Evaluation of Amyotrophic Lateral Sclerosis-Induced Muscle Degeneration Using Magnetic Resonance-Based Relaxivity Contrast Imaging (RCI)
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Cohort
2.2. Image Acquisition
2.3. Image Analysis
2.4. Statistical Analysis
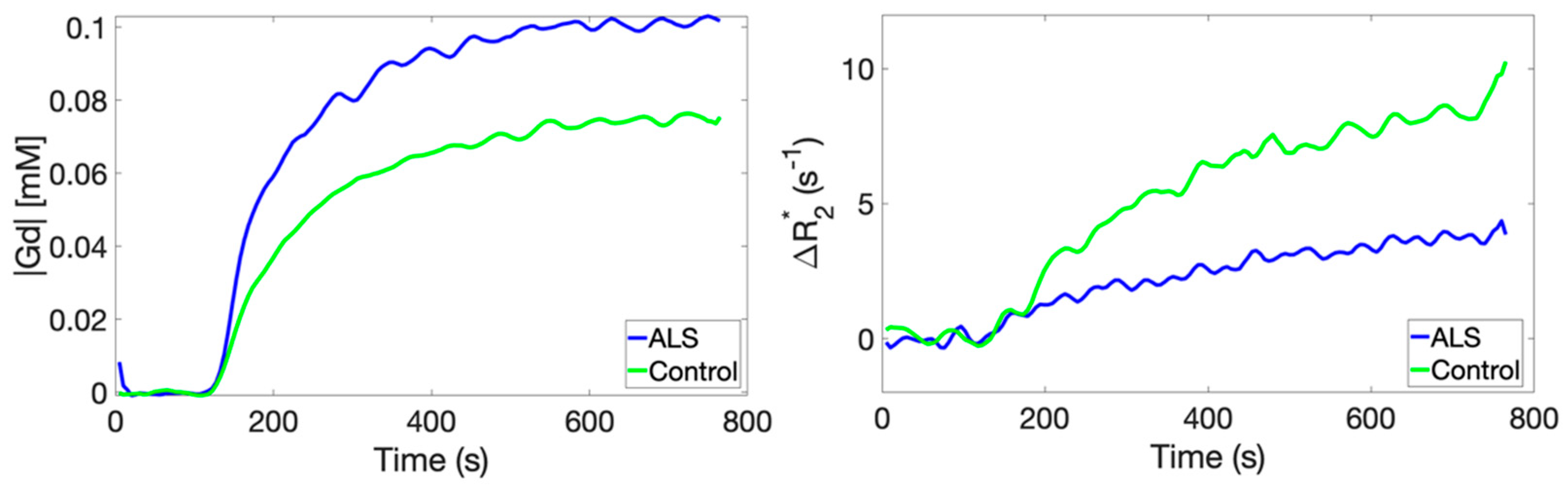
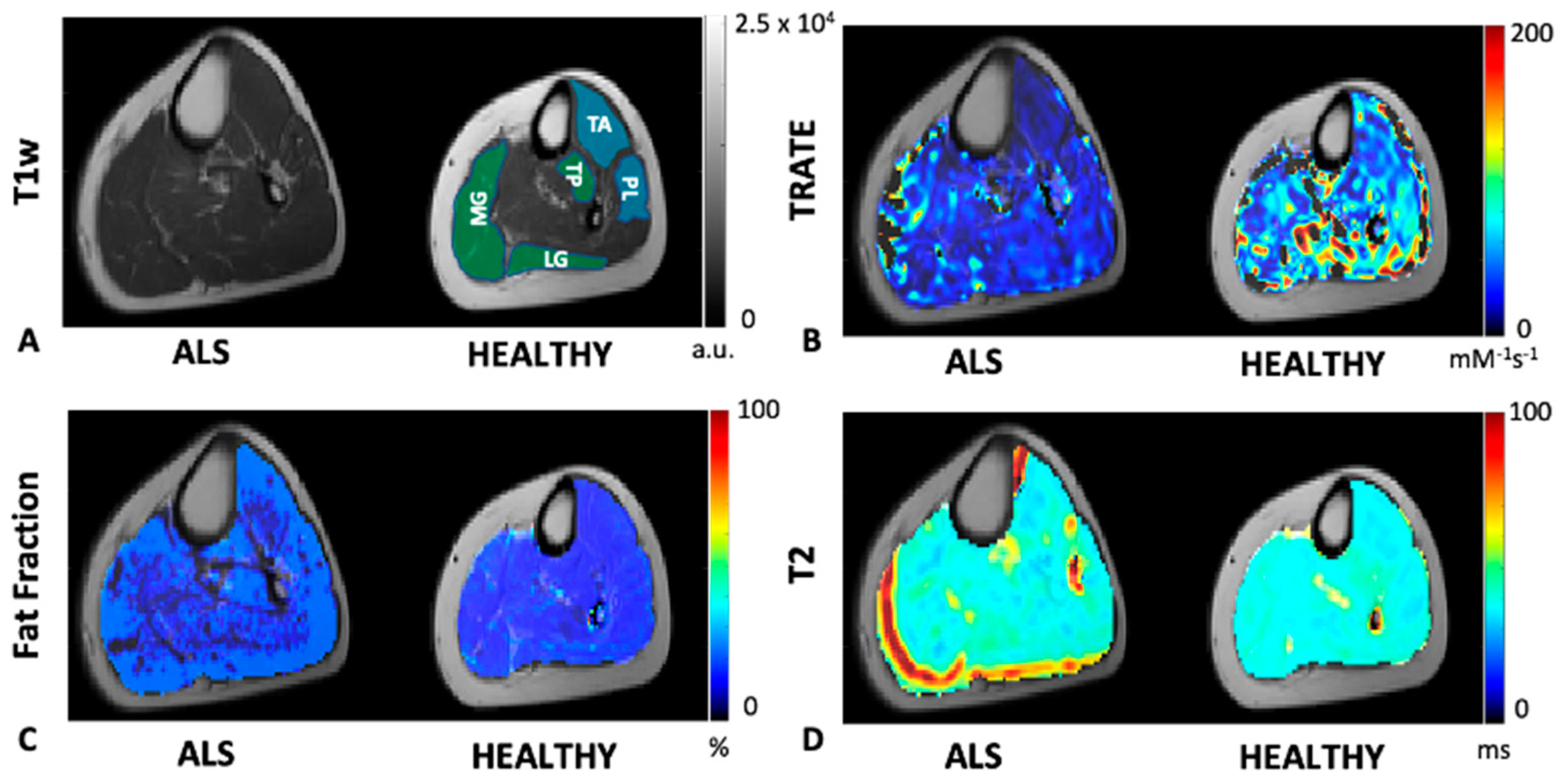
3. Results
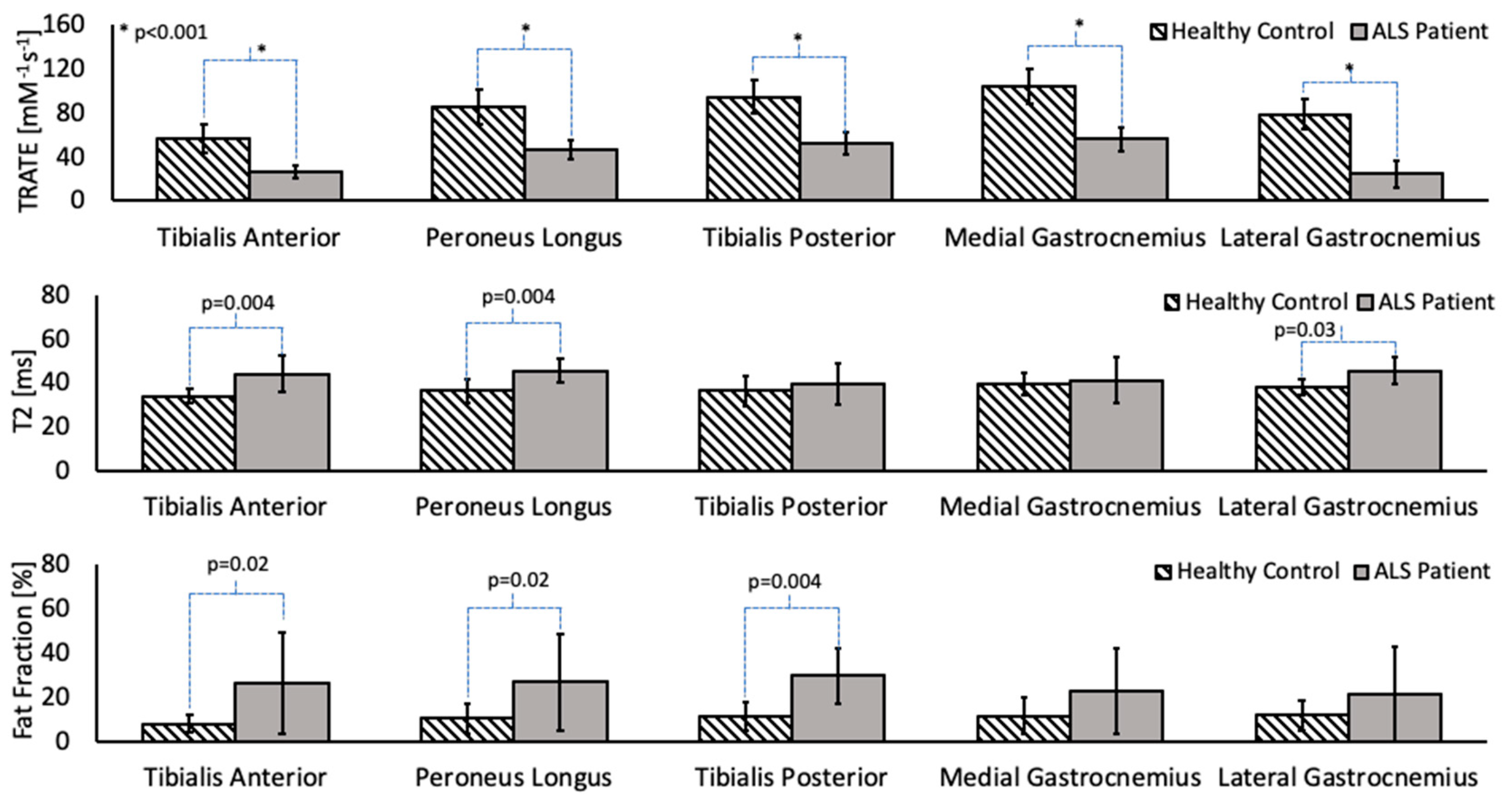
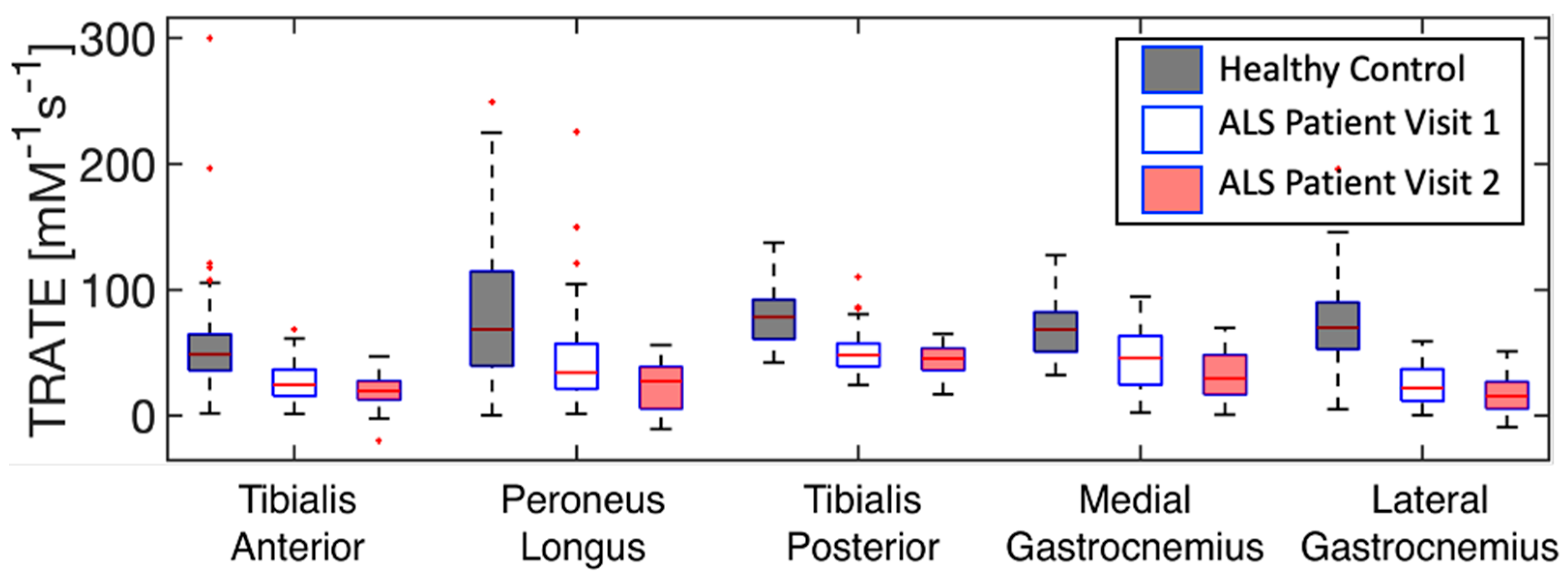
3.1. Comparative Analysis
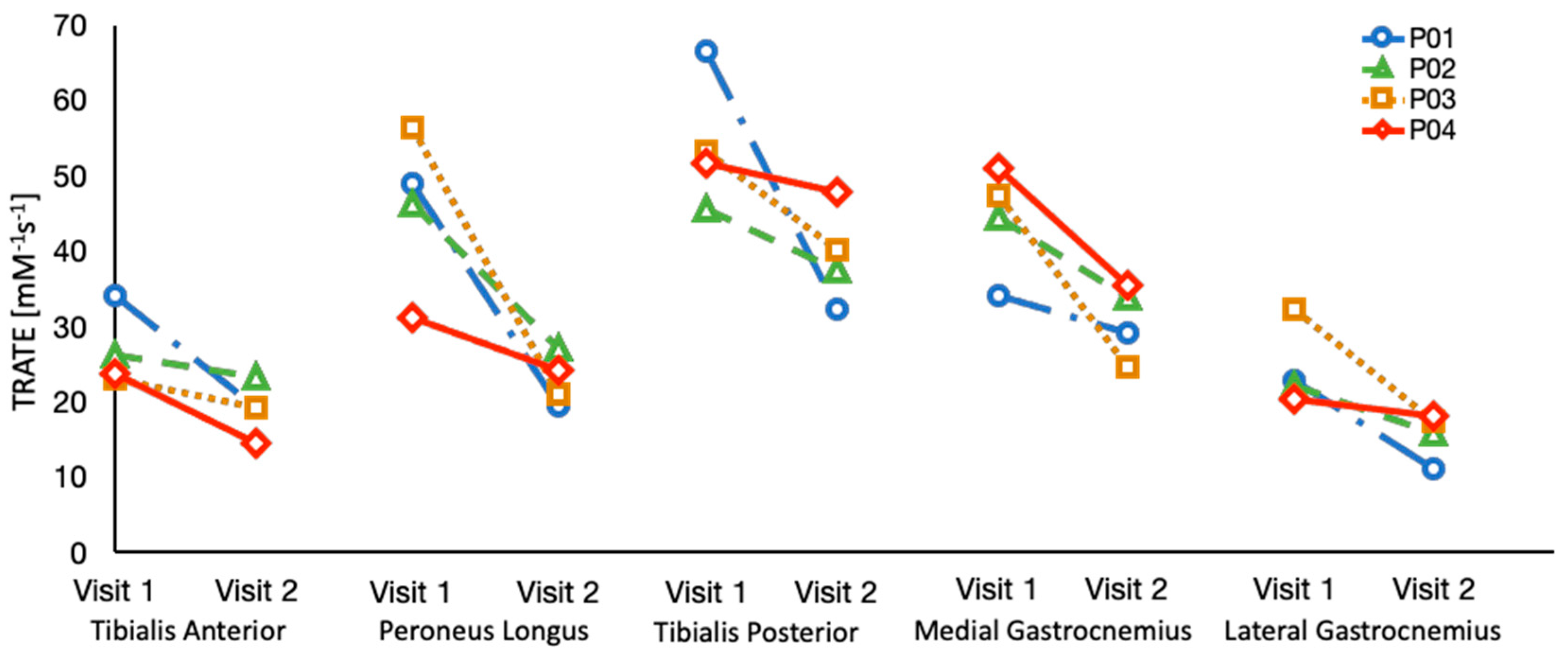
3.2. Longitudinal Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Paganoni, S.; Macklin, E.A.; Lee, A.; Murphy, A.; Chang, J.; Zipf, A.; Cudkowicz, M.; Atassi, N. Diagnostic timelines and delays in diagnosing amyotrophic lateral sclerosis (ALS). Amyotroph. Lateral Scler. Front. Degener. 2014, 15, 453–456. [Google Scholar] [CrossRef] [PubMed]
- Voustianiouk, A.; Seidel, G.; Panchal, J.; Sivak, M.; Czaplinski, A.; Yen, A.; Appel, S.H.; Lange, D.J. ALSFRS and appel ALS scores: Discordance with disease progression. Muscle Nerve 2008, 37, 668–672. [Google Scholar] [CrossRef]
- Cappello, V.; Francolini, M. Neuromuscular Junction Dismantling in Amyotrophic Lateral Sclerosis. Int. J. Mol. Sci. 2017, 18, 2092. [Google Scholar] [CrossRef]
- Williams, J.R.; Fitzhenry, D.; Grant, L.; Martyn, D.; Kerr, D.A. Diagnosis pathway for patients with amyotrophic lateral sclerosis: Retrospective analysis of the US Medicare longitudinal claims database. BMC Neurol. 2013, 13, 160. [Google Scholar] [CrossRef] [PubMed]
