Complicated Liver Cystic Echinococcosis—A Comprehensive Literature Review and a Tale of Two Extreme Cases
Abstract
1. Introduction
2. Materials and Methods
3. Case Reports
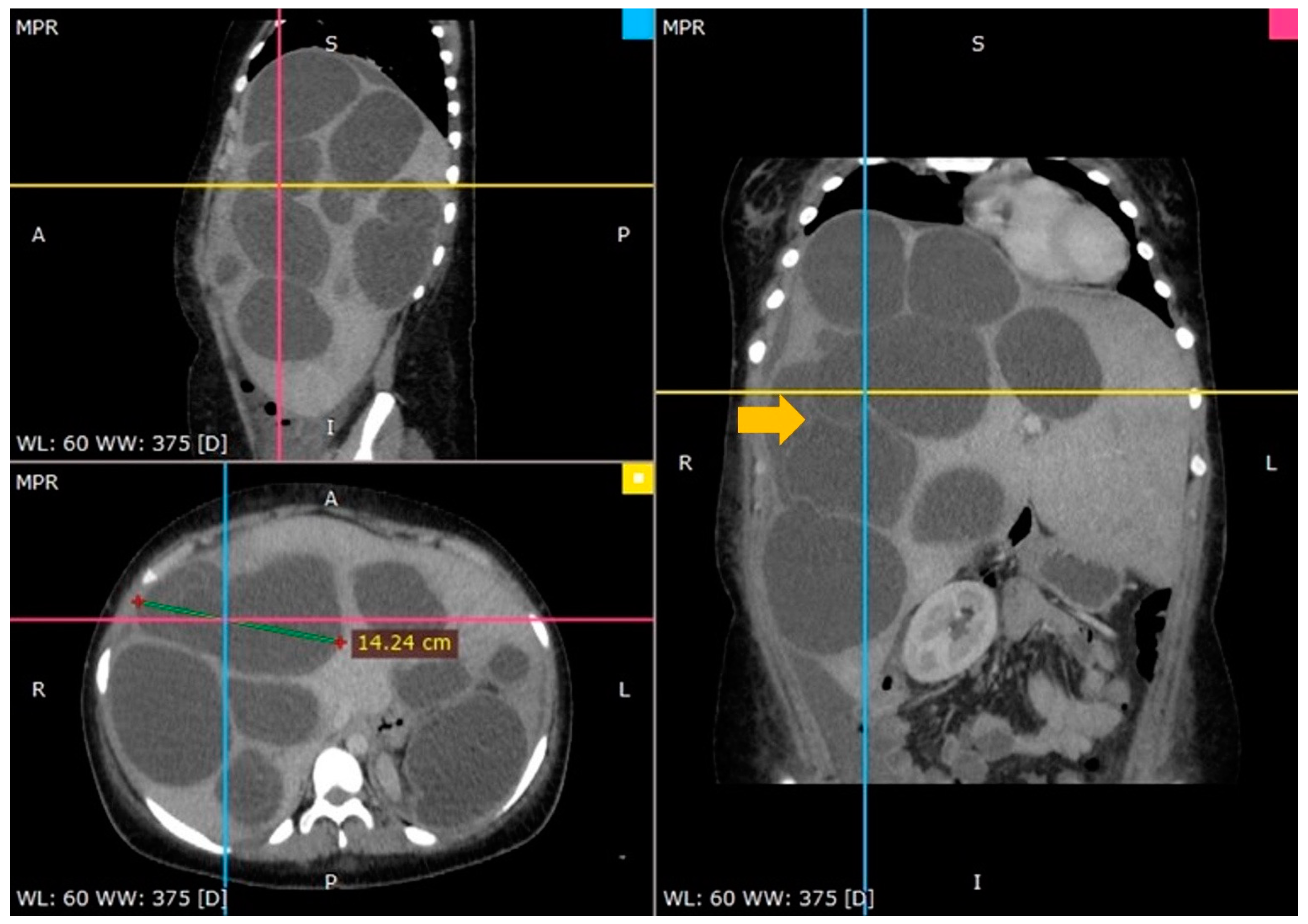
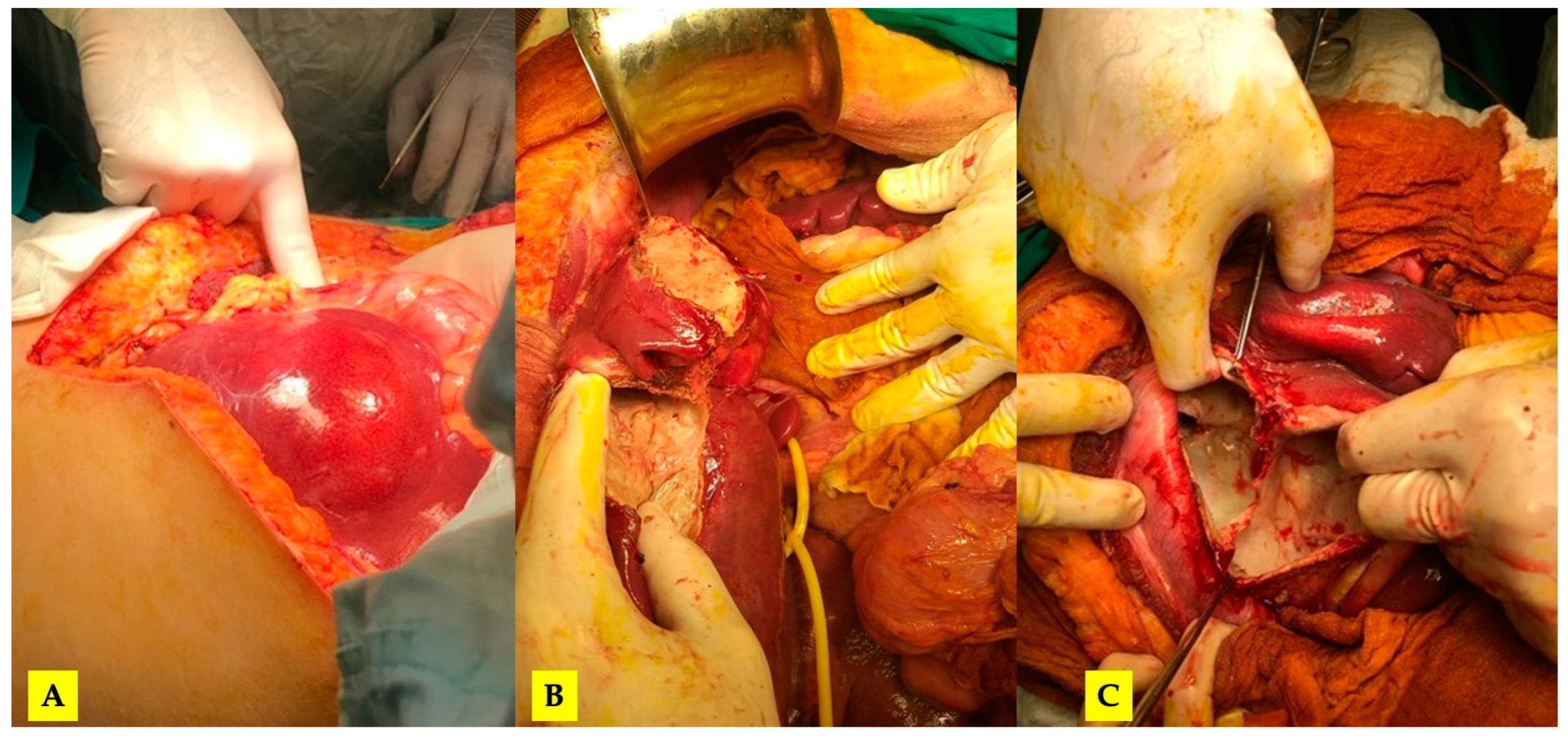
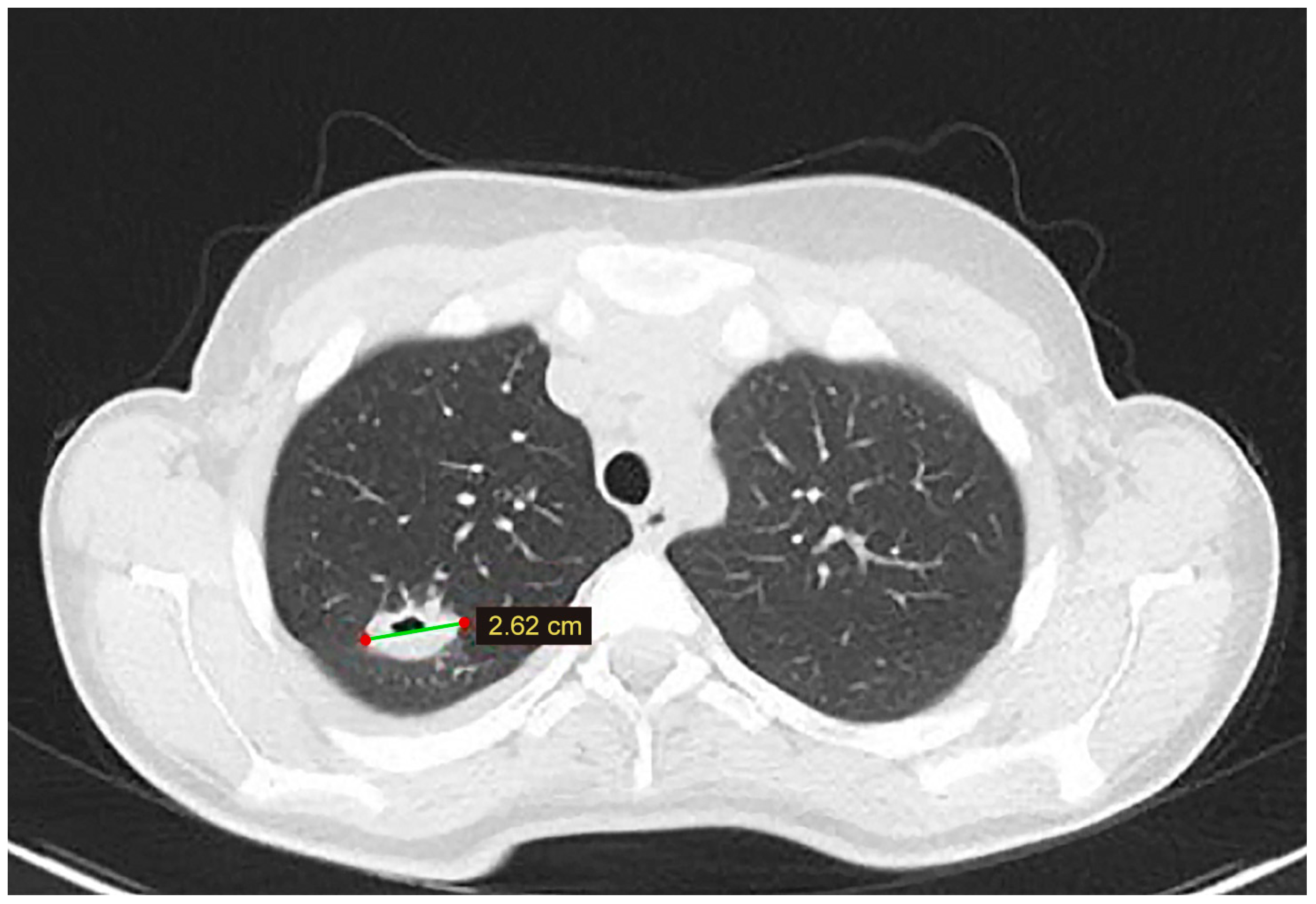
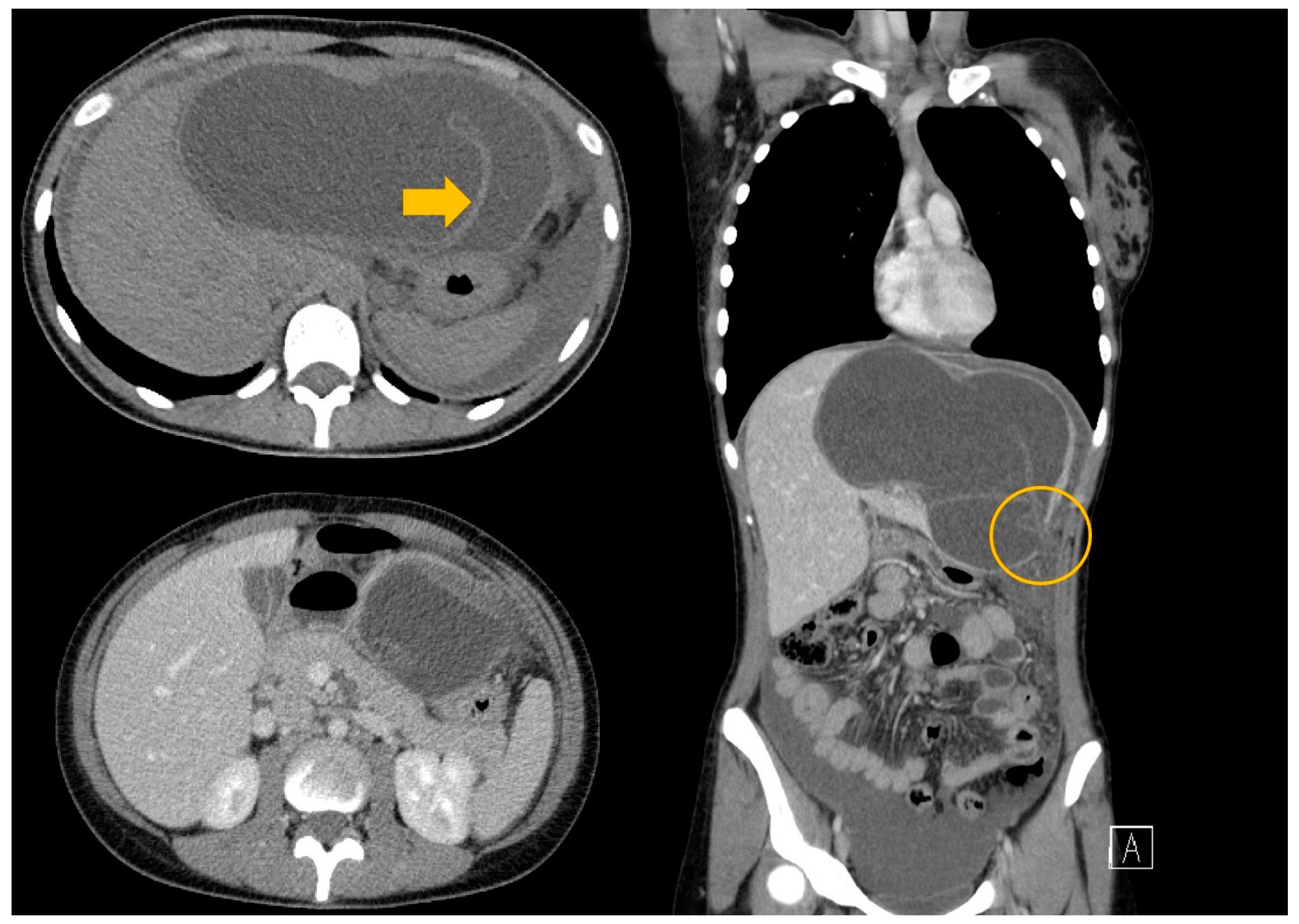
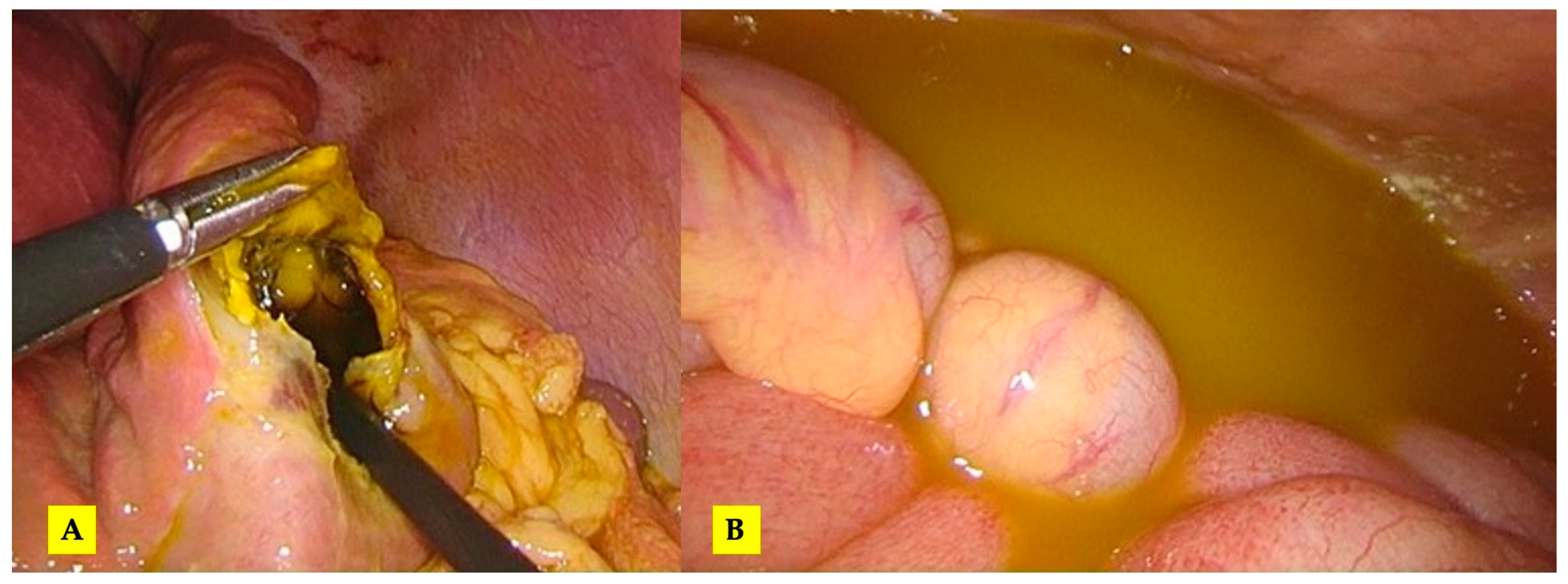
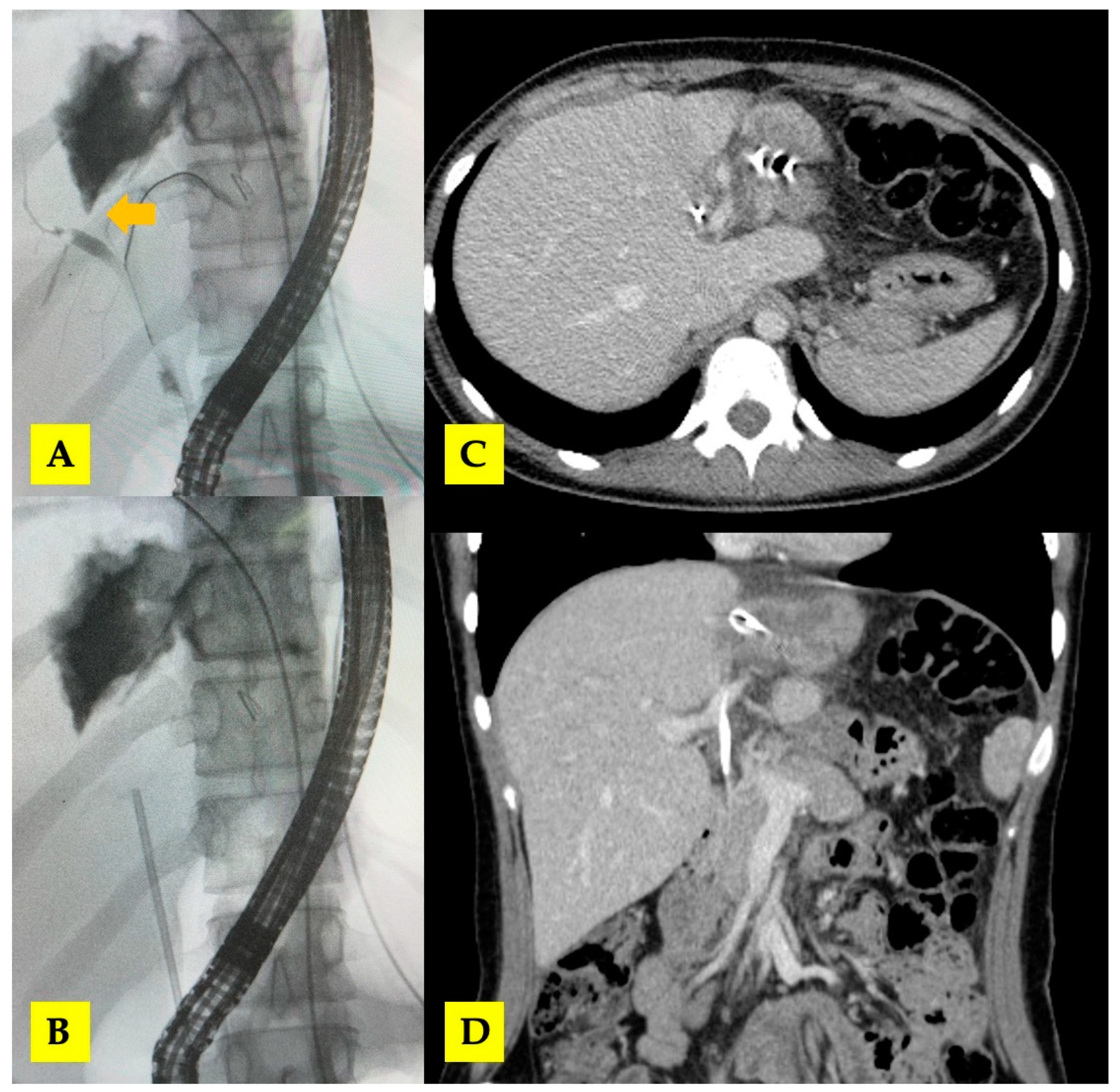
3.1. Case 1
3.2. Case 2
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gessese, A.T. Review on Epidemiology and Public Health Significance of Hydatidosis. Vet. Med. Int. 2020, 2020, 8859116. [Google Scholar] [CrossRef]
- Grosso, G.; Gruttadauria, S.; Biondi, A.; Marventano, S.; Mistretta, A. Worldwide epidemiology of liver hydatidosis including the Mediterranean area. World J. Gastroenterol. WJG 2012, 18, 1425. [Google Scholar] [CrossRef]
- Thompson, R.A.; McManus, D.P. Towards a taxonomic revision of the genus Echinococcus. Trends Parasitol. 2002, 18, 452–457. [Google Scholar] [CrossRef]
- Christodoulidis, G.; Samara, A.A.; Diamantis, A.; Floros, T.; Sgantzou, I.K.; Karakantas, K.S.; Zotos, P.A.; Koutras, A.; Janho, M.B.; Tepetes, K. Reaching the Challenging Diagnosis of Complicated Liver Hydatid Disease: A Single Institution’s Experience from an Endemic Area. Medicina 2021, 57, 1210. [Google Scholar] [CrossRef]
- Acarli, K. Controversies in the laparoscopic treatment of hepatic hydatid disease. HPB 2004, 6, 213–221. [Google Scholar] [CrossRef]
- Derbel, F.; Ben, M.; Hadj Hamida, M.B.; Mazhoud, J.; Youssef, S.; Ben, A.; Jemni, H.; Mama, N.; Ibtissem, H.; Nadia, A.; et al. Hydatid Cysts of the Liver–Diagnosis, Complications and Treatment. In Abdominal Surgery; Fethi, D., Ed.; IntechOpen: Rijeka, Croatia, 2012; Chapter 5. [Google Scholar]
- Peters, L.; Burkert, S.; Grüner, B. Parasites of the liver–epidemiology, diagnosis and clinical management in the European context. J. Hepatol. 2021, 75, 202–218. [Google Scholar] [CrossRef]
