, A.K.; Higuera, C.A.; Krebs, V.E.; Molloy, R.M.; Piuzzi, N.S. An update on venous thromboembolism rates and prophylaxis in hip and knee arthroplasty in 2020. Medicina 2020, 56, 416. [Google Scholar] [CrossRef]
- Fuji, T.; Akagi, M.; Abe, Y.; Oda, E.; Matsubayashi, D.; Ota, K.; Kobayashi, M.; Matsushita, Y.; Kaburagi, J.; Ibusuki, K.; et al. Incidence of venous thromboembolism and bleeding events in patients with lower extremity orthopedic surgery: A retrospective analysis of a Japanese healthcare database. J. Orthop. Surg. Res. 2017, 12, 55. [Google Scholar] [CrossRef] [PubMed]
- JCS Joint Working Group. Guidelines for the diagnosis, treatment and prevention of pulmonary thromboembolism and deep vein thrombosis (JCS 2009). Circ. J. 2011, 75, 1258–1281. [Google Scholar] [CrossRef]
- Ohgi, S.; Tachibana, M.; Ikebuchi, M.; Kanaoka, Y.; Maeda, T.; Mori, T. Pulmonary embolism in patients with isolated soleal vein thrombosis. Angiology 1998, 49, 759–764. [Google Scholar] [CrossRef]
- Kageyama, N.; Ro, A.; Tanifuji, T.; Fukunaga, T. Significance of the soleal vein and its drainage veins in cases of massive pulmonary thromboembolism. Ann. Vasc. Dis. 2008, 1, 35–39. [Google Scholar] [CrossRef]
- Falck-Ytter, Y.; Francis, C.W.; Johanson, N.A.; Curley, C.; Dahl, O.E.; Schulman, S.; Ortel, T.L.; Pauker, S.G.; Colwell, C.W., Jr. Prevention of VTE in orthopedic surgery patients: Antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest 2012, 141, e278S–e325S. [Google Scholar] [CrossRef]
- Mont, M.A.; Jacobs, J.J.; Boggio, L.N.; Bozic, K.J.; Della Valle, C.J.; Goodman, S.B.; Lewis, C.G.; Yates, A.J., Jr.; Watters, W.C., 3rd; Turkelson, C.M.; et al. Preventing venous thromboembolic disease in patients undergoing elective hip and knee arthroplasty. J. Am. Acad. Orthop. Surg. 2011, 19, 768–776. [Google Scholar] [CrossRef] [PubMed]
- Malone, P.C.; Agutter, P.S. The aetiology of deep venous thrombosis. J. Assoc. Phys. 2006, 99, 581–593. [Google Scholar] [CrossRef]
- Brotman, D.J.; Deitcher, S.R.; Lip, G.Y.; Matzdorff, A.C. Virchow′s triad revisited. South. Med. J. 2004, 97, 213–214. [Google Scholar] [CrossRef]
- Kakkos, S.K.; Caprini, J.A.; Geroulakos, G.; Nicolaides, A.N.; Stansby, G.P.; Reddy, D.J. Combined intermittent pneumatic leg compression and pharmacological prophylaxis for prevention of venous thrombo-embolism in high-risk patients. Eur. J. Vasc. Endovasc. Surg. 2009, 37, 364–365. [Google Scholar] [CrossRef][Green Version]
- Rohrer, O.; Eicher, M. Effectiveness of intermittent pneumatic compression (IPC) on thrombosis prophylaxis: A systematic literature review. Pflege 2006, 19, 175–187. [Google Scholar] [CrossRef]