- Walhout, R.; Verstraete, E.; Heuvel, M.P.V.D.; Veldink, J.H.; Berg, L.H.V.D. Patterns of symptom development in patients with motor neuron disease. Amyotroph. Lateral Scler. Front. Degener. 2017, 19, 21–28. [Google Scholar] [CrossRef]
- Agosta, F.; Chiò, A.; Cosottini, M.; De Stefano, N.; Falini, A.; Mascalchi, M.; Rocca, M.; Silani, V.; Tedeschi, G.; Filippi, M. The Present and the Future of Neuroimaging in Amyotrophic Lateral Sclerosis. Am. J. Neuroradiol. 2010, 31, 1769–1777. [Google Scholar] [CrossRef] [PubMed]
- Agosta, F.; Pagani, E.; Petrolini, M.; Caputo, D.; Perini, M.; Prelle, A.; Salvi, F.; Filippi, M. Assessment of White Matter Tract Damage in Patients with Amyotrophic Lateral Sclerosis: A Diffusion Tensor MR Imaging Tractography Study. Am. J. Neuroradiol. 2010, 31, 1457–1461. [Google Scholar] [CrossRef]
- Bede, P.; Bokde, A.L.; Byrne, S.; Elamin, M.; McLaughlin, R.L.; Kenna, K.; Fagan, A.J.; Pender, N.; Bradley, D.G.; Hardiman, O. (2013 Neurology Bede) Multiparametric MRI study of ALS stratified for the C9orf72 genotype. Neurology 2013, 81, 361–369. [Google Scholar] [CrossRef]
- de Carvalho, M.; Dengler, R.; Eisen, A.; England, J.D.; Kaji, R.; Kimura, J.; Mills, K.; Mitsumoto, H.; Nodera, H.; Shefner, J.; et al. Electrodiagnostic criteria for diagnosis of ALS. Clin. Neurophysiol. 2008, 119, 497–503. [Google Scholar] [CrossRef]