- El Fortia, M.; El Gatit, A.; Bendaoud, M. Ultrasound wall-sign in pulmonary echinococcosis (new application). Ultraschall Med. 2006, 27, 553–557. [Google Scholar] [CrossRef]
- Çankaya, B.A.-O.; Yeşilyurt, M. Giant hydatid cysts in pregnancy: A rare presentation. Rev. Soc. Bras. Med. Trop. 2021, 54, e0500-2020. [Google Scholar] [CrossRef]
- Bhutani, N.; Kajal, P. Hepatic echinococcosis: A review. Ann. Med. Surg. 2018, 36, 99–105. [Google Scholar] [CrossRef]
- Zhang, W.; Li, J.; McManus, D.P. Concepts in immunology and diagnosis of hydatid disease. Clin. Microbiol. Rev. 2003, 16, 18–36. [Google Scholar] [CrossRef]
- Ettorre, G.M.; Vennarecci, G.; Santoro, R.; Laurenzi, A.; Ceribelli, C.; Di Cintio, A.; Rizzi, E.B.; Antonini, M. Giant hydatid cyst of the liver with a retroperitoneal growth: A case report. J. Med. Case Rep. 2012, 6, 298. [Google Scholar] [CrossRef] [PubMed]
- Budke, C.M.; Carabin, H.; Ndimubanzi, P.C.; Nguyen, H.; Rainwater, E.; Dickey, M.; Bhattarai, R.; Zeziulin, O.; Qian, M.B. A systematic review of the literature on cystic echinococcosis frequency worldwide and its associated clinical manifestations. Am. J. Trop. Med. Hyg. 2013, 88, 1011–1027. [Google Scholar] [CrossRef] [PubMed]
- Sayek, I.; Yalin, R.; Sanac, Y. Surgical treatment of hydatid disease of the liver. Arch. Surg. 1980, 115, 847–850. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Wen, H.; Li, J.; Lin, R.; McManus, D.P. Immunology and immunodiagnosis of cystic echinococcosis: An update. Clin. Dev. Immunol. 2012, 2012, 101895. [Google Scholar] [CrossRef] [PubMed]
- Moro, P.L.; Bonifacio, N.; Gilman, R.H.; Lopera, L.; Silva, B.; Takumoto, R.; Verastegui, M.; Cabrera, L. Field diagnosis of Echinococcus granulosus infection among intermediate and definitive hosts in an endemic focus of human cystic echinococcosis. Trans. R. Soc. Trop. Med. Hyg. 1999, 93, 611–615. [Google Scholar] [CrossRef] [PubMed]
