Systematic Assessment of the Impact of DTI Methodology on Fractional Anisotropy Measures in Alzheimer’s Disease
Abstract
1. Introduction
2. Methodology
2.1. Subjects
2.2. MRI Protocols
2.3. DTI Processing and Fittings
2.4. Statistical Analysis
3. Results
3.1. Subjects
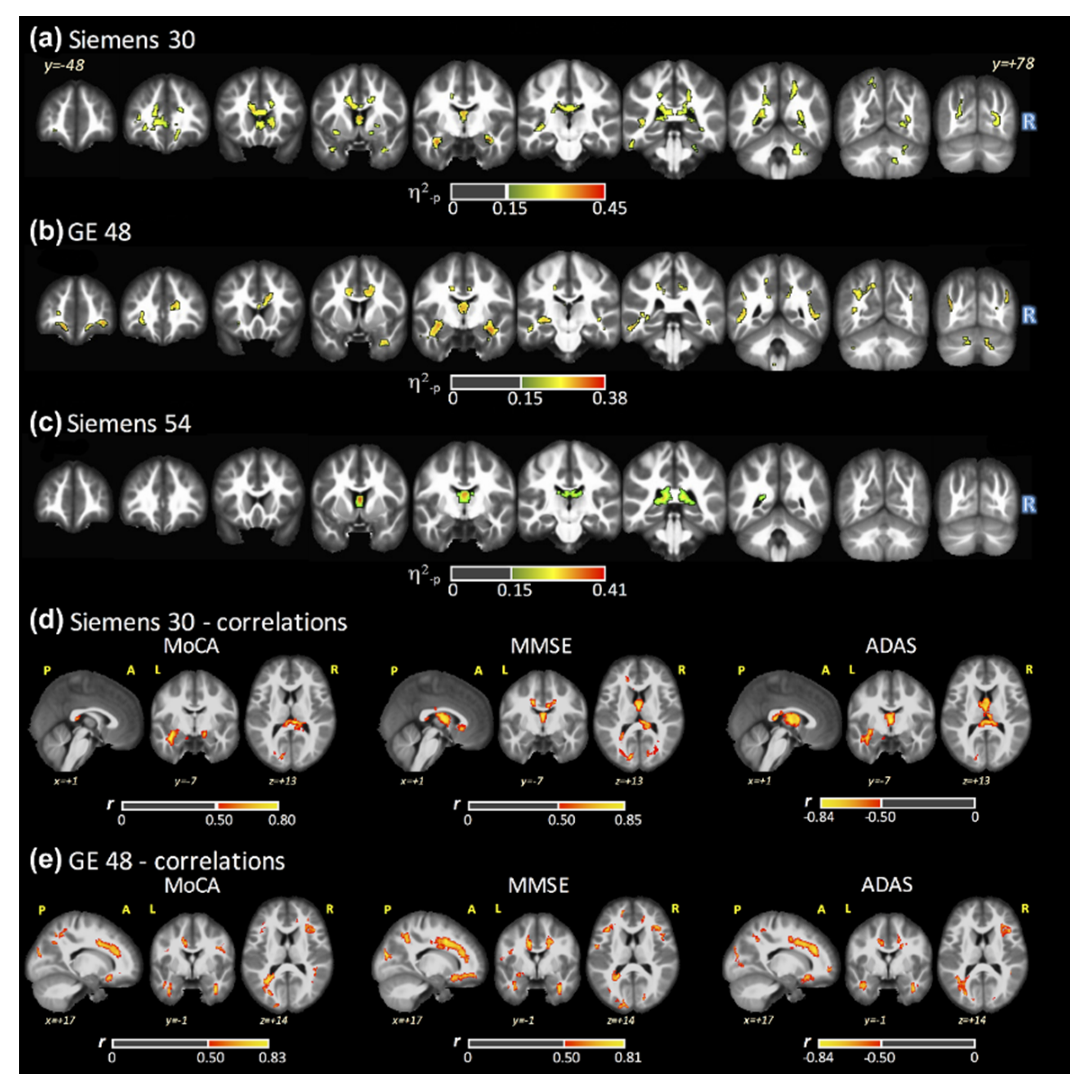
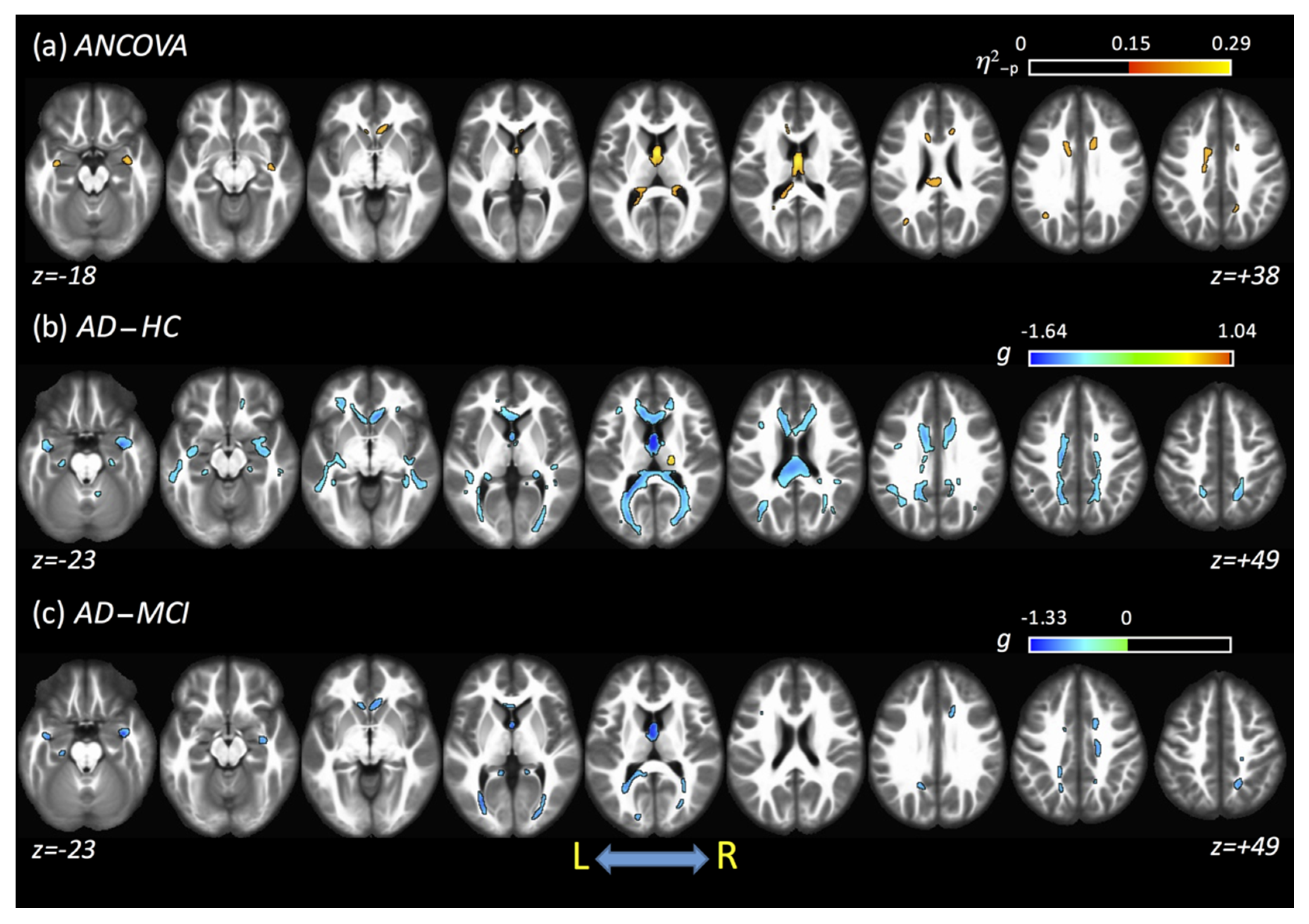
3.2. ANCOVA Results: FSL–LLS
3.3. FA Maps and Cognitive Scores Correlations
3.4. ANCOVA Results: All Fits
3.5. Post Hoc: AD versus HC
3.6. Post Hoc: AD versus MCI
3.7. Post Hoc: MCI versus HC
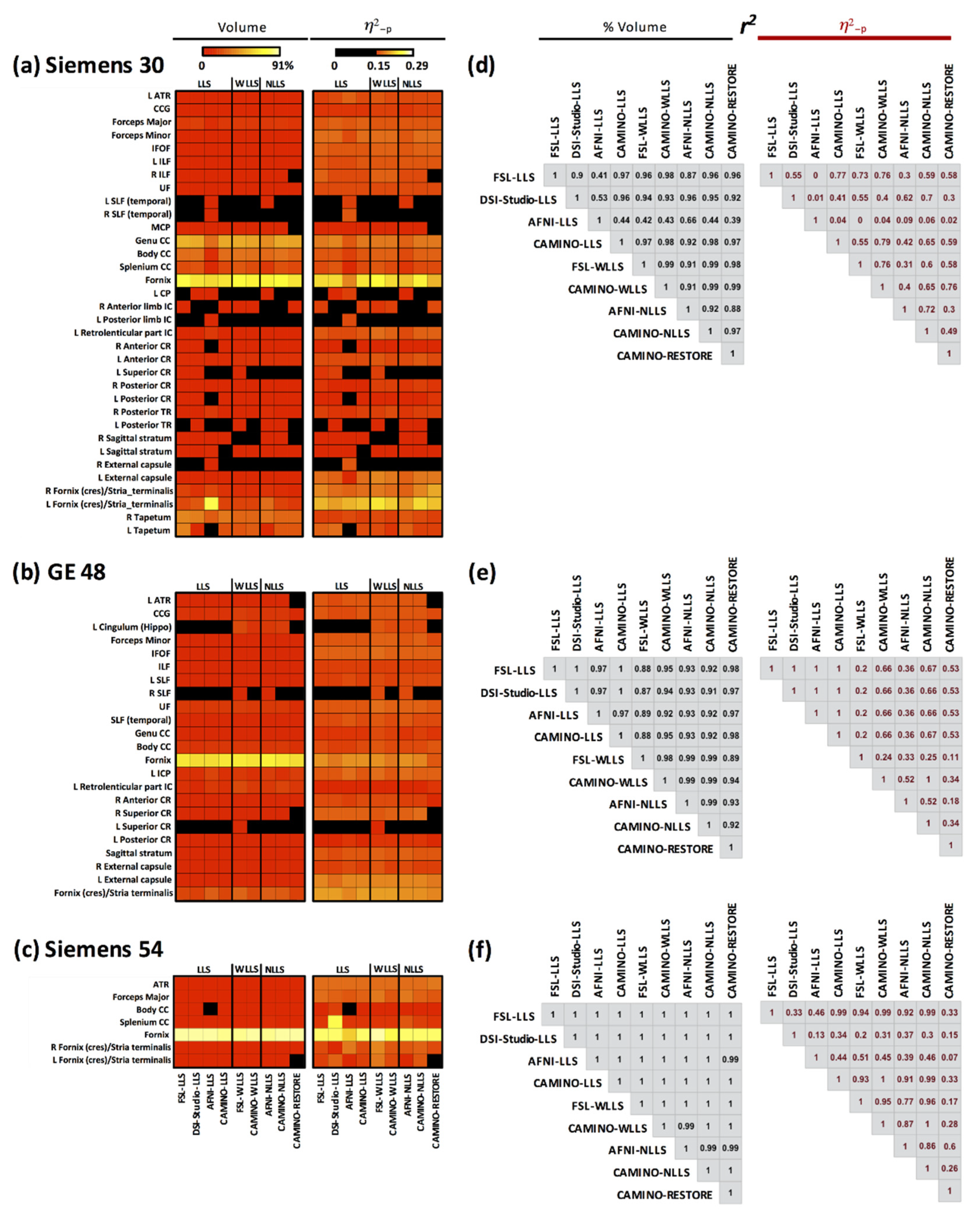
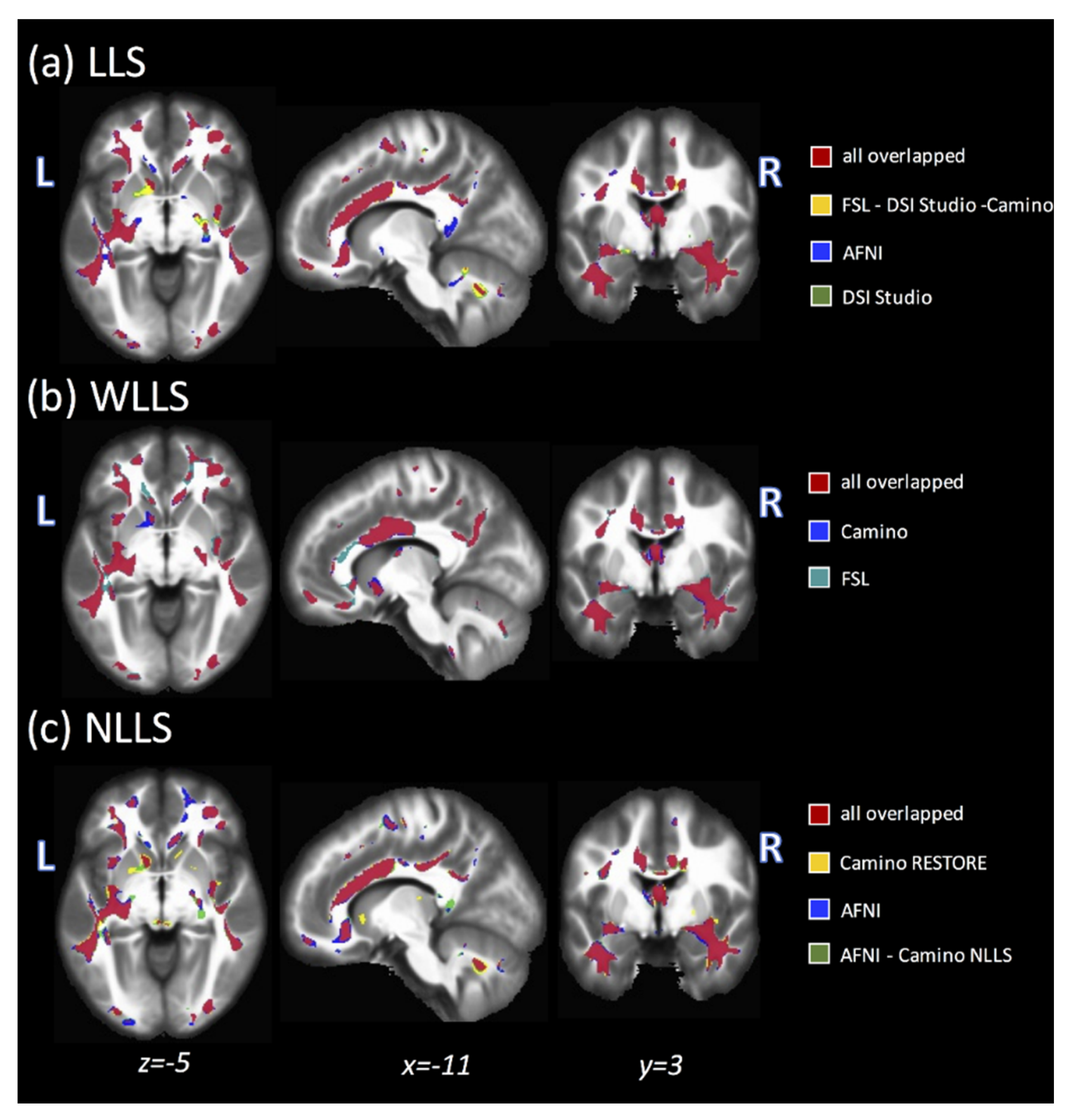
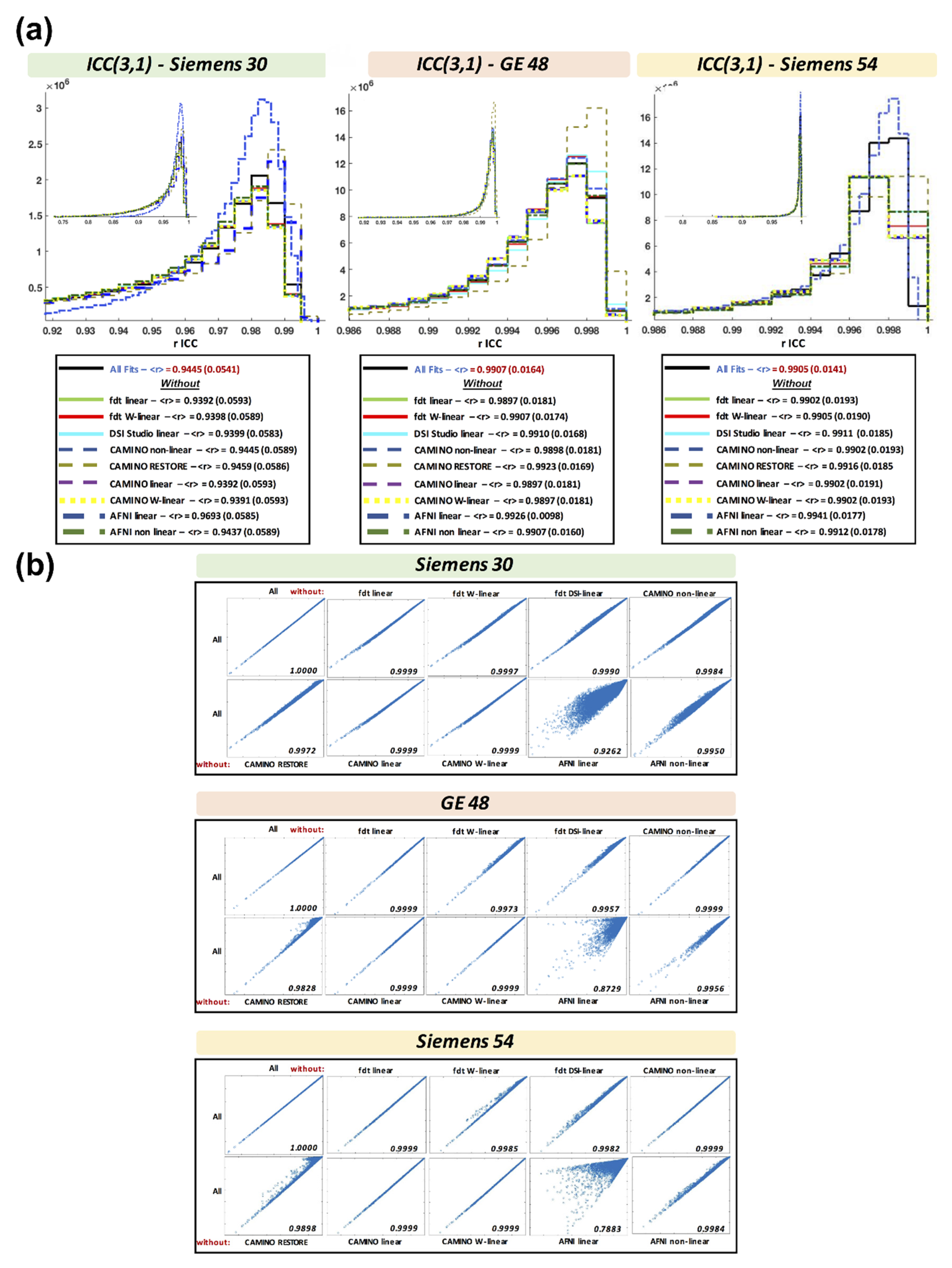
3.8. Intraclass Correlation Coefficient across Fits
3.9. Combined Results Using All Fits and All Acquisitions
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Crous-Bou, M.; Minguillón, C.; Gramunt, N.; Molinuevo, J.L. Alzheimer’s disease prevention: From risk factors to early intervention. Alzheimer’s Res. Ther. 2017, 9, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Jack, C.R., Jr.; Bennett, D.A.; Blennow, K.; Carrillo, M.C.; Dunn, B.; Haeberlein, S.B.; Silverberg, N. NIA-AA research framework: Toward a biological definition of Alzheimer’s disease. Alzheimer’s Dement. 2018, 14, 535–562. [Google Scholar] [CrossRef]
- Kelley, B.J.; Petersen, R.C. Alzheimer’s disease and mild cognitive impairment. Neurol. Clin. 2007, 25, 577–609. [Google Scholar] [CrossRef]
- Lara, E.; Koyanagi, A.; Olaya, B.; Lobo, A.; Miret, M.; Tyrovolas, S.; Haro, J.M. Mild cognitive impairment in a Spanish representative sample: Prevalence and associated factors. Int. J. Geriatr. Psychiatry 2016, 31, 858–867. [Google Scholar] [CrossRef] [PubMed]
- Hampel, H.; Bürger, K.; Teipel, S.J.; Bokde, A.L.; Zetterberg, H.; Blennow, K. Core candidate neurochemical and imaging biomarkers of Alzheimer’s disease. Alzheimer’s Dement. 2008, 4, 38–48. [Google Scholar] [CrossRef] [PubMed]
- Johansen-Berg, H. Behavioural relevance of variation in white matter microstructure. Curr. Opin. Neurol. 2010, 23, 351–358. [Google Scholar] [CrossRef]
- Vasconcelos, L.D.G.; Brucki, S.M.D.; Jackowiski, A.P.; Bueno, O.F.A. Diffusion tensor imaging for Alzheimer’s disease: A review of concepts and potential clinical applicability. Dement. Neuropsychol. 2009, 3, 268–274. [Google Scholar] [CrossRef]