- Rutkove, S.B.; Caress, J.B.; Cartwright, M.S.; Burns, T.M.; Warder, J.; David, W.S.; Goyal, N.; Maragakis, N.J.; Clawson, L.; Benatar, M.; et al. Electrical impedance myography as a biomarker to assess ALS progression. Amyotroph. Lateral Scler. 2012, 13, 439–445. [Google Scholar] [CrossRef]
- Rutkove, S.B.; Caress, J.B.; Cartwright, M.S.; Burns, T.M.; Warder, J.; David, W.S.; Goyal, N.; Maragakis, N.J.; Benatar, M.; Sharma, K.R.; et al. Electrical impedance myography correlates with standard measures of Als severity. Muscle Nerve 2013, 49, 441–443. [Google Scholar] [CrossRef]
- Arts, I.M.; Overeem, S.; Pillen, S.; Kleine, B.U.; Boekestein, W.A.; Zwarts, M.J.; Schelhaas, H.J. Muscle ultrasonography: A diagnostic tool for amyotrophic lateral sclerosis. Clin. Neurophysiol. 2012, 123, 1662–1667. [Google Scholar] [CrossRef]
- Winkler, E.A.; Sengillo, J.D.; Sullivan, J.S.; Henkel, J.S.; Appel, S.H.; Zlokovic, B.V. Blood–spinal cord barrier breakdown and pericyte reductions in amyotrophic lateral sclerosis. Acta Neuropathol. 2012, 125, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, T.M.; Alix, J.J.P.; David, C.; Pearson, E.; Rao, D.G.; Hoggard, N.; O’Brien, E.; Baster, K.; Bradburn, M.; Bigley, J.; et al. Imaging muscle as a potential biomarker of denervation in motor neuron disease. J. Neurol. Neurosurg. Psychiatry 2017, 89, 248–255. [Google Scholar] [CrossRef] [PubMed]