- Ferrer-Inaebnit, E.; Molina-Romero, F.X.; Segura-Sampedro, J.J.; González-Argenté, X.; Canis, J.M.M.; Ester, F.-I.; Xavier, M.-R.F.; José, S.-S.J.; Xavier, G.-A. A review of the diagnosis and management of liver hydatid cyst. Rev. Esp. Enfermedades Dig. 2021, 114, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Mihmanli, M.; Idiz, U.O.; Kaya, C.; Demir, U.; Bostanci, O.; Omeroglu, S.; Bozkurt, E. Current status of diagnosis and treatment of hepatic echinococcosis. World J. Hepatol. 2016, 8, 1169. [Google Scholar] [CrossRef] [PubMed]
- Pascal, G.; Azoulay, D.; Belghiti, J.; Laurent, A. Hydatid disease of the liver. In Blumgart’s Surgery of the Liver, Biliary Tract and Pancreas, 2-Volume Set; Elsevier: Amsterdam, The Netherlands, 2017; pp. 1102–1121.e3. [Google Scholar]
- Secchi, M.A.; Pettinari, R.; Mercapide, C.; Bracco, R.; Castilla, C.; Cassone, E.; Sisco, P.; Andriani, O.; Rossi, L.; Grondona, J.; et al. Surgical management of liver hydatidosis: A multicentre series of 1412 patients. Liver Int. 2010, 30, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Sokouti, M.; Sadeghi, R.; Pashazadeh, S.; Abadi, S.E.H.; Sokouti, M.; Ghojazadeh, M.; Sokouti, B. A systematic review and meta-analysis on the treatment of liver hydatid cyst using meta-MUMS tool: Comparing PAIR and laparoscopic procedures. Arch. Med. Sci. 2019, 15, 284–308. [Google Scholar] [CrossRef] [PubMed]
- Marrero, J.A.; Ahn, J.; Reddy, R.K.; Gastroenterology PPCotACo. ACG clinical guideline: The diagnosis and management of focal liver lesions. Off. J. Am. Coll. Gastroenterol.|ACG 2014, 109, 1328–1347. [Google Scholar] [CrossRef]
- Velasco-Tirado, V.; Romero-Alegría, Á.; Belhassen-García, M.; Alonso-Sardón, M.; Esteban-Velasco, C.; López-Bernús, A.; Carpio-Perez, A.; López, M.F.J.; Bellido, J.L.M.; Muro, A. Recurrence of cystic echinococcosis in an endemic area: A retrospective study. BMC Infect. Dis. 2017, 17, 455. [Google Scholar] [CrossRef] [PubMed]
- Derbel, F.; Mabrouk, M.B.; Hamida, M.B.H.; Mazhoud, J.; Youssef, S.; Ali, A.B.; Jemni, H.; Mama, N.; Ibtissem, H.; Nadia, A. Hydatid cysts of the liver-diagnosis, complications and treatment. Abdom. Surg. 2012, 5, 105–138. [Google Scholar]