- Hanison, E.; Corbett, K. Non-pharmacological interventions for the prevention of venous thromboembolism: A literature review. Nurs. Stand. 2016, 31, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Wickham, N.; Gallus, A.S.; Walters, B.N.; Wilson, A. NHMRC VTE Prevention Guideline Adaptation Committee. Prevention of venous thromboembolism in patients admitted to Australian hospitals: Summary of National Health and Medical Research Council clinical practice guideline. Intern. Med. J. 2012, 42, 698–708. [Google Scholar] [CrossRef] [PubMed]
- Sochart, D.H.; Hardinge, K. The relationship of foot and ankle movements to venous return in the lower limb. J. Bone Jt. Surg Br. 1999, 81, 700–704. [Google Scholar] [CrossRef]
- Anand, S.; Asumu, T. Patient acceptance of a foot pump device used for thromboprophylaxis. Acta Orthop. Belg. 2007, 73, 386–389. [Google Scholar] [PubMed]
- Shimizu, Y.; Kamada, H.; Sakane, M.; Aikawa, S.; Mutsuzaki, H.; Tanaka, K.; Mishima, H.; Ochiai, N.; Yamazaki, M. A novel apparatus for active leg exercise improves venous flow in the lower extremity. J. Sports Med. Phys. Fit. 2016, 56, 1592–1597. [Google Scholar]
- Tanaka, K.; Kamada, H.; Shimizu, Y.; Aikawa, S.; Nishino, T.; Ochiai, N.; Sakane, M.; Yamazaki, M. The use of a novel in-bed active Leg Exercise Apparatus (LEX) for increasing venous blood flow. J. Rural Med. 2016, 11, 11–16. [Google Scholar] [CrossRef]
- Tanaka, K.; Kamada, H.; Shimizu, Y.; Aikawa, S.; Irie, S.; Ochiai, N.; Sakane, M.; Yamazaki, M. Muscle activity in the lower limbs during push-down movement with a new active-exercise apparatus for the leg. J. Phys. Ther. Sci. 2016, 28, 1050–1054. [Google Scholar] [CrossRef]
- Shimizu, Y.; Kamada, H.; Sakane, M.; Aikawa, S.; Mutsuzaki, H.; Tanaka, K.; Mishima, H.; Kanamori, A.; Nishino, T.; Ochiai, N.; et al. A novel exercise device for venous thromboembolism prophylaxis improves venous flow in bed versus ankle movement exercises in healthy volunteers. J. Orthop. Surg. 2017, 25, 230949901773947. [Google Scholar] [CrossRef]
- Borg, G.A. Psychophysical bases of perceived exertion. Med. Sci. Sports Exerc. 1982, 14, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, K.; Yokoyama, T.; Kitaoka, N.; Nishiyama, T.; Manabe, M. Blood flow velocity of the femoral vein with foot exercise compared to pneumatic foot compression. J. Clin. Anesth. 2005, 17, 102–105. [Google Scholar] [CrossRef] [PubMed]
- Mahoney, F.I.; Barthel, D.W. Functional evaluation: The barthel index. Md. State Med. J. 1965, 14, 61–65. [Google Scholar]
- Dohm, M.; Williams, K.M.; Novotny, T. Micro-mobile foot compression device compared with pneumatic compression device. Clin. Orthop. Relat. Res. 2011, 469, 1692–1700. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Maeda, M. Guideline for safety management and promotion in rehabilitation medicine. Jpn. J. Rehabil. Med. 2007, 44, 384–390. (In Japanese) [Google Scholar] [CrossRef][Green Version]