- Klickovic, U.; Zampedri, L.; Sinclair, C.D.; Wastling, S.J.; Trimmel, K.; Howard, R.S.; Malaspina, A.; Sharma, N.; Sidle, K.; Emira, A.; et al. Skeletal muscle MRI differentiates SBMA and ALS and correlates with disease severity. Neurology 2019, 93, e895–e907. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Manera, J.; Llauger, J.; Gallardo, E.; Illa, I. Muscle MRI in muscular dystrophies. Acta Myol. 2015, 34, 95–108. [Google Scholar] [PubMed]
- Sourbron, S.; Heilmann, M.; Walczak, C.; Vautier, J.; Schad, L.R.; Volk, A. T2*-relaxivity contrast imaging: First results. Magn. Reson. Med. 2012, 69, 1430–1437. [Google Scholar] [CrossRef] [PubMed]
- Semmineh, N.B.; Xu, J.; Skinner, J.T.; Xie, J.; Li, H.; Ayers, G.; Quarles, C.C. Assessing tumor cytoarchitecture using multiecho DSC-MRI derived measures of the transverse relaxivity at tracer equilibrium (TRATE). Magn. Reson. Med. 2015, 74, 772–784. [Google Scholar] [CrossRef]
- Brooks, B.R.; Miller, R.G.; Swash, M.; Munsat, T.L. El Escorial revisited: Revised criteria for the diagnosis of amyotrophic lateral sclerosis. Amyotroph. Lateral Scler. Other Mot. Neuron Disord. 2000, 1, 293–299. [Google Scholar] [CrossRef]
- Shefner, J.M.; Liu, D.; Leitner, M.L.; Schoenfeld, D.; Johns, D.R.; Ferguson, T.; Cudkowicz, M. Quantitative strength testing in ALS clinical trials. Neurology 2016, 87, 617–624. [Google Scholar] [CrossRef]
- Jenkinson, M.; Bannister, P.; Brady, M.; Smith, S. Improved Optimization for the Robust and Accurate Linear Registration and Motion Correction of Brain Images. NeuroImage 2002, 17, 825–841. [Google Scholar] [CrossRef] [PubMed]