- Salamone, G.; Licari, L.; Randisi, B.; Falco, N.; Tutino, R.; Vaglica, A.; Gullo, R.; Porrello, C.; Cocorullo, G.; Gulotta, G. Uncommon localizations of hydatid cyst. Review of the literature. Il G. Chir. 2016, 37, 180. [Google Scholar] [CrossRef] [PubMed]
- Alghofaily, K.A.; Saeedan, M.B.; Aljohani, I.M.; Alrasheed, M.; McWilliams, S.; Aldosary, A.; Neimatallah, M. Hepatic hydatid disease complications: Review of imaging findings and clinical implications. Abdom. Radiol. 2017, 42, 199–210. [Google Scholar] [CrossRef] [PubMed]
- Ahrabi, S.Z.; Madani, R.; Bavili, M.M.; Bedoustani, A.B. Incidence of cystic echinococcosis in the East Azerbaijan, Iran, during 2011–2017: A retrospective epidemiological study. Clin. Exp. Health Sci. 2021, 11, 158–162. [Google Scholar] [CrossRef]
- Eisenman, E.J.L.; Uhart, M.M.; Kusch, A.; Vila, A.R.; Vanstreels, R.E.T.; Mazet, J.A.K.; Briceño, C. Increased prevalence of canine echinococcosis a decade after the discontinuation of a governmental deworming program in Tierra del Fuego, Southern Chile. Zoonoses Public Health 2023, 70, 213–222. [Google Scholar] [CrossRef] [PubMed]
- Khachatryan, A.S. Analysis of lethality in echinococcal disease. Korean J. Parasitol. 2017, 55, 549. [Google Scholar] [CrossRef] [PubMed]
- Chautems, R.; Bühler, L.H.; Gold, B.; Giostra, E.; Poletti, P.; Chilcott, M.; Morel, P.; Mentha, G. Surgical management and long-term outcome of complicated liver hydatid cysts caused by Echinococcus granulosus. Surgery 2005, 137, 312–316. [Google Scholar] [CrossRef]
- Gomez, I.G.C.; Lopez-Andujar, R.; Belda Ibanez, T.; Ramia Angel, J.M.; Moya Herraiz, A.; Orbis Castellanos, F.; Pareja Ibars, E.; San Juan Rodriguez, F. Review of the treatment of liver hydatid cysts. World J. Gastroenterol. 2015, 21, 124–131. [Google Scholar] [CrossRef]
- Aghajanzadeh, M.; Shafaghi, A.; Ashoobi, M.T.; Mashhor, M.Y.; Rad, O.M.; Farzin, M. Clinical presentation, diagnosis and surgical treatment of intrabiliary ruptured hydatid disease of the liver. GSC Adv. Res. Rev. 2023, 16, 162–170. [Google Scholar] [CrossRef]