- Chandra, A.; Dervenoulas, G.; Politis, M. Magnetic resonance imaging in Alzheimer’s disease and mild cognitive impairment. J. Neurol. 2019, 266, 1293–1302. [Google Scholar] [CrossRef]
- Hirni, D.I.; Kivisaari, S.L.; Monsch, A.U.; Taylor, K.I. Distinct neuroanatomical bases of episodic and semantic memory performance in Alzheimer’s disease. Neuropsychologia 2013, 51, 930–937. [Google Scholar] [CrossRef]
- Sjöbeck, M.; Elfgren, C.; Larsson, E.-M.; Brockstedt, S.; Latt, J.; Englund, E.M.; Passant, U. Alzheimer’s disease (AD) and executive dysfunction. A case-control study on the significance of frontal white matter changes detected by diffusion tensor imaging (DTI). Arch. Gerontol. Geriatr. 2010, 50, 260–266. [Google Scholar] [CrossRef]
- Mayo, C.D.; Garcia-Barrera, M.A.; Mazerolle, E.L.; Ritchie, L.J.; Fisk, J.D.; Gawryluk, J.R.; Initiative, A.D.N. Relationship Between DTI Metrics and Cognitive Function in Alzheimer’s Disease. Front. Aging Neurosci. 2019, 10, 436. [Google Scholar] [CrossRef] [PubMed]
- Wen, Q.; Mustafi, S.M.; Li, J.; Risacher, S.L.; Tallman, E.; Brown, S.A.; Wu, Y.C. White matter alterations in early-stage Alzheimer’s disease: A tract-specific study. Alzheimer’s Dement. Diagn. Assess. Dis. Monit. 2019, 11, 576–587. [Google Scholar] [CrossRef] [PubMed]
- Bergamino, M.; Kuplicki, R.; Victor, T.A.; Cha, Y.-H.; Paulus, M.P. Comparison of two different analysis approaches for DTI free-water corrected and uncorrected maps in the study of white matter microstructural integrity in individuals with depression. Hum. Brain Mapp. 2017, 38, 4690–4702. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.M.; Jenkinson, M.; Johansen-Berg, H.; Rueckert, D.; Nichols, T.E.; Mackay, C.E.; Watkins, K.E.; Ciccarelli, O.; Cader, M.Z.; Matthews, P.M.; et al. Tract-based spatial statistics: Voxelwise analysis of multi-subject diffusion data. Neuroimage 2006, 31, 1487–1505. [Google Scholar] [CrossRef] [PubMed]
- Bergamino, M.; Keeling, E.G.; Mishra, V.R.; Stokes, A.M.; Walsh, R.R. Assessing white matter pathology in early-stage parkinson disease using diffusion MRI: A systematic review. Front. Neurol. 2020, 11, 314. [Google Scholar] [CrossRef] [PubMed]
- Chang, L.-C.; Jones, D.K.; Pierpaoli, C. RESTORE: Robust estimation of tensors by outlier rejection. Magn. Reson. Med. 2005, 53, 1088–1095. [Google Scholar] [CrossRef] [PubMed]