- Jacobs, J.J.; Mont, M.A.; Bozic, K.J.; Della Valle, C.J.; Goodman, S.B.; Lewis, C.G.; Yates, A.C., Jr.; Boggio, L.N.; Watters, W.C., 3rd; Turkelson, C.M.; et al. American Academy of Orthopaedic Surgeons clinical practice guideline on: Preventing venous thromboembolic disease in patients undergoing elective hip and knee arthroplasty. J. Bone Jt. Surg. Am. 2012, 94, 746–747. [Google Scholar] [CrossRef]
- Shahi, A.; Chen, A.F.; Tan, T.L.; Maltenfort, M.G.; Kucukdurmaz, F.; Parvizi, J. The incidence and economic burden of in-hospital venous thromboembolism in the United States. J. Arthroplast. 2017, 32, 1063–1066. [Google Scholar] [CrossRef]
- Chan, N.C.; Siegal, D.; Lauw, M.N.; Ginsberg, J.S.; Eikelboom, J.W.; Guyatt, G.H.; Hirsh, J. A systematic review of contemporary trials of anticoagulants in orthopaedic thromboprophylaxis: Suggestions for a radical reappraisal. J. Thromb. Thrombolysis 2015, 40, 231–239. [Google Scholar] [CrossRef]
- Tsuda, K.; Kawasaki, T.; Nakamura, N.; Yoshikawa, H.; Sugano, N. Natural course of asymptomatic deep venous thrombosis in hip surgery without pharmacologic thromboprophylaxis in an Asian population. Clin. Orthop. Relat. Res. 2010, 468, 2430–2436. [Google Scholar] [CrossRef]
- Kahn, S.R.; Shivakumar, S. What′s new in VTE risk and prevention in orthopedic surgery. Res. Pract. Thromb. Haemost. 2020, 4, 366–376. [Google Scholar] [CrossRef]
- Husted, H.; Otte, K.S.; Kristensen, B.B.; Orsnes, T.; Wong, C.; Kehlet, H. Low risk of thromboembolic complications after fast-track hip and knee arthroplasty. Acta Orthop. 2010, 81, 599–605. [Google Scholar] [CrossRef] [PubMed]
- Jorgensen, C.C.; Jacobsen, M.K.; Soeballe, K.; Hansen, T.B.; Husted, H.; Kjaersgaard-Andersen, P.; Hansen, L.T.; Laursen, M.B.; Kehlet, H. Thromboprophylaxis only during hospitalisation in fast-track hip and knee arthroplasty, a prospective cohort study. BMJ Open 2013, 3, e003965. [Google Scholar] [CrossRef]
- Khan, S.A.; Logan, P.; Asokan, A.; Handford, C.; Rajgor, H.D.; Khadabadi, N.A.; Moores, T.; Targett, J. The incidence of venous thromboembolism in total joint replacement during COVID-19 pandemic: Has lockdown had an influence? Bone Jt. Open 2020, 1, 751–756. [Google Scholar] [CrossRef] [PubMed]
- Cai, C.; Guo, Y.; You, Y.; Hu, K.; Cai, F.; Xie, M.; Yang, L.; Ling, K.; Ye, D.; Misra, S.; et al. Deep venous thrombosis in COVID-19 patients: A cohort analysis. Clin. Appl. Thromb. Hemost. 2020, 26, 1076029620982669. [Google Scholar] [CrossRef] [PubMed]


| Subject | Sex | Age (y) | Height (cm) | Weight (kg) | BMI (kg/m2) | Surgery | GCS | IPCD | Pharmacological Therapy |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Male | 48 | 161.0 | 77.4 | 29.9 | THA | + | * − | Edoxaban |