- Mahajan, S.; Thapar, S.; Khillan, V.; Gupta, P.; Rastogi, A.; Gupta, E. Comparative evaluation of echinococcus serology with cytology for the diagnosis of hepatic hydatid disease. J. Lab. Physicians 2020, 12, 98–102. [Google Scholar] [CrossRef] [PubMed]
- Bagga, P.K.; Bhargava, S.K.; Aggarwal, N.; Chander, Y. Primary subcutaneous inguinal hydatid cyst: Diagnosis by fine needle aspiration cytology. J. Clin. Diagn. Res. 2014, 8, Fd11–Fd13. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Zhang, T.; Zhao, Y.; Wang, Y.; Lv, Y.; Li, S.; Yang, S.; Zhu, M.; Zhao, W.; Wang, Q. Screening, construction, and serological identification of the diagnostic antigen molecule EG-06283 for the diagnosis of Echinococcus granulosus. Parasitol. Res. 2021, 120, 2557–2567. [Google Scholar] [CrossRef] [PubMed]
- Maleki, F.; Akhlaghi, L.; Tabatabaie, F. Evaluation of Hydatid Cyst Antigen for Serological Diagnosis. Med. J. Islam. Repub. Iran 2023, 37, 87. [Google Scholar] [CrossRef] [PubMed]
- Inan, N.; Arslan, A.; Akansel, G.; Anik, Y.; Sarisoy, H.T.; Ciftci, E.; Demirci, A. Diffusion-weighted imaging in the differential diagnosis of simple and hydatid cysts of the liver. Am. J. Roentgenol. 2007, 189, 1031–1036. [Google Scholar] [CrossRef] [PubMed]
- Kim, A.R.; Park, S.J.; Gu, M.J.; Choi, J.H.; Kim, H.J. Fine needle aspiration cytology of hepatic hydatid cyst: A case study. Korean J. Pathol. 2013, 47, 395–398. [Google Scholar] [CrossRef]
- Stojkovic, M.; Junghanss, T.; Veeser, M.; Weber, T.F.; Sauer, P. Endoscopic Treatment of Biliary Stenosis in Patients with Alveolar Echinococcosis--Report of 7 Consecutive Patients with Serial ERC Approach. PLoS Neglected Trop. Dis. 2016, 10, e0004278. [Google Scholar] [CrossRef] [PubMed]
- Vuitton, D.A. The WHO Informal Working Group on Echinococcosis. Coordinating Board of the WHO-IWGE. Parassitologia 1997, 39, 349–353. [Google Scholar]
- Rinaldi, F.; Brunetti, E.; Neumayr, A.; Maestri, M.; Goblirsch, S.; Tamarozzi, F. Cystic echinococcosis of the liver: A primer for hepatologists. World J. Hepatol. 2014, 6, 293. [Google Scholar] [CrossRef]