- Stokes, A.M.; Quarles, C.C. A simplified spin and gradient echo approach for brain tumor perfusion imaging. Magn. Reson. Med. 2016, 75, 356–362. [Google Scholar] [CrossRef] [PubMed]
- Fram, E.K.; Herfkens, R.J.; Johnson, G.; Glover, G.H.; Karis, J.P.; Shimakawa, A.; Perkins, T.G.; Pelc, N.J. Rapid calculation of T1 using variable flip angle gradient refocused imaging. Magn. Reson. Imaging 1987, 5, 201–208. [Google Scholar] [CrossRef]
- Quarles, C.C.; Gore, J.C.; Xu, L.; Yankeelov, T.E. Comparison of dual-echo DSC-MRI- and DCE-MRI-derived contrast agent kinetic parameters. Magn. Reson. Imaging 2012, 30, 944–953. [Google Scholar] [CrossRef]
- Min, Y.G.; Choi, S.-J.; Hong, Y.-H.; Kim, S.-M.; Shin, J.-Y.; Sung, J.-J. Dissociated leg muscle atrophy in amyotrophic lateral sclerosis/motor neuron disease: The ‘split-leg’ sign. Sci. Rep. 2020, 10, 15661. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Zhang, H.; Chen, Z.; van der Plas, E.; Gutmann, L.; Thedens, D.; Nopoulos, P.; Sonka, M. Fully automated 3D segmentation of MR-imaged calf muscle compartments: Neighborhood relationship enhanced fully convolutional network. Comput. Med. Imaging Graph. 2021, 87, 101835. [Google Scholar] [CrossRef] [PubMed]
- Le Troter, A.; Fouré, A.; Guye, M.; Confort-Gouny, S.; Mattei, J.-P.; Gondin, J.; Salort-Campana, E.; Bendahan, D. Volume measurements of individual muscles in human quadriceps femoris using atlas-based segmentation approaches. Magn. Reson. Mater. Phys. Biol. Med. 2016, 29, 245–257. [Google Scholar] [CrossRef]