| 2 | Female | 70 | 158.0 | 54.8 | 22.0 | THA | + | + | - |
| 3 | Female | 56 | 153.0 | 45.6 | 19.5 | THA | + | + | - |
| 4 | Female | 63 | 151.1 | 62.0 | 27.2 | TKA | + | + | Edoxaban |
| 5 | Female | 43 | 156.0 | 66.0 | 27.1 | THA | + | + | - |
| 6 | Female | 38 | 155.7 | 53.4 | 22.0 | THA | + | + | - |
| 7 | Female | 61 | 151.8 | 56.2 | 24.4 | THA | + | + | - |
| 8 | Male | 38 | 168.5 | 76.8 | 27.0 | THA | + | + | - |
| 9 | Female | 69 | 145.0 | 52.8 | 25.1 | THA | + | + | - |
| 10 | Female | 75 | 141.0 | 39.0 | 19.6 | THA | + | + | - |
| 11 | Female | 59 | 148.1 | 50.2 | 22.9 | THA | + | + | - |
| 12 | Female | 72 | 131.5 | 36.6 | 21.2 | THA | + | + | - |
| 13 | Female | 68 | 155.7 | 53.0 | 21.9 | THA | + | + | - |
| 14 | Female | 55 | 155.0 | 49.4 | 20.6 | THA | + | + | - |
| 15 | Female | 80 | 146.3 | 45.0 | 21.0 | THA | + | + | - |
| 16 | Female | 71 | 151.0 | 63.0 | 27.6 | THA | + | + | - |
| 17 | Female | 69 | 148.6 | 62.8 | 28.4 | THA | + | + | - |
| 18 | Male | 56 | 168.9 | 82.8 | 29.0 | THA | + | + | - |
| 19 | Female | 53 | 158.4 | 50.8 | 20.2 | THA | + | + | - |
| 20 | Male | 67 | 151.4 | 70.7 | 30.8 | THA | + | + | - |
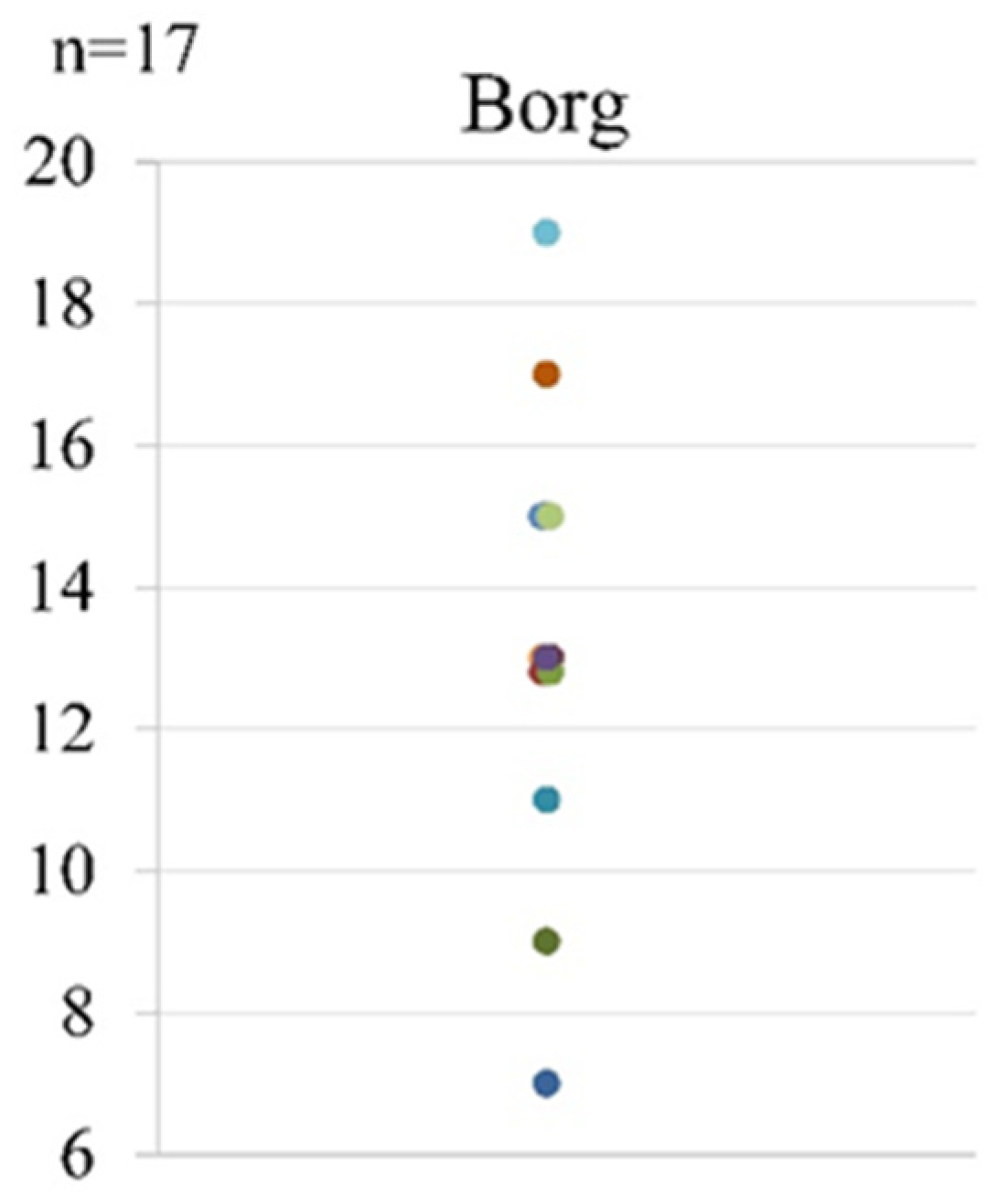
| Borg Scale | |
|---|---|
| 6 | |
| 7 | Very, very light |
| 8 | |
| 9 | Very light |
| 10 | |
| 11 | Fairly light |
| 12 | |
| 13 | Somewhat hard |
| 14 | |
| 15 | Hard |
| 16 | |
| 17 | Very hard |
| 18 | |
| 19 | Very, very hard |
| 20 | |
| Preoperation | Informed Consent D-Dimer Ultrasonography Practice Exercise Using the LEX |
|---|---|
| Operation day | THA/TKA |
| Exercise using the LEX 2 h after surgery and at 5 p.m. with monitoring of ECG, SpO2, and blood pressure VAS·Borg scale | |
| Postoperative day 1–7 | Exercise using the LEX at 7 a.m., 10 a.m., 1 p.m., and 5 p.m. VAS·Borg scale |
| Day 7 | D-dimer Ultrasonography |
| FEEDING | ||
| 0 = unable | ||
| 5 = needs help with cutting, spreading butter, etc., or requires a modified diet | ||
| 10 = independent | ||
| BATHING | ||
| 0 = dependent | ||
| 5 = independent (or in shower) | ||
| GROOMING | ||
| 0 = needs help with personal care | ||
| 5 = independent face/hair/teeth/shaving (implements provided) | ||
| DRESSING | ||
| 0 = dependent | ||
| 5 = needs help but can do about half unaided | ||
| 10 = independent (including buttons, zips, laces, etc.) | ||
| BOWELS | ||
| 0 = incontinent (or needs to be given enemas) | ||
| 5 = occasional accident | ||
| 10 = continent | ||
| BLADDER | ||
| 0 = incontinent or catheterized and unable to manage alone | ||
| 5 = occasional accident | ||
| 10 = continent | ||
| TOILET USE | ||
| 0 = dependent | ||
| 5 = needs some help but can do something alone | ||
| 10 = independent (on and off, dressing, wiping) | ||