- Tamarozzi, F.; Akhan, O.; Cretu, C.M.; Vutova, K.; Akinci, D.; Chipeva, R.; Ciftci, T.; Constantin, C.M.; Fabiani, M.; Golemanov, B.; et al. Prevalence of abdominal cystic echinococcosis in rural Bulgaria, Romania, and Turkey: A cross-sectional, ultrasound-based, population study from the HERACLES project. Lancet Infect. Dis. 2018, 18, 769–778. [Google Scholar] [CrossRef]
- Ran, B.; Aji, T.; Jiang, T.; Zhang, R.; Guo, Q.; Abulizi, A.; Yimiti, Y.; Wen, H.; Shao, Y. Differentiation between hepatic cystic echinococcosis types 1 and simple hepatic cysts: A retrospective analysis. Medicine 2019, 98, e13731. [Google Scholar] [CrossRef]
- Sayek, I.; Onat, D. Diagnosis and treatment of uncomplicated hydatid cyst of the liver. World J. Surg. 2001, 25, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Trigui, A.; Toumi, N.; Fendri, S.; Saumtally, M.S.; Zribi, I.; Akrout, A.; Mzali, R.; Ketata, S.; Dziri, C.; Amar, M.B. Cystic Echinococcosis of the Liver: Correlation Between Intra-Operative Ultrasound and Pre-Operative Imaging. Surg. Infect. 2024, 25, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Elmoghazy, W.; Alqahtani, J.; Kim, S.W.; Sulieman, I.; Elaffandi, A.; Khalaf, H. Comparative analysis of surgical management approaches for hydatid liver cysts: Conventional vs. minimally invasive techniques. Langenbeck’s Arch. Surg. 2023, 408, 320. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.; Mahar, T.; Manan, A.; Ahmad, I.; Qadir, A.; Usman, M.; Waseem, M. Cystobiliary fistula in Hepatic hydatid Cyst disease; A case report and literature review. Med. J. S. Punjab 2021, 2, 44–47. [Google Scholar] [CrossRef]
- Kilic, M.; Yoldas, O.; Koc, M.; Keskek, M.; Karakose, N.; Ertan, T.; Gocmen, E.; Tez, M. Can biliary-cyst communication be predicted before surgery for hepatic hydatid disease: Does size matter? Am. J. Surg. 2008, 196, 732–735. [Google Scholar] [CrossRef]
- Dolay, K.; Akbulut, S. Role of endoscopic retrograde cholangiopancreatography in the management of hepatic hydatid disease. World J. Gastroenterol. WJG 2014, 20, 15253. [Google Scholar] [CrossRef] [PubMed]