- Chang, L.C.; Walker, L.; Pierpaoli, C. Informed RESTORE: A method for robust estimation of diffusion tensor from low redundancy datasets in the presence of physiological noise artifacts. Magn. Reson. Med. 2012, 68, 1654–1663. [Google Scholar] [CrossRef]
- Nasreddine, Z.S.; Phillips, N.A.; Bedirian, V.; Charbonneau, S.; Whitehead, V.; Collin, I.; Cummings, J.L.; Chertkow, H. The Montreal Cognitive Assessment, MoCA: A Brief Screening Tool for Mild Cognitive Impairment. J. Am. Geriatr. Soc. 2005, 53, 695–699. [Google Scholar] [CrossRef] [PubMed]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. Mini-mental state. A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Rosen, W.G.; Mohs, R.C.; Davis, K.L. A new rating scale for Alzheimer’s disease. Am. J. Psychiatry 1984, 141, 1356–1364. [Google Scholar]
- Shrout, P.E.; Fleiss, J.L. Intraclass correlations: Uses in assessing rater reliability. Psychol. Bull. 1979, 86, 420. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.M.; Jenkinson, M.; Woolrich, M.W.; Beckmann, C.F.; Behrens, T.E.; Johansen-Berg, H.; Bannister, P.R.; De Luca, M.; Drobnjak, I.; Flitney, D.E.; et al. Advances in functional and structural MR image analysis and implementation as FSL. Neuroimage 2004, 23, S208–S219. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Routledge: New York, NY, USA, 1988. [Google Scholar]
- Smith, S.M. Fast robust automated brain extraction. Hum. Brain Mapp. 2002, 17, 143–155. [Google Scholar] [CrossRef]
- Avants, B.B.; Epstein, C.L.; Grossman, M.; Gee, J.C. Symmetric diffeomorphic image registration with cross-correlation: Evaluating automated labeling of elderly and neurodegenerative brain. Med Image Anal. 2008, 12, 26–41. [Google Scholar] [CrossRef] [PubMed]
- Rheinheimer, D.C.; Penfield, D.A. The effects of type I error rate and power of the ANCOVA F test and selected alternatives under nonnormality and variance heterogeneity. J. Exp. Educ. 2001, 69, 373–391. [Google Scholar] [CrossRef]
- Sullivan, G.M.; Feinn, R. Using Effect Size—or Why the P Value Is Not Enough. J. Grad. Med Educ. 2012, 4, 279–282. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, L. Statistical methods in psychology journals: Guidelines and explanations. Am. Psychol. 1999, 54, 594. [Google Scholar] [CrossRef]
- Maximov, I.I.; Thönneßen, H.; Konrad, K.; Amort, L.; Neuner, I.; Shah, N.J. Statistical instability of TBSS analysis based on DTI fitting algorithm. J. Neuroimaging 2015, 25, 883–891. [Google Scholar] [CrossRef]