- Essafi, S.; Langs, G.; Deux, J.-F.; Rahmouni, A.; Bassez, G.; Paragios, N. Wavelet-driven knowledge-based MRI calf muscle segmentation. In Proceedings of the 2009 IEEE International Symposium on Biomedical Imaging: From Nano to Macro, Boston, MA, USA, 28 June–1 July 2009; pp. 225–228. [Google Scholar] [CrossRef]
- Rodrigues, R.; Pinheiro, A.M. Segmentation of Skeletal Muscle in Thigh Dixon MRI Based on Texture Analysis. arXiv 2019, arXiv:1904.04747. [Google Scholar]
- Shefner, J.M.; Rutkove, S.B.; Caress, J.B.; Benatar, M.; David, W.S.; Cartwright, M.S.; Macklin, E.A.; Bohorquez, J.L. Reducing sample size requirements for future ALS clinical trials with a dedicated electrical impedance myography system. Amyotroph. Lateral Scler. Front. Degener. 2018, 19, 555–561. [Google Scholar] [CrossRef]
- Sanchez, B.; Rutkove, S.B. Electrical Impedance Myography and Its Applications in Neuromuscular Disorders. Neurotherapeutics 2017, 14, 107–118. [Google Scholar] [CrossRef]
- Rutkove, S.B. Electrical impedance myography: Background, current state, and future directions. Muscle Nerve 2009, 40, 936–946. [Google Scholar] [CrossRef] [PubMed]
| Dual Echo Time Series | Multi-Echo | T1 Map (VFA) | mDixon | T2 Map | T1w TSE | |
|---|---|---|---|---|---|---|
| TR [ms] | 21 | 34 | 7.7 | 8.5 | 3605 | 598 |
| TE(s) [ms] | [1.06, 20.0] | [4.9, 11.4, 17.9, 24.4, 30.9] | 4.6 | 1.4 | [12.0, 18.1, 24.2, 30.3, 36.4, 42.5] | 8 |