- Haralabidis, S.; Diakou, A.; Frydas, S.; Papadopoulos, E.; Mylonas, A.; Patsias, A.; Roilides, E.; Giannoulis, E. Long-Term Evaluation of Patients with Hydatidosis Treated with Albendazole and Praziquantel. Int. J. Immunopathol. Pharmacol. 2008, 21, 429–435. [Google Scholar] [CrossRef] [PubMed]
- Velasco-Tirado, V.; Alonso-Sardón, M.; Lopez-Bernus, A.; Romero-Alegría, Á.; Burguillo, F.J.; Muro, A.; Carpio-Pérez, A.; Bellido, J.L.M.; Pardo-Lledias, J.; Cordero, M. Medical treatment of cystic echinococcosis: Systematic review and meta-analysis. BMC Infect. Dis. 2018, 18, 306. [Google Scholar] [CrossRef]
- Lupia, T.; Corcione, S.; Guerrera, F.; Costardi, L.; Ruffini, E.; Pinna, S.M.; Rosa, F.G.D. Pulmonary echinococcosis or lung hydatidosis: A narrative review. Surg. Infect. 2021, 22, 485–495. [Google Scholar] [CrossRef]
- Usluer, O.; Ceylan, K.C.; Kaya, S.; Sevinc, S.; Gursoy, S. Surgical management of pulmonary hydatid cysts: Is size an important prognostic indicator? Tex. Heart Inst. J. 2010, 37, 429–434. [Google Scholar] [PubMed]
| Gharbi Type | WHO Type | Cyst Morphology |
|---|---|---|
| I | CE 1 | Unilocular anechoic lesion with “double line” sign |
| II | CE 2 | Multiseptated rosette-like honeycomb cyst |
| III | CE 3A | Cyst with detached membranes (“water-lily” sign) |
| III | CE 3B | Cyst with daughter cysts in sold matrix |
| IV | CE 4 | Cyst with heterogenous hypoechoic/hyperechoic contents No daughter cysts |
| V | CE 5 | Solid plus calcified wall |
| WHO Type | Main Characteristics | Viability | Treatment |
|---|---|---|---|
| Cystic Echinococcosis 1 | Uniloculated cyst with “double wall” sign | Active cyst (viable) | ABZ 3-6 months (small cyst < 5 cm) or percutaneous treatment (PAIR) and 1-6 months ABZ (cysts 5–10 cm) or surgery + 1–3 months ABZ (cysts > 10 cm) |
| Cystic Echinococcosis 2 | Uniloculated cyst with regular, vascular “septations” resembling daughter cysts | Active cyst (viable) | Surgery + 1–3 months ABZ or modified percutaneous treatment + 1–3 months ABZ or ABZ alone 3–6 months if cyst is small |