- Hua, K.; Zhang, J.; Wakana, S.; Jiang, H.; Li, X.; Reich, D.S.; Calabresi, P.A.; Pekar, J.J.; Van Zijl, P.C.M.; Mori, S. Tract probability maps in stereotaxic spaces: Analyses of white matter anatomy and tract-specific quantification. Neuroimage 2008, 39, 336–347. [Google Scholar] [CrossRef]
- Wakana, S.; Caprihan, A.; Panzenboeck, M.M.; Fallon, J.H.; Perry, M.; Gollub, R.L.; Mori, S. Reproducibility of quantitative tractography methods applied to cerebral white matter. Neuroimage 2007, 36, 630–644. [Google Scholar] [CrossRef]
- Koo, T.K.; Li, M.Y. A Guideline of Selecting and Reporting Intraclass Correlation Coefficients for Reliability Research. J. Chiropr. Med. 2016, 15, 155–163. [Google Scholar] [CrossRef]
- Jones, D.K. The effect of gradient sampling schemes on measures derived from diffusion tensor MRI: A Monte Carlo study. Magn. Reson. Med. 2004, 51, 807–815. [Google Scholar] [CrossRef] [PubMed]
- Makedonov, I.; Black, S.E.; MacIntosh, B.J. BOLD fMRI in the White Matter as a Marker of Aging and Small Vessel Disease. PLoS ONE 2013, 8, e67652. [Google Scholar] [CrossRef]
- Bergamino, M.; Farmer, M.; Yeh, H.-W.; Paul, E.; Hamilton, J.P. Statistical differences in the white matter tracts in subjects with depression by using different skeletonized voxel-wise analysis approaches and DTI fitting procedures. Brain Res. 2017, 1669, 131–140. [Google Scholar] [CrossRef]
- Maier-Hein, K.H.; Neher, P.F.; Houde, J.-C.; Côté, M.-A.; Garyfallidis, E.; Zhong, J.; Chamberland, M.; Yeh, F.-C.; Lin, Y.-C.; Renjie, H.; et al. The challenge of mapping the human connectome based on diffusion tractography. Nat. Commun. 2017, 8, 1–13. [Google Scholar] [CrossRef]
- Nir, T.M.; Jahanshad, N.; Villalon-Reina, J.E.; Toga, A.W.; Jack, C.R.; Weiner, M.W. Alzheimer’s Disease Neuroimaging Initiative (ADNI. Effectiveness of regional DTI measures in distinguishing Alzheimer’s disease, MCI, and normal aging. Neuroimage Clin. 2013, 3, 180–195. [Google Scholar] [CrossRef] [PubMed]
- Walsh, R.; Bergamino, M.; Stokes, A. Free-Water Diffusion Tensor Imaging (DTI) Improves the Accuracy and Sensitivity of White Matter Analysis in Alzheimer’s Disease. Neurology 2020, 94 (Suppl. 15), 4979. [Google Scholar]
- Oishi, K.; Lyketsos, C.G. Alzheimer’s disease and the fornix. Front. Aging Neurosci. 2014, 6, 241. [Google Scholar] [CrossRef]