| Flip Angle(s) [o] | 25 | 25 | [20,18, 16, 14, 12, 10, 8, 6, 4, 2] | 3 | 90 | 90 |
| Acq. Resolution [mm2] | 3.0 × 3.0 | 1.5 × 1.5 | 2.5 × 2.5 | 1.3 × 1.3 | 3.4 × 3.4 | 1.0 × 1.0 |
| Slice Thickness [mm] | 4.0 | 4.0 | 4.0 | 4.0 | 5.0 | 5.0 |
| # Dynamics | 150 | - | - | - | - | - |
| # Echoes | 2 | 5 | 1 | 6 | 6 | 1 |
| TSE factor | - | - | - | - | 6 | 5 |
| SENSE | 3.5 (RL), 1.5 (AP) | 2 (AP), 1.25 (FH) | 2 (AP) | - | 2 (AP) | - |
| FOV [mm3] | 300 × 230 × 248 | 300 × 230 × 248 | 300 × 230 × 248 | 300 × 230 × 195 | 300 × 230 × 248 | 300 × 230 × 248 |
| Scan Time [mm:ss] | 12:55 | 01:42 | 03:36 | 01:12 | 04:19 | 04:51 |
| Imaging Metric | TRATE [mM−1s−1] ∆R2*(t) [s−1] | T2* [ms] | T1 [ms] | Fat Fraction [%] | T2 [ms] | Signal Magnitude [a.u] |
| Muscle Group | Mean (SD) | 95% Confidence Interval | ||
|---|---|---|---|---|
| ALS Patient | Healthy Control | ALS Patient | Healthy Control | |
| Tibialis Anterior | 27.67 (5.54) | 71.09 (13.52) | [24.7, 30.7] | [62.5, 79.7] |
| Peroneus Longus | 52.75 (8.76) | 80.50 (15.52) | [48.0, 57.9] | [70.6, 90.4] |
| Tibialis Posterior | 43.94 (10.02) | 83.80 (15.14) | [38.5, 49.4] | [74.2, 93.4] |
| Medial Gastrocnemius | 58.72 (10.49) | 89.56 (15.35) | [53.0, 64.4] | [79.8, 99.3] |
| Lateral Gastrocnemius | 50.39 (12.67) | 85.81 (13.71) | [43.5, 57.3] | [77.1, 94.5] |
| Parameters | (Visit 2 − Visit 1)/Visit 1 [%] |
|---|---|
| ALSFRS-R Total [a.u.] | −4.70 * |
| ALSFRS-R Lower Limb [a.u.] | −13.04 * |