| Cystic Echinococcosis 3A | Continuous thin and regular membranes floating in cyst, resembling detached parasite layers | Transitional (variable viability) | ABZ 3-6 months (small cysts < 5 cm) or percutaneous treatment (PAIR) + 1–6 months ABZ (cysts 5–10 cm) or surgery + 1–3 months ABZ (cysts > 10 cm) |
| Cystic Echinococcosis 3B | Predominantly solid with daughter cysts | Transitional (variable viability) | Surgery + 1–3 months ABZ or modified percutaneous treatment + 1–3 months ABZ or ABZ 1–6 months if cyst is small |
| Cystic Echinococcosis 4 | Parasite membranes embedded in heterogenous, avascular solid content (“ball of wool” appearance) | Inactive (low or no viability) | Watch and wait |
| Cystic Echinococcosis 5 | Cysts with solid content with eggshell wall calcifications | Inactive (no viability) | Watch and wait |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Calu, V.; Enciu, O.; Toma, E.-A.; Pârvuleţu, R.; Pîrîianu, D.C.; Miron, A. Complicated Liver Cystic Echinococcosis—A Comprehensive Literature Review and a Tale of Two Extreme Cases. Tomography 2024, 10, 922-934. https://doi.org/10.3390/tomography10060070
Calu V, Enciu O, Toma E-A, Pârvuleţu R, Pîrîianu DC, Miron A. Complicated Liver Cystic Echinococcosis—A Comprehensive Literature Review and a Tale of Two Extreme Cases. Tomography. 2024; 10(6):922-934. https://doi.org/10.3390/tomography10060070
Chicago/Turabian StyleCalu, Valentin, Octavian Enciu, Elena-Adelina Toma, Radu Pârvuleţu, Dumitru Cătălin Pîrîianu, and Adrian Miron. 2024. "Complicated Liver Cystic Echinococcosis—A Comprehensive Literature Review and a Tale of Two Extreme Cases" Tomography 10, no. 6: 922-934. https://doi.org/10.3390/tomography10060070
APA StyleCalu, V., Enciu, O., Toma, E.-A., Pârvuleţu, R., Pîrîianu, D. C., & Miron, A. (2024). Complicated Liver Cystic Echinococcosis—A Comprehensive Literature Review and a Tale of Two Extreme Cases. Tomography, 10(6), 922-934. https://doi.org/10.3390/tomography10060070