- Mielke, M.M.; Kozauer, N.A.; Chan KC, G.; George, M.; Toroney, J.; Zerrate, M.; Albert, M. Regionally-specific diffusion tensor imaging in mild cognitive impairment and Alzheimer’s disease. Neuroimage 2009, 46, 47–55. [Google Scholar] [CrossRef]
- Nowrangi, M.A.; Lyketsos, C.G.; Leoutsakos JM, S.; Oishi, K.; Albert, M.; Mori, S.; Mielke, M.M. Longitudinal, region-specific course of diffusion tensor imaging measures in mild cognitive impairment and Alzheimer’s disease. Alzheimer’s Dement. 2013, 9, 519–528. [Google Scholar] [CrossRef]
- Fellgiebel, A.; Wille, P.; Müller, M.J.; Winterer, G.; Scheurich, A.; Vucurevic, G.; Schmidt, L.G.; Stoeter, P. Ultrastructural Hippocampal and White Matter Alterations in Mild Cognitive Impairment: A Diffusion Tensor Imaging Study. Dement. Geriatr. Cogn. Disord. 2004, 18, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Head, D.; Buckner, R.L.; Shimony, J.S.; Williams, L.E.; Akbudak, E.; Conturo, T.E.; Snyder, A.Z. Differential vulnerability of anterior white matter in nondemented aging with minimal acceleration in dementia of the Alzheimer type: Evidence from diffusion tensor imaging. Cereb. Cortex 2004, 14, 410–423. [Google Scholar] [CrossRef] [PubMed]
- Stahl, R.; Dietrich, O.; Teipel, S.J.; Hampel, H.; Reiser, M.F.; Schoenberg, S.O. White Matter Damage in Alzheimer Disease and Mild Cognitive Impairment: Assessment with Diffusion-Tensor MR Imaging and Parallel Imaging Techniques. Radiology 2007, 243, 483–492. [Google Scholar] [CrossRef]
- Bozzali, M.; Falini, A.; Franceschi, M.; Cercignani, M.; Zuffi, M.; Scotti, G.; Filippi, M. White matter damage in Alzheimer’s disease assessed in vivo using diffusion tensor magnetic resonance imaging. J. Neurol. Neurosurg. Psychiatry 2002, 72, 742–746. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Schuff, N.; Jahng, G.H.; Bayne, W.; Mori, S.; Schad, L.; Weiner, M.W. Diffusion tensor imaging of cingulum fibers in mild cognitive impairment and Alzheimer disease. Neurology 2007, 68, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Teipel, S.J.; Stahl, R.; Dietrich, O.; Schoenberg, S.O.; Perneczky, R.; Bokde, A.L.; Reiser, M.F.; Möller, H.-J.; Hampel, H. Multivariate network analysis of fiber tract integrity in Alzheimer’s disease. Neuroimage 2007, 34, 985–995. [Google Scholar] [CrossRef] [PubMed]