| HHD left [lbs] | −12.18 * |
| HHD Right [lbs] | −16.42 * |
| TRATE [mM−1 s−1] | −21.62 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ragunathan, S.; Bell, L.C.; Semmineh, N.; Stokes, A.M.; Shefner, J.M.; Bowser, R.; Ladha, S.; Quarles, C.C. Evaluation of Amyotrophic Lateral Sclerosis-Induced Muscle Degeneration Using Magnetic Resonance-Based Relaxivity Contrast Imaging (RCI). Tomography 2021, 7, 169-179. https://doi.org/10.3390/tomography7020015
Ragunathan S, Bell LC, Semmineh N, Stokes AM, Shefner JM, Bowser R, Ladha S, Quarles CC. Evaluation of Amyotrophic Lateral Sclerosis-Induced Muscle Degeneration Using Magnetic Resonance-Based Relaxivity Contrast Imaging (RCI). Tomography. 2021; 7(2):169-179. https://doi.org/10.3390/tomography7020015
Chicago/Turabian StyleRagunathan, Sudarshan, Laura C. Bell, Natenael Semmineh, Ashley M. Stokes, Jeremy M. Shefner, Robert Bowser, Shafeeq Ladha, and C. Chad Quarles. 2021. "Evaluation of Amyotrophic Lateral Sclerosis-Induced Muscle Degeneration Using Magnetic Resonance-Based Relaxivity Contrast Imaging (RCI)" Tomography 7, no. 2: 169-179. https://doi.org/10.3390/tomography7020015
APA StyleRagunathan, S., Bell, L. C., Semmineh, N., Stokes, A. M., Shefner, J. M., Bowser, R., Ladha, S., & Quarles, C. C. (2021). Evaluation of Amyotrophic Lateral Sclerosis-Induced Muscle Degeneration Using Magnetic Resonance-Based Relaxivity Contrast Imaging (RCI). Tomography, 7(2), 169-179. https://doi.org/10.3390/tomography7020015