| TRANSFERS (BED TO CHAIR AND BACK) | ||
| 0 = unable, no sitting balance | ||
| 5 = major help (one or two people, physical), can sit | ||
| 10 = minor help (verbal or physical) | ||
| 15 = independent | ||
| MOBILITY (ON LEVEL SURFACES) | ||
| 0 = immobile or <50 yards | ||
| 5 = wheelchair independent, including corners, >50 yards | ||
| 10 = walks with the help of one person (verbal or physical) >50 yards | ||
| 15 = independent (but may use any aid; for example, stick) >50 yards | ||
| STAIRS | ||
| 0 = unable | ||
| 5 = needs help (verbal, physical, carrying aid) | ||
| 10 = independent | ||
| Stop Exercise Criteria | Present Study | |
|---|---|---|
| Systolic blood pressure | Increasing >40 mmHg | Increasing 12 mmHg |
| Diastolic blood pressure | Increasing >20 mmHg | Increasing 19 mmHg |
| Pulse rate | Over 140 bpm | Maximum of 112 bpm |
| SpO2 | Moderate breathing difficulties | No breathing difficulties |
| The criteria were set according to the guidelines for safety management and promotion in rehabilitation medicine, proposed by the Japanese Association of Rehabilitation Medicine [26]. SpO2, blood oxygen saturation level; bpm, beats/min | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tanaka, K.; Shimizu, Y.; Kamada, H.; Aikawa, S.; Mishima, H.; Kanamori, A.; Nishino, T.; Sakane, M.; Ochiai, N.; Yamazaki, M. Feasibility and Safety of a Novel Leg Exercise Apparatus for Venous Thromboembolism Prophylaxis after Total Joint Arthroplasty of the Lower Extremities—A Pilot Study. Tomography 2021, 7, 734-746. https://doi.org/10.3390/tomography7040061
Tanaka K, Shimizu Y, Kamada H, Aikawa S, Mishima H, Kanamori A, Nishino T, Sakane M, Ochiai N, Yamazaki M. Feasibility and Safety of a Novel Leg Exercise Apparatus for Venous Thromboembolism Prophylaxis after Total Joint Arthroplasty of the Lower Extremities—A Pilot Study. Tomography. 2021; 7(4):734-746. https://doi.org/10.3390/tomography7040061
Chicago/Turabian StyleTanaka, Kenta, Yukiyo Shimizu, Hiroshi Kamada, Shizu Aikawa, Hajime Mishima, Akihiro Kanamori, Tomofumi Nishino, Masataka Sakane, Naoyuki Ochiai, and Masashi Yamazaki. 2021. "Feasibility and Safety of a Novel Leg Exercise Apparatus for Venous Thromboembolism Prophylaxis after Total Joint Arthroplasty of the Lower Extremities—A Pilot Study" Tomography 7, no. 4: 734-746. https://doi.org/10.3390/tomography7040061
APA StyleTanaka, K., Shimizu, Y., Kamada, H., Aikawa, S., Mishima, H., Kanamori, A., Nishino, T., Sakane, M., Ochiai, N., & Yamazaki, M. (2021). Feasibility and Safety of a Novel Leg Exercise Apparatus for Venous Thromboembolism Prophylaxis after Total Joint Arthroplasty of the Lower Extremities—A Pilot Study. Tomography, 7(4), 734-746. https://doi.org/10.3390/tomography7040061