- Xie, S.; Xiao, J.X.; Gong, G.L.; Zang, Y.F.; Wang, Y.H.; Wu, H.K.; Jiang, X.X. Voxel-based detection of white matter abnormalities in mild Alzheimer disease. Neurology 2006, 66, 1845–1849. [Google Scholar] [CrossRef]
- Preti, M.G.; Baglio, F.; Laganà, M.M.; Griffanti, L.; Nemni, R.; Clerici, M.; Baselli, G. Assessing corpus callosum changes in Alzheimer’s disease: Comparison between tract-based spatial statistics and atlas-based tractography. PLoS ONE 2012, 7, e35856. [Google Scholar] [CrossRef] [PubMed]
| Across Groups | Dunn’s Test of Multiple Comparisons with Holm–Sidak Adjustment Z; p-Value | ||||||
| AD | HC | MCI | KRUSKAL–WALLIS RANK SUM TEST | AD vs HC | AD vs. MCI | HC vs. MCI | |
| SI30 | |||||||
| N (female) | 7 (3) | 38 (25) | 23 (14) | – | – | – | – |
| AGE | 75.9 (8.6) | 70.1 (4.0) | 70.9 (8.1) | H = 5.8; p = 0.054 | – | – | – |
| MMSE | 19.6 (4.9) | 29.4 (0.9) | 28.3 (1.7) | H = 26.6; p < 0.001 * | Z = −4.96; p < 0.001 * | Z = −3.16; p = 0.002 * | Z = 2.56; p = 0.010 * |
| MoCA | 12.6 (4.1) | 25.7 (2.6) | 22.5 (3.8) | H = 23.9; p < 0.001 * | Z = −4.64; p < 0.001 * | Z = −2.48; p = 0.013 * | Z = 2.55; p = 0.011 * |
| ADAS | 23.1 (4.0) | 8.6 (2.8) | 11.7 (3.0) | H = 26.8; p < 0.001 * | Z = 4.84; p < 0.001 * | Z = 2.54; p = 0.011 * | Z = −2.92; p = 0.003 * |
| GE48 | |||||||
| N (female) | 12 (2) | 41 (27) | 20 (9) | – | – | – | – |
| AGE | 75.6 (5.4) | 70.8 (5.8) | 72.7 (9.3) | H = 4.7; p = 0.094 | – | – | – |
| MMSE | 23.7 (1.9) | 29.3 (0.9) | 27.3 (2.0) | H = 40.6; p < 0.001 * | Z = −5.78; p < 0.001 * | Z = −2.64; p = 0.008 * | Z = 3.42; p = 0.001 * |
| MoCA | 16.1 (3.3) | 25.4 (2.6) | 21.4 (3.4) | H = 35.7; p < 0.001 * | Z = −5.59; p < 0.001 * | Z = −2.09; p = 0.036 * | Z = 3.52; p < 0.001 * |
| ADAS | 22.2 (6.2) | 8.5 (2.3) | 14.6 (5.2) | H = 38.0; p < 0.001 * | Z = 5.68; p < 0.001 * | Z = 1.93; p = 0.054 | Z = −3.85; p < 0.001 * |
| SI54 | |||||||
| N (female) | 11 (3) | 80 (56) | 36 (18) | – | – | – | – |
| AGE | 72.1 (9.2) | 69.6 (6.2) | 71.7 (6.5) | H = 6.13; p = 0.051 | – | – | – |
| MMSE | 24.1 (3.2) | 28.9 (1.2) | 27.3 (2.3) | H = 35.76; p < 0.001* | Z = −5.09; p < 0.001 * | Z = −2.39; p = 0.02 * | Z = 4.06; p < 0.001 * |
| MoCA | 17.6 (3.0) | 25.3 (2.7) | 21.6 (2.8) | H = 45.41; p < 0.001* | Z = −5.65; p < 0.001 * | Z = −2.20; p = 0.03 * | Z = 4.76; p < 0.001 * |
| ADAS | 19.4 (4.6) | 8.5 (2.2) | 12.6 (3.8) | H = 47.34; p < 0.001* | Z = 5.93; p < 0.001 * | Z = 2.54; p = 0.01 * | Z = −4.63; p < 0.001 * |
| Across Scanners | Dunn’s Test of Multiple Comparisons with Holm-Sidak Adjustment Z; p-Value | ||||||
| SI30 | GE48 | SI54 | KRUSKAL–WALLIS RANK SUM TEST | GE48 vs. SI30 | GE48 vs. SI54 | S30 vs. SI54 | |
| HC | |||||||
| N (female) | 38 (25) | 41 (27) | 80 (56) | – | – | – | – |
| AGE | 70.08 (4.04) | 70.79 (5.75) | 69.65 (6.27) | H = 1.50; p = 0.52 | – | – | |
| MMSE | 29.37 (0.94) | 29.33 (0.89) | 28.96 (1.16) | H = 3.91; p = 0.14 | – | – | – |
| MoCA | 25.71 (2.61) | 25.45 (2.63) | 25.27 (2.68) | H = 0.39; p = 0.82 | – | – | – |
| ADAS | 8.59 (2.76) | 8.47 (2.25) | 8.54 (2.16) | H = 0.10; p = 0.95 | – | – | – |
| MCI | |||||||
| N (female) | 23 (14) | 20 (9) | 36 (18) | – | – | – | – |
| AGE | 70.89 (8.14) | 72.68 (9.31) | 71.75 (6.56) | H = 0.21; p = 0.90 | – | – | – |
| MMSE | 28.30 (1.69) | 27.30 (2.00) | 27.30 (2.25) | H = 4.41; p = 0.11 | – | – | – |
| MoCA | 22.47 (3.81) | 21.44 (3.37) | 21.62 (2.82) | H = 1.59; p = 0.45 | – | – | – |
| ADAS | 11.74 (3.02) | 14.58 (5.18) | 12.55 (3.77) | H = 2.38; p = 0.30 | – | – | – |
| AD | |||||||
| N (female) | 7 (3) | 12 (2) | 11 (3) | – | – | – | – |
| AGE | 75.8 (8.6) | 75.6 (5.4) | 72.1 (9.2) | H = 1.14; p = 0.57 | – | – | – |
| MMSE | 19.52 (4.91) | 23.62 (1.92) | 24.14 (3.21) | H = 5.97; p = 0.05 | – | – | – |
| MoCA | 12.57 (4.12) | 16.11 (3.43) | 17.51 (3.03) | H = 6.90; p = 0.03 * | Z = 1.55; p = 0.122 | Z = −1.28; p = 0.201 | Z = −2.62; p = 0.010 * |
| ADAS | 23.11 (4.02) | 22.23 (6.71) | 19.42 (4.61) | H = 4.62; p = 0.10 | – | – | – |
| Fit Algorithm | Tool | Web Page | Command | Option |
|---|---|---|---|---|
| LLS | FSL | https://fsl.fmrib.ox.ac.uk/fsl/fslwiki | dtifit | default |
| LLS | DSI Studio | http://dsi-studio.labsolver.org/ | dsi_studio | default |
| LLS | CAMINO | http://camino.cs.ucl.ac.uk/index.php | modelfit | -inversion 1 (ldt) |
| LLS | AFNI | https://afni.nimh.nih.gov/ | 3dDWItoDT | -linear |
| WLLS | FSL | https://fsl.fmrib.ox.ac.uk/fsl/fslwiki | dtifit | --wls |
| WLLS | CAMINO | http://camino.cs.ucl.ac.uk/index.php | modelfit | -inversion 7 (ldt_wtd) |
| NLLS | CAMINO | http://camino.cs.ucl.ac.uk/index.php | modelfit | -inversion 4 (nldt_pos nldt) |
| NLLS | AFNI | https://afni.nimh.nih.gov/ | 3dDWItoDT | -nonlinear |
| RESTORE | CAMINO | http://camino.cs.ucl.ac.uk/index.php | restore | default |
| ANCOVA | AD vs. HC | AD vs. MCI | ||
| JHU WM Tractography | Volume % | AD < HC Volume % | AD > HC Volume % | AD < MCI Volume % |
| ATR | 0.67 | 1.43 | – | 0.23 |
| Right CST | – | – | 0.34 | 0.68 |
| CCG | 0.89 | 11.11 | – | 0.97 |
| Cingulum Hippo | – | 5.13 | – | 3.30 |
| Forceps Major | 1.57 | 14.05 | – | 8.27 |
| Forceps Minor | 1.05 | 7.13 | – | 1.52 |
| Left IFOF | – | 5.51 | – | 2.14 |
| Right IFOF | 0.36 | 6.74 | – | 2.49 |
| ILF | 0.14 | 6.11 | – | 2.67 |
| UF | 0.86 | 7.74 | – | 3.40 |
| ICBM-DTI-81 WM | Volume % | AD < HC Volume % | AD > HC Volume % | AD < MCI Volume % |
| Genu of CC | 6.05 | 42.04 | – | 10.52 |
| Body of CC | 9.59 | 44.06 | – | 2.30 |
| Splenium of CC | 3.60 | 25.97 | – | 2.27 |
| Fornix | 87.38 | 89.05 | – | 83.11 |
| Right posterior limb of IC | – | 1.77 | – | |
| Left retrolenticular IC | – | 4.71 | – | – |
| Right ACR | – | 5.15 | – | 0.74 |
| Right ACR | 0.39 | 3.85 | – | 7.49 |
| Left ACR | 0.68 | 4.04 | – | – |
| Right PCR | – | 7.76 | – | – |
| Left PCR | – | 7.48 | – | 1.95 |
| PTR | – | 14.70 | – | 7.57 |
| SS | 0.34 | 4.48 | – | – |
| Right EC | 0.77 | 2.82 | – | 0.62 |
| Left EC | – | 5.29 | – | – |
| Fornix (cres)/Stria terminalis | 8.34 | 17.78 | – | 7.03 |
| Right tapetum | – | 24.75 | – | 4.11 |
| Left tapetum | 1.22 | 8.90 | – | 2.56 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bergamino, M.; Keeling, E.G.; Walsh, R.R.; Stokes, A.M. Systematic Assessment of the Impact of DTI Methodology on Fractional Anisotropy Measures in Alzheimer’s Disease. Tomography 2021, 7, 20-38. https://doi.org/10.3390/tomography7010003
Bergamino M, Keeling EG, Walsh RR, Stokes AM. Systematic Assessment of the Impact of DTI Methodology on Fractional Anisotropy Measures in Alzheimer’s Disease. Tomography. 2021; 7(1):20-38. https://doi.org/10.3390/tomography7010003
Chicago/Turabian StyleBergamino, Maurizio, Elizabeth G. Keeling, Ryan R. Walsh, and Ashley M. Stokes. 2021. "Systematic Assessment of the Impact of DTI Methodology on Fractional Anisotropy Measures in Alzheimer’s Disease" Tomography 7, no. 1: 20-38. https://doi.org/10.3390/tomography7010003
APA StyleBergamino, M., Keeling, E. G., Walsh, R. R., & Stokes, A. M. (2021). Systematic Assessment of the Impact of DTI Methodology on Fractional Anisotropy Measures in Alzheimer’s Disease. Tomography, 7(1), 20-38. https://doi.org/10.3390/tomography7010003